Back to Journals » Therapeutics and Clinical Risk Management » Volume 14
An unusual successful treatment with non-sulfonamides: primary cutaneous nocardiosis caused by Nocardia brasiliensis
Authors Chen N, Qin Q, Sun KD, Luo D, Cheng QH
Received 25 March 2018
Accepted for publication 21 June 2018
Published 10 September 2018 Volume 2018:14 Pages 1661—1664
DOI https://doi.org/10.2147/TCRM.S169239
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Deyun Wang
Nian Chen, Qin Qin, Ke-Dai Sun, Dan Luo, Qiong-Hui Cheng
Department of Dermatology, Daping Hospital, Army Medical University, Chongqing 400042, China
Introduction: Primary cutaneous nocardiosis is a rare suppurative or granulomatous inflammation disease caused by Nocardia infection. Because of nonspecific clinical findings, it is always misdiagnosed as common pyogenic infection. Sulfonamides have been the standard treatment for nocardiosis, but the outcome is always poor due to the high rates of misdiagnosis and refractoriness of the disease.
Cases presentations: The presented cases are patients of acute suppurative cutaneous infection without involvement of other organs. We report four cases, of which two cases were localized cutaneous nocardiosis and the other two were lymphocutaneous type of nocardiosis. All cases were diagnosed with pus culture which turned out to be Nocardia brasiliensis. We report an improvement of primary cutaneous nocardiosis symptoms in the four patients treated with non-sulfonamides. All patients were cured and recovered without recurrence during follow-up.
Conclusion: Our results suggest that non-sulfonamides are effective treatment for the patients with primary cutaneous nocardiosis who are resistant or intolerant to sulfonamides.
Keywords: primary cutaneous nocardiosis, treatment, non-sulfonamides
Introduction
Nocardia is a ubiquitous saprophyte that belongs to the class of actinomycetes. It is Gram positive and weakly acid-fast with a filamentous appearance. Despite its low pathogenicity, nocardiosis can cause significantly high morbidity and mortality rates in immunocompromised patients. It is usually transmitted among humans through inhalation or direct skin inoculation. The lung is the most commonly involved site, followed by the brain, the skin and the subcutaneous tissue. Because of its non-specific manifestations and the difficulties in identification through culture, nocardiosis is hard to diagnose. Recently, the increasing number of reported cases with nocardiosis due to the enlarging population of immunocompromised patients and improved diagnostic methods has made Nocardia the second most common opportunistic pathogen.1 Nocardiosis is bringing up a great challenge to clinicians for its increasing prevalence in immunocompromised patients. Although sulfonamides are used as the first-line therapy for nocardiosis, drug resistance has been reported in some studies.2 Besides, some patients are intolerant to the adverse reactions of sulfonamides such as allergy. Therefore, alternative treatment modalities for nocardiosis are indeed in need. Here we report four cases with primary cutaneous nocardiosis who were successfully treated with non-sulfonamides in the past two years. The key details of each case included in this article are summarized in Table 1.
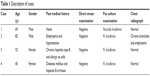  | Table 1 Description of cases |
Written informed consent was obtained from all patients and their next of kin for publication of these case details and any accompanying images. The study was approved by the Ethics Committee of Daping Hospital of Army Medical University.
Case descriptions
Case 1
A 69-year-old male was admitted into our department with a complaint of a painful swelling erythematous lesion on his right face. Five days before hospitalization, he noticed a pimple-like lesion with erythematous swelling on his right face, which quickly ulcerated. There was a 5 × 5 cm2 brightly erythematous, edematous plaque, with a 2 × 3 cm2 central erosion and satellite pustules on the right face. Ipsilateral cervical lymphadenopathies with tender were also palpable. Intravenous piperacillin tazobactam 2.5 g twice daily and skin lesion debridement were given. The lesion gradually diminished in size, permitting discharge on the 14th hospital day, followed by continued oral amoxicillin-clavulanate 0.457 g thrice daily for another 8 days after discharge. The 22-day treatment resulted in closure of the ulcer, leaving pigmentation. A follow-up that lasted 12 months revealed no evidence of recurrence.
Case 2
An 82-year-old male was hospitalized for painful erythematous lesions on his right hand and foot. Four days before hospitalization, a pustule was found on his right foot. An erythematous lesion quickly developed around the pustule and then ruptured. One day before hospitalization, another pustule developed on the root of his right thumb. There was a tender erythematous lesion with a 0.6 × 0.8 cm2 central ulcer on the dorsal aspect of the right foot and a pustule on the root of the right thumb (Figure 1A). Intravenous moxifloxacin 0.4 g once daily was given from the first hospital day with appropriate skin lesion debridement. The lesions on the hand and foot healed, permitting discharge on the 14th hospital day. No recurrence was observed during a follow-up of 12 months (Figure 1B).
