Back to Journals » International Journal of Nanomedicine » Volume 12
Anti-inflammatory activity of niosomes entrapped with Plai oil (Zingiber cassumunar Roxb.) by therapeutic ultrasound in a rat model
Authors Leelarungrayub J, Manorsoi J , Manorsoi A
Received 1 December 2016
Accepted for publication 26 January 2017
Published 29 March 2017 Volume 2017:12 Pages 2469—2476
DOI https://doi.org/10.2147/IJN.S129131
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Thomas Webster
Jirakrit Leelarungrayub,1 Jiradej Manorsoi,2,3 Aranya Manorsoi2,3
1Department of Physical Therapy, Faculty of Associated Medical Sciences, Chiang Mai University, 2Manose Health and Beauty Research Center, Mueng, 3Division of Cosmetic Technology, Faculty of Science and Technology, North Chiang Mai University, Chiang Mai, Thailand
Objective: The aim of this study was to evaluate the antioxidant and anti-inflammatory activities of Plai oil–encapsulated niosomes (Zingiber cassumunar Roxb.) on inflamed subcutaneous Wistar rat skin by therapeutic ultrasound.
Methods: Pure oil from Plai rhizomes was extracted by steam distillation, and antioxidant activities were determined by 2,2-diphenyl-1-picrylhydrazyl radical scavenging assay. Bioactive compounds were analyzed by gas chromatography-mass spectrometry. Niosome particles containing Plai oil were prepared by chloroform film method with sonication before testing for anti-inflammatory activity on locally inflamed subcutaneous rat skin after inducement from lipopolysaccharide with ultrasound once a day for 3 days. Skin temperatures and blood flow were evaluated.
Results: Plai oil presented antioxidant activity that inhibited 2,2-diphenyl-1-picrylhydrazyl radicals. Four active compounds found in the essential oil were sabinene, γ-terpinene, terpinene-4-ol, and (E)-1-(3,4-dimethyoxy phenyl) butadiene. Application of ultrasound (0.2 W/cm2, 20%, 3 min) with gel containing Plai oil–encapsulated niosomes decreased skin temperature and blood flow to the lowest level compared to the application of neurofen drug or gel-based control.
Conclusion: Plai oil, which consists of four main bioactive compounds and possesses antioxidant and anti-inflammatory activities, can be applied against local subcutaneous inflammation when used with therapeutic ultrasound via entrapped niosomes.
Keywords: antioxidant, anti-inflammation, Plai oil, niosomes, therapeutic ultrasound, Zingiber cassumunar Roxb.
Introduction
Today, use of physical therapy to relieve pain or cure inflammation can be modified with various protocols such as rest and ice therapy before any therapeutic device application. Application of ultrasound is one of the many methods that has claimed to benefit the healing process and inflammation.1 Therapeutic ultrasound has been referred to as simple in the long term.2 The biological effects of ultrasound such as compressional, tensile, and shear stresses, including ultrasound energy transmission to improve blood circulation and biomolecule changes in inflammatory processes, are a part of basic knowledge.3 However, phonophoresis or sonophoresis is applicable sometimes when anti-inflammatory drugs, such as neurofen or lidocaine, are mixed with gel-based control during ultrasound application. In this case, application of ultrasound with anti-inflammatory drugs that eliminate pain or inflammation provides better clinical benefit than ultrasound alone. Nevertheless, ultrasound has been applied to eliminate pain or inflammation in clinics for a long time. On the other hand, many traditional plants have been used to release pain and inflammation, for example, Elaeodendron croceum and Calpurnia aurea extracts4 and Poikilacanthus glandulosus (Nees) Ariza leaves,5 including essential oil from Cameroon.6 Also, Thai medicinal plants such as Smilax corbularia Kunth,7 and Sahasthara,8 also present anti-inflammatory and antioxidant activities. Therefore, many medicinal plants present anti-inflammation activity worldwide. Of many Thai medicinal plants, Plai (Zingiber cassumnar Roxb.) belongs to Zingiberaceae family and has specific characteristics such as yellow rhizome and slim leaf. Previous evidence confirmed that it possesses anti-inflammatory,9 anti-histamine,10 local anesthetic,11 and anti-pain12 activities. Each rhizome contains essential oil of ~0.5%–0.9% of total fresh weight that is composed of Sabinene (25%–45%), γ-terpinene (5%–10%), alpha-terinene (2%–5%), terpinene-4-ol (25%–45%), and E-1-(3,4-dimethoxyphenl) butadiene (DMPBD) (1%–10%).13 Toxicity was not presented on rat or rabbit skin14–16 and neither was genetic mutation.9 A previous study showed that the skin absorbed essential oil 1 h after the oil was applied to the skin.17 Plai oil is currently a commercial product in Thailand as are many other products, for example, Plygesal® and Plaitanoids™ that are produced and distributed to relieve muscle pain. Unfortunately, after the oil has been extracted, its physical and chemical properties rapidly undergo changes, for example, a rancid smell and color change develop due to oxidation process when exposed to light or air, which relates to low stability and loss of active compounds. In 2005, a previous review proposed that microemulsion of essential oil had better benefits on skin application when used with ultrasound.18 Moreover, possibly clinical treated efficiency on sonophoresis has been proposed and suggested with modern transdermal delivery systems such as liposomes or niosomes.19 A previous report suggested that liposomes or niosomes, which were synthesized from Tween 80 by thin-film hydration technique, had more stability and high potential vehicles for delivering some compounds to tissues.20 Thus, the objective of this study was to develop a niosome prototype and modify Plai oil by encapsulating niosomes for application in therapeutic ultrasound in order to enhance the anti-inflammation activity of Plai oil.
Materials and methods
Raw material and oil preparation
Two-year-old rhizomes of Plai, without toxic insecticide spraying, were purchased from a local village farmer in Chiang Mai Province, Thailand. Plai rhizomes (Z. cassumnar Roxb.) were characterized by comparing them with a standard voucher specimen that was deposited at the Chiang Mai University Biology Herbarium, Department of Biology, Faculty of Science, Chiang Mai University. Scraping the outermost shell of the fresh rhizome, cleaning, and slicing off pieces were performed before extracting the oil by steam distillation using Clevenger-type apparatus. Then, antioxidant activity and active compounds were analyzed.
Antioxidant assay
1,1-Diphenyl-2-picryl hydrazyl (DPPH) radical scavenging
The antioxidant activity of essential oil was analyzed by DPPH radical scavenging assay.21 After determining the density of Plai oil, the Plai oil diluted in ethanol (50 μL) at 12.5–400 μg/mL was mixed in DPPH solutions and kept in dark for 30 min. Absorbance was read by spectrophotometry at 515 nm. The inhibition percentage of essential oil was compared with standard Trolox (Sigma, St Louis, MO, UK).
Active compound analysis
The major compound in Plai oil was identified by gas chromatography (CG)-mass spectrophotometry (MS) (Wiley7n.l) at the Science and Technology Service Center, Faculty of Science, Chiang Mai University, Thailand. The Agilent GC/MS system consisted of GC6850 and a 5873 MSD, HP-MS 5 column (30 m ×0.25 mm ID ×0.25 μm film thickness). The flow rate of helium carrier gas was 1.0 mL/min with the interface and ion source temperatures at 150°C and 230°C, respectively. The program of GC analysis was set up with an inlet from 80°C to 250°C in 10 min. Finally, the mass spectrum of each compound in the oil was compared to the standard mass spectrum in an MS library (Wiley7n.I).
Oil-encapsulated niosome preparation
Niosomes encapsulated with Plai oil were prepared by film hydration method22 with some modifications. The niosomes comprised Tween 60 and cholesterol at a 3:7 molar ratio, in which pure Plai oil was encapsulated at 0.1%. After dissolving Tween and cholesterol in chloroform, the organic solvent was removed by a vacuum evaporator at 50°C–70°C until a thin film was obtained and kept in a desiccator for 24 h. Then, distilled water was added to dissolve the film and to obtain niosomal suspension, which was then subjected to sonication with four cycles of 2 s and pause of 2 s, using a probe sonicator (Bandeline, Berlin, Germany), followed by a 2 s pause. Finally, the oil-encapsulated niosomes were analyzed to confirm whether they fall within the accepted size range (200–500 nm), and their morphology was confirmed with a Zetasizer (Malvern Instruments Ltd, Malvern, UK) and transmission electron microscope (JEM-1200EX; JEOL Co., Akishima, Tokyo, Japan) with ×12.0 K magnification and optical microscope (HFX-DX; Nikon, Minato, Tokyo, Japan).
