Back to Journals » Patient Preference and Adherence » Volume 10
Acceptability and feasibility of mHealth and community-based directly observed antiretroviral therapy to prevent mother-to-child HIV transmission in South African pregnant women under Option B+: an exploratory study
Authors Nachega J, Skinner D, Jennings L, Magidson J, Altice F , Burke J, Lester R, Uthman O, Knowlton A, Cotton M , Anderson J, Theron G
Received 6 November 2015
Accepted for publication 11 February 2016
Published 28 April 2016 Volume 2016:10 Pages 683—690
DOI https://doi.org/10.2147/PPA.S100002
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Johnny Chen
Jean B Nachega,1,2 Donald Skinner,3 Larissa Jennings,4 Jessica F Magidson,5 Frederick L Altice,6 Jessica G Burke,7 Richard T Lester,8 Olalekan A Uthman,9,10 Amy R Knowlton,11 Mark F Cotton,12 Jean R Anderson,13 Gerhard B Theron14
1Department of Epidemiology, University of Pittsburgh Graduate School of Public Health, Pittsburgh, PA, USA; 2Department of Medicine, Centre for Infectious Diseases, and ACTG Clinical Trial Unit (CTU)/Family Clinical Research Unit (FAMCRU), 3Research on Health and Society, Department of Interdisciplinary Health Studies, Faculty of Medicine and Health Sciences, Stellenbosch University, Cape Town, South Africa; 4Social and Behavioral Interventions Program, Department of International Health, Johns Hopkins Bloomberg School of Public Health, Baltimore, MD, 5Department of Psychiatry, Massachusetts General Hospital, Harvard Medical School, Boston, MA, 6Division of Infectious Diseases, AIDS Program, Yale University School of Medicine, New Haven, CT, 7Department of Behavioral and Community Health, University of Pittsburgh Graduate School of Public Health, Pittsburgh, PA, USA; 8Department of Medicine, Division of Infectious Diseases, University of British Columbia, Vancouver, BC, Canada; 9Warwick Centre for Applied Health Research and Delivery (WCAHRD), Division of Health Sciences, Warwick Medical School, The University of Warwick, Coventry, UK; 10Centre for Evidence-Based Health Care, Faculty of Medicine and Health Sciences, Stellenbosch University, Cape Town, South Africa; 11Department of Health, Behavior and Society, Johns Hopkins Bloomberg School of Public Health, Baltimore, MD, USA; 12Department of Pediatrics and Child Health, Faculty of Medicine and Health Sciences, Stellenbosch University, Cape Town, South Africa; 13Department of Obstetrics and Gynecology, School of Medicine, Johns Hopkins University, Baltimore, MD, USA; 14Department of Obstetrics and Gynecology, Faculty of Medicine and Health Sciences, Stellenbosch University, Cape Town, South Africa
Objective: To examine the acceptability and feasibility of mobile health (mHealth)/short message service (SMS) and community-based directly observed antiretroviral therapy (cDOT) as interventions to improve antiretroviral therapy (ART) adherence for preventing mother-to-child human immunodeficiency virus (HIV) transmission (PMTCT).
Design and methods: A mixed-method approach was used. Two qualitative focus group discussions with HIV-infected pregnant women (n=20) examined the acceptability and feasibility of two ART adherence interventions for PMTCT: 1) SMS text messaging and 2) patient-nominated cDOT supporters. Additionally, 109 HIV-infected, pregnant South African women (18–30 years old) receiving PMTCT services under single-tablet antiretroviral therapy regimen during pregnancy and breastfeeding and continuing for life (“Option B+”) were interviewed about mobile phone access, SMS use, and potential treatment supporters.
Setting: A community primary care clinic in Cape Town, South Africa.
Participants: HIV-infected pregnant women.
Main outcomes: Acceptability and feasibility of mHealth and cDOT interventions.
Results: Among the 109 women interviewed, individual mobile phone access and SMS use were high (>90%), and 88.1% of women were interested in receiving SMS ART adherence support messages such as reminders, motivation, and medication updates. Nearly all women (95%) identified at least one person close to them to whom they had disclosed their HIV status and would nominate as a cDOT supporter. Focus group discussions revealed that cDOT supporters and adherence text messages were valued, but some concerns regarding supporter time availability and risk of unintended HIV status disclosure were expressed.
