Back to Journals » Therapeutics and Clinical Risk Management » Volume 16
A Randomized Controlled Trial Comparing Analgesic Efficacies of an Ultrasound-Guided Approach with and without a Combined Pressure Measurement Technique for Thoracic Paravertebral Blocks After Open Thoracotomy
Authors Choi EK , Kim J , Park SJ
Received 22 May 2020
Accepted for publication 15 July 2020
Published 6 August 2020 Volume 2020:16 Pages 727—734
DOI https://doi.org/10.2147/TCRM.S263353
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Garry Walsh
Eun Kyung Choi, Ji-il Kim, Sang-Jin Park
Department of Anesthesiology and Pain Medicine, Yeungnam University College of Medicine, Daegu, Republic of Korea
Correspondence: Sang-Jin Park
Department of Anesthesiology and Pain Medicine, Yeungnam University College of Medicine, Hyeonchung-ro, Nam-gu, Daegu, Republic of Korea
Tel +82-53-620-3366
Fax +82-53-626-5275
Email [email protected]
Purpose: Ultrasound-guided thoracic paravertebral block (TPVB) is an established means for providing postoperative analgesia in thoracic surgery. However, there are conflicting results regarding the efficacy of post-thoracotomy pain management of ultrasound-guided TPVB when compared with that using traditional landmark approach. We therefore conducted a comparative study to evaluate the analgesic efficacy of TPVB when pressure measurement during needle advancement is combined with an ultrasound-guided approach.
Patients and Methods: The patients scheduled for lobectomy through thoracotomy were randomly allocated to receive either the ultrasound-guided approach only group (U group) or the ultrasound-guided approach combined with pressure measurement group (UP group) (n = 36 per group). Before thoracic muscle closure, 0.375% ropivacaine (20 mL) was administered as a bolus, followed by a continuous infusion of 0.2% ropivacaine (0.1 mL/kg/hr) in both groups. Postoperative pain was assessed using the visual analogue scale (VAS) pain score while resting and coughing. Local anesthetics and pethidine usage and sensory block area were also evaluated.
Results: The UP group showed significantly lower VAS scores, local anesthetics and pethidine usage, and a wider sensory block area than the U group.
Conclusion: A combined technique with ultrasound guidance and pressure measurement provided a superior analgesic effect over that of an ultrasound-guided approach alone for the management of post-thoracotomy pain.
Keywords: postoperative analgesia, pressure measurement, thoracic surgery, ultrasound-guided, visual analogue scale
Introduction
Thoracotomy, among the most painful surgical procedures, causes severe chest pain and impaired respiratory performance in the post-surgical period. Thoracic paravertebral block (TPVB) is an established method for providing postoperative analgesia for thoracic procedures. This method has been shown to provide pain control comparable to that obtained using epidural analgesia but with fewer side effects.1–3 TPVB can be performed using multiple methods including the classic landmark-based approach, neuro-stimulation, and ultrasound-guided approaches. Recent reports have shown that an ultrasound-guided approach is a preferred method due to easy visualization and accurate puncture of the paravertebral space (PVS). However, the sonographic window is limited and often precluding optimal paravertebral space image acquisition while the needle is inserted.4,5 An ultrasound-guided paravertebral approach in human cadavers showed that 6% of needle tips were not at the target5 and catheter insertion was repeated in 20% of the instances.6 A previous clinical study also revealed that the application of ultrasound-guided paravertebral block was unsuccessful in 5–10% of cases.7 This failure rate is similar to the rate observed in paravertebral block using the classic landmark technique.8,9 A substantial improvement in reliability is needed, and thus, we devised the pressure measurement method during needle advancement of TPVB. This technique was first demonstrated by Richardson et al during TPVB using the classic landmark method.10 They showed that the pressure suddenly decreases when the superior costo-transverse ligament is breached by the needle due to the respiratory cycle. Therefore, pressure measurement during needle advancement can provide an objective method for correct paravertebral space localization.
In this study, we investigated whether postoperative analgesia for thoracotomies could be improved when pressure measurement during needle advancement is combined with TPVB using an ultrasound-guided-approach.
