Back to Journals » Infection and Drug Resistance » Volume 12
Antimicrobial susceptibility among Streptococcus pneumoniae and Haemophilus influenzae collected globally between 2015 and 2017 as part of the Tigecycline Evaluation and Surveillance Trial (TEST)
Authors Zhang Z, Chen M, Yu Y, Pan S, Liu Y
Received 27 January 2019
Accepted for publication 8 April 2019
Published 10 May 2019 Volume 2019:12 Pages 1209—1220
DOI https://doi.org/10.2147/IDR.S203121
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Suresh Antony
Zhijie Zhang,1 Meng Chen,2 Ying Yu,3 Sisi Pan,3 Yong Liu1
1Clinical Laboratory Department, Shengjing Hospital of China Medical University, Shenyang, People’s Republic of China; 2Department of Rheumatology, Affiliated Hospital of Hebei University, Baoding, People’s Republic of China; 3Medical Affairs Department, Pfizer Investment Co., Ltd, People’s Republic of China
Purpose: In order to understand the prevalence of Streptococcus pneumoniae and Haemophilus influenzae globally and provide the basis for rational use of antimicrobials in clinical settings, in vitro activity of tigecycline and comparative agents was evaluated against 3929 S. pneumoniae and 4043 H. influenzae isolates obtained from 150 centers globally between 2015 and 2017 as a part of the Tigecycline Evaluation and Surveillance Trial (TEST).
Methods: Broth microdilution methods were performed to determine the minimum inhibitory concentration (MIC) of the isolates according to the guidelines of the Clinical and Laboratory Standards Institute (CLSI). The CLSI breakpoint was used to determine antimicrobial susceptibilities, except for that of tigecycline, for which the US Food and Drug Administration (FDA) breakpoints were used.
Results: More than 99% of S. pneumoniae isolates were susceptible to vancomycin (100%), linezolid (100%), tigecycline (99.9%), and levofloxacin (99.1%). Macrolides (erythromycin, azithromycin, and clarithromycin, 67.1–69.4% susceptibility globally) and penicillin (61.7% in globally) were the drugs with more resisitance to S. pneumoniae. Penicillin-intermediate and -resistant isolates were found in 24.8% and 13.6% of S. pneumoniae isolates. H. influenzae was highly susceptible (>98.7%) to all antibiotics tested except for ampicillin, for which susceptibility was 76.1%. The number of drugs with the lowest susceptibility calculated in Asia were far more than other regions, with 61.5% (8 in 13 drugs) in S. pneumoniae and 70.0% (7 in 10 drugs) in H. influenzae, respectively.
Conclusions: Vancomycin, linezolid, tigecycline, and levofloxacin can be used as the first choice in the empirical therapy of infection disease caused by S. pneumoniae. But macrolides and penicillin should be used prudently in treatment of the infection caused by S. pneumoniae, as well as ampicillin treat the infection caused by H. influenzae. Asia was the region with the most severe resistance in S. pneumoniae and H. influenzae.
Keywords: Streptococcus pneumoniae, Haemophilus influenzae, antimicrobial susceptibility, tigecycline
Introduction
Streptococcus pneumoniae and Haemophilus influenzae are among the major cause of respiratory tract infections, including community-acquired pneumonia (CAP) and acute episodes of chronic obstructive pulmonary disease (COPD), which account for significant mortality.1,2,3,4 In Germany, the incidence of CAP was 0.97% and mortality was 12.9%.2 Lower respiratory infections were the leading cause of death among children younger than 5 years old, accounting for mortality of 11.8%.3 Both pathogens also caused sepsis, meningitis, otitis media, and osteoarthritis.5,6,7
In many countries, antimicrobial therapeutic choices for respiratory tract infections are usually empirical because the limited availability and delay of laboratory identification and antibiotic susceptibility testing result. With the increasing prevalence of resistant organism, the importance of surveillance studies is widely shown.
The Tigecycline Evaluation and Surveillance Trial (TEST) is a global surveillance study focused on monitoring antimicrobial resistance worldwide, which has continued for several years and made a great contribution to the prevalence of bacterial resistance, providing important evidence for empirical therapeutic use of antimicrobial agents in different infectious diseases. This report summarizes data of S. pneumoniae and H. influenzae, the most common pathogens in respiratory tract infections, from TEST between 2015 and 2017. The resistance difference in diverse regions of two bacteria has been compared and the principles of antibiotic use in related diseases has been established.
