Back to Journals » Clinical Ophthalmology » Volume 13
A study protocol for evaluating the efficacy and safety of skin electrical stimulation for Leber hereditary optic neuropathy: a single-arm, open-label, non-randomized prospective exploratory study
Authors Kurimoto T, Ueda K , Mori S , Sakamoto M , Yamada-Nakanishi Y, Matsumiya W, Nakamura M
Received 9 February 2019
Accepted for publication 12 April 2019
Published 28 May 2019 Volume 2019:13 Pages 897—904
DOI https://doi.org/10.2147/OPTH.S204669
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Takuji Kurimoto, Kaori Ueda, Sotaro Mori, Mari Sakamoto, Yuko Yamada-Nakanishi, Wataru Matsumiya, Makoto Nakamura
Division of Ophthalmology, Department of Surgery, Kobe University Graduate School of Medicine, Kobe, Japan
Background: Leber hereditary optic neuropathy (LHON) is a maternally inherited disease caused by three missense mutations of mitochondrial (mt) DNA, ie, m 3460 G>A, m 11778 G>A, or m 14484 T>C in the greater portion of LHON. m 11778 G>A mutation is especially observed in >90% of the cases in Japanese families. Although spontaneous remission of visual function infrequently occurs, effective treatment for LHON remains unestablished. Transcorneal electrical stimulation has been shown to be efficacious in individuals with optic neuropathy. However, due to potential risk of corneal damage, repeated treatments are not permissible. In this exploratory study, we will be conducting skin electrical stimulation (SES) as an intervention for patients with LHON having 11778 missense mutation and investigate effectiveness and safety of SES.
Methods: This is a single-arm, prospective, open-label exploratory trial focused on patients with LHON having 11778 missense mutation. Eleven patients will be enrolled and receive six consecutive SES once every 2 weeks up to 10 weeks. The safety of the SES will be monitored with specular microscopy, slit-lamp biomicroscopy, fundus examinations, and the observation of facial skin. The primary outcome measure will be the averaged l ogarithm of minimum angle resolution (logMAR) converted visual acuity 1 week after the last SES. Secondary outcome measures include changes, in logMAR at 4 and 8 weeks after the last SES, such as visual field indices measured using Humphrey visual field and microperimetry-3, the thickness of peripapillary retinal fiber and macular ganglion cell complex, multifocal visual evoked potentials, critical flicker frequency, and color vision.
Discussion: The results of this proposed proof-of-concept feasibility trial will help plan and execute a larger definitive trial to test SES as an effective strategy for LHON and related optic neuropathies and help establish a beneficial treatment for LHON.
Keywords: skin electrical stimulation, Leber hereditary optic neuropathy, logarithm of minimum angle resolution, single-arm study
Background
Leber hereditary optic neuropathy (LHON) is a rare genetic disease, in which retinal ganglion cells undergo cell death caused by mutation of mitochondrial DNA (mtDNA). LHON typically involves both eyes of young males, between the ages of 10 and 30. The ratio of males to females for this condition is a remarkably imbalanced, at 8:1.1,2 The rate of LHON incidence is currently understood to be 3.22 per 100,000 people.3 Its hereditary pattern is clearly maternal, and most families with LHON cases have either one of the three missense mutations of mtDNA of 16,500 base pairs in length: m 3460 G>A, or m 11778 G>A, or m 14484 T>C. In Japanese families, the m11778 G>A mutation occurs in >90% of the cases,4 and unfortunately this particular mutation has the lowest frequency of spontaneous remission.1
Usually, visual acuity subacutely deteriorates in one eye and then the disease affects an opposite eye within a few weeks after onset. The visual acuity of both eyes then eventually decreases to <0.1. The visual field abnormality typically shows a large central scabbed or perforated scotoma. A fundus examination typically reveals a swollen optic disc, with microvasodilation in the early acute phase. Unlike with optic neuritis or ischemic optic neuropathy, a fluorescence angiography examination does not show fluorescence leakage from the optic disc throughout the course. The optic disc finally reaches inflammatory atrophy within a year. The optical coherence tomography (OCT) usually reveals a loss of the papillary macular fiber bundle, and a thinning of both the temporal papillary retinal nerve fiber thickness, as well as the macular retinal ganglion cell layer complex. Light reflection relativity is preserved even after the severe central scotoma appears.