Patients and Methods
This study was approved by the Yeungnam University Hospital Institutional Review Board (YUMC 2018–09-023) and was registered in a clinical trial registry (NCT03868917) before recruitment of the first subject. Patients who were scheduled to undergo thoracotomy for lung lobectomy were invited to participate and provided written informed consent. Participants receiving American Society of Anesthesiologists Physical Status Classification I–II were randomly assigned to either the ultrasound-guided approach-only group (U group) or the ultrasound-guided approach combined with pressure measurement group (UP group) for postoperative analgesia. Group assignment was performed through computer-generated randomization. The exclusion criteria were as follows: inability to provide adequate informed consent, unstable vertebral and transverse process fractures, any chronic painful conditions or preoperative opioid use, coagulation abnormalities, allergy to any of the drugs used in the study protocol, infection at the injection site, younger than 18 years or older than 85 years, presence of comorbid conditions such as sepsis, unstable angina, congestive heart failure, or patient refusal.
Standard monitoring including noninvasive arterial blood pressure (NIBP), electrocardiogram (EKG), pulse oximetry, and bispectral index (BIS) was performed. General anesthesia was induced with propofol and remifentanil of target control infusion (TCI). Rocuronium was used to facilitate tracheal intubation. All patients were intubated with a double-lumen endobronchial tube and concentrations of remifentanil and propofol were adjusted to maintain systolic blood pressure changes of >20% from the initial values and target BIS values of 40–60. When systolic blood pressure fell below 20% of baseline, 4mg of ephedrine was injected. Patients were placed in the lateral decubitus position. Before thoracotomy, thoracoscopic intrathoracic inspection was performed in all patients according to the following procedure. First, a 5-mm port for the camera was inserted in the 7th intercostal space at the mid-axillary line. After confirming that there was no evidence of pleural dissemination, a thoracotomy was created in the 5th or 6th intercostal space using a posterolateral axillary approach. After completion of the thoracotomy, a chest tube was inserted in the 7th intercostal space through the camera port.
TPVB was performed in the intervertebral space corresponding to the incision using an ultrasound machine (Hitachi healthcare, Prosound alpha 7, OH, USA) after patients were placed in the lateral decubitus position, but before initiation of the thoracotomy. In the U group, the needle entry site was 2.5 cm lateral to the midpoint of the spinous process of the corresponding thoracic vertebrae. A low-frequency 2–5 MHz curved array transducer was placed longitudinally to visualize the transverse processes, costotransverse ligament, and pleura. An 18-gauge Tuohy needle (B. Braun Medical, Inc., Perifix Continuous Epidural Anesthesia Set, Penang, Malaysia) was placed in the paravertebral space puncturing the costotransverse ligament using an in-plane approach under direct ultrasound guidance. Normal saline (10 mL) was injected to prepare the space for catheter insertion, and the catheter was inserted to a depth of 3 cm beyond the tip of the needle. In the UP group, the paravertebral catheter was inserted in the manner that was previously described, and pressure was measured during needle advancement. A pressure transducer was connected through a saline-filled, non-compliant tube and a three-way tap to the proximal end of the Tuohy needle. Pressure was measured when the needle was advanced through the erector spinae muscle, and through the superior costo-transverse ligament into the paravertebral space. Correct needle placement in the paravertebral space was confirmed by the decrease in the pressure. In both groups, the block was activated after the thoracic muscles were closed. Immediately after administering a bolus dose of 0.375% ropivacaine (20 mL), continuous infusion of 0.2% ropivacaine was started at a rate of 0.1 mL/kg/hr in both groups. The bolus infusion volume was 3 mL and the lockout time was set to 30 minutes. In the postanesthesia care unit, the patients were instructed about the use of the visual analogue scale (VAS) pain score and PCA infuser. The patients were instructed to press the PCA button for a bolus injection if their self-reported pain score exceeded 4 at rest. If pain did not subside 30 minutes after the bolus injection, pethidine 12.5 mg was administered intravenously with the dose limited to 75 mg/day. Local analgesic infusion continued until 120 hours after surgery.
An anesthesiologist blinded to the group assignment of each patient evaluated the patients. Postoperative pain was assessed using VAS pain score at 1, 2, 4, 8, 12, 24, 36, 48, 72, 96, and 120 hours after surgery while resting and coughing. The VAS consisted of a 100-mm line (0 mm, no pain; 100 mm, worst pain imaginable). Complications were checked concurrently. The complications included dizziness, sedation, nausea, vomiting, hypotension, hypoxia, epidural spread of the block and urinary retention. Urinary retention was classified as a bladder volume greater than 400 mL with the inability to void spontaneously. Single catheterization was performed. Bladder ultrasonography was undertaken for measuring bladder volume. Hypotension was an over 30% decrease from the mean arterial pressure just before arrival in the operating room. Local anesthetics usage data were obtained from PCA pump infusion records in the 120-hour period after surgery. Pethidine usage data were obtained from medical records. The patient’s sensory level was checked using a pinprick test at 2, 4, and 8 hours after surgery.