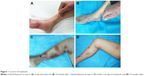  | Figure 1 Course of treatment. |
Case 3
A 52-year-old female was referred to our department for multiple painful erythematous swellings on her right lower limb with intermittent fever and chills for 20 days. She initially found a pimple-like lesion on the dorsal aspect of her right foot after contacting muddy water. Despite treatment with intravenous cephalosporins, the lesion quickly grew in size and subsequently got complicated with painful lymphangitis spreading along the medial side of her right lower leg with multiple erythematous nodules. There was a large cord-like erythematous swelling, with central necrosis and fluctuation on the dorsal aspect of her right foot and the medial side of her lower leg. Multiple erythematous nodules were linearly distributed up on to the right thigh. Intravenous moxifloxacin 0.4 g once a day was begun on the first day of hospitalization when the abscesses were incised and drained. Significant improvement was observed and pus culture turned negative. However, incisions were still discharging and new lesions developed until the 11th day (Figure 1C). The therapy was replaced with intravenous imipenem 500 mg thrice daily and amikacin 400 mg twice daily on the 11th day. Discharge from the incisions stopped and the wounds subsequently closed on the 30th day. After discharge, oral cefixime 0.1 g twice daily was given for two months and minocycline for another month to prevent relapse. The treatment lasted 4 months and a follow-up at 12 months was uneventful (Figure 1D).
Case 4
A 45-year-old female was admitted to our department with multiple painful erythematous lesions on her left hand and forearm. Her hand was slashed in the field 12 days before admission. Two days later, a pimple with an edematous plaque at the same region developed. Despite treatment with intravenous tinidazole and local incision of the abscess, the lesion became more extensive and continued to spread along the left forearm. There was an erythematous lesion with 1 cm skin incision and painful, red, hot, tender nodules linearly distributed on her left hand and forearm. Therapy was begun with intravenous moxifloxacin and incisional drainage of the abscess. When pus culture suggested nocardiosis on the sixth hospital day, trimethoprim and sulfamethoxazole were added but later replaced with amikacin on the ninth day because of drug eruption. The edematous plaques and pain significantly regressed with no new lesions occurring. On the 15th day, moxifloxacin was replaced with imipenem for its limited time of use. The therapy continued until the 30th day on which the patient was discharged when the wounds healed and erythematous lesions disappeared. Treatment with oral cefixime 0.1 g twice daily was continued for 1 month after discharge. The duration of therapy was 2 months. No relapse was noted after 12 months.
Discussion
It was reported that cutaneous nocardiosis accounted for about 7.8%–10% of nocardiosis cases, of which the primary type is relatively rare with no exact prevalence.3,4 But it may have been underestimated because it is always misdiagnosed and treated as cellulitis or other similar diseases when culture is not available or confirmative. Unlike other forms of nocardiosis, primary cutaneous disease usually develops in immunocompetent patients. But patients with impaired cell mediated immunity and geriatric patients with underlying diseases can also develop as an opportunistic infection.3 The fact that three patients in our report had predisposing conditions of pulmonary diseases, diabetes mellitus and liver cirrhosis, respectively, may indicate a susceptibility of the immunocompromised population to primary cutaneous nocardiosis. Although trauma inoculation is reported to be the main way of infection, only one of four cases had suspicious trauma history, and none have evidence of systemic infection, so the infection may have developed through inoculation of pathogens by skin contact as was considered the case in patients with local immunosuppression.2 According the its clinical manifestations, primary cutaneous nocardiosis can be classified into the acute type and the chronic type. The acute type manifests superficial skin and soft tissue infection or lymphocutaneous infection. The chronic type manifests mycetoma. Pustular lesions may be the initial manifestation of skin infection, as in our four cases, which can then progress into cellulitis, abscesses, ulcerative lesions, sporotrichoid lesions, linear/keloid-like lesions or granulomas.5 Acute infections are always difficult to differentiate clinically from cutaneous infections caused by other pyogenic bacteria. The first two cases in our report presented superficial suppurative infection, which were regarded and treated as cellulitis before bacteriodiagnosis. The other two cases were initially diagnosed and treated as cellulitis before hospitalization, but the outcome was so poor that infections quickly progressed and involved their lymphatic systems. When admitted to our department, their clinical characteristics resembled sporotrichosis, but were more acute and inflammatory. Realizing that their infections might have been caused by special pathogens, we performed cultures for the patients which at last confirmed the infection with Nocardia brasiliensis. To learn exactly the species identification of Nocardia before treatment, a combination of real-time qPCR and other techniques like 16S rRNA pyrosequencing is needed in future studies.
