Back to Journals » Drug Design, Development and Therapy » Volume 16
A Potent EGFR Inhibitor, N-Phenyl Pyrazoline Derivative Suppresses Aggressiveness and Cancer Stem Cell-Like Phenotype of Cervical Cancer Cells
Authors Mustofa, Satriyo PB, Suma AAT , Waskitha SSW, Wahyuningsih TD, Sholikhah EN
Received 24 November 2021
Accepted for publication 20 April 2022
Published 20 July 2022 Volume 2022:16 Pages 2325—2339
DOI https://doi.org/10.2147/DDDT.S350913
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Prof. Dr. Tin Wui Wong
Mustofa,1 Pamungkas Bagus Satriyo,1 Artania Adnin Tri Suma,2 Stephanus Satria Wira Waskitha,3 Tutik Dwi Wahyuningsih,3 Eti Nurwening Sholikhah1
1Department of Pharmacology and Therapy, Faculty of Medicine Public Health and Nursing, Universitas Gadjah Mada, Yogyakarta, 55281, Indonesia; 2Department of Pharmaceutical Chemistry, Faculty of Pharmacy, Universitas Gadjah Mada, Yogyakarta, 55281, Indonesia; 3Department of Chemistry, Faculty of Mathematics and Natural Sciences, Universitas Gadjah Mada, Yogyakarta, 55281, Indonesia
Correspondence: Mustofa, Department of Pharmacology and Therapy, Faculty of Nursing Public Health and Nursing, Universitas Gadjah Mada, Yogyakarta, 55281, Indonesia, Tel/Fax +62 274-545188, Email [email protected]
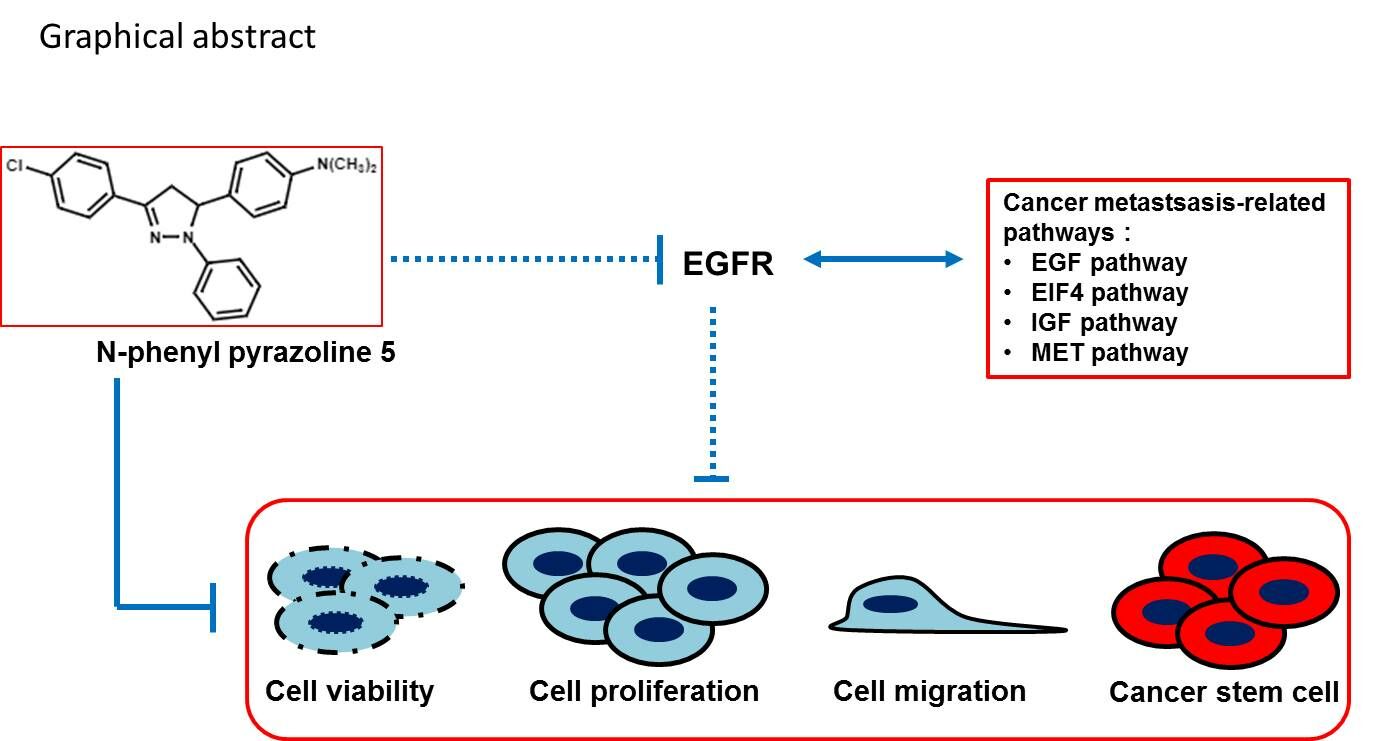
Objective: Metastasis causes approximately 90% of cancer-related deaths, including in cervical cancer patients. Uncontrolled cell proliferation, migration, and cancer stemness act as critical events in primary tumor growth and cancer metastasis progression in cervical cancer. Here, we investigated the anti-proliferative, anti-migration, and cancer stemness inhibition activity of N-phenyl pyrazoline derivatives against cervical cancer cells.