The primary outcome measure was the VAS pain score at rest 2 hours post-surgery. A minimum sample size of 32 patients per group was required to determine a significant difference between the U and UP groups with a probability of type I error of 0.05 and a power of 80% based on our preliminary study. Estimating a dropout rate of 10%, we finally included 72 patients per group. Statistical analyses were performed with SPSS 21.0 (IBM, Armonk, NY, USA). The data are presented as numerical values, the mean (SD), or the median (range) where appropriate. Continuous variables were analyzed with an independent sample t-test. Ordinal and non-normally distributed variables were analyzed using a Wilcoxon rank-sum test. Nominal variables were analyzed using a chi-square test or Fisher’s exact test as applicable. P-values <0.05 were considered to statistical significance.
Results
Seventy-two patients scheduled for thoracotomy for lung lobectomy consented to participate in the study and were randomized into either the U group or the UP group (n=36 for each group). Four patients in the U group were discontinued for the following reasons: disconnection of the block catheter (3 patients), and change in the surgical procedure (1 patient). In the UP group, four patients were excluded from the study for the following reasons: disconnection of the block catheter (2 patients), postoperative occurrence of delirium (1 patient), and ICU transfer accompanied by sedation (1 patient). Figure 1 shows the CONSORT flow diagram for patients enrolled in the study.
We compared the baseline characteristics of the patients, duration of surgery and anesthesia, total dose of remifentanil during anesthesia, length of skin incision, and time required for catheter insertion and found no significant differences between the groups (Table 1). The total consumption of PCA and additional analgesia (pethidine) in the UP group were lower than those in the U group (Table 1). In the sensory threshold tests, patients in the UP group exhibited significantly wider dermatomal distributions than those in the U group at both 2 and 8 h after surgery (Table 1). The pain scores (VAS) after surgery are shown in Tables 2 and 3. The mean VAS scores in the UP group were significantly lower than those in the U group at 1, 2, 4, 8, and 12 hours during resting and at 1, 2, and 4 hours during coughing. There was no significant difference between two groups in the incidence of dizziness, sedation, nausea, vomiting, hypotension, hypoxia, epidural spread of the block and urinary retention (Table 4).
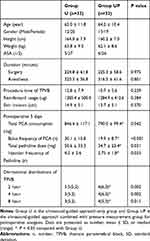 |
Table 1 Demographic and Baseline Characteristics |
 |
Table 2 Visual Analog Scale Scores at Rest During the First 120 Hours Postoperatively |
 |
Table 3 Visual Analog Scale Scores at Cough During the First 120 Hours Postoperatively |
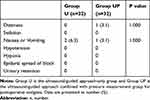 |
Table 4 Side Effects |
Discussion
This study showed that VAS score was lower in the UP group than in the U group during 12 and 4 hours postoperatively at rest and coughing, respectively. Further, the total usage of local anesthetics (PCA pump and additional pethidine injections) over 120 hours postoperatively was higher in the U group than in the UP group. Sensory distribution of TPVB was wider in the pressure measurement group at 2, 4, and 8 hours post-operation.
Postoperative pain after thoracotomy is very severe, and thus, adequate analgesia is required. If not, it can cause an increased stress response in patients and may further compromise pulmonary functions. Among the several postoperative care such as thoracic epidural analgesia, nerve block, systemic opioid, epidural analgesia has been most widely used to control postoperative pain after thoracotomy. In addition to effective pain relief, thoracic epidural analgesia may provide favorable homeostatic conditions including endocrine and immune functions.11 Previous studies have suggested that epidural analgesia was superior to TPVB in the postoperative period,12,13 although the extent of sensory blockade was comparable. However, sympatholytic complications such as hypotension, epidural hematoma, nerve injury, and even pulmonary compromise relating to respiratory muscle weakness can be significant drawbacks when it is applied. TPVB may avert these potentially serious problems or may be associated with fewer complications due to a unilateral segmental block.