Conclusion: mHealth and/or cDOT supporter as interventions to improve ART adherence are feasible in this setting. However, safe HIV status disclosure to treatment supporters and confidentiality of text messaging content about HIV and ART were deemed crucial.
Keywords: HIV, AIDS, pregnancy, short messaging system, social support
Introduction
Annually, nearly 1.4 million human immunodeficiency virus (HIV)-infected women give birth worldwide, and nearly all (>90%) reside in low- or middle-income countries where breastfeeding is common and access to prevention of mother-to-child HIV transmission (PMTCT) services may be limited.1 Indeed, in some sub-Saharan settings, due to lack of or late access to or suboptimal antiretroviral therapy (ART), HIV mother-to-child HIV transmission (MTCT) rates range from 25% to 48%.1,2 In contrast, in high-income countries, MTCT rates as low as 1%–2% or less have been achieved through effective combination of ART and breastfeeding avoidance.3 The success of this approach, coupled with a once-daily single-pill combination regimen, the increasing acknowledgment of the benefits of early treatment and the role of ART in preventing sexual transmission, evidence of protection against MTCT during breastfeeding, and simplification of program implementation have led the World Health Organization to recommend a single once-daily regimen during pregnancy and breastfeeding and continuing for life (Option B+), especially in generalized epidemics, regardless of CD4+ T-cell count.4 But concerns have been raised regarding the risk of ART adherence fatigue, with resulting virological failure and drug resistance among women enrolled in Option B+ regimens, as well as questions regarding the need to continue ART in the postpartum period among women in relatively good health.5
Low ART adherence among pregnant and recently delivered women impairs the effectiveness of PMTCT programs and may compromise long-term maternal health. A meta-analysis of ART adherence studies conducted among pregnant women in low-, middle- and high-income settings (51 studies, N=20,153) in the pre-Option B+ era revealed that only 75.7% had optimal (≥80%) antepartum adherence, declining to 50.3% during the postpartum period.5 Furthermore, women progressing through the postpartum period become more susceptible to “falling off the cliff” of the PMTCT treatment cascade.6 Evidence suggests that barriers to ART adherence among pregnant and recently delivered women include physical and depressive symptoms, forgetfulness, substance abuse, fear of HIV status disclosure, regimen fatigue, competing priorities, and lack of social support.7 Other recently identified barriers include concerns of gender inequity in treatment, food insecurity, and HIV disclosure.8,9 Despite the critical need for PMTCT during pregnancy and the postpartum period, evidence-based interventions to promote ART adherence during this period, particularly in the context of Option B+, are lacking.10
Mobile health (mHealth) and directly observed therapy (DOT) improve ART adherence in HIV-infected adults, in general.11–16 For example, weekly two-way interactive short message service (SMS) text messages significantly improved ART adherence and virological suppression in Kenyan HIV-infected adults.17 In South Africa, community-based directly observed antiretroviral therapy (cDOT) provided by patient-nominated community supporters such as friends and family, was associated with improved CD4+ T-cell count at 6 months follow-up and reduced AIDS-related mortality at 24 months follow-up.13 Coupling real-time mobile SMS with patient-nominated cDOT supporters may provide two powerful interventions to improve ART outcomes among women enrolled in PMTCT services.17 Little is known, however, regarding the acceptability of mobile SMS and cDOT, individually or in combination, among pregnant and recently delivered women.18 Attention to adherence support may be of particular importance in these women, who may have first learned of their HIV infection in pregnancy and have not been afforded the time for acceptance and treatment readiness.
This study used a mixed-methods approach to examine the acceptability and feasibility of mHealth and cDOT as possible interventions to improve ART adherence for PMTCT outcomes in HIV-infected South African pregnant women. We explored access to mobile phones, preferences for patient-nominated cDOT supporters, and use of mHealth in this population, with the ultimate aim to improve their ART adherence, virological, and immunological outcomes, and to decrease the risks of HIV transmission to their infants.
Design and methods
Study population
The study population was recruited from the Michael Mapongwana Community HIV primary care clinic in Khayelitsha, a township outside the city of Cape Town, South Africa, from December 2014 to January 2015. PMTCT services, including ART initiation and adherence support, are provided each month at the clinic to ~1,000 HIV-infected pregnant and postpartum women. Of note, Western Cape is the only province in South Africa where Option B+ (once-daily single tablet antiretroviral regimen during pregnancy and breastfeeding and continuing for life) is currently being implemented, pending countrywide expansion.
