Back to Journals » Clinical, Cosmetic and Investigational Dermatology » Volume 16
Acrodermatitis Continua of Hallopeau Successfully Treated with Ustekinumab: A Case Report and Literature Review
Authors Shi L , Zeng L , Huang X, Wang B , Zhang Y, Zhang G
Received 14 January 2023
Accepted for publication 28 March 2023
Published 6 April 2023 Volume 2023:16 Pages 951—957
DOI https://doi.org/10.2147/CCID.S404860
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 5
Editor who approved publication: Dr Jeffrey Weinberg
Liping Shi,1,2 Linxi Zeng,1,2 Xin Huang,1,2 Bin Wang,1,2 Yuli Zhang,1,2 Guoqiang Zhang1,2
1Department of Dermatology, The First Hospital of Hebei Medical University, Shijiazhuang, People’s Republic of China; 2Candidate Branch of National Clinical Research Center for Skin Diseases, Shijiazhuang, People’s Republic of China
Correspondence: Guoqiang Zhang, Department of Dermatology, The First Hospital of Hebei Medical University, 89 Donggang Road, Yuhua District, Shijiazhuang, Hebei Province, People’s Republic of China, Tel +8618633888122, Email [email protected]
Abstract: Acrodermatitis continua of Hallopeau (ACH) is a rare variant of pustular psoriasis that presents as a sterile pustular eruption of the periungual and subungual regions. It affects the skin and nail bed and can lead to distal phalangeal destruction as the disease progresses. ACH is an incurable disease that requires long-term maintenance therapy to prevent any complications. Because ACH is a variant of pustular psoriasis, it is commonly treated with anti-psoriatic therapies. Unfortunately, it is resistant to many available anti-psoriatic therapies, and there are no clinical guidelines for it; the treatment is therefore highly challenging. Current treatment strategies are mostly based on a few case reports and case series. In this study, we present a case of ACH in a 24-year-old man with a long history of severe skin lesions and overt onychodystrophy, successfully treated with Ustekinumab. In this patient, we observed rapid improvement in skin lesions and symptoms. Ustekinumab can considerably improve other symptoms besides plaque psoriasis. The treatment and excellent outcomes of Ustekinumab may provide clinical benefits to more patients and serve as a reference for other dermatologists.
Keywords: pustular psoriasis, sterile pustules, anti-psoriatic therapies, biological agent
Introduction
Acrodermatitis continua of Hallopeau (ACH) is a rare and disabling inflammatory skin disease that usually involves the distal parts of the hands and feet. This disease is characterized by periungual and subungual sterile pustules, paronychia, onychodystrophy, distal phalangeal lysis, and atrophy of the skin of the fingers and toes.
ACH is a variant of pustular psoriasis, and it is commonly treated with anti-psoriatic therapies. As is well-known, psoriasis management is challenging. Traditional treatments often have a slow onset, limited clearance of lesions, or side effects. ACH is also resistant to many conventional topical and systemic anti-psoriatic therapies, such as calcineurin inhibitors, phototherapy and methotrexate.1,2 However, the introduction of biological agents has revolutionized the treatment of psoriasis, even in children and elderly patients, in whom they have demonstrated excellent efficacy and safety,3,4 thus making them viable for the therapy of ACH.
Case Presentation
A 24-year-old male patient without a personal or family history of psoriasis visited our department because of a 13-year history of recurrent redness, swelling, pustules and tenderness on the tips of several digits on both the hands. Pustules were initially found around the nails, followed by scales and crusts, leading to onychodystrophy and progressive deterioration of the nails. Four years ago, he suffered unbearable burning pain in the fingers, which seriously affected his sleep quality, and white exudate appeared when the hands dropped, accompanied by swelling. Two years ago, the right forearm was sprained, and one day later, a large edematous cutaneous lesion with erythema appeared on the dorsum of the forearm from the wrist to the elbow. The local dermatologist diagnosed it as erysipelas, and the patient was treated with intravenous flucloxacillin (500 mg, four times per day). The erythema highly subsided after 15 days; however, the pustules persisted on the forearm, and the lesions recurred thereafter.
Dermatologic examination revealed severe erythema with clear boundaries on all tips of the digits of his right hand and the thumb, ring finger, and little finger of his left hand, with several erosions and covered with a thick yellow crust. A similar skin lesion measuring 2.5×2 cm in size was seen on the right forearm. Further examination revealed that anonychia occurred on five fingers, three fingernails were dystrophic, and seven fingers could not be fully extended (Figure 1A–C). The patient reported a pain score of 8 out of 10 on the visual analog scale (VAS) pain scale and 24 points out of 30 on the Dermatology Life Quality Index (DLQI). According to the clinical manifestations, the patient was diagnosed with ACH.