All Nocardial infection should be treated. A combination of antibiotics and appropriate surgical drainage is the preferred treatment.6 Sulfonamides have been the first-line drug therapy for nocardiosis. However, the resistance of the bacteria and the patients’ intolerance to the drugs necessitate the use of alternative antibiotics. Drug sensitivity test can help us selecting proper antibiotics, but it may bring challenges when they are unavailable. Minocycline, amikacin, imipenem and amoxicillin-clavulanic acid have been proved to be effective on primary cutaneous nocardiosis.7 But the relevant literature and experience are still limited. It is reported that penicillins are ineffective against N. brasiliensis infection, for that the pathogen produces β-lactamases.3 But piperacillin tazobactam showed good effect on the treatment of superficial cutaneous infection in our first patient. Moxifloxacin was reported to have cured brain abscesses caused by Nocardia farcinica.8 In our practice, moxifloxacin cured one superficial cutaneous nocardiosis and improved another two. Therefore, it is indicated that moxifloxacin can be an alternative drug for N. brasiliensis. In patients with a lymphocutaneous infection or other severe clinical symptoms, combined drug therapy is recommended. Although the third patient was significantly improved by moxifloxacin and her second pus culture turned negative, new lesions still developed. When moxifloxacin was used in combination with amikacin in case 4, the effect was excellent. Combination therapy can be continued until the patient is clinically improved and then be followed by single-drug therapy. Early intravenous administration may better control the disease and shorten the duration of disease. Our first two patients were cured with a shorter course than the previously reported duration of 2–4 months.5 Parenteral therapy can be changed to an oral regimen when clinical improvement is achieved. Third-generation cephalosporins are active against Nocardia spp.6 In our latter two cases, oral cefixime is also proved to be effective to N. brasiliensis. Because the disease has a tendency to recur, all the patients need to be closely followed up.
Conclusion
This is the first case series to evaluate the efficacy of non-sulfonamides on primary cutaneous nocardiosis patients. Although it is rarely seen, cutaneous nocardiosis should be considered in patients with an infection that rapidly progresses from a pimple to an abscess or ulcerated lesion, involve lymphatic system or poorly responds to treatment for common bacterial infections. Specific investigations such as pus culture or biochemical tests are key to diagnosis. In patients who are resistant or intolerant to sulfonamides, combined or single use of alternative antibiotics such as moxifloxacin and third-generation cephalosporins can achieve good outcomes.
Disclosure
The authors report no conflicts of interest in this work.
References
Salinas-Carmona MC, Rocha-Pizaña MR. Construction of a Nocardia brasiliensis fluorescent plasmid to study Actinomycetoma pathogenicity. Plasmid. 2011;65(1):25–31. | ||
Chen KW, Lu CW, Huang TC, et al. Cutaneous manifestations of Nocardia brasiliensis infection in Taiwan during 2002–2012-clinical studies and molecular typing of pathogen by gyrB and 16S gene sequencing. Diagn Microbiol Infect Dis. 2013;77(1):74–78. | ||
Cy S, Lin CS, Yan SH, Wang CK. Cutaneous Type of Nocardiosis Caused by Nocardia brasiliensis in an Elderly Patient. Int J Gerontol. 2012;6:150–153. | ||
Soma S, Saha P, Sengupta M. Cutaneous Nocardia brasiliensis infection in an immunocompetent host after ovarian cystectomy: A case study. Australas Med J. 2011;4(11):603–605. | ||
Dodiuk-Gad R, Cohen E, Ziv M, et al. Cutaneous nocardiosis: report of two cases and review of the literature. Int J Dermatol. 2010;49(12):1380–1385. | ||
Palmieri JR, Santo A, Johnson SE. Soil-acquired cutaneous nocardiosis on the forearm of a healthy male contracted in a swamp in rural eastern Virginia. Int Med Case Rep J. 2014;7:41–47. | ||
Maraki S, Scoulica E, Alpantaki K, Dialynas M, Tselentis Y. Lymphocutaneous nocardiosis due to Nocardia brasiliensis. Diagn Microbiol Infect Dis. 2003;47(1):341–344. | ||
Fihman V, Berçot B, Mateo J, et al. First successful treatment of Nocardia farcinica brain abscess with moxifloxacin. J Infect. 2006;52(4):e99–e102. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