Paravertebral regional anesthesia blocks the somatic and sympathetic nervous systems by injecting a local anesthetic (LA) into the PVS where the thoracic nerve and its branches are located after exiting the intervertebral foramen. PVS is medially connected to the epidural space through the intervertebral foramen; therefore, LA into this space causes an analgesic equivalent to a unilateral epidural block. Given the advent and development of US guidance for TPVB, the ease and safety of this approach have contributed to increased clinical relevance. Under US guidance, more exact anatomical landmarks can be verified and accurate needle positioning and LA spread can be monitored in real time. Thus, TPVB could be a safer method than epidural analgesia with a lower risk of injury to the thoracic medulla. Moreover, unlike epidural analgesia, TPVB produces a primarily unilateral sensory block, which is preferable for unilateral surgery such as thoracotomy and breast surgery. Biswas et al have shown the analgesic efficacy of TPVB13 and in a previous meta-analysis, the analgesic effects of TPVB were comparable to those obtained using thoracic epidural analgesia, with fewer complications.14 Therefore, TPVB is a reliable block and may be a proper alternative in patients undergoing thoracotomy, especially in cases in which a unilateral sensory block is required.
Although an ultrasound-guided approach produces real visualization of anatomical landmarks and the inserted needle, there are conflicting findings related to the efficacy of post-thoracotomy pain management of TPVB for lung surgery in comparison with those of thoracic epidural analgesia.15,16 The studies in human cadavers and human patients also showed that the application of ultrasound-guided TPVB was unsuccessful in 4–7% of cases.4–7 A potential explanation for the unsuccessful results is the technical difficulty, even under ultrasound guidance, that can occur with potential image loss when the needle tip is advanced to the adjacent transverse process.4,5 Therefore, we tried to improve the accurate placement of the needle tip by combining a pressure measurement method with an ultrasound-guidance approach while also intending to assess the analgesic efficacy through this more delicate technique. Richardson et al suggested a pressure measurement method during needle advancement for TPVB, which improved the success rate of thoracic paravertebral analgesia.10 In our study, TPVB with a pressure measurement technique provided superior post-thoracotomy pain relief for lung surgery in comparison with that with only an ultrasound-guided technique. Additionally, this can be detected clinically by sensory testing. The sensory distribution was wider when the pressure measurement method was applied with ultrasound visualization. It may be suggested that the combination of pressure measurements with ultrasound-guided produces the high probability of delicate and correct placement of the needle tip during TPVB. On the other hand, a discrepancy in the analgesic efficacy between both situations (at rest and during coughing) may explain why pain during coughing is a more significant parameter for analgesic efficacy, particularly after thoracotomy.17
Using the TPVB technique, needle tip was positioned immediately after puncturing the superior costotransverse ligament and then catheter was inserted into the thoracic PVS. Some studies reported that endothoracic fascia divides the thoracic PVS into two compartments; anterior extrapleural and posterior subendothoracic compartment.18 According to Naja et al study, injection of local anesthetics with dye into anterior compartment allowed a wider spread of longitudinally than that into posterior compartment.19 However, a recent evaluation in human cadavers demonstrated that internal subdivision of thoracic PVS was not observed and the endothoracic fascia was indistinguishable from the parietal pleural.20 In the present study, we could not identify the endothoracic fascia although using an ultrasound-guided method. However, irrespective of catheter tip location in any compartment, 4 dermatomes was achieved in the TPVB used a pressure measurement technique, which is consistent with previous finding of 4.2 dermatomes with meaningful analgesia via TPVB when the catheter was inserted into the posterior subendothoracic compartment under US guidance.21 Four dermatomes sensory block is enough to cover all incisions occurred during thoracotomy. Moreover, in the US technique, we used parasagittal in-plane approach. Some studies reported that this approach is reproducible with minimal complications and less likely catheter migration into the intervertebral foramen compared to transverse approach which has an increased incidence of epidural spread.22,23
Based on published data, there was no optimal concentration and dose of local anesthetics for bolus and/or continuous TPVB. Commonly reported dosage in adults is 0.25–0.5% bupivacaine, a 15–20 mL bolus, 1–2% lidocaine, a 15–20 mL bolus dose, and 0.375–0.5% ropivacaine, a 10–20 mL bolus dose postoperatively.24 By Kotze et al systemic review from 25 randomized trials, higher dose regimens and continuous infusion of local anesthetics are predictive of better analgesic effect.25 In the present study, we used 0.375% ropivacaine (20 mL) as a bolus, followed by 0.2% ropivacaine 0.1 mL/kg/hr. Our results of comparable analgesic effect (unilateral somatic block over 4 with range, 3–6) and very few side effects suggest that this dosing regimen would be adequate as post-thoracotomy analgesia.