Methods: The chalcone and phenylhydrazine were used to synthesize the N-phenyl pyrazoline 2/5 (P2 and P5). The MTT, colony formation, and wound healing assays were performed to evaluate the N-phenyl pyrazoline effect in HeLa cells. The N-phenyl pyrazoline’s protein target was predicted using SwissTargetPrediction and AutoDock Vina software. The Western blotting assay was performed to evaluate the target proteins. The public dataset analysis was used to confirm the clinical relevance of target protein in cervical cancer patients.
Results: N-phenyl pyrazoline 2 and 5 were successfully synthesized. The N-phenyl pyrazolines 2 and 5 exhibit cytotoxic effect in HeLa cell line with 20.26 μM, 4.708 μM of IC50, respectively. Further study shows that the N-phenyl pyrazoline 5 suppresses the cell proliferation and migration ability of HeLa cell line in a dose-dependent manner. Target prediction and molecular docking reveal that EGFR and ERBB2 protein as the main target of the N-phenyl pyrazoline 5 compound. The N-phenyl pyrazoline 5 suppresses the EGFR expression level but not the total ERK1/2. Public data and GSEA analysis found that the EGFR high expression level is positively associated with poor survival, cancer metastasis-related signaling pathways, and cancer stem cell markers in cervical cancer patients. In addition, the N-phenyl pyrazoline 5 reduces the HeLa’s tumorsphere size and cancer stem cell marker, CD133.
Conclusion: N-phenyl pyrazoline 5 suppresses the cell viability, proliferation, migration, and cancer stem cell-like phenotype of cervical cancer cells via EGFR inhibition.
Keywords: N-phenyl pyrazoline, cervical cancer, cancer stem cell-like phenotype, EGFR
Graphical Abstract:
Introduction
In 2020, cervical cancer was the fourth most common cancer diagnosed in women worldwide, with approximately 604.000 new cases. Along with 342.000 death cases, cervical cancer is the fourth leading cause of cancer-related deaths in women worldwide. In several countries located in South-Eastern Asia, South America, Melanesia, and sub-Saharan Africa, cervical cancer was found as the most common cancer diagnosed and the most cancer-related deaths in women.1 The general term of cancer metastasis describes as the spreading of cancer cells from the primary site to surrounding tissues and distant organs. Cancer metastasis is responsible for approximately 90% of cancer-related deaths.2 The metastatic cervical cancer patients have a lower 5-year survival rate than localized cervical cancer patients (16.5% and 91.5%, respectively).3 Another study found that the 5-year overall survival rate of metastatic cervical cancer was declined up to 12.6%.4 Moreover, a specific retrospective study in late-stage (stage IVB) cervical cancer cases found that the 5-year overall survival rate was only 9.9%.5 In applying standard therapy such as surgery, radiotherapy, and chemotherapy, the prognosis of the metastatic cervical cancer patient is still worst.