Several clinical trials have been attempted to improve vision in the context of this disease, including the use of idebenone, the derivatives of coenzyme Q105,6 EPI-473,7 and also various forms of gene therapy using the adeno-associated virus vector8; however, the effectiveness of all these approaches is still undetermined and firm evidence in favor of any beneficial treatment of LHON has not yet been established.
Transcorneal electrical stimulation (TES) involves applying a weak current to the eyes after mounting a Burian-Allen type electrode on the cornea. When a retina is electrically stimulated phosphene can be induced in front of the patient. In 2005 Morimoto et al discovered the neuroprotective effects of retinal ganglion cells by electrical stimulation of the cornea, using rat optic nerve transection models.9 The electrical activation of the retina promotes the production of insulin-like growth factor-1 in the retinal Müller cells, resulting in neuroprotective effects, which were later demonstrated to also defend against retinal pigmentosa in rat and rabbit models.
Regarding clinical trials of TES on human ophthalmic diseases, Fujikado et al reported the effectiveness of TES in five cases of old traumatic optic neuropathy and three cases of nonarteritic anterior ischemic optic neuropathy patients.10 Their study revealed how a single TES treatment was able to increase visual acuity and sensitivity of the isoptor, as measured by Goldmann perimetry (GP). Furthermore, in 2011 a randomized controlled trial (RCT) of TES with 24 cases of retinitis pigmentosa (RP) was successfully conducted,11 in which the treatment was applied once a week for 6 weeks. Participants were divided into three groups, according to how the current intensity elicited phosphene: a 0% group (sham stimulation), as well as 66%, and 150% groups, respectively. As a result, the amplitude of the scotopic b wave in the 150% group was significantly increased and the visual field size of GP was significantly enlarged at 2 weeks after the last TES treatment. In another RCTs, the TES approach was administered once a week for 52 weeks, in 52 patients with RP. The amplitudes of the flash b wave and the photopic b wave in the 150% stimulation group were significantly increased compared to the results observed in the sham cohort.12 In another study, Inomata et al reported case studies showing the effectiveness of TES, including two cases of central retinal artery occlusion (CRAO), and one case of branch occlusion of the central retinal artery (BRAO). Administering TES once a month for 3 months increased the logarithm of minimum angle resolution (LogMAR) results by more than two lines in two of three cases and materially decreased visual field defects in all cases.13 About 6 years later, an RCT of TES for CRAO and BRAO cases was conducted in Germany, in which TES was applied for six consecutive weeks in 12 cases of CRAO and one case of BRAO. At 2 and 11 weeks after the last TES session, a significant improvement of scotopic a-waves was observed in the 150% group, although the number of enrolled cases was small.14 Unfortunately, there is at present no report verifying the effectiveness of TES for LHON, although basic and clinical evidence for the neuroprotective effects of TES on intractable retinal and optic nerve degenerative diseases is accumulating. However, when employing TES it is important to recognize that there is a risk of corneal epithelial damage, since the stimulating electrode is placed directly on the cornea, and for this reason, TES is impossible to apply for cases associated with corneal diseases and to repeatedly apply. In contrast, skin electrical stimulation (SES), where a stimulating electrode and a reference electrode are placed on the skin around the eyes to stimulate the eyeball, can elicit phosphene as well as TES; furthermore, it can prevent corneal complications, which allow electrical stimulation to be repeatedly applied to the eyeball more safely than by the TES approach.15 Regarding the adverse effect of SES, it is possible that SES may be involved in contact dermatitis caused by the electrode pad and the pain induced by the activation of sensory nerves unlike TES.
LHON is one of the intractable optic neuropathies for which effective treatments have not yet been established. In the proposed study, we will investigate whether improvement of visual function in patients with LHON can be achieved by consecutive SES treatments more safely than TES, and investigate the safety of SES.
Methods
Study design
This will be an exploratory non-randomized single-arm feasibility trial to determine the safety and effectiveness of SES, which will be designed to enroll 11 patients with LHON. The present study’s protocol received approval from the Institutional Review Board at Kobe University, Japan (approval No. 290080).
Trial registration
University Hospital Medical Information Network-Clinical Trial Registry: UMIN-CTR, registration identifier: UMIN000031057. Registered 20 March 2018,
Informed consent
Written and verbal versions of the participant information sheet and informed consent will be presented to the participants by the attending investigator detailing: the exact nature of the study, the implications and constraints of the protocol and the known side effects and any risks involved in taking part. It will be clearly stated that the participant is free to withdraw from the study at any time for any reason without prejudice to future care, and with no obligation to give the reason for withdrawal. The investigator will not undertake any study specific procedures until valid consent is obtained.
