Back to Journals » Neuropsychiatric Disease and Treatment » Volume 10
A prospective naturalistic study of antidepressant-induced jitteriness/anxiety syndrome
Authors Harada T, Inada K , Yamada K, Sakamoto K, Ishigooka J
Received 4 July 2014
Accepted for publication 29 August 2014
Published 10 November 2014 Volume 2014:10 Pages 2115—2121
DOI https://doi.org/10.2147/NDT.S70637
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Roger Pinder
Video abstract presented by Dr Tsuyoto Harada
Views: 2168
Tsuyoto Harada, Ken Inada, Kazuo Yamada, Kaoru Sakamoto, Jun Ishigooka
Department of Psychiatry, Tokyo Women’s Medical University School of Medicine, Tokyo, Japan
Objective: Patients often develop neuropsychiatric symptoms such as anxiety and agitation after they have started taking an antidepressant, and this is thought to be associated with a potentially increased risk of suicide. However, the incidence of antidepressant-induced jitteriness/anxiety syndrome has not been fully investigated, and little has been reported on its predictors. The aim of this study was to survey the incidence of antidepressant-induced jitteriness/anxiety syndrome and clarify its predictors in a natural clinical setting.
Materials and methods: Between January 2009 and July 2012, we prospectively surveyed 301 patients who had not taken any antidepressants for 1 month before presentation, and who were prescribed antidepressants for 1 month after their initial visit. Patients were classified as developing antidepressant-induced jitteriness/anxiety syndrome if they experienced any symptoms of anxiety, agitation, panic attacks, insomnia, irritability, hostility, aggressiveness, impulsivity, akathisia, hypomania, or mania during the first month.
Results: Among the 301 patients, 21 (7.0%) developed antidepressant-induced jitteriness/anxiety syndrome. Major depressive disorder and a diagnosis of mood disorder in first-degree relatives of patients were significantly associated with induction of antidepressant-induced jitteriness/anxiety syndrome (odds ratio 10.2, P=0.001; odds ratio 4.65, P=0.02; respectively). However, there was no such relationship for sex, age, class of antidepressant, combined use of benzodiazepines, or diagnosis of anxiety disorder.
Conclusion: The findings of this study suggest that major depressive disorder and a diagnosis of mood disorder in first-degree relatives may be clinical predictors of antidepressant-induced jitteriness/anxiety syndrome.
Keywords: side effects, antidepressants, selective serotonin-reuptake inhibitors, tricyclic antidepressive agents, suicide
Introduction
Patients often experience worsening of neuropsychiatric symptoms, such as anxiety and agitation, after they have started taking an antidepressant. Even in the 1980s, when tricyclic antidepressants (TCAs) were mainly used for the treatment of depression, such phenomena were well known clinically1 and often referred to as jitteriness syndrome,2–6 although the relationship between jitteriness syndrome and suicidal thoughts and suicidal behavior had not been discussed. More recently, after selective serotonin-reuptake inhibitors (SSRIs) had been widely introduced into clinical practice, the issue of a direct relationship between SSRIs and suicidal thoughts and suicidal behavior began to be highlighted. In 2003, the US Food and Drug Administration (FDA) warned of the possibility of suicidal thoughts and suicidal behavior associated with the use of antidepressants in the pediatric population,7 and in 2004 the FDA warned physicians of “activation syndrome”, characterized by such symptoms as anxiety, agitation, panic attacks, insomnia, irritability, hostility, aggressiveness, impulsivity, akathisia, hypomania, and mania, which might lead to potential suicide risk.8 After a series of warnings about the relationship between antidepressant use and suicidal thoughts and suicidal behavior, many studies covering several related aspects were published. However, the results of those studies were inconsistent, as some maintained that SSRIs might increase the risk of suicidal thoughts and suicidal behavior,9 whereas others did not.10–12 A review of all of the 24 placebo-controlled trials evaluating the effectiveness of antidepressants in pediatric patients that had been submitted to the FDA concluded that antidepressant use was associated with a modestly increased risk of suicidal thoughts and suicidal behavior, although no instances of death occurred in the trials.13 Therefore, although it can be assumed that SSRIs may increase the risk of suicidal thoughts and suicidal behavior, most such cases do not have a fatal outcome, and thus SSRI use might not be associated with an increased risk of suicide itself. Although many studies on the relationship between SSRI use and suicidal thoughts and suicidal behavior have been published, only a very small number of studies on jitteriness/anxiety syndrome induced by antidepressants have been published. The neuropsychiatric symptoms emerging after introduction of antidepressants are sometimes referred to as “activation syndrome”, but this term has not yet been medically defined and has remained inconsistent. Accordingly, in the present article, we have avoided using the term “activation syndrome”, and instead adopted the term “jitteriness/anxiety syndrome” to maintain consistency and to cover several neuropsychiatric symptoms emerging after the start of antidepressant administration.