During the first 9 years of the disease course, he was treated at several hospitals with multiple oral and topical medications, with little or no response. Four years ago, he was admitted to a tertiary hospital in Beijing, where topical medications and systemic traditional Chinese medicine were prescribed, but without any success. He was later treated with acitretin (20–30 mg/day for 3 years) and narrow-band ultraviolet B phototherapy (84 sessions). Although these drugs did not cause apparent side effects, the lesions remained unaffected and worsened.
Laboratory tests and imaging examinations were conducted, including blood and urine tests, testing of the liver function, kidney function, hepatitis B and hepatitis C antibodies, the interferon-gamma release assay (IGRA), chest computed tomography (CT) as well as anteroposterior and oblique position X-rays for the hands. The X-rays of both the hands showed no bony abnormalities, but the swelling of the surrounding soft tissue with uneven density and edges at the distal phalanx of all fingers of the right hand, and the thumb, ring finger, and little finger of the left hand. The IGRA test showed that the tuberculosis T cells (T-N) were at a level of 172.8 pg/mL, indicating that the T-cell test for tuberculosis infection was positive. Chest CT showed localized areas of thickening, adhesions, and calcifications in the pleura on the right side. Active tuberculosis was excluded after a visit to the local chest hospital. No other abnormalities were found.
On March 21, 2022, we first administered the patient the recommended dose of 45 mg of Ustekinumab to the patient, followed by a second injection of 45 mg 4 weeks later, and the subsequent two injections were given in the intervals of 12 weeks. So far, the patient has received four injections. There were no adverse effects during the treatment. The pain was completely eliminated half a month after the first injection, and improvement in the skin lesions was observed 4 weeks after the treatment, and the skin lesions were almost completely resolved after the third treatment (28 weeks after the initiation of the treatment) (Figure 1D–F; for details of the timeline, see Figure 2). We observed progressive improvement in DLQI from the first to the fourth injection, with the scores of 27, 23, 14, and 2 points, respectively. We noted a considerable improvement in the nails before the third injection (16 weeks after the initiation of the treatment). During a telephone-based follow-up 20 days after the fourth injection, the patient reported that new pustules occasionally appeared, but they quickly crusted and healed, having no adverse effect on daily life.
 |
Figure 2 The timeline of the course of disease. |
Discussion
ACH is a disorder that most commonly affects adult women, but it also affects men and women of all ages but rarely in children.5 Due to its low prevalence, there are no treatment guidelines or randomized controlled clinical trials, so the treatment for ACH is extremely challenging. The treatment of ACH is guided by its similarity to the other types of pustular psoriasis, as it is considered a variant of pustular psoriasis,6 Topical therapy (calcineurin inhibitors, corticosteroids and vitamin D derivatives, or a combination of these drugs) and phototherapy (ultraviolet A and narrowband ultraviolet B therapies) are often ineffective. Systemic therapy is usually necessary. Conventional systemic therapy, mainly including cyclosporine, tretinoin and methotrexate, be effective in some cases,7 but often loses efficacy over time.2 Biologic agents (IL-17 inhibitors, IL-12/23 inhibitors, IL-23 inhibitors, and anti-TNF agents), as well as small molecule compounds such as apremilast,8 seem to be a promising option for ACH. Starting with acitretin or methotrexate can serve as the first-line treatment for ACH, followed by biological agents, whereas cyclosporine contributes to short-term control.9 For patients with primary non-response, a timely, individualized administration of switch treatment should be conducted to avoid irreversible complications.
Biologics that can regulate the immunological pathways involved in psoriasis have long been available, and they can prevent the progression of inflammatory or immune cascades at an early stage, thereby reducing the number of side effects.10 They last longer in the body than conventional anti-psoriatic drugs, thereby reducing the need for frequent dosing. Several scholars have shared the experience of using biological agents for ACH based on case reports and small case series, with the anti-TNF agents being the most commonly reported. However, anti-TNF agent therapy often loses efficacy over time, prompting a switch to other agents11,12 or requiring an increased dose and frequency to maintain efficacy.13,14 In addition, IL-17 and IL-1 inhibitors have also been reported to be successful in treating ACH.