Lipopolysaccharide (LPS)-induced subcutaneous inflammatory assay in rats
This animal study was conducted in accordance with the guidelines for care and use of laboratory animals. This study approved by the Animal Care Committee, Faculty of Medicine, Chiang Mai University, Thailand. The study protocol was in accordance with a previous study.23 Male Wistar rats (aged 5–8 weeks, weight 250–300 g) were purchased from the National Laboratory Animal Center, Mahihol University, Bangkok, Thailand. They were housed in individual cages and maintained at an ambient temperature of 24°C±1°C at the Faculty of Medicine, Chiang Mai University, Thailand. All the rats were selected and placed into five cages by sampling method and fasted for 16 h before the experiment. The experimental animals were divided into five groups: group 1 was injected with normal saline solution (control; n=5) and groups 2–5 were injected with LPS from Porphyromonas gingivalis at a concentration of 0.1 mg/0.1 mL per area, gently in the subcutaneous area. At 48 h or day 2 postinjection, the rats in group 2 were not treated, those in group 3 were treated with ultrasound via non-oil-encapsulated niosomes gel, those in group 4 were treated with oil-encapsulated niosomes gel, and those in group 5 were treated with ultrasound via neurofen gel.
Ultrasound program
Ultrasound (Intelect®; Chattanooga Medical Supply, Inc, TN, USA) was used in accordance to the treatment guideline,24 with head sound sizes of 2 cm2, 0.2 w/cm2 and 3 MHz for 3 min, with circulating direction technique on the inflamed area (Figure 1A). Nearly 400 mg of gel was used for treatment on head sound in groups 3–5. All treatment was repeated daily for 3 days. The skin temperature (°C) and blood flow (flux per min) were detected with an optical probe and the inflamed skin area was treated directly each day by a laser Doppler blood flow meter (TDR4; MOOR Instrument, Axminster, UK) (Figure 1B).
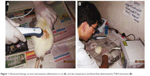  | Figure 1 Ultrasound therapy on local subcutaneous inflammation in rat (A), and skin temperature and blood flow determined by TDR4 instrument (B). |
Statistical analysis
The results were expressed as mean and standard error of mean. Repeated measurement one-way ANOVA in SPSS version 16.0 (SPSS Inc., Chicago, IL, USA) was used to compare between groups statistically on each day, and the significant level was P<0.01.
Results
Results of DPPH radical inhibitory activity by standard Trolox showed a liner correlation (R2=0.989) between concentration and percentage inhibition (Figure 2A). Whereas the activity of Plai oil in inhibiting the DPPH radical had no linear response compared to standard Trolox, but activity increased with an increase in the doses: 12.5 μg/mL (2.5%±1.1%), 25 μg/mL (7.5%±1.4%), 50 μg/mL (12.3%±1.3%), 100 μg/mL (21.2%±1.8%), 150 μg/mL (26.6%±1.6%), 200 μg/mL (32.2%±2.1%), and 400 μg/mL (51.3%±1.3%) (Figure 2B). GC-MS analysis found four main active compounds: terpenen-4-ol (48%), sabinene (19%), DMPBD (15%), and γ-terpinene (2.8%) (Figure 3).
  | Figure 2 The percentage inhibition of DPPH radicals between standard Trolox (0.2–1.2 mmol/L) (A) and Plai oil (12.5–400 μg/mL) (B). |
The maximum load of Plai oil in niosomes was 0.1%, which is the best concentration for physical stability compared to higher concentrations of 0.5%, 1.0%, 1.5%, and 2.0%, in room temperature without particle precipitation. Figure 4 shows the characteristics of niosomes encapsulated with and without Plai oil. The particle size of niosomes was evaluated by transmission electron microscopy at 20,000× and compared with a 100 nm scale (Figure 4A and B). The particle size also was observed by an optical microscope at 1,000× and compared with the microscope scale (nm) (Figure 4C). After preliminary niosomes preparation with various conditions, Tween 60 and cholesterol at 3:7 molar ratio at a final solution of 1.0% (w/v), Plai oil-encapsulated niosomes was suitably applied in therapeutic ultrasound in this study (Figure 5).