Antidepressants activate brain monoamines, which are responsible for many forms of active behavior,14,15 and it is assumed that in the case of jitteriness/anxiety syndrome, antidepressants abnormally stimulate the production of brain monoamines and trigger emotional excitement and exaggeration, although the actual biological mechanism remains unclear. Some previous studies have suggested an association between mood disorders and the emergence of neuropsychiatric symptoms after the start of antidepressant administration.16,17
Clinically, it is assumed that benzodiazepine may prevent the mood lability that occurs after starting an antidepressant, but no previous study has considered this. A number of studies on jitteriness/anxiety syndrome have covered such aspects as the incidence, types of drugs, and diagnosis, but most of them have focused on only one antidepressant in comparison with a placebo, and only a single diagnosis.18 Only a few studies have investigated the classes of antidepressant and the types of diagnosis that are more frequently associated with jitteriness/anxiety syndrome, and possible predictors of jitteriness/anxiety syndrome have received little attention. Furthermore, previous studies of jitteriness/anxiety syndrome induced by antidepressants19,20 focused on only one disorder, compared patients taking a specific antidepressant with those taking a different one or a placebo, and made no reference to any predictors of jitteriness/anxiety syndrome.
Therefore, the aims of the present study were 1) to investigate the incidence of jitteriness/anxiety syndrome, and 2) to examine its association with any relevant independent demographic or clinical variables.
Materials and methods
Patients
The study population comprised patients who newly presented at the psychiatric department of our hospital between January 2009 and July 2012. We prospectively surveyed 301 patients who had not taken any antidepressants for 1 month before initial presentation, and who were prescribed antidepressants for 1 month thereafter. The policy at our hospital is basically not to prescribe antidepressants to patients with bipolar disorder. The outpatients were interviewed in a semistructured manner based on the Structured Clinical Interview for DSM (Diagnostic and Statistical Manual of Mental Disorders)-IV-TR Axis I Disorders, research version, patient edition. The study was approved by the ethical committee of Tokyo Women’s Medical University.
Evaluation of antidepressant-induced jitteriness/anxiety syndrome
Patients who experienced any of the following ten symptoms during the first month were classified as having jitteriness/anxiety syndrome: anxiety, agitation, panic attacks, insomnia, irritability, hostility, aggressiveness, impulsivity, akathisia, hypomania, and mania. However, patients were excluded if they already had these symptoms at the first visit and if the symptoms persisted or became aggravated during the first month. These symptoms are those covered in the 2004 FDA warning. The emergence of jitteriness/anxiety syndrome was judged by the attending psychiatrists clinically. No standardized scale was used. The following clinical variables were examined: sex, age, class of antidepressant, combined use of benzodiazepine, DSM-IV-TR diagnosis, and diagnosis of mood disorder in first-degree relatives of the patients.
With regard to antidepressants, we investigated all kinds of antidepressants, including SSRIs, selective serotonin norepinephrine-reuptake inhibitors, and TCAs. We categorized patients who took any benzodiazepine for at least 1 week after the first visit as those whose treatment was combined with benzodiazepine.