Materials and methods
Isolate collection
Each participating center was required to contribute at least 135 gram-negative and 65 gram-positive organisms in every study year. Fifteen S. pneumoniae and 15 H. influenzae were included in that total. All isolates had to be collected from patients with infection and the probable causative organism identified using specific laboratory criteria. Only one isolate per patient was accepted. Banked, stored, or duplicate isolates were not accepted.8 All strains were isolated from routine hospital laboratory procedure. They were not specifically obtained for this research.
International Health Management Associates (IHMA, Schaumburg, USA), the reference laboratory, was responsible for the coordination of organism collection, transport, confirmation of identity, storage, and management of the database. Between 10% and 15% of isolates were tested randomly each year by IHMA to verify isolate identity and minimum inhibitory concentration (MIC).
Antimicrobial susceptibility testing
MIC testing was implemented in all centers using broth microdilution methods9 and MicroScan®panels (Dade MicroscanInc, West Sacramento, CA, USA). For S. pneumoniae, the panel included the following antimicrobial agents: amoxicillin-clavulanate, ampicillin, azithromycin, ceftriaxone, clarithromycin, clindamycin, erythromycin, levofloxacin, linezolid, meropenem, minocycline, penicillin, piperacillin-tazobactam, tigecycline, and vancomycin. The panel for H. influenzae testing included amikacin, amoxicillin-clavulanate, ampicillin, cefepime, ceftriaxone, levofloxacin, meropenem, minocycline, piperacillin-tazobactam, and tigecycline.
Quality control was performed using S. pneumoniae ATCC49619, H. influenzae ATCC49247, and H. influenzae ATCC49766 each testing day.
Antimicrobial resistance determination
Breakpoints of the Clinical and Laboratory Standards Institute (CLSI)10 were used to determine the susceptibility of antimicrobial agents, with the exception of tigecycline, for which the US Food and Drug Administration (FDA) breakpoints were used.11The penicillin oral breakpoints (susceptible ≤0.06 mg/L, intermediate 0.125-1, resistant ≥2 mg/L) were used for S. pneumoniae according to a previous study.8 β-lactamase produced by H. influenzae was detected using the preferred local methodology at each center.
Statistical analysis
SPSS Statistics 17.0 (SPSS Inc., Chicago, IL, USA) was used to analyze the data. A comparison of susceptibility rates was conducted using Pearson chi-squared test or continuity-adjusted chi-squared test.
Results
From 2015 to 2017, a total of 3,929 S. pneumoniae and 4,043 H. influenzae isolates were obtained from 150 centers in 47 countries (Table 1). Europe was the main contributor with 62.2% (2442/3929) in S. pneumoniae and 62.6% (2529/4043) in H. influenzae, followed by North America (19.7% and 18.4%, respectively), Asia (7.8% and 11.6%, respectively), and Latin America (6.2% and 3.8%, respectively). The Middle East and Africa only contributed 69–92 isolates from 6 centers of 5 countries (Table 1).
 | Table 1 Number of isolates (centers) contributing S. pneumoniae or H. influenzae between 2015 and 2017 |
From all patients, the proportion of subjects of 61–80 years of age was 33.0% and 31.5% in S. pneumoniae and H. influenzae, respectively, following by 0–20 and 41–60 year-old patients with the proportion of 21.2–28.0%. Sputum and blood were the main specimen source in S. pneumoniae, with proportions of 27.7% and 27.5%, respectively. But in H. influenzae, sputum (42.0%) was the major source followed by trachea/lung (27.2%) samples including specimens from trachea, bronchials or lungs, such as bronchoalveolar lavage fluid (BALF) and bronchialbrush. There were 3.3% S. pneumoniae and 0.5% H. influenzae isolated from cerebrospinal fluid (Table 2).
