Back to Journals » International Medical Case Reports Journal » Volume 13
A Teenager with Vernal Keratoconjunctivitis and Pellucid Marginal Degeneration, Presenting with Exotropia
Authors Bajracharya L , Agrawal N , Dhungel S , Parajuli R, Adhikari S
Received 27 May 2020
Accepted for publication 14 August 2020
Published 7 September 2020 Volume 2020:13 Pages 399—408
DOI https://doi.org/10.2147/IMCRJ.S262999
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Leena Bajracharya,1 Nisha Agrawal,2 Shashwat Dhungel,3 Rojeeta Parajuli,4 Srijana Adhikari4
1Cornea Department, Tilganga Institute of Ophthalmology, Kathmandu, Nepal; 2Pediatric Ophthalmology and Strabismus Department, Taparia Eye Care, Biratnagar, Nepal; 3Department of Refractive Surgery, Tilganga Institute of Ophthalmology, Kathmandu, Nepal; 4Pediatric Ophthalmology and Strabismus Department, Tilganga Institute of Ophthalmology, Kathmandu, Nepal
Correspondence: Leena Bajracharya; Nisha Agrawal Tel +977 1449 3775
; +977 98 5204 9870
Email [email protected]; [email protected]
Background: Vernal keratoconjunctivitis (VKC), an allergic disease, has a known association with ectatic disorders of the cornea. Pellucid marginal degeneration (PMD) is a bilateral, asymmetrical, ectatic disorder of the cornea characterized by inferior corneal thinning. We report a case of sensory exotropia due to PMD in association with VKC.
Case Details: A 19-year old boy with a history of VKC presented with exotropia of the right eye of 3 years’ duration. His unaided vision in his right eye was 1/60 and in his left eye it was 6/36. On examination, both eyes had high against the rule astigmatism, which was more in the right eye. There was exotropia of 15º (40 prism diopter base in) in the right eye with suppression (Worth four dot test). Corneal examination in both eyes showed inferior band thinning, 2 mm above the inferior limbus, extending from 4 to 8 clock hours, with bulging of the cornea just above the thinning. The clinical features were suggestive of PMD, which was supported by his corneal scans – Atlas, Pentacam, and Optovue. Although he was undergoing treatment for VKC, the onset of PMD and decrease in vision went unnoticed. The asymmetric error which was not corrected during the sensitive period of visual development led to sensory exotropia.
Conclusion: A child with VKC should undergo regular refraction so as not to miss any ectatic changes occurring in the cornea. A delay in diagnosing corneal ectasia may negate the possibility of collagen cross-linking which prevents progression of ectasia. If visual rehabilitation is delayed beyond the age of visual maturation, it can lead to strabismus, suppression, and loss of binocular function.
Keywords: vernal keratoconjunctivitis, pellucid marginal degeneration, exotropia, suppression
Introduction
Vernal keratoconjunctivitis (VKC), an allergic disease which is common in young children and adolescents, has a known association with ectatic disorders of the cornea, especially keratoconus.1 Pellucid marginal degeneration (PMD) is rarely associated with VKC. A study by Cameron et al2 showed that the types of ectasia present in VKC were keratoconus (86.8%), followed by PMD (8%) and keratoglobus (3%).
Pellucid marginal degeneration is a rare, bilateral, asymmetrical, ectatic disorder of the cornea, which is characterized by thinning of the inferior cornea. It usually starts later in life and progresses slowly.3 It can induce high astigmatism, which, if left uncorrected during the sensitive period of visual development, may inflict sensory strabismus and amblyopia.
Exotropia is the most common form of strabismus.4,5 It has sensory implications on visual function but medical attention is usually sought for cosmetic purposes.6 Sensory strabismus is a less common form of strabismus, the prevalence of which has been reported to be 5−9%.7 Temporary or permanent loss of vision in one or both eyes can lead to sensory strabismus. We report a case of PMD and sensory exotropia in the background of VKC. This report aims to emphasize the importance of detailed history taking and meticulous examination of eyes of allergic children to reveal associated corneal ectasia which can lead to decreased vision.