Interventions
The participants will receive six consecutive SES treatments once every 2 weeks for 10 weeks. The instrument to be used in delivering these treatments is produced by the Mayo© Corporation and is a non-approved medical instrument. It enables one to visualize the electric current being applied to the body, while also preventing any excessive electrical stimulation. Two electrode pads will be placed on the skin of the forehead above the eyebrow and below the margin of the lower lid. Participants will notice the phosphene elicited by the SES procedure. The threshold current of phosphene is defined as the level of current such that participants can experience phosphene in the entire visual field. After determining this threshold the participant will be treated by using the following SES protocol: the biphasic square wave, 1 mA amplitude, 10 ms duration, 20 Hz frequency, for a duration of 30 mins. In cases where the SES threshold exceeds 1 mA, the current needed to determine the exact level will be found and adopted into the treatment. The investigator who accompanies the participant will monitor the condition of the participant during SES for 30 mins.
During the course of the investigation, logMAR results will be measured at 1, 4, and 8 weeks after the last SES treatment. Other visual function capabilities will also be measured, as detailed in Figure 1.
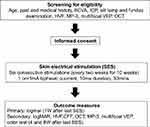 | Figure 1 Trial flow diagram. |
Study population
Participants were selected that fulfill all conditions of both the inclusion and exclusion criteria, as shown in Figure 2. The rationale that we adopted for the inclusion criteria are as follows: (1) the age of onset varies in younger and older patients; (2) in LHON, transient damage to the optic nerve is revealed to be terminated at 8 months or more after onset;16 (3) the spontaneous recovery of this mutation is the lowest; (4) most patients with LHON have a final vision of <0.1.
 | Figure 2 Inclusion and exclusion criteria for the study. |
The rationale for the exclusion criteria we adopted are as follows: (1) smoking is a risk factor for the occurrence and the progression of LHON; (2) safety of SES compared with other electronic medical instruments has not been established; (3) the degree of visual acuity largely varies after the intraocular surgery; (4) other eye diseases that accompany this may make it difficult for us to evaluate the effect of SES because it is impossible to exclude the influence of other eye disease to visual function; (5) the effects of idebenone treatment are excluded; (6) these drugs have the risk of impairing the optic nerve; (7) SES may induce epilepsy; (8) the effects of SES on the formation stage of eyes have not been established; (9) the adhesive pad with electrode put on the skin around forehead and cheeks when delivering electrical stimulation to the patients; (10,11) these are considered to be as the general exculsions.
Sample size determination and power
The averaged logMAR results for 14 cases of LHON with a 11778 mutation that occurred naturally are 1.65±0.49 (mean ± SD), based on preliminary data from our institute. For the feasibility of SES to improve visual acuity of LHON, it is anticipated that these treatments will achieve an improvement of LogMAR results to 1.0. Therefore, assuming that the null hypothesis is that the difference of the averaged LogMAR values before the study compared to 1 week after the final session is 0.65±0.64 (mean ± SD), we will use the paired Student’s t-test with a two-sided significance level of 5% to determine whether the difference between the before and after LogMAR values is zero. In this context, we will need 10 cases to keep the power of the test at 80%. But, considering the possible withdrawal of consent, or that one patient might drop out after participating in the study, the use of 11 cases was considered to be optimal. Because the potential risk of the SES approach to the cornea is low compared with the TES approach, fewer cases are expected to drop out due to SES safety. Even if more than one participant drops out, the statistical power of 70% for eight participants can be maintained. Thus, we set out one participant as a dropout. In the case of more than two dropouts, the efficacy endpoint will be analyzed based on the data obtained from nine cases who completed the protocol according to each protocol set and the safety endpoint will be analyzed based on the data obtained from 11 cases that include dropouts by the full analysis set.
Outcome measures
There is no prior empirical data about the possible efficacy of using SES to treat any optic neuropathy. However, the safety and effectiveness of TES, a technique similar to SES, have been proven in several clinical trials.10,17 Other previous study revealed that the SES of an eyelid with a ring-shaped electrode could unharmfully elicit phosphene, and when the duration of an SES session is long (>2 ms), phosphene is raised with less current intensity that is required for the somatosensory nerve.15 Based on these previous studies we have designed the proposed investigation as a proof-of-concept effort. The main goals of the study will be to determine the potential efficacy and safety of SES. Table 1 shows all the planned assessments and procedures that will be performed before and after the SES treatments. The primary analysis is scheduled for 1 week after the last SES treatment. The averaged values of the LogMAR results at 1 week after the last SES session will be estimated and be statistically evaluated compared with those before the treatments. The time points of the secondary outcome measures will be at 4 and 8 weeks after the last SES treatments. To evaluate the effectiveness of the SES approach, the logMAR, Humphrey visual field (HVF), critical fusion frequency (CFF), and microperimetry-3 (MP-3), as well as the multifocal VEP will be performed to confrim whether functional improvements are materially observable. The slit-lamp, fundus, specular microscope and OCT examinations will also be performed to confirm the safety and adverse effects of the SES treatments.
