Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 14
An Oral Whole-Cell Killed Nontypeable Haemophilus influenzae Immunotherapeutic For The Prevention Of Acute Exacerbations Of Chronic Airway Disease
Received 27 May 2019
Accepted for publication 16 September 2019
Published 25 October 2019 Volume 2019:14 Pages 2423—2431
DOI https://doi.org/10.2147/COPD.S217317
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Richard Russell
Robert L Clancy,1 Allan W Cripps2
1School of Medicine and Public Health, University of Newcastle, Newcastle, NSW, Australia; 2School of Medicine, Menzies Health Institute Queensland, Griffith University, Gold Coast Campus, Gold Coast, QLD, Australia
Correspondence: Allan W Cripps
School of Medicine, Menzies Health Institute Queensland, Griffith University, Gold Coast Campus, Gold Coast, QLD 4222, Australia
Tel +61 7 56780321
Email [email protected]
Abstract: In subjects with chronic bronchitis, protection against acute bronchitis following oral administration of a whole-cell killed nontypeable Haemophilus influenzae (NTHi) preparation was demonstrated in the mid-1980s. Subsequently, studies aiming to validate clinical efficacy of this oral treatment were complicated by a number of factors, including the modification of clinical definitions, the implications of which were not recognized at that time. The objective of this review is to integrate our pre-clinical and clinical research in this field conducted over the past 30 years to demonstrate the evolution of the idea of communication between mucosal surfaces through the common mucosal immune system and the development of an effective oral NTHi immunotherapy. Our earliest studies recruited subjects with chronic sputum production and high levels of culture-positive sputum for Gram-negative bacteria but by 2000, the clinical diagnostic focus had switched from “chronic bronchitis” to “chronic obstructive pulmonary disease” (COPD), which was functionally defined using spirometry. This change led to variable clinical trial results, confirming the importance of chronic sputum production and culture-positive sputum. Additional conditioning factors such as patient age and gender were influential in study populations with low culture-positive sputum production. Through this period, studies in human and in rodent models provided new insights into airway protection mechanisms and the pathogenesis of airway inflammation. Key findings were the importance of a dysbiosis within the airway microbiome, and the critical role of an interdependence between the bronchus and the gut, with a Peyer’s patch-dependent extra-bronchus “loop” controlling the composition of the bronchus microbiome. Within this context, intercurrent virus infections initiate a microbiome-dependant hypersensitivity reaction involving Peyer’s patch-derived Th17 cells. We conclude that whole-cell killed NTHi immunotherapy has consistent and significant benefits when examined in the context of changing clinical disease definitions, age and gender, and has the potential to change the natural history of chronic airway disease.
Keywords: lung infection, airway inflammation, oral immunotherapy, lung immunity, nontypeable Haemophilus influenzae, COPD, chronic obstructive pulmonary disease
Introduction
In 1985, we reported that an orally administered whole-cell killed nontypable Haemophilus influenzae (NTHi) preparation protected against acute exacerbations in subjects with chronic bronchitis.1 This study was not primarily designed to test the oral treatment, but rather was part of a program investigating whether the Common Mucosal Immune System (CMIS) found in animal models was clinically relevant in humans.
During the following 30 years, a combination of studies in chronic bronchitis and in rodent models focussed on NTHi as a probe to better understand the physiology of airway protection and the pathophysiology of endobronchitis. This review aims to integrate this research to document the evolution of the concept of communication between mucosal surfaces and the development of an effective oral immunotherapy that has the potential to change the natural history of chronic airway disease. These studies support the importance of a dysbiosis within damaged airways as a critical contributor to progression of airway damage in COPD.
Research that our group has undertaken dominates the field with respect to the development of an oral whole-cell killed NTHi immunotherapeutic for the prevention of acute exacerbations of chronic airway disease. This review utilises a narrative approach, scaffolded on our previously published research, supplemented with unpublished data from the same studies to provide a more comprehensive data set and to further illustrate the clinical outcomes of this intervention. Human research ethics approval for the collection of the previously unpublished data was granted in accordance with the original published studies.