Ustekinumab is a fully human monoclonal antibody that specifically binds to the shared subunit p40 of IL-12 and IL-23, thereby blocking the differentiation of Th1 and Th17 cells and the subsequent inflammatory cascade mediated by IL-12 and IL-23.15 We have identified five case reports of patients with ACH treated with IL-12/23 inhibitors and four case reports of patients with ACH treated with IL-23 inhibitors in the literature, involving a total of twelve patients with ACH, as detailed in Table 1.11,16–23 Five patients were treated with Guselkumab, two with Risankizumab and five with Ustekinumab. Ustekinumab was reported to be effective, either as monotherapy or along with acitretin,11,20–23 but only one patient achieved a satisfactory clinical response at the recommended dose and frequency.11 One article compared the efficacy of anti-TNF agents and Ustekinumab in the treatment of ACH, and no difference was observed.24 However, Ustekinumab along with acitretin or as monotherapy can achieve good efficacy even after the failure of anti-TNF agents.11,21,22 The persistence of the response to Ustekinumab in ACH treatment is uncertain, with only one study reporting a poor and slower response to Ustekinumab monotherapy after a 7-month interruption than the initial regimen.22
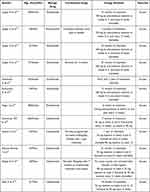 |
Table 1 Cases Reported in the Literature of Patients with ACH Treated with IL23 and IL12/23 Inhibitors |
Our patient responded exceptionally well to the treatment with Ustekinumab and was very satisfied with its efficacy. Two weeks after the first dose, the patient reported a decrease in pain score from 8 to 0, which indicated a complete remission. At 4 weeks, a considerable alleviation of the erythema, cutaneous edema, and psoriatic scales was observed. At 16 weeks, the skin lesions were almost completely cleared in all affected fingers, and the nail dystrophy began to improve. During the fourth dose (28 weeks), the deck pustules had all disappeared and new nails had grown. There were no adverse effects during the treatment. Large-scale and long-term clinical studies must confirm the efficacy and safety of this agent and to identify other effective treatments in the future.
The response of our patient to the treatment reflected that: 1) patients with refractory ACH can still benefit from Ustekinumab after the failure of conventional anti-psoriatic drugs and traditional Chinese medicine; 2) Ustekinumab can quickly and effectively relieve the burning pain and skin lesions and improve the quality of life of patients with ACH; 3) nail improvement is a relatively slow process, and therefore it is necessary to adhere to the treatment for a long time.
Conclusion
Ustekinumab has an excellent therapeutic efficacy in patients with ACH and is a reasonable option for the treatment of ACH. The onset of ACH in our patient was in childhood, which is very rare, and he is a perfect example of refractory characteristics. The patient’s quality of life was affected so seriously that he willingly underwent various treatments without satisfying outcomes over 13 years. The ability of Ustekinumab to induce remission in such a refractory disease is encouraging. Large-scale, long-term clinical studies are still needed to further determine its utility.
Abbreviations
ACH, Acrodermatitis continua of Hallopeau; VAS, visual analog scale; DLQI, Dermatology Life Quality Index; IGRA, interferon-gamma release assay; CT, computed tomography; T-N, tuberculosis T cells.
Ethics Approval and Informed Consent
All procedures performed in this study involving human participants was approved by the Ethics Committee of the First Hospital of Hebei Medical University.
Consent for Publication
Written informed consent was obtained from the patient for publication of this manuscript and any accompanying images.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Funding
This work is supported by the Graduate Education and Teaching Reform Project of Hebei Medical University (2022-20) and Medical science research project of Hebei Province (20190470).
Disclosure
All authors have no conflicts of interest in this work.
References
1. Sotiriadis D, Patsatsi A, Sotiriou E, Sotiriou E, Papagaryfallou I, Chrysomallis F. Acrodermatitis continua of Hallopeau on toes successfully treated with a two-compound product containing calcipotriol and betamethasone dipropionate. J Dermatolog Treat. 2007;18(5):315–318. doi:10.1080/09546630701367118
2. Frew JW, Murrell DF. Highly resistant acrodermatitis continua of Hallopeau and pustular psoriasis. Skin Appendage Disord. 2017;3(4):179–181. doi:10.1159/000464140