Procedures
A mixed-methods approach was used, including 1) two focus group discussions (FGDs) with 20 HIV-positive clinic-attending pregnant women on the acceptability and feasibility of a proposed SMS/cDOT adherence intervention (qualitative) and 2) individual structured interviews with HIV-positive pregnant women (n=109) to assess mobile phone access, SMS literacy, HIV disclosure, and interest in and availability of patient-nominated cDOT supporters (quantitative).
Qualitative phase
Pregnant women were recruited purposively by a clinic-based study nurse, based on the following characteristics: ≥18 years old, documented HIV seropositive, on ART or about to initiate ART, and able to give consent. The study nurse explained the study to potential participants and referred interested women to a study coordinator who obtained full informed consent and recruited them into one of the two focus groups. Both FGDs were led by a trained social scientist with qualitative research experience, using an open-ended interview guide to explore the following domains: 1) potential benefits and barriers of using an mHealth and patient-nominated cDOT supporter intervention to improve ART adherence and 2) how the context of pregnancy and the post-childbirth period may affect the use of mobile or cDOT supporter components of the proposed intervention. Each session, ~45 minutes in duration, was conducted in the local language (Xhosa), audio recorded, and transcribed for analysis.
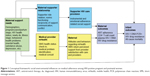
The interview guide was based upon a proposed conceptual framework, focusing on social support, reciprocity, and social network and role theories to elucidate the social dynamics of introducing and reinforcing norms of ART adherence and assistance in existing salient roles (support network members and medical providers). By bolstering the adherence support functioning of existing support networks and clinic providers, the study addresses key influences on ART adherence. In addition, the conceptual model includes text messages as a form of social support as well as information (Figure 1).
Quantitative phase
HIV-positive pregnant women aged 18 years or older, on ART or about to start ART and able to give consent, were recruited sequentially from the Michael Mapongwana HIV care clinic to complete an individual structured interview assessing 1) the use of cell phones; 2) acceptance of the use of text messaging for health messages intervention purposes; and 3) availability of support person(s) as a possible intervention for ART adherence. The survey took ~15 minutes to complete and was administered in the local language (Xhosa) by a skilled female field worker with previous clinical and survey research experience. The target sample size was derived to achieve precision of ±5% around proportions with 80% power and a type 1 error of 5%.
Statistical analysis
All qualitative data were transcribed, translated to English, and coded in Atlas-Ti Version 7.0 software (ATLAS.ti Scientific Software Development GmbH, Berlin, Germany; http://atlasti.com/). The FGD transcripts were iteratively reviewed and a codebook was developed based on the study’s objective and emerging themes in the data. A content thematic analysis approach was then used to identify the common preferences and concerns among women regarding the proposed intervention.19 Descriptive statistics (eg, mean, frequency, proportions) were used to summarize data from the structured survey using STATA, Version 11 (StataCorp LP, College Station, TX, USA).
Ethical approvals
Ethical approval was provided by the Stellenbosch University Research Ethics Committee and the Western Cape Province Government, Department of Health/Ministry of Health. Written informed consent was obtained from all study participants prior to enrollment.
Results
Qualitative phase
In FGDs, topics addressed included 1) acceptability and preferred content of SMS messaging; 2) concerns related to confidentiality of messaging; and 3) acceptability of patient-nominated cDOT supporters.