Data analysis
Differences in demographic and clinical variables between patients with and without jitteriness/anxiety syndrome were compared by means of χ2 analysis for categorized variables and t-tests for continuous variables.
Multiple logistic regression analysis was conducted to examine any association between relevant independent demographic and clinical variables and the appearance of jitteriness/anxiety syndrome. The following factors were introduced as independent variables: sex, age, class of antidepressant, combined use of benzodiazepine, diagnoses of major depressive disorder and anxiety disorder, and diagnosis of mood disorder in first-degree relatives of the patients. Statistical significance was set at P<0.05, and SPSS (version 11.5) was used for all statistical analyses.
Results
Characteristics of the patients
The age (mean ± standard deviation) of the patients was 45.3±17.8 years, and 70.4% (212 of 301) of the patients were female (Table 1). The DSM-IV-TR diagnoses of the patients were mood disorders in 150 patients (49.8%) including major depressive disorder in 99 (32.9%), pain disorder in 56 (18.6%), panic disorder in 32 (10.6%), undifferentiated somatoform disorder in 14 (4.7%), adjustment disorders in 12 (4.0%), and other disorders in 37 (12.3%), while none of the patients had bipolar disorder. The prescribed antidepressants were sertraline in 100 patients (33.2%), paroxetine in 79 (26.2%), amitriptyline in 51 (16.9%), escitalopram in 18 (6.0%), mirtazapine in 14 (4.7%), fluvoxamine in eleven (3.7%), nortriptyline in nine (3.0%), milnacipran in nine (3.0%), duloxetine in five (1.7%), clomipramine in two (0.6%), trazodone in two (0.6%), and sulpiride in one (0.3%).
Incidence and course of jitteriness/anxiety syndrome
Of the 301 patients, 21 (7.0%) developed jitteriness/anxiety syndrome. Twelve patients showed one symptom, six patients showed two, and three patients showed three. The symptoms of jitteriness/anxiety syndrome were insomnia in nine patients (42.9%), irritability in seven (33.3%), anxiety and agitation in five (23.8%), panic attack in three (14.3%), impulsivity in two (9.5%), and akathisia and hypomania in one (4.8%). None of the patients showed hostility or mania. Among the 21 patients with jitteriness/anxiety syndrome, although one developed suicidal ideation, none showed self-harm and none attempted suicide.
The DSM-IV-TR diagnoses of patients with jitteriness/anxiety syndrome were major depressive disorder in eleven patients (52.4%), panic disorder in four (19.0%), adjustment disorder in two (9.5%), and somatoform disorder, depressive disorder not otherwise specified, and anxiety disorder not otherwise specified in one (4.8%).
Antidepressants taken by the patients with jitteriness/anxiety syndrome were paroxetine in eight patients (38.1%), sertraline in seven (33.3%), fluvoxamine in two (9.5%), and escitalopram, milnacipran, clomipramine, and trazodone in one (4.8%).
Predictors of jitteriness/anxiety syndrome
As shown in Tables 1 and 2, mood disorder in first-degree relatives of patients was significantly associated with the induction of jitteriness/anxiety syndrome by antidepressants in both χ2 analysis (P=0.001), and multiple logistic regression analysis (odds ratio 10.2, P=0.001). Among the five patients who had a family history of mood disorder in first-degree relatives and developed jitteriness/anxiety syndrome, four patients had a family history of major depressive disorder in first-degree relatives, and one patient had a family history of bipolar disorder in first-degree relatives.
The incidence of jitteriness/anxiety syndrome among the patients with major depressive disorder was 11.1% (eleven of 99), and as shown in Table 2, multiple logistic regression analysis demonstrated that this diagnosis was significantly associated with the induction of jitteriness/anxiety syndrome by antidepressants (odds ratio 4.65, P=0.02), although χ2 analysis demonstrated that this association was not significant (P=0.057).