3. Megna M, Camela E, Battista T, et al. Efficacy and safety of biologics and small molecules for psoriasis in pediatric and geriatric populations. Part I: focus on pediatric patients. Expert Opin Drug Saf. 2023;22(1):25–41. doi:10.1080/14740338.2023.2173170
4. Megna M, Camela E, Battista T, et al. Efficacy and safety of biologics and small molecules for psoriasis in pediatric and geriatric populations. Part II: focus on elderly patients. Expert Opin Drug Saf. 2023;22(1):43–58. doi:10.1080/14740338.2023.2173171
5. Kiszewski AE, De Villa D, Scheibel I, Ricachnevsky N. An infant with acrodermatitis continua of Hallopeau: successful treatment with thalidomide and UVB therapy. Pediatr Dermatol. 2009;26(1):105–106. doi:10.1111/j.1525-1470.2008.00838.x
6. Navarini AA, Burden AD, Capon F, et al. European consensus statement on phenotypes of pustular psoriasis. J Eur Acad Dermatol Venereol. 2017;31(11):1792–1799. doi:10.1111/jdv.14386
7. Sehgal VN, Verma P, Sharma S, et al. Acrodermatitis continua of Hallopeau: evolution of treatment options. Int J Dermatol. 2011;50(10):1195–1211. doi:10.1111/j.1365-4632.2011.04993.x
8. Megna M, Potestio L, Di Caprio N, Tajani A, Fabbrocini G, Annunziata A. Acrodermatitis continua of Hallopeau: is apremilast an efficacious treatment option? Dermatol Ther. 2022;35(5):e15423. doi:10.1111/dth.15423
9. Kromer C, Loewe E, Schaarschmidt ML, et al. Treatment of acrodermatitis continua of Hallopeau: a case series of 39 patients. J Dermatol. 2020;47(9):989–997. doi:10.1111/1346-8138.15466
10. Kipnis CD, Myers WA, Opeola M, Gottlieb AB. Biologic treatments for psoriasis. J Am Acad Dermatol. 2005;52(4):671–682. doi:10.1016/j.jaad.2004.12.032
11. Palacios-Álvarez I, Simal-Gómez G, Mas-Vidal A, Bernad-Alonso I. Treatment of acrodermatitis continua of Hallopeau with ustekinumab as monotherapy after failure of anti-TNF agents. J Dtsch Dermatol Ges. 2018;16(5):611–613. doi:10.1111/ddg.13506
12. Gokdemir G, Kutlu S, Köşlü A. Acrodermatitis continua resistant to etanercept: therapeutic challenge and unfortunate outcome. J Eur Acad Dermatol Venereol. 2009;23(3):345–347. doi:10.1111/j.1468-3083.2008.02849.x
13. Tobin AM, Kirby B. Successful treatment of recalcitrant acrodermatitis continua of Hallopeau with Adalimumab and Acitretin. Br J Dermatol. 2005;153(2):445–446. doi:10.1111/j.1365-2133.2005.06759.x
14. Ryan C, Collins P, Kirby B, Rogers S. Treatment of acrodermatitis continua of Hallopeau with Adalimumab. Br J Dermatol. 2009;160(1):203–205. doi:10.1111/j.1365-2133.2008.08893.x
15. Koutruba N, Emer J, Lebwohl M. Review of ustekinumab, an interleukin-12 and interleukin-23 inhibitor used for the treatment of plaque psoriasis. Ther Clin Risk Manag. 2010;6:123–141. doi:10.2147/tcrm.s5599
16. Langer N, Wilsmann-Theis D, Kromer C, Mohr J, Mössner R. Successful therapy of acrodermatitis continua of Hallopeau with IL-23 blockers -two new cases. J Dtsch Dermatol Ges. 2021;19(10):1504–1507.
17. Yamamoto H, Kamiya K, Okada H, Maekawa T, Komine M, Ohtsuki M. A case of acrodermatitis continua of Hallopeau evolving into generalized pustular psoriasis successfully treated with guselkumab. Int J Dermatol. 2023;62(2):269–270. doi:10.1111/ijd.16153
18. Buononato D, Licata G, Gambardella A, De Rosa A, Calabrese G, Argenziano G. A case of acrodermatitis continua of Hallopeau successfully treated with guselkumab. Dermatol Ther. 2022;35(7):e15514. doi:10.1111/dth.15514
19. Hugo J, Gkalpakioti P, Arenbergerova M, Arenberger P, Gkalpakiotis S. Acrodermatitis continua Hallopeau successfully treated by risankizumab. Int J Dermatol. 2021;60(4):e144–e145. doi:10.1111/ijd.15391
20. Cymerman RM, Cohen DE. Treatment of acrodermatitis Continua of Hallopeau with ustekinumab as monotherapy. JAMA Dermatol. 2016;152(3):346–348. doi:10.1001/jamadermatol.2015.3444
21. Saunier J, Debarbieux S, Jullien D, Garnier L, Dalle S, Thomas L. Acrodermatitis continua of Hallopeau treated successfully with ustekinumab and Acitretin after failure of tumour necrosis factor blockade and anakinra. Dermatology. 2015;230(2):97–100. doi:10.1159/000367690
22. Adışen E, Özer İ, Temel B, Gürer MA. Ustekinumab for the treatment of acrodermatitis continua of Hallopeau refractory to anti-TNF agents. Dermatol Ther. 2017;30(2):e12460. doi:10.1111/dth.12460
23. Adas A, Dadban A, Arnault JP, Chaby G, Lok C. Acrodermatitis continua of Hallopeau response to optimized biological therapy. Dermatol Online J. 2019;25(2). doi:10.5070/D3252042897
24. Husson B, Barbe C, Hegazy S, et al. Efficacy and safety of TNF blockers and of ustekinumab in palmoplantar pustulosis and in acrodermatitis continua of Hallopeau. J Eur Acad Dermatol Venereol. 2020;34(10):2330–2338. doi:10.1111/jdv.16265
 © 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