A limitation of this study is that, even with the aid of pressure measurements with ultrasound visualization, correct location of the catheter tip may not be certain. The use of a paravertebral catheter during TPVB is often described as the limited extent of the block within only a few segments,26,27 most probably due to catheter misplacement; embedment into the muscle, mediastinal, epidural space, or through the pleural, although there is a low probability. However, the accurate insertion of the needle into the PVS through more objective and feasible methods such as ultrasound combined with pressure measurements may be helpful for precise catheter placement. Another limitation is related to small sample size. Although we acquired the number of enrolled patients from power analysis through pilot study, additional data are required to validating our results.
Conclusions
We showed that a combination technique of ultrasound guidance and pressure measurements provided a superior analgesic effect over that with an ultrasound-guided approach alone. It may be suggested that a more delicate placement of the needle during TPVB is possible using the above-described technique.
Data Sharing Statement
All datasets analyzed for this study are included in the article. The figures and tables will be available by contacting the corresponding author. The data can be used permanently after the article is published.
Ethics and Consent
This trial was conducted in accordance with the Declaration of Helsinki. This study was approved by the Yeungnam University Hospital Institutional Review Board (YUMC 2018-09-023) and was registered in a clinical trial registry (NCT03868917) before recruitment of the first subject. All the study participants provided written informed consent for the study.
Acknowledgments
This work was supported by the 2018 Yeungnam University Research Grant. We would like to thank Editage for English language editing.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Kosiński S, Fryźlewicz E, Wiłkojć M, Ćmiel A, Zieliński M. Comparison of continuous epidural block and continuous paravertebral block in postoperative analgaesia after video-assisted thoracoscopic surgery lobectomy: a randomised, non-inferiority trial. Anaesthesiol Intensive Ther. 2016;48(5):280–287. doi:10.5603/AIT.2016.0059
2. Andreae MH, Andreae DA. Regional anaesthesia to prevent chronic pain after surgery: a Cochrane systematic review and meta-analysis. Br J Anaesth. 2013;111(5):711–720. doi:10.1093/bja/aet213
3. Casati A, Alessandrini P, Nuzzi M, et al. A prospective, randomized, blinded comparison between continuous thoracic paravertebral and epidural infusion of 0.2% ropivacaine after lung resection surgery. Eur J Anaesthesiol. 2006;23(12):999–1004. doi:10.1017/S0265021506001104
4. Cowie B, McGlade D, Ivanusic J, Barrington MJ. Ultrasound-guided thoracic paravertebral blockade: a cadaveric study. Anesth Analg. 2010;110(6):1735–1739. doi:10.1213/ANE.0b013e3181dd58b0
5. Luyet C, Herrmann G, Ross S, et al. Ultrasound-guided thoracic paravertebral puncture and placement of catheters in human cadavers: where do catheters go? Br J Anaesth. 2011;106(2):246–254. doi:10.1093/bja/aeq309
6. Luyet C, Eichenberger U, Greif R, Vogt A, Szücs Farkas Z, Moriggl B. Ultrasound-guided paravertebral puncture and placement of catheters in human cadavers: an imaging study. Br J Anaesth. 2009;102(4):534–539. doi:10.1093/bja/aep015
7. Župčić M, Graf Župčić S, Duzel V, et al. A combination of levobupivacaine and lidocaine for paravertebral block in breast cancer patients undergoing quadrantectomy causes greater hemodynamic oscillations than levobupivacaine alone. Croat Med J. 2017;58(4):270–280. doi:10.3325/cmj.2017.58.270
8. Coveney E, Weltz CR, Greengrass R, et al. Use of paravertebral block anesthesia in the surgical management of breast cancer: experience in 156 cases. Ann Surg. 1998;227(4):496–501. doi:10.1097/00000658-199804000-00008
9. Pusch F, Freitag H, Weinstabl C, Obwegeser R, Huber E, Wildling E. Single-injection paravertebral block compared to general anaesthesia in breast surgery. Acta Anaesthesiol Scand. 1999;43(7):770–774. doi:10.1034/j.1399-6576.1999.430714.x
10. Richardson J, Cheema SP, Hawkins J, Sabanathan S. Thoracic paravertebral space location. A new method using pressure measurement. Anaesthesia. 1996;51(2):137–139. doi:10.1111/j.1365-2044.1996.tb07700.x
11. Liuboshevskiĭ PA, Artamonova NI, Ovechkin AM. [Haemostasis disturbances as the component of the surgical stress-response and possibilities of their correction]. Anesteziol Reanimatol. 2012;3:44–48. Russian.