Uncontrolled cell proliferation is an important event in the initiation of cancer progression in the primary tumor site. Sustained proliferation is also required for generating the new tumor tissues during the distant organ metastasis.6 The ability of migration and invasion allows the cancer cells to move from the primary site to surrounding tissue and infiltrate the lymph/blood vessel to distant organs.2 The cancer stem cells are a small subpopulation of cells within tumors that exhibit self-renewal and multi-lineage differentiation ability. In solid cancer including cervical cancer, the cancer stem cells play a pivotal role in tumor development, metastasis, and drug-resistant.7 Pyrazoline and its derivatives are heterocyclic compounds with broad biological activities.8 Pyrazoline derivatives exhibited anti-cancer activities against cervical cancer, hepatocellular carcinoma, lung cancer, and breast cancer cells.8–11 A previous study found that our synthesized N-phenyl pyrazolines showed anti-cancer activity against breast cancer and colorectal cancer cells. The last five years’ studies found that the pyrazoline and N-phenyl pyrazoline derivatives targeted the EGFR.12,13 The EGFR signaling pathway is a well-known signaling pathway that regulates cancer cell proliferation, tumor growth, and metastasis in cancers. Further, the human papillomavirus infection is well known as the main cause of cervical cancer. The HPV oncoproteins (E5, 6, and 7) induce the cell transformation to cancerous cells by inhibiting the tumor suppressor protein (p53 and Rb). Furthermore, these oncoproteins also induce EGFR activation and its downstream targets lead to increasing the cancer cell proliferation, angiogenesis, and anti-apoptotic effect.15,16 Targeting the EGFR in cervical cancer using N-phenyl pyrazoline treatment may give a chance to inhibit cancer distant metastasis and improve the patient’s prognosis. However, the mechanisms of N-phenyl pyrazolines against cervical cancer cells are poorly understood. In the present study, we synthesized two N-phenyl pyrazoline derivative compounds and performed cell functional assays to evaluate their effects on the cervical cancer cells’ viability, proliferation, migration, and cancer stem cell-like phenotype.
Materials and Methods
N-Phenyl Pyrazolines Synthesis
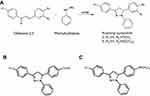
The chalcone 2/5 was diluted in 5 mL ethanol and added into boiling three-necked flask. The 5 mL of glacial acetic acid was added by dropping to the mixture. Phenylhydrazine (0.075 mL/0.76 nmol) was added, and the mixture was refluxed for 6.5 hours. Later, the mixture was poured into a block of ice and was incubated at 4°C for 24 hours. The precipitate was filtered and washed using pre-cooling aquadest (pH 6 −7). The filtrates were dried using a vacuum desiccator. Purifications were performed using column chromatography with hexane-dichloromethane as eluent. The final dried products, N-phenyl pyrazolines 2 and 5 (P2 and P5), were analyzed using H-NMR and C-NMR spectrometry.
Cell Culture
In this study, we used the cervical adenocarcinoma cell line, HeLa cells. The HeLa cell was purchased from ATCC (American Type Culture Collection, USA). HeLa cell line was derived from cervical adenocarcinoma of 31 years old female patient. This cell line was cultured in RPMI medium (#31800022, Thermo Fisher Scientific.Inc, USA) supplemented with 10% FBS (fetal bovine serum) (#GibcoTM 10270106, Thermo Fisher Scientific.Inc), 100 IU/mL and 100 µg/mL of Penicillin/Streptomycin, and 0.5% of amphotericin B. The culture conditions were carried out in a 5% CO2 incubator with a humidified atmosphere at 37°C. The sub-cultures were performed every 48–72 hours for maintenance.
MTT Assay
Harvested HeLa cells were seeded in the 96-well plates with 1×104 cells/well density. After 24 hours of incubation in RPMI medium (#31800022, Thermo Fisher Scientific. Inc, USA) without any supplementation, the cells were treated with different doses of P2, and P5 compounds. The control groups were treated with the highest concentration of DMSO that was used to dilute the highest concentration of the compounds. After 24 or 48 hours of treatment, the cells were added with 100 µL of 10% 3-(4,5-dimethylthiazol-2yl)-2,5-diphenyltetrazolium bromide (MTT reagent) for each well. The cells were incubated for 4 hours in a 5% CO2 incubator in the humidified atmosphere at 37°C. The 10% sodium dodecyl sulfate (100 µL) were added to each well and were incubated overnight in the darkroom at room temperature. To quantify the viable cells, the absorbance values were measured using spectrophotometry at 565 nm wavelength. In addition, to investigate the cancer chemoresistant phenotype as one of the cancer stem cell characteristics, the HeLa cells isolated from hanging drop tumorsphere culture were seeded in the 96-well plates with 1×104 cells/well density. After 24 hours incubation in RPMI medium (#31800022, Thermo Fisher Scientific. Inc, USA) without any supplementation, the cells were treated with different doses of paclitaxel or P5-paclitaxel combination. After 48 hours, the MTT assay was carried out as previously described.
Wound Healing Assay
HeLa cells were seeded onto 24-well plates with a density of 4×105 cells for each well. The cells were incubated in a complete medium without FBS for 24 hours. The approximately 100% monolayer confluent wells were scratched with a sterile yellow pipette tip to create the wound area without cells. Later, the cells were treated with different concentrations of the P5 compound. The control group was treated with DMSO. To evaluate the migration rates, the cell images for each well were captured at 0, 24, and 48 hours after scratching time. The wound enclosure areas were quantified using ImageJ version 1.518j software (Wayne Rasband National Institutes of Health, Bethesda, MD, USA).