The CMIS And The Lung
A paradigm shift in the field of mucosal immunology occurred in 1971 when Craig and Cebra showed, by allogenic cell transfer into irradiated rabbits, that the Peyer’s patches were an enriched source of precursors for IgA-producing immunocytes within the rabbit gut mucosa.2 Bienenstock described aggregated lymphoid collections in the rabbit bronchus (bronchus-associated lymphoid tissue or BALT) with characteristics similar to those of the Peyer’s patch. In 1975, when BALT lymphocytes were shown to re-populate bronchial and intestinal mucosa with IgA-containing cells,3 Bienenstock proposed the concept of a CMIS based on selective cell traffic between mucosal sites.
The concept of a CMIS underpinning the distribution of mucosal protection, including the lung, has gained widespread acceptance over the past 40 years, although many additional discoveries including regional and environmental influences4 have modified and expanded the concept. Historically, the 1970s was an early period in the field of mucosal immunology with many unanswered questions. For example, mucosal T cells were thought to be down regulated with only a transient response to an immunizing antigen; that is, they were “non-responsive”.5 Interestingly, it was not known then whether T cells as well as B cells participated in the CMIS, or what directional flow of inter-mucosal cell traffic was dominant or how effector mechanisms at distant mucosa connected with trafficking lymphocytes. Over the next 20 years, studies in animal models provided greater clarity on these questions regarding CMIS connectivity and function.6 Proof that T cells could participate in inter-mucosal cell traffic was confirmed by mucosa-seeking T cells being detected following feeding of allogenic lymphocytes using the mixed lymphocyte reaction as readout for antigen-specific T cells.7 The importance of the dominant directional flow of B-lymphocytes from gut-associated lymphocyte tissue to the bronchus mucosa was confirmed using sheep models.8 The phenomenon of “non-responsive” mucosal T cells, described above and in various formats and at a number of different mucosal sites, was resolved when mucosal T cells prepared from resected human bronchus downregulated autologous blood T cell responses to “memory” antigens in co-cultures.9 At that time, this control mechanism was considered to be mediated by “suppressor T cells” which are now specifically recognised as “CD4+ T reg” cells. Importantly, this finding was the first observation of “down regulating” T cells outside of the circulating blood compartment.
Chronic Bronchitis – Changing Clinical Definition And Greater Understanding Of Mucosal Immune Mechanisms In The Lung
Planning of our first oral NTHi immunotherapy study in chronic bronchitis was occurring when the current state of knowledge of the CMIS with respect to the lung was as summarized above.1 Selection of chronic bronchitis with frequent acute exacerbations as a study model to determine the relevance of the CMIS to humans was influenced by the British experience with the “Great Smog” in 1952, the CIBA Guest Symposium in 1959 and the Medical Research Council classifications in 1965.10 The failure of Fletcher’s group at the Postgraduate Medical School in London to recognize the impact of acute exacerbations on the natural history of chronic bronchitis shifted diagnosis towards support for the functional diagnosis of COPD. These influential early ideas and their re-assessment due to numerous observations were revisited by Nicholas Anthonisen in 2005.10 By 2000, the selection of subjects for clinical trials was based on spirometry measurements, which particularly suited large studies assessing the value of inhaled steroid/beta-agonist combinations.11 This shifting criterion for trial entry is important in interpreting trial outcomes with oral NTHi immunotherapy. The three published studies we conducted before 2000 included subjects with chronic bronchitis (identified using the Medical Research Council criteria at that time), whereas in studies published later, the primary inclusion criterion was COPD based on limited airflow as determined by spirometry.
At the time of the initial oral immunotherapy study in 1985, it was postulated that in chronic bronchitis, inhaled toxins damaged airways. Following an intercurrent virus infection, bacteria resident in the upper airways, specifically NTHi, would descend into the normally sterile bronchi to elicit an acute inflammatory response, detected clinically as an increase in the volume and purulence of sputum. The hypothesis tested was that oral administration of whole-cell killed NTHi to subjects with chronic bronchitis would, via the CMIS, stimulate IgA antibody secretion in the upper airways to prevent descent of bacteria into the bronchial system. Between 1985 and 1990, three studies at different medical sites in subjects with chronic bronchitis were published (Table 1).