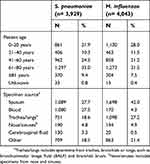 | Table 2 Age distribution and specimen source for culture of S. pneumoniae and H. influenzae isolates between 2015 and 2017 |
S. pneumoniae
More than 99% isolates of S. pneumoniae were susceptible to vancomycin (100%), linezolid (100%), tigecycline (99.9%), and levofloxacin (99.1%) (Table 3). Four tigecycline nonsusceptibility isolates, MIC 0.25–2 μg/mL, from Asia, Canada, and Florida, USA. There are 36 levofloxacin nonsusceptible S. pneumoniae isolates (11 intermediate, 25 resistance) from diverse regions. Ninteen isolates (52.8%) from ≥61 -year-old patients, 15 isolates (41.7%) from 30 to 60-year-olds and 2 isolates (5.6%) from patients <18 years.
 | Table 3 Antimicrobial susceptibility (MIC90, mg/L, and % susceptibility) among S. pneumoniae, PISP, and PRSP isolates in various regions and global between 2015 and 2017 from TEST study |
Amoxicillin-clavulanate susceptibility were above 92% in most regions except for Africa and Asia, which showed 82.6% and 88.0%, respectively. Ceftriaxone susceptibility was observed in 97.8% of isolates globally and more than 96.7% of isolates in many regions other than Africa (92.8%) and Asia (91.6%). Meropenem susceptibility was below 60.9% in Africa, Asia, and the Middle East, but above 81.6% in Europe, North America, and Latin America (Table 3).
Minocycline susceptibility was 74.5% globally, over 79.2% in Europe and North America, but only 30.8% in Asia (Table 3).
With the exception of Latin America, for which susceptibility of azithromycin (33.1%) was lower than that of clarithromycin (66.8%) and erythromycin (66.4%), similar susceptibility was observed in three macrolides (azithromycin, clarithromycin, and erythromycin) in each region. But differences were observed between regions. The isolates with the most susceptibility were obtained in Europe (78.5–78.7%), followed by Latin America (66.4–66.8%, except for azithromycin), and North America (61.2–61.6%). The susceptibility of lincosamide clindamycin (45.9–85.2%) was 4.9–37.3% higher than that of erythromycin (31.4–78.6%) in each region and globally (Table 3).
The susceptibility of penicillin (61.7%) to S. pneumoniae globally was the lowest one in all antibiotics in the TEST panel. Africa, Asia, and the Middle East were the sources of isolates with 43.5%, 29.9%, and 20.7% susceptibility, respectively, which were lower than those in Europe (67.6%), North America (65.2%) and Latin America (52.2%).
In total, 973 (24.7%) and 533 (13.6%) isolates of S. pneumoniae showed intermediate and resistance to penicillin—penicillin-intermediate S. pneumoniae (PISP) and penicillin-resistant S. pneumoniae(PRSP), respectively. The susceptibility of macrolides, β-lactams, clindamycin and minocycline were strongly influenced by penicillin nonsusceptibility among S. pneumoniae. The susceptibility of PISP isolates to macrolides, minocycline, clindamycin and meropenem lower than PSSP (penicillin-susceptible S. pneumoniae) isolates with the decrease of susceptibility of 32.9–34.4%, 28.9%, 26.3% and 18.2% respectability. Susceptibility was reduced by 96.4% in meropenem, about 60% in macrolides among PRSP. Statistical analysis showed significant difference in all drugs between PSSP and PISP or between PISP and PRSP although only 0.6% and 1% reduction in Amoxclav and ceftriaxone compared PSSP to PISP (P=0.003, P<0.0001 respectively) (Figure 1).
Susceptibility of eight drugs (61.5%, 8/13) in Asia, two drugs (15.4%, 2/13) in the Middle East, and one drug (7.8%,1/13) in Africa were the lowest sensitive drugs in six regions in 13 antibiotics with breakpoint.
H. influenzae
H. influenzae isolates were highly susceptible (>98.7%) to all antibiotics in the TEST panel except for ampicillin, for which susceptibility ranged from 57.6% in Asia to 86.1% in Africa (Table 4).
In total, 877 (21.7%) β-lactamase-positive H. influenzae and 110 (2.7%) BLNAR (β-lactamase negative, ampicillin-intermediate or resistant) isolates were reported between 2015 and 2017. The highest prevalence of β-lactamase-positive H. influenzae was observed in Asia (37.3%) and the North America (28.7%) (Table 4). Susceptibility of β-lactamase-positive H. influenzae isolates to antibiotics in the TEST panel were higher than 97%, except for amoxicillin-clavulanate in Asia, which showed 89.1% susceptibility.