 | Table 1 Schedule for the study |
Statistical analysis
The findings from this study will be published in a peer-reviewed journal, and the data will also be included in subsequent larger trials if appropriate. Baseline data to be collected and summarized will include: participant gender and age, logMAR converted from decimal visual acuity, mean deviation of Humphrey visual field Swedish Interactive Thresholding Algorithm program 30–2 (Carl Zeiss Meditec, Inc., Dublin, CA, USA) using a size V visual stimuli, the average of CFF test results over three trials, the amplitude of the multifocal VEP (VERIS 5.2 software; Electro-Diagnostic Imaging, Inc., Redwood City, CA, USA), the thickness of the circumpapillary retinal nerve fiber layer and ganglion cell complex (GCC) obtained by spectral-domain OCT (3D-OCT, Topcon Corporation, Tokyo, Japan), the mean deviation of MP-3 (Nidek Co., Ltd, Aichi, Japan) in the macula area and cell densities of corneal endothelial cells obtained by specular microscope (Konan Medical, Nishinomiya, Japan), as well as the quality of color vision, obtained by using standard pseudoisochromatic plates, part 2 for acquired color vision defects (SPP-2; JFC Sales Plan Co. Ltd., Tokyo, Japan).
Statistical analyses were performed using Med Calc (version 18.2.1, MedCalc Software, Mariakerte, Belgium) and IBM SPSS statistics (SPSS, Chicago, IL, USA).
Continuous baseline data will be presented as means and SDs for normally distributed variables, but as medians and interquartile ranges (IQR) for non-normally distributed variables, and as frequencies and percentages for categorical baseline variables. For the primary outcome, the averaged logMAR before and 1 week after last SES treatment will be calculated and statistically analyzed using the paired Student’s t-test. Similarly, averaged logMAR 4 and 8 weeks after the last SES as secondary endpoints will be statistically analyzed compared with the last SES using the paired Student’s t-test. Concerning other secondary endpoints, it will be considered of value in the following cases, namely, the mean deviation, pattern deviation, and the foveal threshold of HVF (center 30–2, stimulus size III or V) statistically improved in 1, 4, and 8 weeks after the last SES compares with before SES using paired Student’s t-test or the total number of 1% or 5% of probability plot with pattern deviation increased >20%. CFF increases >20% in 1, 4, and 8 weeks after the last SES compared with before SES. Concerning color test with SPP-2, color abnormality detectable before SES disappeared at 8 weeks after the last SES. The thickness of peripapillary retinal nerve fiber and macular GCC detected by OCT decreased by <20% compared with that of the last SES. Concerning MP-3, the number of measurement points where the actual measurement values increase >5 dB exceeds compared with those before and 8 weeks after the last SES. Concerning multifocal VEP, the amplitude of VEP at 8 weeks after the last SES increased compared with that in the last SES.
All participants will receive the same treatment and we will only analyze the data from the participants who actually receive all consecutive SES treatments. Changes from the baseline in logMAR, MD of HVF and MP-3, averaged CFF, color vision, and amplitude of multifocal VEP will be summarized as means and SDs if normally distributed, or as medians and IQRs if the data do not demonstrate normality, and as frequencies and percentages for categorical baseline variables.
Confidentiality, ethics and dissemination
The present study will be conducted in accordance with the current Declaration of Helsinki. All patients will be informed that they can withdraw from the study at any time. The authors are committed to publishing the study results as widely as possible in peer-reviewed journals, and to ensuring that appropriate recognition is provided to everyone who will have worked on this study.
Monitoring
There is no Data and Safety Monitoring Committee for the present study. The responsible doctor for monitoring the safety and progress of the trial is appointed and will meet approximately twice per year.