Age was not significantly associated with the induction of jitteriness/anxiety syndrome (P=0.057 in t-test and P=0.07 in multiple logistic regression analysis). Other independent variables, such as sex, class of antidepressant, combined use of benzodiazepine, and a diagnosis of anxiety disorder, were not significantly associated with the induction of jitteriness/anxiety syndrome.
Period of onset of jitteriness/anxiety syndrome
The period of onset of jitteriness/anxiety syndrome was 7.3±8.07 days. About half of the 21 patients developed jitteriness/anxiety syndrome within 3 days, and 16 did so within 1 week, suggesting that most symptoms emerged within a very short time after patients had started to take antidepressants.
Interventions for jitteriness/anxiety syndrome and their outcome
Table 3 shows the types of interventions used for treatment of anxiety syndrome and their outcome. Antidepressants were stopped in about half of the patients who developed anxiety syndrome. This approach appeared to be effective, as eight of the nine patients whose antidepressants were stopped improved.
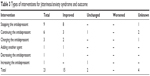  | Table 3 Types of interventions for jitteriness/anxiety syndrome and outcome |
Discussion
The incidence of jitteriness/anxiety syndrome in this prospective cohort was 7%. Previous studies have reported that the incidence of jitteriness/anxiety syndrome induced by antidepressants ranges widely from 4% to 65%.18 This wide range may have been the result of differences in the numbers of the symptoms included, study duration, the methods used for evaluating symptoms, the diagnoses included, or the types of antidepressants used. Another problem was the difficulty in detecting neuropsychiatric symptoms, as symptoms considered to be adverse effects, such as anxiety and insomnia, are generally seen in patients with depression or anxiety disorders. Therefore, a certain proportion of symptoms that might be considered adverse effects would actually be due simply to worsening of the extant illness, thus explaining the wide range of incidence. Several years ago, we performed a retrospective study and found that the incidence of jitteriness/anxiety syndrome was 4.3%,21 ie, about half the incidence demonstrated in the present study. This suggests that in our previous retrospective study, we might have overlooked about half of the symptoms. The fact that our study included no patients with bipolar disorder may also have accounted for the low incidence of jitteriness/anxiety syndrome in our study. Takeshima and Oka reported that patients with bipolar disorder develop “activation syndrome” more frequently than patients with major depressive disorder.16 If we had prescribed antidepressants even to patients with bipolar disorder, the incidence of jitteriness/anxiety syndrome in our study would likely have been higher.
We found that induction of jitteriness/anxiety syndrome was significantly associated with the presence of major depressive disorder and a diagnosis of mood disorder in first-degree relatives of the studied patients. To our knowledge, this is the first prospective study to have highlighted a possible predictor of neuropsychiatric symptoms induced by antidepressants. Kraus et al conducted a meta-analysis of randomized placebo-controlled trials, and concluded that in patients with major depressive disorder the incidence of definitive suicidal behavior was higher for paroxetine (0.32% versus 0.05%), whereas there were no differences in the incidence of definitive suicidal behavior for all indications. This suggests that patients with major depressive disorder develop definitive suicidal behavior more frequently than patients with other psychiatric disorders during paroxetine therapy. However, it was pointed out that because a higher proportion of young patients with major depressive disorder had taken paroxetine, the risk of suicidal behavior in this cohort might have been higher.17 Akiskal et al reported that of 52 patients who developed hypomania solely as a result of taking antidepressants, 24 (47.1%) had a family history of unipolar depression, and six (11.8%) had a family history of bipolar disorder, suggesting a high familial psychiatric burden,22 in accordance with our present findings.
In this study, age was not significantly associated with induction of jitteriness/anxiety syndrome (P=0.057 in t-test and P=0.07 in multiple logistic regression analysis). Takeshima and Oka reported that early age at onset of a first major depressive episode was associated with development of activation syndrome,16 and data from the FDA indicate that the odds ratios for suicidal ideation and behavior tend to be higher in younger individuals,23 which is inconsistent with our present results. This may be because only a few young patients were included in our study, and thus our sample size for young patients may not have been large enough for statistical evaluation: among the 301 patients, only four were under 18 years old, and 21 were 18–24 years of age.