Colony Formation Assay
To evaluate the effect of the P5 compound on cancer cell proliferation, the HeLa cell line (100 cells/well) was seeded onto 6-well plates with a complete medium. After 24 hours of incubation, the cells were treated with different doses of P5 compound, and the control group was treated with DMSO. During seven days of incubation time after the treatment, the medium was replaced with a new medium with the same responsible compound concentration for every 48 hours. Then, the cells were fixed with ice-cold methanol absolute for 15 minutes at −20°C. After being washed with PBS, the cells were stained with 1% methylene blue diluted in methanol for 4 hours. The cells were washed by PBS to remove excessive staining dye and dried at room temperature. The numbers of colonies were counted under the microscope.
Western Blotting Assay
The HeLa cells from the control and treated groups were harvested by scraping method. The harvested cells were added with 1X RIPA lysis buffer (ab156034, Abcam) and were incubated on ice for 30 minutes. The mixtures were centrifuged for 10 minutes at 12.000 rpm at 4°C. The supernatants were carefully collected to 1.5 mL tubes as total protein samples. The total protein quantification was performed using PierceTM BCA Protein Assay Kit (23227, Thermo Fisher Scientific). After being added with sample buffer, the samples were boiled for 10 minutes and centrifuged at 12.000 rpm for 10 minutes. The samples were loaded onto and were run by SDS-PAGE. The blots were transferred onto the nitrocellulose membrane. The blocking step was carried out using 5% of BSA for 1 hour. The membranes were washed out using PBS-Tween-20 (PBST), then incubated with primary antibodies specific against EGFR (A11352, Abclonal), ERK1/2 (A16686, Abclonal), CD133 (A0219, Abclonal), and β-actin (AC026, Abclonal) for overnight at 4°C. After the membranes were washed using PBST, the membranes were incubated with HRP Goat Anti-Rabbit IgG secondary antibody (AS014, Abclonal) for 1 hour at room temperature. To visualize the protein bands, the UVP Biospectrum Imaging System using SuperKinaseTM West Femto Maximum – ECL reagent (BMU102-EN, Abbkine).
Prediction and Molecular Docking
The N-phenyl pyrazoline 5 was modelized in 3D structure and optimized using DFT/B3LYP method. Geometry optimization was carried out using Gaussian 09W software. The optimized structure of N-phenyl pyrazoline 5 was converted into Sybyl MOL2 format. The target-ligand prediction was performed using SwissTargetPrediction online platform through http://www.swisstargetprediction.ch/ website. The EGFR protein data (PDB: 1M17) was downloaded from https://www.rcsb.org/ website. The standard ligand (erlotinib) and water molecule were removed using Discovery Studio software. The polar hydrogen atoms were added to the EGFR structure using AutoDockTools 1.5.6. The active site of EGFR was recognized by amino acid interaction with the standard ligand (erlotinib). Molecular docking was performed using AutoDock Vina software.
Public Dataset Analysis
The TCGA Cervical Cancer (CESC) dataset was used to evaluate the prognostic value of EGFR and ERBB2 expression levels in cervical cancer patients (https://xenabrowser.net/heatmap/). The 308 cervical cancer patients were included, and analysis was performed using the available platform on that website. Further analysis, the TCGA-PanCancer Atlas dataset was downloaded from cBioPortal website. This dataset consists of information from 297 cervical cancer patients, including genomic and clinical characteristics for each patient. Three cervical patients who lack either gene expression or overall survival information were excluded from this study. The Kaplan Meier (KM) overall survival analysis was carried out using the “R2: Genomics Analysis and Visualization Platform” and GraphPad Prism 7.00. The patients were divided into two groups by the automated “Kaplan scan” cutoff function of the R2 platform. This function generated a KM plot with the most optimal cutoff value between two groups (high and low expression groups). The Log-rank (Mantel-Cox) test was used to determine the association between the gene expression and the overall survival rate of cervical cancer patients. To investigate the enriched signaling pathway related to the gene expression level, Gene Set Enrichment Analysis software from Broad Institute was used. To analyze the correlation between EGFR and cancer stem cell markers, we perform Pearson correlation analysis using the TCGA-PanCancer Atlas dataset.