  | Figure 5 Oil-encapsulated niosomes and gel-based control (A) and pilot gels containing oil-entrapped niosomes (B). |
The results show that the skin temperature and blood flow increased significantly after LPS injection on days 1 and 2 (Table 1 and Figure 6) compared to the non-LPS-injected group and control rats (P<0.01). Application of therapeutic ultrasound, with gel-based control, gel-based nerofen, and gel-based nioplai (Figure 1), showed significantly lower skin temperatures and blood flow after treatment on days 2–4 (Figure 6; P<0.01) compared to the LPS group. Groups that received ultrasound with gel-based nioplai showed lower skin temperature and blood flow compared to those that received gel-based nerofen.
Discussion
This was a preliminary study that analyzed the anti-inflammatory,9 anti-histamine,10 and anti-pain activities of natural oil from Plai (Z. cassumunar Roxb.).12 Moreover, this study evaluated the antioxidant activity of Plai oil by scavenging DPPH radicals. Although methods such as ferric reducing antioxidant power (FRAP) and DPPH protocols have been documented in previous study for evaluating the antioxidant activity of essential oil, DPPH method responded greatly to the antioxidant activity of essential oil.25 DPPH results of this study confirmed the antioxidant activity of Plai oil. But the concentrations of each compound compared to previous studies were different: sabinene (25%–45%), γ-terpinene (5%–10%), α-terpinene (2%–5%), terpinene-4-ol (25%–45%), and DMPBD (1%–10%),13 sabinene (27%–34%), γ-terpinene (6%–8%), α-terpinene (4%–5%), terpinene-4-ol (30%–35%), and DMPBD (12%–19%).26 In addition, a 2016 study found that Plai rhizome contains terpinene-4-ol (40.5%±6.6%) and sabinene (17.4%±1.4%)18 as active compounds. However, this study identified four active compounds: terpenen-4-ol (48%), sabinene (19%), DMPBD (15%), and γ-terpinene (2.8%) by GC-MS. Different results may be possible due to age, growing area, and material preparation before oil extraction.
Plai oil–encapsulated niosomes were prepared following a well-known pharmaceutical protocol. Niosomes are nonionic surfactant–based vesicles, similar to liposomes that are able to encapsulate either hydrophilic or lipophilic compounds, but with lower cost and greater stability.27 Moreover, protection of active compounds in Plai oil can be improved when encapsulated in niosomal membrane composed of Tween 60 and cholesterol. In addition, the results of this study are in agreement with that of a 2016 study, which shows that essential oil–encapsulated microemulsions, composed of Tween 20 and propylene glycol (2:1) in water, could stabilize active compounds such as sabinene. In addition, previous reports also have claimed that niosomes are able to promote any compound passing through the skin.28 Transmission electron microscope and optical microscope analysis indicated that niosomes fall within a range of 100 nm. However, niosome permeability theory proposes that not only small-sized particles penetrate through the skin,29 but also an optimum ratio of lipid or surfactant may increase the flexibility of the bilayer membrane.30 On the other hand, both liposomes and niosomes revealed significant permeability and nontoxicity on mammalian skin.14–16 Thus, application of niosomes to eliminate subcutaneous inflammation is possibly safe.
Application of therapeutic ultrasound in combination with physical therapy was summarized in an application and safety considerations report,31 which proposes that ultrasound can assist in transporting compound, especially a drug, into the skin via a phonophoresis technique or sonophoresis method.1 Moreover, the ultrasound program with 0.2 W/cm2, 3 MHz for 3 min and circulating technique around the inflamed area was designed following the universal anti-inflamed program in the physical therapy clinic.24 Similar evidence on skin healing discussed in previous reports shows the preferred intensity to be 0.2 W/cm2 when a 1–3 MHz machine is used.32 A previous report suggested that drug delivered can pass the transdermal stratum corneum (10–30 μm) and form a barrier that stops passive drug diffusion by applying low-frequency ultrasound (<100 kHz).33 In addition, a recent study proposed that the cavitation mechanism of ultrasound is possible for drug delivery by sonophoresis,34 which relates to a previous report on drug absorption in capillary vessels after crossing the stratum corneum.35 Furthermore, the ultrasound program in this study followed the universal anti-inflamed program in the clinic.