The most commonly preferred adherence-related messages were reminders to take ART medication and attend clinic appointments, credible information on ART drug regimens for HIV-infected pregnant women, and motivational messages to let them know they were not alone. As three women described:
If they [SMS messages] can remind us about the times of taking the pills or the dates of coming to the clinic. The SMS’s must also be educational about taking your pills and if you don’t take them, this and this will happen to you. [FGD woman]
Things like [information on] support groups like this one also helping because it’s where you see that you are not alone. There are people that are going through the same problem as you. [FGD woman]
I think it [SMS] will help a lot because sometimes after finding out about your status you still feel lost and don’t have any information about HIV. The only thing you know is the stuff you hear from other people so if you get SMS’s that support you it will help you. [FGD woman]
Women in FGDs revealed concerns that other people would see the ART adherence text messages and find out about their HIV status. For some women, their diagnosis of HIV was relatively new, and they had not yet fully absorbed what their diagnosis meant for them and their families. The move to a new community, as some women reported, constituted a “protection” and an impetus for the move was that no one would know their HIV status. These women felt they could restart their social lives and keep their status hidden. As one woman described:
It’s when you didn’t disclose your status and then someone get hold of your phone. I used to stay in the Eastern Cape but then I moved to Cape Town. It was easier [to take ART] because they don’t know me here. But if people don’t know about your status, it’s hard because you’re scared that people are going to see you take them. [FGD woman]
Several findings also emerged regarding the acceptability of patient-nominated cDOT supporters during the study’s FGDs. Women liked the idea of being supported by patient-nominated relatives or friends. When asked about the potential advantages/benefits of using a patient-nominated treatment supporter to provide daily cDOT, two women commented:
I think that person will be good for supporting you; like she said some people get dizzy after taking ART so if you faint and the person who knows about you taking ART will make things easier. [FGD woman]
The person will also make sure that you go to the clinic and you take your ART on time. [FGD woman]
Some women were uncertain whether all cDOT supporters could be relied upon to be consistently available and actively engaged. As one woman described:
I think that SMS system will help more than the patient-nominated supporter; people have their own problems. What if they have a problem and they can’t make it to me? But both are good in their own different ways. And you can’t really trust people because that person can talk about you to other people. [FGD woman]
Finally, the discussions revealed some confusion about participants’ knowledge about HIV and the role of ART, including why adherence is important. They recognized the role of ART in protecting HIV-exposed babies but were often unclear of the impact of ART on HIV-infected pregnant and recently delivered women, including concerns regarding drug-related side effects and confusion about the effects of ART on tuberculosis and other sexually transmitted infections. For example, one woman explained:
I think as a person who is HIV positive there are many things that you can catch, so if you take your ART you can protect the baby. And at the same time, you keep the virus under control so that it cannot spread more. You can also get STIs [sexually transmitted infections] so you must take ART to protect your child. [FGD woman]
I haven’t experienced anything, like I heard people say if you take ARVs you will get side effects but I never experienced that. [FGD woman]
Some people say it’s because of the side effects; because some vomit, others get a rash or nauseous. And not getting enough counselling and education or explanation about the ARVs. [FGD woman]
Quantitative phase
Participants (n=109) in the quantitative phase ranged from 18 to 30 years of age. Nearly all (99.1%) owned a mobile phone, and nearly all (88.1%) women were interested in receiving SMS adherence support messages for reminders, motivation, and drug updates. SMS literacy was high with 107 (98.2%) women regularly using SMS text messages to communicate with family (83.5%), male partners (53.2%), and friends (41.3%). No participants reported using SMS regularly to correspond with community health workers. Only 20 (18.3%) women reported having received health educational text messages. All women reported having at least one person in their network to whom they felt comfortable disclosing their HIV status and discussing ART adherence. One hundred and six (97.5%) were prepared to share the name and contact details of a potential nominated cDOT supporter for the proposed intervention. Additional interview results, including those related to preferences for SMS messages and individuals with whom they would feel comfortable discussing HIV and adherence-related issues, are reported in Table 1.