Study management and patient safety
A trial management group involving the principal investigators (TK, MN, KU), co-investigators (MS, SM, YN), and investigator responsible for monitoring (WM) will review, implement, and supervise all aspects of this trial. The monitoring doctor, under the supervision of the principal investigators (TK, MN), will be responsible for receiving, processing, editing, and storing all the data. All investigational product supplies in the study will be stored in a secure, safe place, under the responsibility of the principal investigators. The statistical analysis will be conducted by the investigators (led by KU).
To ascertain the patient safety at every visit throughout the trial, slit-lamp biomicroscopy, fundus examination, and Goldmann applanation to measure intraocular pressure will be performed to verify whether the intraocular inflammatory changes or other ophthalmic adverse effects occur or not. On days 1, 3, 6, and 10 of the visits, specular microscopy will be performed to verify whether the corneal endothelial cells decrease in number or not. Face photographs will be taken to observe whether dermatitis occurs where the electrode pad has been attached through the period of treatment from 2 to 7 of the visits. In the present study, the evident decrease in the density of the corneal endothelial cells, the marked progression of cataract, induction of intraocular inflammation, and dermatitis where the electrode pad is attached has been considered as adverse events.
Discussion
The evidence on the efficacy for LHON has not been established, although some of clinical trials have successfully revealed improvement in visual function.5,6,18 Presently, there is a need for innovative treatment methods to save LHON patients, who become blind during adolescence. The most striking characteristics of SES are the ones that make it minimally invasive and easily feasible for repeated application in patients. According to this protocol, SES is beneficial for LHON patients and holds the potential to be a novel and effective approach to the treatment of not only LHON cases with a 11778 missense mutation but also other optic nerve diseases, including anterior ischemic optic neuropathy, traumatic optic neuropathy, and retinal pigmentosa. It has already been revealed that TES has the potential to improve such intractable optic and retinal diseases based on previous studies. Alternatively, positive results also could have major implications for the treatment of LHON with other missense mutations, as well as for LHON patients in the acute phase. This treatment would be easily available to a family ophthalmologist or employed as daily self-stimulation after a modest training regime that met a constant standard. In animal experiments consecutive daily TES promoted RGC survival much better than a single TES treatment in a rat optic nerve crush model.19 This degree of effectiveness might also be relevant to other hereditary optic neuropathies, such as autosomal dominant optic atrophy.
Limitations of the present study are that it is unclear whether the effectiveness of SES depends on the intensity of the electrical current. Also, the present study does not include a control group because of the rarity of LHON. If successful, this approach will be tested in multicenter randomized trials that compare the group receiving SES treatments with other groups experiencing sham stimulation.
Abbreviation list
LogMAR, logarithm of minimum angle resolution; SES, skin electrical stimulation; BCVA, best corrected visual acuity; mtDNA, mitochondrial DNA; LHON, Leber hereditary optic neuropathy; TES, Transcorneal electrical stimulation; HVF, Humphrey visual field; CFF, critical fusion frequency; MP-3, microperimetry-3; cpRNFL, circumpapillary retinal nerve fiber layer; GCC, ganglion cell complex; OCT, Optical coherence tomography.
Availability of data and material
Data from the trial will be made available for the institutional policies.
Consent for publication
The consent for publication will be obtained from eligible patients before they participate in the trial.
Ethics approval and consent to participate
The present study’s protocol received approval from the Institutional Review Board at Kobe University, Japan (approval No. 290080) and will be conducted in accordance with the current Declaration of Helsinki. Eligible patients will undergo informed consent before participating in the trial.
Author contributions
TK and MN contributed to the study concept. KU contributed to the analytic plan. SM and TK drafted the first paper and wrote revision and the final manuscript. All authors made substantial contributions to the concept and design of the trial. All authors contributed to data analysis, drafting and revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Hotta Y, Fujiki K, Hayakawa M, et al. Clinical features of Japanese Leber‘s hereditary optic neuropathy with 11778 mutation of mitochondrial DNA. Jpn J Ophthalmol. 1995;39(1):96–108.
2. Ueda K, Morizane Y, Shiraga F, et al. Nationwide epidemiological survey of Leber hereditary optic neuropathy in Japan. J Epidemiol. 2017;27(9):447–450. doi:10.1016/j.je.2017.02.001
3. Man PY, Griffiths PG, Brown DT, Howell N, Turnbull DM, Chinnery PF. The epidemiology of Leber hereditary optic neuropathy in the North East of England. Am J Hum Genet. 2003;72(2):333–339. doi:10.1086/346066
4. Mackey DA, Oostra RJ, Rosenberg T, et al. Primary pathogenic mtDNA mutations in multigeneration pedigrees with Leber hereditary optic neuropathy. Am J Hum Genet. 1996;59(2):481–485.