An animal study that focused on subcutaneous inflammation in accordance to a previous study23 showed that subcutaneous injection of 100 μL of LPS at 0.1 mg to generate an inflammatory condition resulted in an inflamed knob after 24–48 h of postinjection. LPS induces inflammation and involves specific intracellular events of transduction; for example, protein kinase C, tyrosine kinase, and mitogen-activated protein kinase.36 During the inflammation period, signs such as local edema, high temperature, and redness are presented,37 as well as increase in skin blood flow due to microvascular remodeling and release of histamine responses.38 Thus, highest skin temperatures and blood flow were presented by the LPS-treated rat groups, whereas no responses occurred in the non-LPS-injected group.
Group that received ultrasound with niosome placebo on days 2–4 showed decreased skin temperature and blood flow, but those that received ultrasound treatment with Plai–oil entrapped niosomes showed significantly lower skin temperature and blood flow compared to those that received ultrasound, placebo, and neurofen drug (P<0.01). Neurofen is a positive anti-inflammatory drug that is used as a standard comparison to Plai oil. It contains ibuprofen that has been reported as a nonsteroidal anti-inflammatory drug and works by acting on a group of compounds called prostaglandins. It also acts as a non-selective inhibitor of cyclooxygenase-1 (COX)-1 and -2.39 Neurofen containing ibuprofen together with ultrasound treatment presents anti-inflammation by reducing skin temperature and blood flow, but it has less activity than that of Plai oil–encapsulated niosomes. Possible mechanisms on some ingredients or active compounds in neurofen involve skin permeability, and this is still unclear. This study could not confirm the concentration of Plai oil in the inflamed subcutaneous area, but positive results may reflect some benefits of Plai oil in the niosome that can pass through the skin from ultrasound application. Moreover, compounds of Plai oil that act on the prostaglandin pathway are different from neurofen. Plai oil contains more active compounds and has various functions, for example, DMPBD40 and phenylbutenoids inhibit COX-2 activity.41 Furthermore, a study by Chaiyana et al also reported that sabinene and terpinene-4-ol could reduce the expression of nuclear factor-kappa and interleukin-6 released in human peripheral blood mononuclear cells, which reflected the compounds’ anti-inflammatory activity.18 Unfortunately, this study did not evaluate the effect of application of neurofen and Plai oil on anti-inflammatory pathways, which may be studied more in the future.
Conclusion and limitations
This preliminary study showed the possibility of applying a natural medicinal plant product, such as Plai oil with active compounds in a niosomal system, for inhibiting inflammation via therapeutic ultrasound. However, this study could not confirm skin permeability, active compound distribution, or pharmacokinetics in human skin, and more research are needed to confirm these results.
Acknowledgments
This study received a grant from the Thailand Research Fund (TRF) (MRG5080140). We thank a corresponding author, Jirakrit Leelarungrayub, for providing consent to publish Figure 1B.