  | Table 1 Results of quantitative phase (structured interviews; n=109) |
Discussion
This study provides preliminary data on acceptability and feasibility of an mHealth and/or cDOT treatment supporter intervention for women receiving PMTCT in South Africa. Important findings from this mixed-methods study showed that individual mobile phone access and cell phone literacy were high (>90%) and that nearly all (88.1%) women were interested in receiving SMS adherence support messages as reminders, motivation, and drug updates. The high penetration of mobile phone technology in HIV-infected pregnant women and its acceptability suggest its utility to improve PMTCT outcomes. Nevertheless, only a small minority (<20%) had already used mobile phone communications for their health management, suggesting an opportunity for a structured mHealth intervention. In addition, qualitative findings provided important insights into how message reminders must be tailored to avoid disclosing someone’s HIV status to others. In several other studies on HIV-infected persons, text-based messaging has been supported as a platform for providing prompt and confidential behavioral change and motivational support messages, but this has not been evaluated in HIV-infected pregnant and breastfeeding women as an additional intervention for PMTCT.20–23
Certain mHealth interventions using SMS are effective in promoting adherence to ART medications.24–26 Tailored message content and timing appear to have a critical role in the impact of SMS interventions.26 Current evidence suggests superiority of two-way text messaging interventions over a one-way (more passive) mode of providing ART medication reminders.27,28 Our study shows that tailored messages also appear promising for PMTCT, although most existing research has yet to demonstrate the effectiveness of one-way messages.26 Pop-Eleches et al14 tested both daily and weekly messages and found that weekly messages improved ART adherence, whereas daily messages did not. WelTel Kenya participants reported that their weekly two-way messages made them feel “like someone cares”.11
An important issue revealed by the study participants was the risk of unintended disclosure of HIV status when using mHealth to promote ART adherence. This can be understood in the context of variable HIV disclosure rates among pregnant and postpartum women in sub-Saharan Africa; a recent systematic review found that although across 47 studies, 67% of HIV-infected pregnant and postpartum women in sub-Saharan Africa had disclosed their HIV status to another person, these rates varied widely (ie, from 5% to 97%).29 Therefore, in order to promote acceptability, text messages should use neutral language (not specifically mention ART/HIV), as well as use euphemistic terms for ART, other medications, and types of clinic visits. This is a primary reason why nonmedical language such as “Mambo?”/“Are You OK” was/is used in the WelTel Kenya trial by Lester et al.12 Participants were/are instructed to indicate within 48 hours of receiving the message either that they are doing well (eg, “OK”) or that they have a problem (eg, “Problem”). If a problem is reported, the participant will receive a follow-up support/ART adherence counseling call from the study team. With this approach, no accidental disclosures have been reported with over 3,000 participants thus far.12 Also, our data are in agreement with Jennings et al18 in Kenya who found that although PMTCT adherence text messages would need to protect against unintended HIV disclosure, women were eager to receive real-time support messages for adherence and motivation.
Unintended disclosure was also a concern in a recently published qualitative study aiming to develop text message content in Kenya for an mHealth study to improve postpartum retention in PMTCT and early infant HIV diagnosis. This study also suggested that the SMS messages incorporate motivational messages to engage women in care, particularly by focusing on the child’s health needs (eg, child’s clinic-based immunizations).30 Also, the implementation of mHealth in western Kenya has proven feasible and effective in tracking and improving the treatment-seeking behavior of pregnant women, during pregnancy, until 18 months following delivery, and could therefore effectively complement the ongoing PMTCT efforts to reach the goal of virtual elimination in sub-Saharan Africa.31
Regarding patient-nominated cDOT supporters, the qualitative data illustrate the important role of social support to meet maternal health and HIV-related needs. Social support typically includes emotional support (ie, providing love, empathy), instrumental or “tangible” support, informational support (ie, problem solving and guidance), and/or social companionship, which can provide a feeling of connection, belonging, and/or affirmation.32,33 Our qualitative data point specifically to the need for instrumental and emotional support during pregnancy, which is reflected in our conceptual model (Figure 1). This finding is in line with prior qualitative research pointing to the role of social support in understanding successful adherence in sub-Saharan Africa, referring to how individuals can overcome obstacles to adherence in a large part due to resources made available by treatment supporters, family, friends, and health care providers; further, this support is reciprocal, such that adherence follows to fulfill a perceived responsibility to one’s supporters.34