There has been some discussion as to whether newer antidepressants, such as SSRIs, are more likely to induce suicidal thoughts and suicidal behavior than older antidepressants, such as TCAs, and this issue remains inconclusive, as some studies have reported that there are no differences in induction of suicidal thoughts and suicidal behavior among the various classes of antidepressants,24,25 whereas others have maintained that newer drugs are more likely to have such effects.9 To our knowledge, no previous study has directly compared newer with older antidepressants in terms of the risk of jitteriness/anxiety syndrome. In the present study, we found no association between the class of antidepressant employed and the induction of jitteriness/anxiety syndrome. However, as this was a prospective cohort study that was not controlled, there was a difference in background between patients who were treated with SSRIs first and those who were treated with TCAs first, eg, SSRIs were used more for patients with anxiety disorders, and TCAs more for older patients with pain disorders. In view of this issue, a controlled study will be needed in order to compare SSRIs and TCAs directly in terms of their risk of inducing jitteriness/anxiety syndrome.
Another important issue is the type of SSRI that is more likely to induce jitteriness/anxiety syndrome. Hammad presented data pertaining to the overall relative risks of antidepressant-related agitation or hostility in trials involving patients with major depressive disorder, and indicated that paroxetine was the only agent with a significant risk of inducing adverse effects (relative risk 7.69, 95% confidence interval 1.80–32.99).26 In the present study, we conducted χ2 analysis of the effects of paroxetine and sertraline for which sufficient samples were available, and found that these two agents were not significantly associated with induction of jitteriness/anxiety syndrome, and also that there was no difference in the incidence of jitteriness/anxiety syndrome between patients who took paroxetine and those who took sertraline. Although this result is inconsistent with Hammad’s data, ours was a cohort study, and we tended to avoid administering paroxetine to patients who were thought to be at particularly high risk of developing jitteriness/anxiety syndrome, such as those who were young, or those with agitation.
In this study, the period of onset of jitteriness/anxiety syndrome was 7.3±8.07 days; about half of the patients developed it within 3 days, and about two-thirds did so within 1 week. Yeragani et al studied 52 patients with panic disorder who were treated with imipramine in a randomized double-blind test, and found that the jitteriness scores for affected patients were highest during week 1.27 Beasley et al studied the activating effects of fluoxetine using data from double-blind clinical trials, and found that the first occurrences peaked during week 1 and then declined thereafter.28 These results are in agreement with the early onset of jitteriness/anxiety syndrome found in our present study.
Our findings concerning interventions for jitteriness/anxiety syndrome suggested that stopping antidepressants was effective, as eight of nine patients whose antidepressants were stopped showed improvement. Although it would seem natural to stop antidepressants if anxiety symptoms appeared soon after the start of medication, in a clinical situation it is sometimes difficult to distinguish the appearance of jitteriness/anxiety syndrome from worsening of the existing illness, especially in patients with insomnia or anxiety, which are typical symptoms of depression. If the neuropsychiatric symptoms are due to the adverse effects of an antidepressant, then the medication should be stopped, but if the symptoms are due to the existing illness, then the antidepressant should be continued or the dose increased. In this study, six patients continued taking antidepressants even after neuropsychiatric symptoms had appeared, and three of them showed improvement. Therefore, the symptoms in these three cases might have been attributable to the existing illness.
In our study, only one patient developed suicidal thoughts, and no patient developed suicidal behavior. The association between taking antidepressants and the emergence of suicidal behavior is unclear, and the number of patients who developed suicidal thoughts and suicidal behavior was too low to merit productive discussion.
This study had two limitations. First, it was a naturalistic cohort study, so we did not use antidepressants for patients with bipolar disorder or for patients who might potentially have had bipolar disorder, such as those with onset at a young age, and this might have been the reason for the low incidence (7.0%) of jitteriness/anxiety syndrome. Second, there were 27 patients who started to take antidepressants but did not return after the first visit, and therefore it is possible that some of them might have developed jitteriness/anxiety syndrome later.