Author contributions
JL was responsible for obtaining funding, experimental design, establishing all protocols, and performing all testing and data analysis. JM and AM prepared and established the preparation of niosomes and also assisted in rechecking the original and final versions of the manuscript. All authors contributed toward data analysis, drafting and revising the paper and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
Machet L, Boucaud A. Phonophoresis; efficiency, mechanisms and skin tolerance. In J Pharm. 2002;243(1–2):1–15. | ||
Robertson VJ, Baker KG. A review of therapeutic ultrasound; effectiveness studies. Phys Ther. 2001;81(7):1339–1350. | ||
Silberstein J, Lakin CM, Kellogg Parsons J. Shock wave lithotripsy and renal hemorrhage. Rev Urol. 2008;10(3):236–241. | ||
Elisha IL, Dzoyem JP, McGaw LJ, Botha FS, Eloff JN. The anti-arthritic, anti-inflammatory, antioxidant activity and relationships with total phenolics and total flavonoids of nice South African plants used traditionally to treat arthritis. BMC Complement Altern Med. 2016;16:307. | ||
de Brum TF, Camponogara C, da Silva Jesus R, et al. Ethnopharmacological study and topical anti-inflammatory activity of crude extract from Poikilacanthus glandulosus (Nees) Ariza leaves. J Ethnopharmacol. 2016;193:60–67. | ||
Ndoye Foe FM, Tchinang TF, Nyegue AM, et al. Chemical composition, in vitro antioxidant and anti-inflammatory properties of essential oils of four dietary and medicinal plants from Cameroon. BMC Complement Altern Med. 2016;16:117. | ||
Ruangnoo S, Jaiaree N, Makchuchit S, Panthong S, Thongdeeying P, Itharat A. An in vitro inhibitory effect on raw 264.7 cells by anti-inflammatory compounds from Smilax corbularia Kunth. Asian Pac J Allergy Immunol. 2012;30(4):268–274. | ||
Kakatum N, Jaiarree N, Makchucit S, Itharat A. Antioxidant and anti-inflammatory activities of Thai medicinal plants in Sahasthara remedy for muscle pain treatment. J Med Assoc Thai. 2012;95(Suppl 1):S120–S126. | ||
Wasuwat S, Nandhasri P, Suntorntanasat T, Rojjanapothi W. Antiinflammatory action of Plai oil, Zingiber cassumunar Roxb. Presented at: The First Princess Chulabhorn Science Congress; December 10–13; 1987; Bangkok, Thailand. | ||
Piromrat K, Tuchinda M, Geadsomnuig S, Koysooko R, Bunjob M. Antihistamic effect of Plai (Zingiber cassumunar Roxb.) on histamine skin test in asthmatic children. Siriraj Hosp Gaz. 1986;38(4):251–256. | ||
Anantasan V, Asayakun S. Study on the local anesthetic effect of squeenzed Zingeber cassumuna Robx. Chiangmai Med Bull. 1971;10(1):10–23. | ||
Panthong A, Kanjanapothi D, Niwatananun V, Tuntiwachwuttikul P, Reutrakul V. Anti-inflammatory activity of compounds isolated from Zingiber cassumnar. Planta Med. 1990;56(6):655. | ||
Masuda T, Jitoe A. Antioxidative and anti-inflammatory compounds from tropical gingers; Isolation, structure determination, and activities of cassumunins A, B, and C, new complex curcuminoids from Zingiber cassumunar. J Agr Food Chem. 1994;42(9):1850–1856. | ||
Kiatyingunsulee N, Wangmad M, Swasdimongkul D, Mokkhasmit M. Some pharmacological studies of active constitute in Plai (Zingiber cassumunar Roxb.) Bull Dep Med Sci (Thailand). 1979;21:13–24. | ||
Panyathanya R, Gnamwat W, Chawalidthumrong P, Permpipat U, Lellakulthanit O, Chantachaya C. Study of acute and chronic toxicity of Plai (Zingiber cassumunar Roxb.) in rats. Siriraj Hosp Gaz. 1986;38(6):413–416. | ||
Mokkhasmit M, Swatdimongkol K, Satrawaha P. Study on toxicity of Thai medicinal plants. Bull Dept Med Sci. 1971;12(2/4):36–65. | ||
Pongsakorn S, Koysooko R, Pinthong T, Morasakul B. Pharmacokinetic studies of compound D isolated from Plai (Zingiber cassumunar Roxb.) in rat. Vajira Med J. 1988;32:101. | ||
Sivakumar M, Tachibana K, Pandit AB, et al. Transdermal drug delivery using ultrasound-theory, understanding and critical analysis. Cell Mol Biol (Noisy-le-grand). 2006;51:OL767–OL784. | ||
Ita K. Recent progress in transdermal sonophoresis. Pharm Dev Technol. 2015;25:1–9. | ||