Case Report
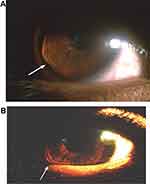
A 19-year old boy presented with a desire to have his eyes aligned. He complained of outward deviation of the right eye noted for 3 years, which was gradually increasing. There was no history of diurnal variation, trauma, ocular surgery, or use of glasses in the past. There was a history suggestive of recurrent vernal keratoconjunctivitis which started at the age of 5 years. He was treated with topical steroids, non-steroidal anti-inflammatory drugs, and mast cell stabilizer in the past. The patient was right handed but he was not aware of rubbing his right eye more than his left eye. At presentation, he was on mast cell stabilisers and did not complain of itching. There was no family history of atopy or deviation of eyes. The patient has one elder brother aged 24 years whose ocular examination findings were not suggestive of allergy or ectatic disorders of the cornea. On examination, uncorrected visual acuity in the right eye was 1/60 and that of the left eye was 6/36. There was absence of scissor’s reflex on retinoscopy and refraction showed high against the rule astigmatism in both eyes. Vision did not improve with refractive correction of +2DS/-12 DC at 90° in the right eye, whereas the left eye improved to 6/12 with +0.0 DS/-4.5DC at 90°. The patient had a normal head posture, and Hirschberg test revealed 15º exotropia in the right eye with full range of version and duction movements. Prism base cover test revealed a 40 prism diopter base in exotropia for near vision. The right eye was suppressed (Worth four dot test). There was mild papillary reaction in the upper tarsal conjunctiva, suggesting the presence of allergic conjunctivitis. On slit lamp examination, there was a band of thinning in the inferior cornea extending from 4 to 8 clock hours in both eyes. The band of thinning was located 2 mm above the inferior limbus. The cornea just above the thinning was steep and bulging, which could be elicited when light was shone to the limbus from the opposite side (Figure 1). In these figures, a typical cone shaped bulge (like in keratoconus) was not appreciated in the light reflex. Central corneal thickness looked normal in both eyes and there was no striae, scarring, or vascularization in either eye. Munson’s sign (conical indentation of the inferior eyelid margin on down gaze, a feature of keratoconus) was absent in both eyes. Dilated fundus examination was normal. These findings were consistent with corneal ectasia in both eyes, with the right eye more affected than the left. A rigid gas permeable contact lens trial could not be done because the corneas could not hold the lens. With a trial of miniscleral contact lens, vision improved to 6/9 in both eyes. Corneal scans showed against the rule astigmatism. A crab claw pattern with central flattening of the vertical meridian was seen in the Atlas (Figure 2). In the right eye, the pentacam showed that the steepest part of the cornea was just below the thinnest part of the cornea (Figure 3A–F). In the Belin Ambrosio Display (BAD) map relatively lower BAD D values were observed, especially in the left eye (Figure 4). The thinnest part of the cornea was not coinciding with the thinnest part of the epithelium in the optovue, especially in the right eye (Figure 5). All of these findings suggest PMD.8 The right eye was more severely affected, leading to high myopia and astigmatism. This asymmetric error went unnoticed and uncorrected during his sensitive period of visual development, which led to sensory exotropia.
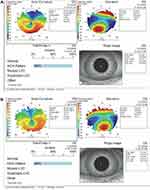 |
Figure 2 Corneal topography map of the right (A) and left (B) eyes showing a crab claw pattern in axial curvature. |
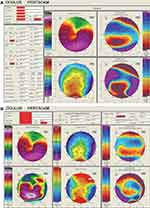 |
Figure 3 Continued. |
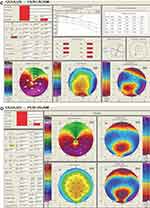 |
Figure 3 Continued. |
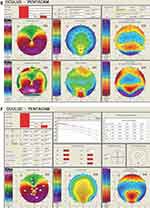 |
Figure 3 Pentacam reports show thinning inferiorly, with steep keratometry below the region of thinning in the right eye (A–C) and the left eye (D–F). |
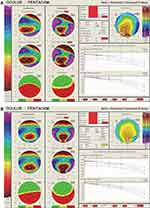 |
Figure 4 Belin Ambrosio Display values in the right (A) and left eyes (B). It is showing relatively lower values, especially in the left eye. |
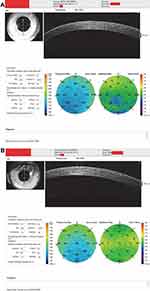 |
Figure 5 Pachymetric and epithelial pachymetric map shows minimum thickness of the cornea and that of the epithelium are not corresponding to the same location in the right (A) and left (B) eyes. |
The patient was advised for follow-up for progression of PMD. If any signs of progression of PMD would be observed, collagen cross-linking (CXL) would be the treatment plan to halt further thinning and to prevent hydrops or the need of keratoplasty. For the visual rehabilitation, the patient was advised to use mini scleral lens. When the cornea stabilizes (with or without CXL), the patient will be planned for strabismus surgery.