 |
Table 1 Summary Of Published Data Of Oral Whole-Cell Killed NTHi Immunotherapy Studies In Chronic Bronchitis Conducted Before 1990 |
These three studies had similar design characteristics. Each showed a significant reduction of acute episodes of bronchitis defined as “an increase in volume and purulence of sputum” without qualification as to change in therapy. Additional clinical observations included a reduction in antibiotic use,1,12 reduced admission to hospital1 and an increase in quality of life.12 In all three studies, the effect on bacterial colonization was examined, but different microbiological reporting methods were used. Two studies showed a significant reduction in bacterial colonization using sputum culture12,13 with both studies reporting reduction in NTHi and Streptococcus pneumonia, reflecting non-specific clearance from the airways by phagocytosis. Of specific interest was a 3-log reduction (P<0.05) in NTHi count following oral therapy with NTHi, with a treatment effect lasting about 10 months.13 The third study reported a reduction in pharyngeal colonization with NTHi.1
We completed two further clinical studies before 1990. The first compared the effects of a single species killed-NTHi oral preparation and a polybacterial immune stimulant that contained twenty-fold less NTHi. This and other whole-cell and lysate polybacterial products had been on the European market (as “immune stimulants”) since the 1950s. The claims of clinical benefit were short term but unclear.14 We showed that in subjects with low secretory IgA antibody that a whole-cell polybacterial product (Buccaline BernaTM) was an adjuvanted vaccine, with an increase in specific H. influenzae saliva antibody. However, in subjects with a high baseline level of anti-NTHi antibody, there was no increase in antibody levels above the baseline pre-study level due to the presence of regulatory cells as discussed above.15 In the same study, the high dose of the single bacterial NTHi preparation did not stimulate an antibody response, a finding that had also been noted in the clinical studies in chronic bronchitis.1 The comparative clinical trial between Buccaline BernaTM and single species NTHi preparation showed only the latter induced protection, as judged by a reduction in acute episodes of bronchitis.14 The concept that the primary mechanism of immunological protection was not antibody-mediated but instead mediated through Th17 T cells was subsequently proven in both human and animal studies, described below. The second study selected subjects with two or more acute episodes of bronchitis each year for at least two consecutive years.12 Most had unrecognized chronic bronchitis and COPD. There was a significant reduction in incidence of acute wheezy bronchitis in the active group, associated with a selective inhibition of “winter associated” colonization of the upper airways with NTHi. Reduction of upper airway NTHi colonization following oral immunotherapy not only confirmed the gut-lung link through a CMIS but suggested that this response is specific in the upper airways, in contrast with the non-specific reduction of bacteria in sputum as previously discussed.12,13 These observations also suggest that oral immunotherapy may stimulate different immune mechanisms in the upper and lower airways. The link with wheeze and possible reversible airway obstruction may relate to detection of IgE anti-NTHi antibody, and the concept that an allergic reaction to colonizing bacterial proteins may play a role in late onset airway disease.16 Importantly, these studies raised further questions since there was no detectable increase in IgA antibody, as postulated in the hypothesis being tested. To resolve this, studies to determine mechanisms of airway protection and disease pathogenesis were conducted in animal models and human clinical investigations. In summary, studies in the rat model of lung acute infection showed that T cells obtained from the thoracic duct, but not B cells, transferred immunity following oral administration with whole-cell killed NTHi.17 These cells were later identified as CD4+Th17 T cells18 that recruited and activated neutrophils into the bronchus,19 where they created a phenotypically unique population (in humans) of long-lived, metabolically active phagocytes.19 Indeed, antigen reactive T cells were detected in transit in blood following oral administration of whole-cell killed NTHi.20 To complete a physiological loop, by confirming an afferent limb of this “extra bronchial on demand” protection mechanism, it was shown that antigen was aspirated into the gut for delivery to Peyer’s patches.20 The role of virus, in initiating exacerbations, was studied in the mouse with a co-infection model involving NTHi and influenza virus.21 There was a mutual stimulation of microbes, both of which were reduced by prior oral treatment with whole-cell killed NTHi.21
Taken together, studies in animal models and humans (many using orally administered whole-cell killed NTHi as a probe) support a construct that oral NTHi immunotherapy acts by reinforcing a physiological pathway. In summary, the bronchus microbiome is controlled by delivering aspirated antigen to Peyer’s patches, which in turn stimulate Th17 T cells that home to the bronchus. Within the bronchus Th17 T cells recruit and activate phagocytic cells that reduce the load of colonizing bacteria.18 This mucosal immune protection within the airways would appear to involve two main pathways:
- The first pathway, for particulate antigens (such as bacteria) that involves Peyer’s patches as described above.