Hanging-Drop Tumorsphere Assay
The ± 70% confluence HeLa cells were harvested and were counted using a hemocytometer. The cells were resuspended in the complete growth medium with a density of 50 cells/20 µL and 100 cells/20 µL. The 20 µL of cell suspension drop was seeded in the lid of 100 mm culture dishes. All hanging drops of both cell densities were divided into four groups. The control groups contained DMSO, while the treated groups contained three doses of N-phenyl pyrazoline 5 (1 µM, 10 µM, and 20 µM). The hanging drops were performed in 10 replicates for each group. The 5 mL of PBS were added to the bottom side of the culture dishes. After being incubated in a 5% CO2 incubator in the humidified atmosphere at 37°C for seven days, the tumorspheres were observed under the microscope. The tumorspheres size was quantified using ImageJ version 1.51j8 (Wayne Rasband National Institutes of Health, Bethesda, MD, USA).
Statistical Analysis
The in vitro cell-based assays were carried out at least three times, and results were provided as Mean ± SD. Data quantification of wound healing and colony formation assays were performed using ImageJ version 1.51j8 (Wayne Rasband National Institutes of Health, Bethesda, MD, USA). Quantification data were visualized using SigmaPlot software.
Results
The N-Phenyl Pyrazoline 5 Suppresses the HeLa Cells’ Viability in Doses and Time-Dependent Manner
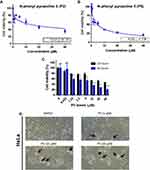
In the beginning, we synthesized the N-phenyl pyrazoline from chalcone and phenylhydrazine (Figure 1A–C). The N-phenyl pyrazolines were analyzed with H-NMR and C-NMR spectroscopy (Table 1). To investigate the pharmacological effects of N-phenyl pyrazoline 2 and 5 against the cervical cancer cells, we performed MTT assay using HeLa cell line. The HeLa were treated with several doses of N-phenyl pyrazoline 2 and 5. The DMSO-treated cells were used as the control groups. Treatment with 0.625–40 µM of each compound suppressed the HeLa cell viability in doses dependent manner with 48 hours IC50 of 20.26 µM, 4.708 µM for P2 and P5, respectively (Figure 2A and B). Considering that P5 exhibits the lowest IC50 against the HeLa cells, we used the P5 compound for further study. We performed MTT assays using P5 (0.625–40 µM) with 24 and 48 hours of incubation time to explore whether the P5 inhibits HeLa cell viability in a time-dependent manner or not. Markedly, the P5 inhibited the cell viability of HeLa cells in a time-dependent manner. The 48 hours treatment group showed significantly lower cell viability relative to 24 hours treatment group with the doses of P5, 0.625 to 40 µM (Figure 2C). We observed that increasing doses of P5 decreased the HeLa cell number and changed the cell morphology (Figure 2D). All those findings suggest the anti-cancer activity of the P5 compound against cervical cancer cells.
 |
Table 1 The H-NMR and C-NMR Spectrometry Results of N-Phenyl Pyrazolines |
The N-Phenyl Pyrazoline 5 Treatment Inhibits the Cell Migration and Proliferation in the Cervical Cancer Cells
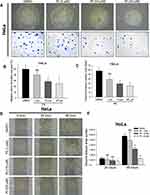
Considering sustained uncontrolled cell proliferation plays a pivotal role in cancer progression, including cervical cancer, we performed the colony formation assay to evaluate the P5 treatment on cell proliferation of cervical cancer cells. The 1 µM doses of P5 compound suppressed the colony formation size and number. However, the statistical calculation showed the value was not significant (Figure 3A–C). The 10 and 20 µM doses of P5 treatments significantly suppressed the colony formation size and number relative to control groups (Figure 3A–C). The migration ability of cancer cells is required for cancer spreading from the primary to surrounding tissues and distant organs during cancer metastasis. To explore the anti-migratory effect of P5 compound against cervical cancer cells, we performed the wound healing assay. In the 24 hours of P5 treatment, we observed that the 1 and 10 µM doses of P5 compounds did not significantly suppress the wound closure area relative to DMSO groups (Figure 3D and E). The 20 µM dose of P5 compound was significantly suppressed the wound enclosure area. In the 48 hours of treatment, the 10 and 20 µM doses of P5 compound were significantly decreased the wound enclosure areas (Figure3D and E). Furthermore, we performed the wound healing assay using the concentration of P5 lower than the IC50 dose. We found that 3 µM of P5 significantly reduced the migration ability of HeLa cells after 24 hours of treatment (Supplementary Figure S1A and B). After 48 hours of treatment, the examined concentrations of compound P5, 2 µM, and 3 µM significantly decreased the wound enclosure areas. All findings suggest that P5 compound suppressed the cell proliferation in HeLa cell line in a doses-depended manner. Further, the P5 compound treatments inhibit the HeLa cell migration in doses and time-depended manner.