Discussion
Allergic conditions are common ailments of young children and adolescents. Constant eye rubbing leads to release of cytokine like tear metalloproteinase, interleukin-6, and tumor necrosis factor-alpha, which induces apoptosis or controlled cell death of stromal keratocytes. This is the inciting cause of ectasia.9 The association of VKC with ectatic disorders of the cornea has been studied in various parts of the world. A study by Cameron et al2 showed that the types of ectasia present in the VKC were keratoconus (86.8%), followed by PMD (8%), and kertoglobus (3%). In a hospital-based study done in Nepal, Keratoconus-like topography was present in 11.3% of VKC.10 A study from India reported that 6% of VKC patients had keratoconus.11 Caputo et al12 reported only 0.77% of VKC subjects having topographic changes of keratoconus in a sub-group of population in Italy. Literature regarding the association of PMD with VKC is comparatively scarce. In a retrospective review of cases of PMD by Sridhar MS,13 1.7% of PMD cases were associated with VKC. Vandana et al14 noted PMD in a 45 years old male but with atopic keratoconjunctivitis and not VKC. Screening for ectatic disorders should be performed promptly whenever an allergic child presents with progressive visual blurring, decrease in best corrected visual acuity (BCVA) by spectacles, irregular astigmatism, or with frequent change in glass prescription.
PMD is characterized by inferior thinning of the cornea and is a rare condition.3 It usually starts later in life than keratoconus and has slower progression.3 The average age of presentation of PMD in general is 34±14 years (range=8–66 years) with male predominance (77.6%).13,15 In the study done by Cameron et al2 the age of subjects with PMD and VKC together ranged from 13 –24 years and the age of onset of PMD ranged from 10 –14 years. Our patient had onset of allergic symptoms at the age of 5 years. But the exact age of onset of decrease in vision is not known to the patient or his parents. Since vision after scleral lens correction has been 6/9, onset of PMD could have been after crossing the amblyogenic age.
The presence of exotropia associated with ectatic disorder of the cornea is also rarely reported in the literature. There are few reports of exotropia with keratoconus but no case of PMD has been reported to present with ocular deviation. Ciftci et al7 reported a case of a boy of 17 years with advanced keratoconus, BCVA of 1/60, and exotropia of the eye. Sherafat et al6 evaluated 350 patients with keratoconus and found that 20 of them had abnormal binocular function. Another study by Khan et al16 reported that six out of 103 patients with keratoconus had abnormal binocular function and exotropia. These patients may have developed suppression from uncorrected high refractive error followed by sensory strabismus.6,16 Likewise, in our patient, poor vision due to high astigmatism, and long standing asymmetrical uncorrected refractive error might have led to sensory exotropia. With the miniscleral lens trial, our patient did not feel any diplopia. This could be due to suppression of the worse eye. In the study by Khan et al,16 after correction of the vision of the exotropic eyes, the subjects felt diplopia, which was later corrected with strabismus surgery.
Poor socio-economic strata, lack of awareness about the symptoms, and poor access to higher healthcare facilities could be reasons the patient did not receive any medical attention or refractive correction prior to consulting us. The benefits of riboflavin and ultraviolet-A induced CXL are well established for keratoconus to halt the progression.17 Although there have not been many studies regarding the role of CXL for treatment of PMD, the available studies show promising results of CXL in stabilizing PMD.17,18 On this basis, the plan for our patient would be to perform CXL in either eye if they show signs of progression. Close follow-up is very important to detect signs of progression at the earliest. Strabismus surgery would be needful for a teenager like him for cosmesis and for diplopia if he would develop any.
Conclusion
Frequent evaluation of vision and refraction as well as careful examination of the cornea is mandatory in a child with VKC and, if there is any doubt, screening for ectasia of the cornea should be undertaken. The patient party should be counseled about the importance of regular vision tests in an allergic child. This should be included in the protocol of management of VKC. Similarly a child with sensory exotropia due to high myopic astigmatism at adolescent age with subnormal BCVA should also be evaluated, keeping in mind the possibility of corneal ectasia in that age group. If any decrease in vision is not detected in the early years, it can lead to suppression, strabismus, and loss of binocular function. Also, if corneal ectasia is not diagnosed in time or if thinness of the cornea reaches below a threshold value then CXL cannot be done and its treatment might necessitate keratoplasty, which is more costly and complex.
Ethics and Consent
Ethical approval regarding publication of the case details was obtained from the Institutional Review Board of Tilganga Institute of Ophthalmology. Written informed consent for patient information and images to be published was obtained from the patient.