These findings are consistent with other studies examining the acceptability of cDOT interventions in the general HIV population in which a family member such as a mother, sibling, or friend (irrespective of HIV status) was a more commonly preferred ART treatment supporter, compared to sexual partners, who were least mentioned as a preferred supporter.35 Only in long-term, stable relationships, with HIV status mutually disclosed, were sexual partners reported as important sources of social support and potentially effective candidates for community-based ART DOT.35 This evidence suggests that effective cDOT interventions among pregnant and postpartum women should be family based, rather than couples based, in order to improve maternal and newborn ART outcomes. Our study provides important clinical implications to guide the design and evaluation of future and novel HIV treatment adherence interventions in pregnant and postpartum women. mHealth and/or cDOT supporter interventions for PMTCT should address issues related to safe disclosure of HIV status to treatment supporter. Maintenance of confidentiality in SMS messages intended to educate patients about HIV and ART will promote acceptability and feasibility of this intervention. There is a critical need to evaluate the effectiveness and cost-effectiveness of these interventions singularly or in combination (eg, by factorial design to allow precise determination of component intervention and interaction effects) through well-controlled randomized trials in this underresearched HIV-infected pregnant and postpartum women.36
Pregnancy and the immediate postpartum period (eg, breastfeeding) is a key opportunity for using community- or home (family)-based DOT-ART by providing targeted social support needed to address specific individual, structural, and social barriers to ART adherence experienced by this vulnerable population. In a mathematical simulation model, McCabe et al37 found that DOT was associated with a relative risk of HIV transmission of 0.39 compared to conventional ART, highly cost-effective in the cohort as a whole (cost–utility ratio $14,233 per QALY), and cost-saving in women whose plasma HIV-1 RNA on self-administered ART would have exceeded 1,000 copies/mL. The results were stable in wide-ranging sensitivity analyses, with DOT being cost-saving or highly cost-effective in almost all cases.37
This study is limited due to its cross-sectional nature. This does not allow us to determine how relationships and feelings about the proposed interventions may change over time or how acceptability and feasibility may change after birth since all women were still pregnant. Also, our study draws upon a small number of focus groups for which we used convenience sampling. Therefore, some preferences among women for PMTCT-related text messaging and cDOT supporter interventions may not have been captured. To mitigate this potential limitation, we invited enrolled women to share as many of their opinions as possible, enabling the study to report on a range of important considerations for effective mHealth and cDOT components for PMTCT. In addition, the study did neither assess women’s perceptions of training needs for effective cDOT supporters nor explore the preferred methods of using SMS for PMTCT. The study’s findings are also representative of HIV-positive women who are currently seeking treatment and engaged in PMTCT care, therefore providing more positive responses than from noncare-seeking pregnant and postpartum women. Nevertheless, findings from this study include timely and valuable acceptability and feasibility data to support developing and designing these two novel low-cost ART adherence interventions. The implementation of these two interventions, either alone or in combination, may provide important support for this vulnerable population. These home- and community-based interventions shift point of care and tasks from health clinics and health care providers to friends and/or family members in communities, and therefore, are likely to be easily scalable and cost-effective.
Conclusion
Our findings indicate that mHealth and patient-nominated treatment supporters are acceptable and potentially promising interventions to support adherence to PMTCT. However, issues related to confidential SMS messaging require attention to avoid unintended disclosure. These include orientating family-based adherence supporters on issues of privacy, correct ART use, and timely engagement. Future research may also examine how best to link SMS adherence messaging among pregnant and postpartum women with cDOT supporter communication to interventions targeting other structural and psychosocial barriers (ie, depression, substance abuse) in support of lifelong ART adherence.
Acknowledgments
The authors thank the study participants who participated in this research, and the field worker, Ms Tembi Koliswa, who conducted the interviews and moderated the focus group discussions, and Elizabeth D Lowenthanl, MD, for critical reading and advice on this manuscript.
Dr Nachega receives research grant support from the National Institutes of Health/National Institutes for Allergy and Infectious Disease, the AIDS Clinical Trial Group (ACTG)/Stellenbosch University Clinical Trial Unit (2UM1AI069521-08); the US President Emergency Plan for AIDS Relief (PEPFAR; T84HA21652-01-00) for Medical Education Partnership Initiative; the European Developing Countries Clinical Trial Partnership Senior Fellowship Award (TA-08-40200-021); and the Wellcome Trust Southern Africa Consortium for Research Excellence (WT087537MA). Professor Altice is funded by the National Institute on Drug Abuse for research (R01-DA10186, R01 DA13805, and R01 DA017072) and career development (K24 DA017072). Dr Lester is supported in part by research grants from the National Institutes of Health/National Institutes for Mental Health (R01-MH09755), the Michael Smith Foundation for Health Research, and Grand Challenges Canada. Dr Uthman acknowledges support from the FAS Marie Curie International PostDoc (2012-0064). Dr Magidson acknowledges support from the National Institute of Mental Health (T32MH093310).
Disclosure
The authors report no conflicts of interest in this work.