Asia was the region with the highest prevalence of BLNAR isolates (8.3%), with 1.1–2.8% prevalence in other regions. Susceptibility was calculated only for isolates from Asia, Europe, and globally because there were inadequate (<10) isolates from other regions. The susceptibility of BLNAR isolates over 93% to all antibiotics except for ampicillin (Table 4).
Susceptibility of seven drugs (70.0%, 7/10) in Asia, one drug (10.0%,1/10) in Africa were the lowest sensitive drugs in six regions in 10 antibiotics with breakpoint.
Discussion
Europe and North America were the two largest contributors of S. pneumoniae and H. influenzae between 2015 and 2017, representing 81.8% and 80.9% of the total samples, respectively. The data may be could not reflect the truth of Africa and the Middle East because only 69–92 isolates were collected in these regions. We should analysis the results of our study combined with other investigations.
In this research, about one third of isolates in S. pneumoniae and H. influenzae were sourced from 61–80-year-old patients, followed by 0–20 and 41–60-year-olds, very similar to the investigation of Tian et al.12 About one half of S. pneumoniae from lower respiratory tract (sputum and trachea/lungs), and one quarter from blood indicated the common infection site of S. pneumoniae. The familiar relationship of H. influenzae with infection of respiratory tract reflected through two thirds of H. influenzae sourced from sputum and trachea/lungs. Compared with H. influenzae, more infections of the central nervous system were caused by S. pneumoniae.
As the most susceptible antibiotics against S. pneumoniae, vancomycin, linezolid, tigecycline, and levofloxacin must play an important role in empirical therapy of clinical infection disease caused by S. pneumoniae. We should pay attention to four nonsusceptible isolates to tigecycline in our study. There is no report about the resistant mechanism of S. pneumoniae collected from clinical disease against tigecycline. Cattoir et al13 analysis of five isolates of Enterococcus feacium which tigecycline nonsusceptible (three in vitro mutants and two clinical isolates). One amino acid substitution in RpsJ protein(S10 ribosomal protein) caused by four modifications was regarded as the main reason of increasing MIC of tigecycline in E.feacium. Lupien et al14 detected the resistance mutant isolates of S. pneumoniae against tigecycline induced in the laboratory. They found the resistance of tigecycline to gram-positive coccus correlated with ribosome constituents, such as ribosomal protein, 16S rRNA and rRNA methyltransferase, different from resistance to gram-negative bacilli mainly related with the efflux. There were 36 S. pneumoniae exhibited levofloxacin-nonsusceptible isolates in this study. Research of Schmitz et al15 verified that levofloxacin-resistant S. pneumoniae associated with 1 or 2 point-mutations of quinolone resistance determining regions (QRDRs). In accordance with our results, these resistant isolates sourced from adult total, possibly because quinolone cannot be used in children and the resistant isolates have not been screened.
The superactivity of tigecycline against S. pneumoniae and H. influenzae in the current study have shown consistent results in many recent studies. In China, 881 S. pneumoniae isolates obtained from 26 hospitals between 2011 and 2016 showed 100% tigecycline susceptibility.16 Pfaller et al observed significant tigecycline activity against S. pneumoniae, with a mean MIC90 of 0.06 mg/L, from North America, Europe, Latin America, and Asia-Pacific.17 Data from the SENTRY Antimicrobial Surveillance Program indicated tigecycline MIC90 values against S. pneumoniae below 0.06 mg/L in Latin America,18 the US,19 and Asia,20 and a MIC90 value of 0.5 mg/L against H. influenzae in the US.19
Lower susceptibilities of amoxicillin-clavulanate, ceftriaxone, and meropenem were observed in Africa and Asia compared to those in other regions in our study. There is a systematic review21 about antimicrobial resistance in Africa collected from 2013–2016 showing 17.4% and 2.2% median resistance to amoxicillin-clavulanate and ceftriaxone, similarly with our investigation (82.6% and 92.8% susceptibility in Africa, respectively). A report from China22 showed resistances of S. pneumoniae that were greater than 30% to amoxicillin-clavulanate and ceftriaxone, more serious than our study (88.0% and 91.6% susceptibility in Asia, respectively). By contrast, the susceptibility to these two drugs in Korea23and Iran24 were similar to those found in our study. The same resistance trends about amoxicillin-clavulanate and ceftriaxonein diverse study shows the severe drug resistance may be the real status in Africa and Asia.