- The second pathway, for soluble antigens, involves an extensive network of high turnover dendritic cells (DCs). These DCs interact with T cells in regional nodes, activating regulatory T cells resulting in down regulation as the predominant response.
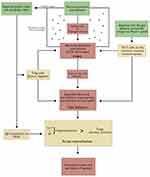
Importantly, for the first pathway, particulate antigen has a threshold, below which is protected from interaction with the mucosal epithelial DC network.22 How then does an acute exacerbation come about? It can be best understood by considering the dynamic between the abnormal biota within the damaged airways and the restraining “bronchus → gut → bronchus” immune response that follows infection with an intercurrent virus. An increased delivery of Th17 cell into the bronchus lumen causes an excessive and inappropriate flux of neutrophils, detected clinically as an increase in volume and purulence of sputum. The DC “carpet” is, in this circumstance, exposed to multiple stimuli with a consequent reduction in inhibition and even an additional pro-inflammatory effect contributing to sputum purulence.18,23 Our current understanding the immunological responses in the airways of subjects with COPD and how these responses are modulated is shown in Figure 1.
After the year 2000, two multi-centre, Phase II, whole-cell killed NTHi oral immunotherapy trials were completed. It is important to recognize that the structure of clinical trials in COPD had undergone major changes since the earlier studies in chronic bronchitis. These changes largely reflected the development of inhalation therapy around the turn of the century, based on objective measurements of airways obstruction. That is, the recruitment criteria for these clinical studies had shifted from “chronic cough and sputum” to a spirometric definition of irreversible airways obstruction.
The importance of acute exacerbations to both the immediate and long-term patient outcomes was also better recognized and reduction in acute exacerbations became a primary objective for these studies. The classification of acute exacerbations now included a management component: specifically the use of oral/parenteral corticosteroids (moderate exacerbations) or admission to hospital (severe exacerbations). The effect of this change can be seen in the 2006 study24 where protection against untreated exacerbations (17%) was not significant but became significant when qualified as requiring hospitalization or corticosteroid treatment.
2006 Multisite Studies In COPD
Two studies with identical design, conduct and statistical analysis were performed in subjects with COPD. Subjects for both studies were recruited at the same time and then assigned to either study according to their forced expiratory volume (FEV1) score; Study 1: Mild COPD (FEV1, <80% but >50% predicted normal); Study 2: Moderate–Severe COPD (FEV1, <50% predicted normal). Study 2 has been published.24 A combined analysis of the two studies was planned and agreed prior to the onset of the trials. The results are combined below in Table 2. The details of design, ethics approval, study sites and management, methodology, statistical analysis and clinical definitions were as described in the publication of Study 2.24
 |
Table 2 Protection Against Acute Exacerbations Of COPD By An Oral Whole-Killed Cell NTHI Immunotherapy. This Study Was Conducted In 2 Parts With Only Data From 38 Subjects With Moderate/severe Exacerbations (FEV1 ≤50% Of Normal) Published Previously.24 This Table Includes Previously Unpublished Data From All Subjects, Including Those With FEV1 ≤70% Are Included. The Demographic Data For Both Studies Has Also Been Merged. The Methodology For All Subjects Studied Was Identical And Is Described In Reference.24 |
Published in 2006, Study 2 included 38 subjects with moderate–severe COPD. In the active treatment group, there was a significant reduction in both moderate and severe exacerbations. In addition, the duration of moderate exacerbations was reduced (P=0.01) and antibiotic usage reduced by 72% fewer days (P=0.01).24 The combined data for both studies are presented in Table 2 and the following observations were made:
- Colonization with NTHi was less in this COPD group than had been reported in the earlier pre-1990 studies of subjects with chronic bronchitis (see Table 1). Only 30% of the COPD group had a positive sputum sample and only 60% could give a sputum sample at any visit during the study. Thus, culture-positive data for NTHi was in any specimen collected over the study period. This result contrasted with the earlier chronic bronchitis studies where sputum could be obtained on most visits from all subjects, and multiple samples were culture positive.