The EGFR Which is Associated with Poor Prognosis of Cervical Cancer Patients is Inhibited by N-Phenyl Pyrazoline 5 Treatment
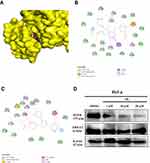
Previously, we confirmed the P5 compound treatment suppressed the cervical cancer cell (HeLa cell line) cell viability, proliferation, and migration ability. For further study, we explored the possible mechanism behind the P5 compound effect against cervical cancer cells. We performed ligand-protein target prediction using SwissTargetPrediction online platform. We found the EGFR, ERBB2, PTK2, KDR, and PRKCZ as the top 5 proteins targeted by the P5 compound (Table 2). Since the EGFR expression was positively associated with the shorter survival rate of cervical cancer patients but not the ERBB2 (Supplementary Figure S2A and B), we used the EGFR as the target protein for further study. Using AutoDock Vina, we analyzed the binding site and affinity between P5 compound to the EGFR and erlotinib as a standard ligand for EGFR protein. The N-phenyl pyrazoline 5 binding affinity to EGFR was lower than the binding affinity of erlotinib to EGFR (Table 3). The binding conformation of P5 was overlapped with the binding conformation of erlotinib (Figure 4A). The amino acid residues involved in Erlotinib-EGFR and N-phenyl pyrazoline 5-EGFR interaction were provided in Figures 4B and C. The Western blotting results revealed that EGFR expression levels were suppressed by N-phenyl pyrazoline 5 in doses dependent manner, but not the total ERK1/2 (Figure 4D). After confirming that EGFR was the target of P5 compound, we explored the role of EGFR in cervical cancer patients. Based on the TCGA-PanCancer Atlas dataset analysis, we found a significant positive association between the EGFR expression level and the overall survival rate of cervical cancer patients. The EGFR high group showed a lower overall survival rate relative to the EGFR low group (Figure 5A). Using the same groups of cervical cancer patients, we found that EGF, EIF4, IGF1, MET, and IGFR1signaling pathways were enriched in the EGFR high group (Figure 5B–G). Our findings confirm that the EGFR is targeted by P5 compound. Furthermore, the EGFR showed a clinically important role in cervical cancer patients.
 |
Table 2 Protein Target Prediction Results of N-Phenyl pyrazoline 5 Using SwissTargetPrediction Platform |
 |
Table 3 Binding Affinity of Ligand-Target Protein |
The N-Phenyl Pyrazoline 5 Attenuates the Cervical Cancer Stem Cell-Like Phenotype via EGFR Inhibition
The cancer stem cells are a subpopulation of cancer cells that exhibit self-renewal ability and multilineage differentiation. This type of cancer cell contributes to cancer progression, including cervical cancer metastasis. Based on the Pearson correlation test, we found the EGFR mRNA expression level was positively significantly associated with cancer stem cell markers (CD44, MYC, ALDH1A3, and KLF4) (Figure 6A). The P5 suppressed the cervical cancer stem cell-like phenotype. The 10µM and 20 µM of P5 significantly reduced the tumorsphere sizes in HeLa cells (Figure 6B and C). The 1 µM of P5 reduced the mean of tumorsphere size, although the statistical p-values are not significant. Using Western blotting assay, we confirmed that P5 treatment suppressed the cancer stem cell marker, CD133 (Figure 6D). Chemoresistant is one of the characteristics of cancer stem cells including in cervical cancer stem cells. The MTT assay revealed that HeLa cells generated from hanging drop tumorsphere assay exhibit chemoresistant against paclitaxel compared to the HeLa parental cells (Supplementary Figure S1C and D). Further, the P5 sensitized the HeLa cancer stem cell against paclitaxel. Those findings suggest the P5 attenuates the cervical cancer stem cell-like phenotype via EGFR inhibition.