5. Klopstock T, Metz G, Yu-Wai-Man P, et al. Persistence of the treatment effect of idebenone in Leber‘s hereditary optic neuropathy. Brain. 2013;136(Pt 2):e230. doi:10.1093/brain/aws279
6. Klopstock T, Yu-Wai-Man P, Dimitriadis K, et al. A randomized placebo-controlled trial of idebenone in Leber‘s hereditary optic neuropathy. Brain. 2011;134(Pt 9):2677–2686. doi:10.1093/brain/awr170
7. Sadun AA, Chicani CF, Ross-Cisneros FN, et al. Effect of EPI-743 on the clinical course of the mitochondrial disease Leber hereditary optic neuropathy. Arch Neurol. 2012;69(3):331–338. doi:10.1001/archneurol.2011.2972
8. Guy J, Feuer WJ, Davis JL, et al. Gene therapy for Leber hereditary optic neuropathy: low- and medium-dose visual results. Ophthalmology. 2017;124(11):1621–1634. doi:10.1016/j.ophtha.2017.05.016
9. Morimoto T, Miyoshi T, Matsuda S, Tano Y, Fujikado T, Fukuda Y. Transcorneal electrical stimulation rescues axotomized retinal ganglion cells by activating endogenous retinal IGF-1 system. Invest Ophthalmol Vis Sci. 2005;46(6):2147–2155. doi:10.1167/iovs.04-1339
10. Fujikado T, Morimoto T, Matsushita K, Shimojo H, Okawa Y, Tano Y. Effect of transcorneal electrical stimulation in patients with nonarteritic ischemic optic neuropathy or traumatic optic neuropathy. Jpn J Ophthalmol. 2006;50(3):266–273. doi:10.1007/s10384-005-0304-y
11. Schatz A, Rock T, Naycheva L, et al. Transcorneal electrical stimulation for patients with retinitis pigmentosa: a prospective, randomized, sham-controlled exploratory study. Invest Ophthalmol Vis Sci. 2011;52(7):4485–4496. doi:10.1167/iovs.10-6932
12. Schatz A, Pach J, Gosheva M, et al. Transcorneal electrical stimulation for patients with retinitis pigmentosa: a prospective, randomized, sham-controlled follow-up study over 1 year. Invest Ophthalmol Vis Sci. 2017;58(1):257–269. doi:10.1167/iovs.16-19906
13. Inomata K, Shinoda K, Ohde H, et al. Transcorneal electrical stimulation of retina to treat longstanding retinal artery occlusion. Graefes Arch Clin Exp Ophthalmol. 2007;245(12):1773–1780. doi:10.1007/s00417-007-0610-9
14. Naycheva L, Schatz A, Willmann G, et al. Transcorneal electrical stimulation in patients with retinal artery occlusion: a prospective, randomized, sham-controlled pilot study. Ophthalmol Ther. 2013;2(1):25–39. doi:10.1007/s40123-013-0012-5
15. Delbeke J, Pins D, Michaux G, Wanet-Defalque MC, Parrini S, Veraart C. Electrical stimulation of anterior visual pathways in retinitis pigmentosa. Invest Ophthalmol Vis Sci. 2001;42(1):291–297.
16. Hwang TJ, Karanjia R, Moraes-Filho MN, et al. Natural history of conversion of Leber‘s hereditary optic neuropathy: a prospective case series. Ophthalmology. 2017. doi:10.1016/j.ophtha.2017.01.002
17. Naycheva L, Schatz A, Rock T, et al. Phosphene thresholds elicited by transcorneal electrical stimulation in healthy subjects and patients with retinal diseases. Invest Ophthalmol Vis Sci. 2012;53(12):7440–7448. doi:10.1167/iovs.12-9612
18. Koilkonda R, Yu H, Talla V, et al. LHON gene therapy vector prevents visual loss and optic neuropathy induced by G11778A mutant mitochondrial DNA: biodistribution and toxicology profile. Invest Ophthalmol Vis Sci. 2014;55(12):7739–7753. doi:10.1167/iovs.14-15388
19. Tagami Y, Kurimoto T, Miyoshi T, Morimoto T, Sawai H, Mimura O. Axonal regeneration induced by repetitive electrical stimulation of crushed optic nerve in adult rats. Jpn J Ophthalmol. 2009;53(3):257–266. doi:10.1007/s10384-009-0657-8
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