- Treatment-related protection was limited to those exacerbations classed as moderate–severe.24
- Analysis of subsets focused on age and gender demonstrated that both of these characteristics affected response to the oral treatment (Table 3) with women responding better to the oral treatment than did men, a difference that was particularly notable in men >65 years.
 |
Table 3 Effect Of Age And Gender On Protection Induced By Whole-Cell Killed NTHi Oral Immunotherapy Against Moderate–Severe Acute Exacerbations Of COPD |
2011 Multisite Study In COPD25
The second multi-site whole-cell killed NTHi oral immunotherapy study was performed at 21 sites across Australia in 2011. A total of 320 subjects were recruited to the study based on a power analysis from the results of the 2006 study in moderate–severe COPD.3 For this study, the switch to a diagnosis of the COPD was more significant with respect to a reduction in numbers of subjects with chronic bronchitis, with only 28% of subjects capable of giving a sputum sample at any visit and the number of subjects in the active group culture positive for NTHi at the beginning of the trial being only 2.5%. Analysis at the end of the 9-month study showed no significant benefit in the active treatment group compared with the placebo group. However, those subjects less than 65 years old had a 54% reduction in moderate-severe exacerbations (P=0.01) with significantly less use of corticosteroids and antibiotics and a 60% reduction in duration of episodes. These findings are consistent with “all age” results from earlier studies where cohort recruitment utilised the “chronic bronchitis” criteria. In this younger group, an impact on daily living using the validated St George Questionnaire showed significant and meaningful improvement of symptoms of cough, sputum, wheeze and breathlessness at 12 (P=0.02) and 24 (P=0.01) weeks post-treatment in the active group. This latter observation supports the concept that there are mechanisms in common that underpin acute exacerbations and background symptoms.
Conclusions
This review of clinical studies on respiratory tract immunotherapy using oral whole-cell killed NTHi demonstrates that:
- Where NTHi is the predominant organism, subjects with chronic bronchitis and regular sputum production are those most likely to benefit from oral immunotherapy with killed NTHi.
- Subjects with COPD and no symptoms of chronic cough and sputum may benefit from this immunotherapeutic treatment, but there are conditioning factors – specifically, age, gender and results of sputum culture – that impact on effectiveness. Traditional bacteriological assessment, using culture methods, is an insensitive marker of the presence of NTHi within the bronchus26 of an individual patient. However, at a cohort level, clinical sputum culture has value, perhaps by detecting bacteria above a threshold required for re-stimulation of Peyer’s patch-derived T cells, as is needed and observed in the animal model studies.17
- Immuno-senescence operates within the mucosal immune system, as has been reported with parenteral vaccines.27 The impact of patient age is important when the cohort colonization with NTHi is low. Furthermore, as demonstrated for systemic vaccines, use of different dosing requirements may improve outcome, although simply adding adjuvants is unlikely to be of benefit given the probability that regulatory T cells would be disproportionately expanded.22
- Clinical benefits from oral administration of a whole-cell killed NTHi immunotherapy using a single pre-winter course include a significant reduction in the number and duration of exacerbations (there is a shift from more severe to less severe episodes) and in the use of antibiotics and corticosteroids and, admission into hospital.1,12,13,24,25
- Oral immunotherapy with bacterial preparations may have a role in the management of other chronic airway disease where chronic inflammation is likely driven by an abnormal microbiome. For example, in subjects with bronchiectasis, we have demonstrated oral immunotherapy using a killed whole cell Pseudomonas aeruginosa preparation significantly reduced the load of bacteria in sputum.28
- Oral bacterial products have proven safe and inexpensive immunotherapy.
Concluding Remarks
With small patient cohorts, clear, consistent and significant benefits have been demonstrated by whole-cell killed NTHi oral immunotherapy studies, and far exceed the marginal benefits from current inhaled therapies. Comparatively, these have required substantially greater cohort sizes to demonstrate any significant effect. This focuses attention on the important contribution to disease by the dysbiota associated with damaged airways. Furthermore, it is noted that the treatment benefits described in these clinical studies are additional to any benefit resulting from inhalation therapy. Given COPD is a major reason for antibiotic use (and given that inhaled steroids increase the amount of antibiotics taken), a significant advantage of oral NTHi immunotherapy is halving the use of antibiotics, in subjects with chronic sputum production.