Discussion
Metastasis in cancer patients significantly decreases the survival rate, including in cervical cancer patients. The metastatic cervical patients have approximately 8 to 13 months of median survival time. The cervical patients who develop multiple organ metastasis possess a worse median overall survival time of 5 months. Rapid cell proliferation has been found as one of the critical processes in the initial tumorigenesis, including in cervical cancer. Mostly, the HPV infection was found in cervical cancer patients. In the initiation, the oncoviral proteins disrupt the DNA repair system and apoptosis, leading the uncontrolled cell proliferation. This uncontrolled cell proliferation is required for tumor growth in the primary site as well as distant organ metastasis. Recently, some studies have revealed a critical regulator and potent inhibitor of cell proliferation in cervical cancer.20–22 Pyrazoline and its derivatives are kinds of interesting compounds that possess anti-proliferative and cytotoxic effects in several types of cancer. Previous studies showed that pyrazoline derivatives exhibit anti-cancer activity against other cancer types such as prostate cancer, hepatocellular carcinoma, and breast cancer with 1.3–7.8 µM of IC50 range. The 1,3,5-trisubstituted pyrazolines (pyrazoline derivatives) that were generated from chalcone and hydrazine derivatives also showed cytotoxic effects again breast cancer cells. In this study, we prepared the pyrazoline derivatives called N-phenyl pyrazoline. We also investigated two N-phenyl pyrazoline activities against cervical cancer cells. We found that N-phenyl pyrazoline 5 significantly suppressed the cell viability and cell proliferation of cervical cancer cells with 4.708 µM of IC50.
Metastasis is a serial process involved in cancer cell dissemination from the primary site to the distant organs. The cancer cells undergone EMT could migrate from the primary site to the surrounding tissues, blood vessels and recolonize in the distant organ. Cell proliferation is required for migrated cells to generate the new bulk of tumor tissue in distant organs.24,25 Taken together, the migration ability is important during cancer metastasis, including in cervical cancer. Here, we provide the basic evidence that our N-phenyl pyrazoline 5 inhibits cervical cancer migration. Furthermore, we found that N-phenyl pyrazoline 5 exhibits a better binding affinity to the EGFR than the EGFR inhibitor (erlotinib). This finding is consistent with previous studies that showed that most pyrazoline derivatives act as EGFR inhibitors in molecular docking and in vitro studies.23,26–28
Overexpression of tyrosine kinase receptor, EGFR associated with cancer progression, treatment response, and metastasis in various types of cancer. Non-small cell lung cancer (NSCLC) patients with overexpression of EGFR have the worst prognosis with a shorter survival time. The EGFR overexpression is associated with poor prognosis and a predictive factor for HER2 inhibitor (trastuzumab) treatment. The EGFR overexpression is associated with the decreased survival rate of cervical cancer patients. Consistent with the previous study, our finding showed that cervical cancer patients with high expression of EGFR significantly associated with lower overall survival rates relative to the low EGFR cervical cancer patients. In another study, the overexpression and activation of EGFR are associated with poor response to chemotherapy in cervical cancer. Our GSEA results revealed the positive association of high expression to several signaling pathways such as EGF, ElF4, IGF, and MET signaling pathways. Interestingly, these signaling pathways play a key role in cancer metastasis. Activation of EGF signaling pathway induces migration and epithelial-mesenchymal transition, which are required for cancer cell dissemination in solid tumors, including cervical cancer.33–35 The EIF4 activation induces EMT and metastasis of breast cancer cells. Meanwhile, EIF4 transcription induced by E6 oncoviral protein from HPV increases the cell proliferation and migration in cervical cancer cells. The insulin-like growth factor regulates the invasiveness, proliferation, and cancer stem cells in cervical cancer.38,39 The c-met expression is significantly associated with disease-free survival, lymph node involvement, and lymphovascular space invasion in cervical cancer. Taken together, targeting the EGFR may improve the prognosis of cervical cancer patients with primary and metastasis tumors.
The cancer stem cells are a subset of cancer cells that possess self-renewal ability and multilineage differentiation capacity. These cells are responsible for the progression of cervical cancer, including metastasis. Undergone the EMT, the cancer stem cells invade surrounding tissue and spread to distant organs to generate new bulk of tumor tissue. The EGFR kinase plays an important role in solid tumor cancer stem cells such as breast cancer, head, and neck squamous cell carcinoma, non-small-cell lung cancer, and cervical cancer.41–44 Consistently, we found that EGFR expression is positively associated with cancer stem cell markers. Further, inhibition of EGFR kinase activation markedly reduces the cancer stem cell’s enrichment in cervical cancer cells. The EGFR inhibition also sensitizes the resistant cervical cancer stem cells to paclitaxel based on in vitro and in vivo studies. Interestingly, in the present study, we observed that the N-phenyl pyrazoline treatments also reduce the tumorsphere size in a dose-dependent manner against cervical cancer cells.