This study has demonstrated that major depressive disorder and a diagnosis of mood disorder in first-degree relatives are predictors of jitteriness/anxiety syndrome in patients taking antidepressants. Clinicians should carefully monitor patients with major depressive disorder and a diagnosis of mood disorder in first-degree relatives for possible development of jitteriness/anxiety syndrome during antidepressant therapy. Our sample did not include a sufficient number of young patients or patients with bipolar disorders to merit statistical evaluation. Further studies will therefore be needed to evaluate the possible risk of antidepressant-induced jitteriness/anxiety syndrome in such patients.
Acknowledgment
This work was supported by JSPS KAKENHI (21791149).
Disclosure
During the past 3 years, TH has received speakers bureau honoraria from GlaxoSmithKline, Otsuka Pharmaceuticals, Meiji Seika Pharma, and Tanabe Mitsubishi. KI has received grant/research support and speakers bureau honoraria from Pfizer Pharmaceuticals, and has received speakers bureau honoraria from Otsuka Pharmaceuticals, Ely Lilly, Janssen, Dainihon Sumitomo, Mochida, Novartis, and GlaxoSmithKline. KY has received grant/research support and speakers bureau honoraria from Tsumura, Eisai, and GlaxoSmithKline, and has received speakers bureau honoraria from Astellas, Shionogi, Pfizer Pharmaceuticals, Meiji Seika Pharma, Otsuka Pharmaceuticals, Yoshitomi, Tanabe Mitsubishi, Mochida, Ely Lilly, Kyowa Kirin, and Kracie. KS has received speakers bureau honoraria from Eli Lilly, GlaxoSmithKline, Otsuka Pharmaceuticals, Meiji Seika Pharma, and Pfizer Pharmaceuticals. JI has received speakers bureau honoraria from Dainihon Sumitomo, Mochida, Shionogi, and Otsuka Pharmaceuticals.
References
King RA, Riddle MA, Chappell PB, et al. Emergence of self-destructive phenomena in children and adolescents during fluoxetine treatment. J Am Acad Child Adolesc Psychiatry. 1991;30(2):179–186. | ||
Friedman EH. Tricyclic-induced activation/jitteriness. J Clin Psychopharmacol. 1991;11(6):394–395. | ||
Jackson CW, Lydiard RB. Two cases of activation/jitteriness associated with TCAs. J Clin Psychopharmacol. 1990;10(6):439–440. | ||
Pohl R, Yeragani VK, Ortiz A, Rainey JM Jr, Gershon S. Response of tricyclic-induced jitteriness to a phenothiazine in two patients. J Clin Psychiatry. 1986;47(8):427. | ||
Yeragani VK, Pohl R, Balon R. Tricyclics, akathisia, and jitteriness. J Clin Psychopharmacol. 1988;8(4):295. | ||
Yeragani VK, Pohl R, Balon R, Kulkarni A, Keshavan M. Low serum iron levels and tricyclic antidepressant-induced jitteriness. J Clin Psychopharmacol. 1989;9(6):447–448. | ||
US Food and Drug Administration. Reports of suicidality in pediatric patients being treated with antidepressant medications for major depressive disorder (MDD). 2003. Available from: http://www.fda.gov/drugs/drugsafety/postmarketdrugsafetyinformationforpatientsandproviders/ucm168828.htm. Accessed October 24, 2014. | ||
US Food and Drug Administration. Worsening depression and suicidality in patients being treated with antidepressants. 2004. Available from: http://www.fda.gov/Drugs/DrugSafety/PostmarketDrugSafetyInformationforPatientsandProviders/ucm161696.htm. Accessed October 24, 2014. | ||
Juurlink DN, Mamdani MM, Kopp A, Redelmeier DA. The risk of suicide with selective serotonin reuptake inhibitors in the elderly. Am J Psychiatry. 2006;163(5):813–821. | ||