The purpose of this review has been to review and present all the studies that administered an oral, inactivated, bacterial immunotherapeutic, in a context of studies on the mechanisms of airways protection and pathogenesis of acute exacerbations.
During a period of important changes in key patient inclusion criteria (eg, functional diagnosis rather than clinical, gender and age impacting on immune senescence and responsiveness), understanding variable results in the different studies provides a basis for future studies measuring changes in the airway microbiome using quantitative molecular methodology which is now available.
The laboratory and clinical studies described highlight, for the first time, the importance of airway dysbiosis in progressing damage in COPD subjects, and identify a new and effective treatment that can change the natural history of the disease. Future studies are needed to determine whether augmenting natural protection with oral whole-cell killed NTHi immunotherapy can extend the duration and quality of patients’ life. It is anticipated that oral NTHi immunotherapy represents a relatively early prototype treatment, as more effective analogues are developed, with application in a wider range of bronchitic disorders.
Author Contributions
Both authors contributed to data analysis, drafting and revising the article, gave final approval of the version to be published and agree to be accountable for all aspects of the work.
Disclosure
The authors declare that they have no competing interests.
References
1. Clancy RL, Murree-Allen K, Cripps AW, Yeung S, Engel M. Oral immunisation with killed haemophilus influenzae for protection against acute bronchitis in chronic obstructive lung disease. Lancet. 1985;326:1395–1397. doi:10.1016/S0140-6736(85)92559-0
2. Craig SW, Cebra JJ. Peyer’s patches: an enriched source of precursors for IgA-producing immunocytes in the rabbit. J Exp Med. 1971;134:188–200. doi:10.1084/jem.134.1.188
3. Rudzik R, Clancy RL, Perey DYE, Day RP, Bienenstock J. Repopulation with IgA-containing cells of bronchial and intestinal lamina propria after transfer of homologous Peyer’s patch and bronchial lymphocytes. J Immunol. 1975;114:1599–1604.
4. Brandtzaeg P, Kiyono H, Pabst R, Russell MW. Terminology: nomenclature of mucosa-associated lymphoid tissue. Mucosal Immunol. 2008;1:31–37. doi:10.1038/mi.2007.9
5. Clancy RL, Bienenstock J. The proliferative response of bronchus-associated lymphoid tissue after local and systemic immunization. J Immunol. 1974;112:1997–2001.
6. Wallace FJ, Clancy RL, Cripps AW. An animal model demonstration of enhanced clearance of nontypable haemophilus influenzae from the respiratory tract after antigen stimulation of gut-associated lymphoid tissue. Am Rev Respir Dis. 1989;140:311–316. doi:10.1164/ajrccm/140.2.311
7. Husband AJ, Dunkley ML, Cripps AW, Clancy RL. Antigen-specific response among T lymphocytes following intestinal administration of alloantigens. Aust J Exp Biol Med Sci. 1984;62:687–699. doi:10.1038/icb.1984.65
8. Scicchitano R, Husband AJ, Clancy RL. Contribution of intraperitoneal immunization to the local immune response in the respiratory tract of sheep. Immunology. 1984;53:375–384. doi:10.1164/arrd.1978.117.3.513
9. Clancy RL, Pucci A, Jelihovsky T, Bye P. Immunologic “memory” for microbial antigens in lymphocytes obtained from human bronchial mucosa 1–3. Am Rev Respir Dis. 1978;117:513–518. doi:10.1164/arrd.1978.117.3.513
10. Anthonisen NR. The British hypothesis revisited. Eur Respir J. 2004;23:657–658. doi:10.1183/09031936.04.00013004
11. Calverley P, Pauwels R, Vestbo J, et al. Combined salmeterol and fluticasone in the treatment of chronic obstructive pulmonary disease: a randomised controlled trial. Lancet. 2003;361:449–456. doi:10.1016/S0140-6736(03)12459-2
12. Tandon MK, Gebski V. A controlled trial of a killed haemophilus influenzae vaccine for prevention of acute exacerbations of chronic bronchitis. Intern Med J. 1991;21:427–432.