There are fewer reports about the prevalence of meropenem-resistance S. pneumoniae. A study about a Chinese hospital25 showed a 15.4% resistance of meropenem, lower than Asia in this study (40.3% nonsusceptible). Similar to the present investigation, a susceptibility of 84.4% for meropenem against 450 S. pneumoniae collected from the US and Europe in 2014 shown by Pfaller et al.26 Meropenem-resistance S. pneumoniae 15A-ST63 clone has been found in Japan.27 Miyazaki et al28found serotype 35B were the most prevalent in Japan with the highest nonsusceptibility of 81.0% in these S. pneumoniae. 15A and 35B is not included in the currently used PCV-13 vaccine (pneumococcal conjugate valent) in Japan. The two serotype of S. pneumoniae may be the main serotype of infection disease of S. pneumoniae in future Japan even in other regions.
The susceptibility of minocycline in 2015–2017 was higher than 2009–2012 TEST study in S. pneumoniae8 increasing from 51.7–74.5% globally and 20.9–64.8% to 30.8–81.6% in six regions. More observation and analysis should be made in future to determine whether these trends are significant.
The susceptibility of S. pneumoniae to macrolides were comparable in 2015–2017 and 2009–20128 except for those from the Middle East, for which susceptibility decreased from 65.9–66.3% in 2009–2012 to 34.1–36.3% in 2015–2017. In the present investigation, the global susceptibility to macrolides was the lowest in all antibiotics tested except for penicillin. The Middle East and Asia were the regions showing the lowest susceptibility to macrolides.
Similar results have been reported in other research. Grace et al29 summarized results from the Middle East since 2003, which showed a rapid increase in resistance of S. pneumoniae against macrolides from below 30% before 2007 to 77% after 2007. The findings of community-acquired respiratory tract infection pathogen surveillance (CARTIP) in the Asia-Pacific region30 and a study of 17 cities in China22 showed that resistance of macrolides was approximately 90%. Results from the Survey of Antibiotic Resistance (SOAR) in 2012–2014 indicated that the nonsusceptibility of macrolides to S. pneumoniae was 80% in South Korea and 50% in India, Singapore, and Thailand.31 However, meta-analysis data of Iroh Tam et al from South Africa32 and children in Africa33showed that susceptibility of erythromycin were 97.6% and 94.0%, which were far more sensitive than the 50.7% found in our study. The number of S. pneumoniae isolates from Africa were the least of those obtained from the six regions in our study. To reflect the real situation in Africa, more isolates should be collected. Results consistent with those from our study were found in SENTRY18 in Latin America, with an observed susceptibility to erythromycin of 65.5%. The results from an investigation of the European Antimicrobial Resistance Surveillance Network (EARS-Net) and Antibiotic Resistance and Prescribing in European Children (ARPEC) showed resistance to macrolides of 15.3% and 33.1%.34 The reports from Latin America and Europe were all similar to our findings (about 66% and 78% susceptibility, respectively).
The susceptibility of clindamycin to S. pneumoniae 4.9–37.3% higher than that of erythromycin in each region and globally, indicated that there are a portion of isolates resistant to macrolides meanwhile sensitive to clindamycin. These S. pneumoniae may be the M phenotype isolates carrying mef(E)/mel efflux pump gene, which is resistance to 14- and 15-membered macrolides but susceptible to lincosamides, and streptogramin B. These isolates display low level resistance (MICs 1–8 μg/mL) to erythromycin usually.35 The most common macrolides resistance mechanism in S. pneumoniae is ribosomal methylation coded by erm(B) gene which confer resistance to macrolides, lincosamides, and streptogramin B. This phenotype is characterized as the MLSB.35 From our investigation, The propotion of MLSB was about 20% in globally which were the major reason lead to the resistance of macrolides against S. pneumoniae. The propotion of M phenotype is about 10% globally.