References
De Cock KM, Fowler MG, Mercier E, et al. Prevention of mother-to-child HIV transmission in resource-poor countries: translating research into policy and practice. JAMA. 2000;283:1175–1182. | ||
Wiktor SZ, Ekpini E, Nduati RW. Prevention of mother-to-child transmission of HIV-1 in Africa. AIDS. 1997;11(Suppl B):S79–S87. | ||
Townsend CL, Cortina-Borja M, Peckham CS, de Ruiter A, Lyall H, Tookey PA. Low rates of mother-to-child transmission of HIV following effective pregnancy interventions in the United Kingdom and Ireland, 2000–2006. AIDS. 2008;22:973–981. | ||
World Health Organization (WHO). Antiretroviral Drugs for Treating Pregnant Women and Preventing HIV Infections in Infants: Recommendations for a Public Health Approach. Geneva, Switzerland: World Health Organization; 2013. | ||
Nachega JB, Uthman OA, Anderson J, et al. Adherence to antiretroviral therapy during and after pregnancy in low-income, middle-income, and high-income countries: a systematic review and meta-analysis. AIDS. 2012;26:2039–2052. | ||
Psaros C, Remmert JE, Bangsberg DR, Safren SA, Smit JA. Adherence to HIV care after pregnancy among women in sub-Saharan Africa: falling off the cliff of the treatment cascade. Curr HIV/AIDS Rep. 2015;12:1–5. | ||
Mills EJ, Nachega JB, Bangsberg DR, et al. Adherence to HAART: a systematic review of developed and developing nation patient-reported barriers and facilitators. PLoS Med. 2006;3:e438. | ||
Global Network of People living with HIV (GNP+). Understanding the Perspectives and/or Experiences of Women Living with HIV Regarding Option B+ in Uganda and Malawi. Amsterdam, The Netherlands: Global Network of People living with HIV; 2013. | ||
Katirayi L, Namadingo H, Bobrow E, et al. Acceptability of lifelong treatment (Option B+) among HIV-positive pregnant and lactating women in selected sites in Malawi. Abstract A-641-0287-09318. In: International AIDS Society Conference on HIV Pathogenesis, Treatment and Prevention. Melbourne, Australia; 2014. | ||
Thompson MA, Mugavero MJ, Amico KR, Cargill VA, Chang LW, Gross R, et al. Guidelines for improving entry into and retention in care and antiretroviral adherence for persons with HIV: evidence-based recommendations from an International Association of Physicians in AIDS Care panel. Ann Intern Med. 2012;156:817–833, w-284, w-285, w-286, w-287, w-288, w-289, w-290, w-291, w-292, w-293, w-294. | ||
Altice FL, Maru DS, Bruce RD, Springer SA, Friedland GH. Superiority of directly administered antiretroviral therapy over self-administered therapy among HIV-infected drug users: a prospective, randomized, controlled trial. Clin Infect Dis. 2007;45:770–778. | ||
Lester RT, Ritvo P, Mills EJ, et al. Effects of a mobile phone short message service on antiretroviral treatment adherence in Kenya (WelTel Kenya1): a randomised trial. Lancet. 2010;376:1838–1845. | ||
Mitty JA, Macalino GE, Bazerman LB, et al. The use of community-based modified directly observed therapy for the treatment of HIV-infected persons. J Acquir Immune Defic Syndr. 2005;39:545–550. | ||
Nachega JB, Chaisson RE, Goliath R, et al. Randomized controlled trial of trained patient-nominated treatment supporters providing partial directly observed antiretroviral therapy. AIDS. 2010;24:1273–1280. | ||
Pop-Eleches C, Thirumurthy H, Habyarimana JP, et al. Mobile phone technologies improve adherence to antiretroviral treatment in a resource-limited setting: a randomized controlled trial of text message reminders. AIDS. 2011;25:825–834. | ||
Mills EJ, Lester R, Thorlund K, et al. Interventions to promote adherence to antiretroviral therapy in Africa: a network meta-analysis. Lancet HIV. 2015;1:e104–e111. | ||
Duwell MM, Knowlton AR, Nachega JB, et al. Patient-nominated, community-based HIV treatment supporters: patient perspectives, feasibility, challenges, and factors for success in HIV-infected South African adults. AIDS Patient Care STDS. 2013;27:96–102. | ||