13. Lehmann D, Coakley KJ, Coakley CA, et al. Reduction in the incidence of acute bronchitis by an oral haemophilus influenzae vaccine in patients with chronic bronchitis in the highlands of Papua New Guinea. Am J Respir Crit Care Med. 1991;144:324–330.
14. Clancy RL, Cripps AW, Pang G, Yeung S, Murree-Allen K. The paradox of immunization against haemophilus influenzae-related endobronchitis: protection restricted to ingestion of’non-adjuvenated’vaccine. Adv Exp Med Biol. 1986;216:1759–1764.
15. Clancy RL, Cripps AW, Husband AJ, Buckley D. Specific immune response in the respiratory tract after administration of an oral polyvalent bacterial vaccine. Infect Immun. 1983;39:491–496.
16. Otczyk DC, Clancy RL, Cripps AW. Haemophilus influenzae and smoking-related obstructive airways disease. Int J Chron Obstruct Pulmon Dis. 2011;6:345–351. doi:10.2147/COPD.S19359
17. Wallace FJ, Cripps AW, Clancy RL, Husband AJ, Witt CS. A role for intestinal T lymphocytes in bronchus mucosal immunity. Immunology. 1991;74:68–73.
18. Clancy RL, Dunkley ML. Acute exacerbations in COPD and their control with oral immunization with non-typeable haemophilus influenzae. Front Immunol. 2011;2:1–6. doi:10.3389/fimmu.2011.00007
19. Pang G, Ortega M, Zighang R, Reeves G, Clancy RL. Autocrine modulation of IL-8 production by sputum neutrophils in chronic bronchial sepsis. Am J Respir Crit Care Med. 1997;155:726–731. doi:10.1164/ajrccm.155.2.9032219
20. Clancy RL, Dunkley ML. Oral non‐typable haemophilus influenzae enhances physiological mechanism of airways protection. Clin Exp Immunol. 2010;161:127–133. doi:10.1111/j.1365-2249.2010.04142.x
21. Dunkley ML, Husband AJ, Beagley KW, Clancy RL, Collins AM, Cripps AW, Emery DL, . A rodent model of concurrent respiratory infection with influenza virus and gram-negative bacteria: synergistic infection and protection by oral immunization. In: Mucosal Solutions: Advances in Mucosal Immunology. Vol. 1. University of Sydney, Sydney Australia. 1997;261–268.
22. Kopf M, Schneider C, Nobs SP. The development and function of lung-resident macrophages and dendritic cells. Nat Immunol. 2015;16:36–44. doi:10.1038/ni.3052
23. Clancy RL. Towards a vaccine for chronic obstructive pulmonary disease. Intern Med J. 2012;42:607–613. doi:10.1111/j.1445-5994.2012.02752.x
24. Tandon MK, Phillips M, Waterer G, Dunkley ML, Comans P, Clancy RL. Oral immunotherapy with inactivated nontypeable haemophilus influenzae reduces severity of acute exacerbations in severe COPD. Chest. 2010;137:805–811. doi:10.1378/chest.09-1382
25. Clancy RL, Dunkley ML, Sockler J, McDonald CF. Multi‐site placebo‐controlled randomised clinical trial to assess protection following oral immunisation with inactivated non‐typeable haemophilus influenzae in chronic obstructive pulmonary disease. Internal Medicine Journal. 2016;46:684–693. doi:10.1111/imj.13072
26. Dickson RP, Erb-Downward JR, Martinez FJ, Huffnagle GB. The microbiome and the respiratory tract. Annu Rev Physiol. 2016;78:481–504. doi:10.1146/annurev-physiol-021115-105238
27. DiazGranados CA, Dunning AJ, Kimmel M, et al. Efficacy of high-dose versus standard-dose influenza vaccine in older adults. N Engl J Med. 2014;371:635–645. doi:10.1056/NEJMoa1315727
28. Grimwood K, Kyd JM, Owen SJ, Massa HM, Cripps AW. Vaccination against respiratory pseudomonas aeruginosa infection. Hum Vaccin Immunother. 2015;11:14–20. doi:10.4161/hv.34296
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.