Susceptibility of S. pneumoniae to penicillin is very important, only 61.7% in globally. Latin America, Africa, Asia, and the Middle East yielded S. pneumoniae isolates with lower susceptibility to penicillin compared with Europe and North America. Similar results have been found in other studies. In Asia, from 17 hospitals of China, the resistance of S. pneumoniae to penicillin was 50%,22 similar to that reported from Guangzhou, China.25The susceptibility of penicillin was just 21.2% in Korea and 49.3%, 63.8%, and 49.0% in India, Singapore, and Thailand, respectively.31 The resistance of penicillin to S. pneumoniae increased to 40–70% after 2008 in the Middle East,29 and the susceptibility was 68.6% and 78.0% in Africa.32,33 These results approximated those data of Asia, the Middle East, and Africa in our study. However, reports from SENTRY in Latin America indicated a susceptibility of 87.2%, which was higher than 51.7% reported in the current study for this region.18
A significant decrease in susceptibility was observed in PISP and PRSP against three β-lactams, three macrolides, clindamycin, and minocycline (Figure 1), that is to say, the multidrug resistance (MDR) isolates increasing significantly with the resistance of penicillin. MDR was observed in 59.3% of isolates from Asian countries in ANSORP Surveillence, reflecting the severe resistance of S. pneumoniae. Clinical therapy of infection due to MDR S. pneumoniae is difficult and often ineffective. The misuse and overuse of antimicrobials may be responsible for this rise in MDR isolates.
With the susceptibility of over 98.7% in all antibiotics except for ampicillin in H. influenzae manifested that these antimicrobial drugs can be used as the first choice in the empirical treatment of H. influenzae. Reports on the susceptibility of H. influenzae to ampicillin differ greatly, from 16.7% in Korea31to 52.5–63.3% in Singapore,31 Thailand,31 and China36 to 91.1% in India.31 So, surveillance in different regions, such as diverse countries, provinces, cities even hospitals is necessary for the empirical therapy of infectious disease.
In Asia, except for ampicillin, six drugs were the lowest sensitive drug in all regions in H. influenzae from 2015–2017. Highest prevalence of BL-Pos and BLNAR H. influenzae appeared in Asia also, 37.3% and 8.3%, respectively, significantly increased compared with the same TEST study of 2004–2012 (27.8% and 3.5%, respectively).8 We should pay attention to the relatively severe resisitance of H. influenzae in Asia. It is extremely urgent to control the development of the resistance of antimicrobial drugs.
Conclusions
In summary, the high activities determined for vancomycin, linezolid, tigecycline, and levofloxacin against S. pneumoniae manifested that these antibiotics can be used as the first choice in the empirical therapy of infection disease caused by S. pneumoniae. However, macrolides and penicillin should be used prudently in treatment the infection caused by S. pneumoniae, as well as ampicillin to H. influenzae.
Asia was the region with the most severe resistance in S. pneumoniae and H. influenzae, followed by Africa and the Middle East. Insufficient resource of isolates maybe lead to the deviation of resistance in these regions. But the likely trends found by many other surveillance studies indicated that our research has value. With the serious trend toward drug resisitance in S. pneumoniae and H. influenzae, it is imperative to implement rational and normative use of antibacterial agents at full scale and to take practical actions and adopt effective measures to limit the progress of bacterial resistance in all countries.
Acknowledgments
The authors should like to acknowledge and thank all TEST investigators and laboratories for their participation in the study, as well as the staff at IHMA for their coordination of TEST and at Pfizer, who were involved in study design and the decision to submit this article for publication.
Disclosure
Sisi Pan and Ying Yu are employees of Pfizer Investment Co., Ltd. The authors report no other conflicts of interest in this work.
References
1.
2. Kolditz M, Tesch F, Mocke L, Höffken G, Ewig S, Schmitt J. Burden and risk factors of ambulatory or hospitalized CAP: a population based cohort study. Respir Med. 2016;121:32–38. doi:10.1016/j.rmed.2016.10.015
3.