Ruckmani K, Sankar V, Sivakumar M. Tissue distribution, pharmacokinetics and stability studies of zidovudine delivered by niosomes and proniosomes. J Biomed Nanotechnol. 2010;6(1):43–51. | ||
Thitilertdecha N, Teerawutgulraq A, Kilburn JD, Rakariyatham N. Identification of major phenolic compounds from Nephelium lappaceum L. and their antioxidant activities. Molecules. 2010;15(3):1453–1465. | ||
Baillie AJ, Florence AT, Hume LR, Muirhead GT, Rogerson A. The preparation and properties of niosomes – non-ionic surfactant vesicles. J Pharm Pharmacol. 1985;37(12):863–868. | ||
Shapira L, Soskolne WA, Houri Y, Barak V, Halabi A, Stabholz A. Protection against endotoxic shock and lipopolysaccharide-induced local inflammatory by tetracycline: correlation with inhibition of cytokine secretion. Infect Immun. 1996;64(3):825–828. | ||
de Brito Vieira WH, Aguiar KA, da Silva KM, Canela PM, da Silva FS, Abreu BJ. Overview of ultrasound usage trends in orthopedic and sports physiotherapy. Crit Ultrasound J. 2012;4(1):11. | ||
Belabbes R, Dib ME, Djabou N, et al. Chemical variability, antioxidant and antifungal activities of essential oils and hydrosol extract of Calendula arvensis L. from western Algeria. Chem Biodivers. Epub 2017 Jan 21. | ||
Pongprayoon U, Tuchinda P, Claeson P, Sematong T, Reutrakul V, Soontornsaratune P. Topical antiinflammatory activity of the major lipophilic constituents of the rhizome of Zingiber cassumunar Roxb. Part II: Hexane extractives. Phytomedicine. 1997;3(4):323–326. | ||
Rajera R, Nagpal K, Singh SK, Mishra DN. Niosomes: a controlled and novel drug delivery system. Biol Pharm Bull. 2011;34(7):945–953. | ||
Fang JY, Hong CT, Chiu WT, Wang YY. Effect of liposomes and niosomes on skin permeation of enoxacin. Int J Pharm. 2001;219(1–2):61–72. | ||
Ceve G, Schätzlein A, Blume G. Transdermal drug carrier; basic properties, optimization and transfer efficiency in the case of epicutaneously-applied peptide. J Control Release. 1995;36(1–2):3–16. | ||
Hofer C, Hartung R, Göbel R, Deering P, Lehmer A, Breul J. New ultradeformable carrier for potential transdermal application of interleukin-2 and interferon-α: theoretic and practical aspects. World J Surg. 2000;24(10):1187–1189. | ||
Miller DL, Smith NB, Balley MR, et al. Overview of therapeutic ultrasound applications and safety considerations. J Ultrasound Med. 2012;31(4):623–634. | ||
Dyson M, Franks C, Suckling J. Stimulation of healing of varicose ulcers by ultrasound. Ultrasonics. 1976;14(5):232–236. | ||
Pitt WG, Husseini GA, Staples BJ. Ultrasonic drug delivery – a general review. Expert Opin Drug Deliv. 2004;1(1):37–56. | ||
Park D, Park H, Seo J, Lee S. Sonophoresis in transdermal drug deliverys. Ultrasonics. 2014;54(1):56–65. | ||
Mitragotri S, Kost J. Low-frequency sonophoresis: a review. Adv Drug Deliv Rev. 2004;56(5):589–601. | ||
Dong Z, Qi X, Fidler IJ. Tyrosine phosphorylation of mitogen-activated protein kinase is necessary for activation of murine macrophages by natural and synthetic bacterial products. J Exp Med. 1993;177:1071–1077. | ||
Taylor PM, Robertson SA, Dixon MJ. Evaluation of the use of thermal thresholds to investigate NSAID analgesia in a model of inflammatory pain in cats. J Feline Med Surg. 2007;9:313–318. | ||
Huggenberger R, Detmar M. The cutaneous vascular system in chronic skin inflammation. J Investig Dermatol Symp Proc. 2011;15(1):24–32. | ||
Bushra R, Aslam N. An overview of clinical pharmacology of ibuprofen. Oman Med J. 2010;25(3):155–1661. | ||
Jeenapongsa R, Yoovathaworn K, Sriwatanakul KM, Pongprayoon U, Sriwatanakul K. Anti-inflammatory activity of (E)-1-(3,4-dimethoxyphenyl) butadiene from Zingiber cassumunar Roxb. J Ethnopharmacol. 2003;87(2–3):143–148. | ||
Han AR, Kim MS, JeongYH, Lee SK, Seo EK. Cyclooxygenase-2 inhibitory phenylbutenoids from the rhizomes of Zingiber cassumunar. Chem Pharm Bull (Tokyo). 2005;53(11):1466–1468. |
 © 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.