Funding
No funding or grant support.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Martínez-Abad A, Piñero DP. Pellucid marginal degeneration: detection, discrimination from other corneal ectatic disorders and progression. Cont Lens Anterior Eye. 2019;42(4):341–349. doi:10.1016/j.clae.2018.11.010
2. Cameron JA, Al-Rajhi AA, Badr IA. Corneal ectasia in vernal keratoconjunctivitis. Ophthalmology. 1989;96(11):1615–1623. doi:10.1016/S0161-6420(89)32677-7
3. Jinabhai A, Radhakrishnan H, Donnell CO. Pellucid corneal marginal degeneration: a review. Cont Lens Anterior Eye. 2011;34:56–63. doi:10.1016/j.clae.2010.11.007
4. Attada TR, Deepika M, Laxmi S. Strabismus in paediatric age (3–16 year): a clinical study. Int J Res Med Sci. 2016;4(6):1903–1909. doi:10.18203/2320-6012.ijrms20161731
5. Garvey KA, Dobson V, Messer DH, Joseph MM, Harvey EM. Prevalence of strabismus among preschool, kindergarten, and 1st grade tohono o’odham children. Optometry. 2010;81(4):194–199. doi:10.1016/j.optm.2009.10.010
6. Sherafat H, White JE, Pullum KW, Adams GG, Sloper JJ. Anomalies of binocular function in patients with longstanding asymmetric keratoconus. Br J Ophthalmol. 2001;85(9):1057–1060. doi:10.1136/bjo.85.9.1057
7. Ciftci S, Simsek A, Dogan E, Ciftci L. Sensory exotropia due to keratoconus and review of the literature. Clin Ophthalmol. 2013;7:2069–2072. doi:10.2147/OPTH.S53397
8. Koc M, Tekin K, Inanc M, Kosekahya P, Yilmazbas P. Crab claw pattern on corneal topography: pellucid marginal degeneration or inferior keratoconus? Eye (Lond). 2018;32(1):11–18. doi:10.1038/eye.2017.198
9. Mukhtar S, Ambati BK. Pediatric keratoconus: a review of the literature. Int Ophthalmol. 2018;38(5):2257–2266. doi:10.1007/s10792-017-0699-8
10. Gautam V, Chaudhary M, Sharma AK, Shrestha GS, Rai PG. Topographic corneal changes in children with vernal keratoconjunctivitis: a report from Kathmandu, Nepal. Cont Lens Anterior Eye. 2015;38(6):461–465. doi:10.1016/j.clae.2015.05.013
11. Saboo US, Jain M, Reddy JC, Sangwan VS. Demographic and clinical profile of vernal keratoconjunctivitis at a tertiary eye care center in India. Indian J Ophthalmol. 2013;61(9):486–489. doi:10.4103/0301-4738.119431
12. Caputo R, Versaci F, Pucci N, et al. Very low prevalence of keratoconus in a large series of vernal keratoconjunctivitis patients. Am J Ophthalmol. 2016;172:64–71. doi:10.1016/j.ajo.2016.09.009
13. Sridhar MS, Mahesh S, Bansal K, Nutheti R, Rao GN. Pellucid marginal corneal degeneration. Ophthalmology. 2004;111(6):1102–1107. doi:10.1016/j.ophtha.2003.09.035
14. Jain V, Nair AG, Jain MK, Shome D. Pellucid marginal corneal disease in a case of atopic keratoconjunctivitis. Ocul Immunol Inflamm. 2010;18(3):187–189. doi:10.3109/09273940903572538
15. Shimazaki J, Maeda N, Hieda O, et al. Japan pellucid marginal corneal degeneration study group y7 national survey of pellucid marginal corneal degeneration in Japan. Jpn J Ophthalmol. 2016;60(5):341–348. doi:10.1007/s10384-016-0462-0
16. Khan AO, Al-Shamsi H. Binocular diplopia following vision restoration in patients with keratoconus and sensory strabismus. Strabismus. 2008;16(1):19–22. doi:10.1080/09273970701863149
17. Yong J, Hatch K. Corneal cross-linking: an effective treatment option for pellucid marginal degeneration. Semin Ophthalmol. 2019;34(7–8):1–6. doi:10.1080/08820538.2019.1659832
18. Pircher N, Lammer J, Holzer S, Gschließer A, Schmidinger G. Corneal crosslinking for pellucid marginal degeneration. J Cataract Refract Surg. 2019;45(8):1163–1167. doi:10.1016/j.jcrs.2019.03.018
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.