4. Maddi S, Kolsum U, Jackson S, et al. Ampicillin resistance in Haemophilus influenzae from COPD patients in the UK. Int J Chron Obstruct Pulmon Dis. 2017;12:1507–1518. doi:10.2147/COPD.S150887
5. Zuníno C, Vomero A, Pandolfo S, et al. Etiology and evolution of the osteoarticular infections 2009–2015:pediatric Hospital Pereira Rossell Uruguay. Rev Chilena Infectol. 2017;34(3):235–242. doi:10.4067/S0716-10182017000300005
6. Jiang H, Su M, Kui L, et al. Prevalence and antibiotic resistance profiles of cerebrospinal fluid pathogens in children with acute bacterial meningitis in Yunnan province, China, 2012–2015. PLoS One. 2017;12(6):e0180161. doi:10.1371/journal.pone.0180161
7. Cho YC, Chiu NC, Huang FY, et al. Epidemiology and antimicrobial susceptibility of non-typeable Haemophilus influenzae in otitis media in Taiwanese children. J Micro Immu and Infec. 2017;S1684–S1182(17):30203–30207.
8. Viktorija T, Michael JD. Regional and global antimicrobial susceptibility among isolates of Streptococcus pneumoniae and Haemophilus influenzae collected as part of the Tigecycline Evaluation and Surveillance Trial (T.E.S.T.) from 2009 to 2012 and comparison with previous years of T.E.S.T. (2004–2008). Anna Clin Micro and Antimicrob. 2014;13:52–59.
9.
10.
11.
12. Tian L, Sun Z, Zhang Z. Antimicrobial resistance of pathogens causing nosocomial bloodstream infectionin Hubei Province, China, from 2014 to2016: a multicenter retrospective study. BMC Public Health. 2018;8(1):1121–1128. doi:10.1186/s12889-018-6013-5
13. Cattoir V, Isnard C, Cosquer T, et al. Genomic analysis of reduced susceptibility to tigecycline in Enterococcus faecium. Antimicrob Agents Chemother. 2015;59(1):239–244. doi:10.1128/AAC.04174-14
14. Lupien A, Gingras H, Leprohon P, Ouellette M. Induced tigecycline resistance in Streptococcus pneumoniae mutants reveals mutations in ribosomal proteins and rRNA. J Antimicrob Chemother. 2015;70(11):2973–2980. doi:10.1093/jac/dku445
15. Schmitz J, van der Linden M, Al-Lahham A, Levina N, Pletz MW
16. Li S, Guo Y, Zhao C, et al. In vitro activities of tedizolid compared with other antibiotics against Gram-positive pathogens associated with hospital-acquired pneumonia, skin and soft tissue infection and bloodstream infection collected from 26 hospitals in China. J Med Microbiol. 2016;65(10):1215–1224. doi:10.1099/jmm.0.000347
17. Pfaller MA, Huband MD, Streit J, Flamm RK, Sader HS. Surveillance of tigecycline activity tested against clinical isolates from a global (North America, Europe, Latin America and Asia-Pacific) collection (2016). Inter J Antimicrol Agents. 2018;51(6):848–853. doi:10.1016/j.ijantimicag.2018.01.006
18. Sader HS, Castanheira M, Farrell DJ, Flamm RK, Mendes RE, Jones RN. Tigecycline antimicrobial activity tested against clinical bacteria from Latin American medical centres:results from SENTRY Antimicrobial Surveillance Program(2011–2014). Int J Antimicrob Agents. 2016;48(2):144–150. doi:10.1016/j.ijantimicag.2016.04.021
19. Helio SS, David JF, Robert KF, Ronald NJ. Variation in potency and spectrum of tigecycline activity against bacterial strains from U.S. medical centers since its approval for clinical use (2006 to 2012). Antimicrob Chemother. 2014;58(4):2274–2280. doi:10.1128/AAC.02684-13
20. Mendes RE, Mendoza M, Banga SK, et al. Regional resistance surveillance program results for 12 Asia-Pacific Nations (2011). Antimicrob Chemother. 2013;57(11):5721–5726. doi:10.1128/AAC.01121-13
21. Tadesse BT, Ashley EA, Ongarello S, et al. Antimicrobial resistance in Africa: a systematic review. BMC Infect Dis. 2017;17(1):616–632.
22. Zhao C, Li Z, Zhang F, et al. Serotype distribution and antibiotic resistance of Streptococcus pneumoniae isolates from 17 Chinese cities from 2011 to 2016. BMC Infect Dis. 2017;17(1):804–815. doi:10.1186/s12879-017-2880-0
23. Kim SH, Bae IK, Park D, et al. Serotype distribution and antimicrobial resistance of streptococcus pneumoniae isolates causing invasive and noninvasive pneumococcal diseases in Korea from 2008 to 2014. BioMed Research Inter. 2016;2016:6950482.