Simon GE, Savarino J. Suicide attempts among patients starting depression treatment with medications or psychotherapy. Am J Psychiatry. 2007;164(7):1029–1034. | ||
Tondo L, Lepri B, Baldessarini RJ. Suicidal status during antidepressant treatment in 789 Sardinian patients with major affective disorder. Acta Psychiatr Scand. 2008;118(2):106–115. | ||
Simon GE, Savarino J, Operskalski B, Wang PS. Suicide risk during antidepressant treatment. Am J Psychiatry. 2006;163(1):41–47. | ||
Hammad TA, Laughren T, Racoosin J. Suicidality in pediatric patients treated with antidepressant drugs. Arch Gen Psychiatry. 2006;63(3):332–339. | ||
Rotenberg VS. Functional brain asymmetry as a determinative factor in the treatment of depression: theoretical implications. Prog Neuropsychopharmacol Biol Psychiatry. 2008;32(8):1772–1777. | ||
Rotenberg VS. Search activity concept: relationship between behavior, health and brain functions. Activitas Nervosa Superior. 2009;51(1):12–44. | ||
Takeshima M, Oka T. Association between the so-called “activation syndrome” and bipolar II disorder, a related disorder, and bipolar suggestive features in outpatients with depression. J Affect Disord. 2013;151(1):196–202. | ||
Kraus JE, Horrigan JP, Carpenter DJ, Fong R, Barrett PS, Davies JT. Clinical features of patients with treatment-emergent suicidal behavior following initiation of paroxetine therapy. J Affect Disord. 2010;120(1–3):40–47. | ||
Sinclair LI, Christmas DM, Hood SD, et al. Antidepressant-induced jitteriness/anxiety syndrome: systematic review. Br J Psychiatry. 2009;194(6):483–490. | ||
Beasley CM Jr, Potvin JH. Fluoxetine: activating and sedating effects. Int Clin Psychopharmacol. 1993;8(4):271–275. | ||
Amsterdam JD, Hornig-Rohan M, Maislin G. Efficacy of alprazolam in reducing fluoxetine-induced jitteriness in patients with major depression. J Clin Psychiatry. 1994;55(9):394–400. | ||
Harada T, Sakamoto K, Ishigooka J. Incidence and predictors of activation syndrome induced by antidepressants. Depress Anxiety. 2008;25(12):1014–1019. | ||
Akiskal HS, Hantouche EG, Allilaire JF, et al. Validating antidepressant-associated hypomania (bipolar III): a systematic comparison with spontaneous hypomania (bipolar II). J Affect Disord. 2003;73(1–2):65–74. | ||
Friedman RA, Leon AC. Expanding the black box – depression, antidepressants, and the risk of suicide. N Engl J Med. 2007;356(23):2343–2346. | ||
Schneeweiss S, Patrick AR, Solomon DH, et al. Comparative safety of antidepressant agents for children and adolescents regarding suicidal acts. Pediatrics. 2010;125(5):876–888. | ||
Schneeweiss S, Patrick AR, Solomon DH, et al. Variation in the risk of suicide attempts and completed suicides by antidepressant agent in adults: a propensity score-adjusted analysis of 9 years’ data. Arch Gen Psychiatry. 2010;67(5):497–506. | ||
Hammad TA. Results of the analysis of suicidality in pediatric trials of newer antidepressants. 2004. Available from: http://www.fda.gov/ohrms/dockets/ac/04/slides/2004-4065S1_08_FDA-Hammad.ppt. Accessed September 19, 2014. | ||
Yeragani VK, Pohl R, Balon R, Ramesh C, Weinberg P. Imipramine-induced jitteriness and decreased serum iron levels. Neuropsychobiology. 1992;25(1):8–10. | ||
Beasley CM Jr, Sayler ME, Weiss AM, Potvin JH. Fluoxetine: activating and sedating effects at multiple fixed doses. J Clin Psychopharmacol. 1992;12(5):328–333. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.