Jennings L, Ong’ech J, Simiyu R, Sirengo M, Kassaye S. Exploring the use of mobile phone technology for the enhancement of the prevention of mother-to-child transmission of HIV program in Nyanza, Kenya: a qualitative study. BMC Public Health. 2013;13:1131. | ||
Skinner D. Qualitative research methodology: an introduction. In: Ehrlich R, Joubert G, editors. Epidemiology: A Research Manual for South Africa. Cape Town: Oxford University Press; 2014. | ||
Belzer ME, Naar-King S, Olson J, et al. The use of cell phone support for non-adherent HIV-infected youth and young adults: an initial randomized and controlled intervention trial. AIDS Behav. 2014;18:686–696. | ||
Lewis MA, Uhrig JD, Bann CM, et al. Tailored text messaging intervention for HIV adherence: a proof-of-concept study. Health Psychol. 2013;32:248–253. | ||
Noar SM, Willoughby JF. eHealth interventions for HIV prevention. AIDS Care. 2012;24:945–952. | ||
Swendeman D, Rotheram-Borus MJ. Innovation in sexually transmitted disease and HIV prevention: internet and mobile phone delivery vehicles for global diffusion. Curr Opin Psychiatry. 2010;23:139–144. | ||
Coomes CM, Lewis MA, Uhrig JD, Furberg RD, Harris JL, Bann CM. Beyond reminders: a conceptual framework for using short message service to promote prevention and improve healthcare quality and clinical outcomes for people living with HIV. AIDS Care. 2012;24:348–357. | ||
Finitsis DJ, Pellowski JA, Johnson BT. Text message intervention designs to promote adherence to antiretroviral therapy (ART): a meta-analysis of randomized controlled trials. PLoS One. 2014;9:e88166. | ||
Horvath T, Azman H, Kennedy GE, Rutherford GW. Mobile phone text messaging for promoting adherence to antiretroviral therapy in patients with HIV infection. Cochrane Database Syst Rev. 2012;3:Cd009756. | ||
Robbins RN, Spector AY, Mellins CA, Remien RH. Optimizing ART adherence: update for HIV treatment and prevention. Curr HIV/AIDS Rep. 2014;11:423–433. | ||
Saberi P, Johnson MO. Technology-based self-care methods of improving antiretroviral adherence: a systematic review. PLoS One. 2011;6:e27533. | ||
Tam M, Amzel A, Phelps BR. Disclosure of HIV serostatus among pregnant and postpartum women in sub-Saharan Africa: a systematic review. AIDS Care. 2015;27:436–450. | ||
Odeny TA, Newman M, Bukusi EA, McClelland RS, Cohen CR, Camlin CS. Developing content for a mHealth intervention to promote postpartum retention in prevention of mother-to-child HIV transmission programs and early infant diagnosis of HIV: a qualitative study. PLoS One. 2014;9:e106383. | ||
Mushamiri I, Luo C, Iiams-Hauser C, Ben Amor Y. Evaluation of the impact of a mobile health system on adherence to antenatal and postnatal care and prevention of mother-to-child transmission of HIV programs in Kenya. BMC Public Health. 2015;15:102. | ||
Sherbourne CD, Stewart AL. The MOS social support survey. Soc Sci Med. 1991;32:705–714. | ||
Langford CP, Bowsher J, Maloney JP, Lillis PP. Social support: a conceptual analysis. J Adv Nurs. 1997;25:95–100. | ||
Ware NC, Idoko J, Kaaya S, et al. Explaining adherence success in sub-Saharan Africa: an ethnographic study. PLoS Med. 2009;6:e11. | ||
Nachega JB, Knowlton AR, Deluca A, et al. Treatment supporter to improve adherence to antiretroviral therapy in HIV-infected South African adults. A qualitative study. J Acquir Immune Defic Syndr. 2006;43(Suppl 1):S127–S133. | ||
Chaiyachati KH, Ogbuoji O, Price M, Suthar AB, Negussie EK, Bärnighausen T. Interventions to improve adherence to antiretroviral therapy: a rapid systematic review. AIDS. 2014;28(Suppl 2):S187–S204. | ||
McCabe CJ, Goldie SJ, Fisman DN. The cost-effectiveness of directly observed highly-active antiretroviral therapy in the third trimester in HIV-infected pregnant women. PLoS One. 2010;5:e10154. |
 © 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.