24. Houri H, Tabatabaei SR, Saee Y, Fallah F, Rahbar M, Karimi A. Distribution of capsular types and drug resistance patterns of invasive pediatric Streptococcus pneumoniae isolates in Teheran, Iran. Inter J Infec Dis. 2017;57:21–26. doi:10.1016/j.ijid.2017.01.020
25. Songyin H, Xiaoqiang L, Weisi L, et al. Serotype distribution and antibiotic resistance of Streptococcus pneumoniae isolates collected at a Chinese hospital from 2011 to 2013. BMC Infect Dis. 2015;15:312–321. doi:10.1186/s12879-015-1042-5
26. Pfaller MA, Sader HS, Rhomberg PR, Flamm RK. In vitro activity of delafloxacin against contemporary bacterial pathogens from the United States and Europe, 2014. Antimicrob Agents Chemother. 2017;61(4):e02609–e02616. doi:10.1128/AAC.02609-16
27. Nakano S, Fujisawa T, Ito Y, et al. Spread of meropenem-resistant Streptococcus pneumoniae Serotype 15A-ST63 Clone in Japan, 2012–2014. Emerg Infect Dis. 2018;24(2):275–283. doi:10.3201/eid2402.171268
28. Miyazaki H, Shibuya R, Midorikawa N, Chang B, Ohnishi M, Matsumoto T. Serotype distribution and antimicrobial susceptibility of Streptococcus pneumoniae strains isolated in Japan after introduction of the routine immunization program. J Infect Chemother. 2017;23(4):234–240. doi:10.1016/j.jiac.2016.12.016
29. Grace EM, Marwan O, Rayane R, Fouad D, Monzer H. Molecular mechanisms and epidemiology of resistance in Streptococcus pneumoniae in the Middle East region. J Med Micro. 2017;66(7):847–858. doi:10.1099/jmm.0.000503
30. Zhang Y, Zhang F, Wang H, et al. Antimicrobial susceptibility of Streptococcus pneumoniae, Haemophilus influenzae and Moraxella catarrhalis isolated from community-acquired respiratory tract infections in China: results from the CARTIPS antimicrobial surveillance program. J Glob Antimicrob Resist. 2016;5:36–41. doi:10.1016/j.jgar.2016.03.002
31. Torumkuney D, Chaiwarith R, Reechaipichitkul W, et al. Results from the Survey of Antibiotic Resistance (SOAR) 2012–14 in Thailand, India, South Korea and Singapore. J Antimicrob Chemother. 2016;71(Suppl1):i3–i19. doi:10.1093/jac/dkw073
32. Iroh Tam PY, Sadoh AE, Obaro SK. A meta-analysis of antimicrobial susceptibility profiles for Pneumococcal pneumonia in sub-Saharan Africa. Paedia Inter Child Health. 2018;38(1):7–15. doi:10.1080/20469047.2017.1298700
33. Iroh Tam PY, Thielen BK, Obaro SK, et al. Childhood pneumococcal disease in Africa – A systematic review and meta-analysis of incidence, serotype distribution, and antimicrobial susceptibility. Vaccine. 2017;35(15):1817–1827. doi:10.1016/j.vaccine.2017.02.045
34. Bielicki JA, Lundin R, Sharland M. Antibiotic resistance prevalence in routine bloodstream isolates from children’s hospitals varies substantially from adult surveillance data in Europe. Pediatr Infect Dis J. 2015;34(7):734–741. doi:10.1097/INF.0000000000000652
35. Schroeder MR, Stephens DS. Macrolide resistance in Streptococcus pneumoniae. Front Cell Infect Microbiol. 2016;6:98 eCollection 2016. doi:10.3389/fcimb.2016.00098
36. Li JP, Hua CZ, Sun LY, Wang HJ, Chen ZM, Shang SQ. Epidemiological features and antibiotic resistance patterns of Haemophilus influenzae originating from respiratory tract and vaginal specimens in pediatric patients. J Pediatr Adolesc Gynecol. 2017;30(6):626–631. doi:10.1016/j.jpag.2017.06.002
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.