12. Tamura T, Mori S, Mori A, et al. A randomized controlled trial comparing paravertebral block via the surgical field with thoracic epidural block using ropivacaine for post-thoracotomy pain relief. J Anesth. 2017;31(2):263–270. doi:10.1007/s00540-017-2307-5
13. Biswas S, Verma R, Bhatia VK, Chaudhary AK, Chandra G, Prakash R. Comparison between thoracic epidural block and thoracic paravertebral block for post thoracotomy pain relief. J Clin Diagn Res. 2016;10(9):UC08–UC12. doi:10.7860/JCDR/2016/19159.8489
14. Davies RG, Myles PS, Graham JM. A comparison of the analgesic efficacy and side-effects of paravertebral vs epidural blockade for thoracotomy – a systematic review and meta-analysis of randomized trials. Br J Anaesth. 2006;96(4):418–426. doi:10.1093/bja/ael020
15. Scarci M, Joshi A, Attia R. In patients undergoing thoracic surgery is paravertebral block as effective as epidural analgesia for pain management? Interact Cardiovasc Thorac Surg. 2010;10(1):92–96. doi:10.1510/icvts.2009.221127
16. Helms O, Mariano J, Hentz JG, et al. Intra-operative paravertebral block for postoperative analgesia in thoracotomy patients: a randomized, double-blind, placebo-controlled study. Eur J Cardiothorac Surg. 2011;40(4):902–906. doi:10.1016/j.ejcts.2011.01.067
17. Niemi G, Breivik H. The minimally effective concentration of adrenaline in a low-concentration thoracic epidural analgesic infusion of bupivacaine, fentanyl and adrenaline after major surgery. A randomized, double-blind, dose-finding study. Acta Anaesthesiol Scand. 2003;47(4):439–450. doi:10.1034/j.1399-6576.2003.00077.x
18. Stopar Pintaric T, Veranic P, Hadzic A, Karmakar M, Cvetko E. Electron-microscopic imaging of endothoracic fascia in the thoracic paravertebral space in rats. Reg Anesth Pain Med. 2012;37(2):215–218. doi:10.1097/AAP.0b013e31824451cb
19. Naja MZ, Ziade MF, El Rajab M, El Tayara K, Lönnqvist PA. Varying anatomical injection points within the thoracic paravertebral space: effect on spread of solution and nerve blockade. Anaesthesia. 2004;59(5):459–463. doi:10.1111/j.1365-2044.2004.03705.x
20. Bouman EAC, Sieben JM, Balthasar AJR, et al. Boundaries of the thoracic paravertebral space: potential risks and benefits of the thoracic paravertebral block from an anatomical perspective. Surg Radiol Anatomy. 2017;39(10):1117–1125. doi:10.1007/s00276-017-1857-4
21. Richardson J, Jones J, Atkinson R, Richardson J. The effect of thoracic paravertebral blockade on intercostal somatosensory evoked potentials. Anesth Analg. 1998;87(2):373–376. doi:10.1097/00000539-199808000-00025
22. Karmakar MK. Ultrasound-guided thoracic paravertebral block. Tech Reg Anesth Pain Manage. 2009;13:142–149. doi:10.1053/j.trap.2009.06.020
23. Fanelli A, Montoya M, Ghisi D. Ultrasound-guided thoracic paravertebral block: classic approach. In: Bigeleisen PE, Orebaugh SL, Moayeri N, editors. Ultrasound-Guided Regional Anesthesia and Pain Medicine: Tech- Niques and Tips. Philadelphia, PA: Lippincott Williams & Wilkins; 2009:150–155.
24. D’Ercole F, Arora H, Kumar PA, et al. J paravertebral block for thoracic surgery. Cardiothorac Vasc Anesth. 2018;32(2):915–927. doi:10.1053/j.jvca.2017.10.003
25. Kotze A, Scally A, Howell S. Efficacy and safety of different techniques of paravertebral block for analgesia after thoracotomy: a systematic review and metaregression. Br J Anaesth. 2009;103:626–636. doi:10.1093/bja/aep272
26. Klein SM, Bergh A, Steele SM, Georgiade GS, Greengrass RA. Thoracic paravertebral block for breast surgery. Anesth Analg. 2000;90(6):1402–1405. doi:10.1097/00000539-200006000-00026
27. Marhofer P, Kettner SC, Hajbok L, Dubsky P, Fleischmann E. Lateral ultrasound-guided paravertebral blockade: an anatomical-based description of a new technique. Br J Anaesth. 2010;105(4):526–532. doi:10.1093/bja/aeq206
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