In summary, N-phenyl pyrazoline 5 suppressed the cell proliferation and migration of cervical cancer cells. The EGFR is associated with the worst prognosis of cervical cancer patients. The molecular docking model and in vitro assays suggested the N-phenyl pyrazoline 5 acts as an EGFR inhibitor against cervical cancer cells. The N-phenyl pyrazoline 5 attenuates the cancer stem cell-like phenotypes of cervical cancer cells.
Conclusions
Taken together, all findings suggest that N-phenyl pyrazoline 5 acts as an EGFR inhibitor lead to attenuating the aggressiveness and metastasis ability of cervical cancer cells. Further, the N-phenyl pyrazoline 5 treatment sensitizes the cervical cancer stem cell to the chemotherapy agent, paclitaxel. Since this study used one cervical adenocarcinoma cell line (HeLa cell line), these results require further evaluation using different cervical cancer cell lines.
Ethical Approval
Ethical approval for the study was obtained from Medical Health Research Ethics Committee (MHREC) of Faculty of Medicine, Public Health, and Nursing, Universitas Gadjah Mada – Dr. Sardjito General Hospital (approval number: KE/FK/1101/EC/2020).
Acknowledgment
This study was supported by UGM Research Directorate and UGM Reputation Improvement Team towards World-Class Universities-UGM Quality Assurance Office.
Disclosure
The authors declare no conflicts of interest in this work.
References
1. Bray F, Ferlay J, Soerjomataram I, et al. Erratum: Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2020;70(4):313. doi:10.3322/caac.21609
2. Seyfried TN, Huysentruyt LC. On the origin of cancer metastasis. Crit Rev Oncog. 2013;18(1–2):43–73. doi:10.1615/CritRevOncog.v18.i1-2.40
3. Li H, Wu X, Cheng X. Advances in diagnosis and treatment of metastatic cervical cancer. J Gynecol Oncol. 2016;27(4). doi:10.3802/jgo.2016.27.e43
4. Yin Z, Tang H, Li L, et al. Impact of sites versus number of metastases on survival of patients with organ metastasis from newly diagnosed cervical cancer. Cancer Manag Res. 2019;11:7759–7766. doi:10.2147/CMAR.S203037
5. Oishi S, Kudaka W, Toita T, et al. Prognostic factors and treatment outcome for patients with stage ivb cervical cancer. Anticancer Res. 2016;36(7):3471–3475.
6. Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144(5):646–674. doi:10.1016/j.cell.2011.02.013
7. Mendoza-Almanza G, Ortíz-Sánchez E, Rocha-Zavaleta L, Rivas-Santiago C, Esparza-Ibarra E, Olmos J. Cervical cancer stem cells and other leading factors associated with cervical cancer development (review). Oncol Lett. 2019;18(4):3423–3432. doi:10.3892/ol.2019.10718
8. Yusuf M, Jain P. Synthetic and biological studies of pyrazolines and related heterocyclic compounds. Arab J Chem. 2014;7(5):553–596. doi:10.1016/j.arabjc.2011.09.013
9. Kumari P, Mishra VS, Narayana C, Khanna A, Chakrabarty A, Sagar R. Design and efficient synthesis of pyrazoline and isoxazole bridged indole C-glycoside hybrids as potential anticancer agents. Sci Rep. 2020;10(1). doi:10.1038/s41598-020-63377-x
10. Wang H, Zheng J, Xu W, et al. A new series of cytotoxic pyrazoline derivatives as potential anticancer agents that induce cell cycle arrest and apoptosis. Molecules. 2017;22(10). doi:10.3390/molecules22101635
11. Chaudhary M, Kumar N, Baldi A, Chandra R, Babu MA, Madan J. 4-Bromo-4’-chloro pyrazoline analog of curcumin augmented anticancer activity against human cervical cancer, HeLa cells: in silico-guided analysis, synthesis, and in vitro cytotoxicity. J Biomol Struct Dyn. 2020;38(5):1335–1353. doi:10.1080/07391102.2019.1604266
12. Suma AAT, Wahyuningsih TD, Mustofa M. Synthesis, cytotoxicity evaluation and molecular docking study of N-phenylpyrazoline derivatives. Indones J Chem. 2019;19(4):1081–1090. doi:10.22146/ijc.45777
13. Matiadis D, Sagnou M. Pyrazoline hybrids as promising anticancer agents: an up-to-date overview. Int J Mol Sci. 2020;21(15):1–41. doi:10.3390/ijms21155507
14. Sasaki T, Hiroki K, Yamashita Y. The role of epidermal growth factor receptor in cancer metastasis and microenvironment. Biomed Res Int. 2013;2013:1–8. doi:10.1155/2013/546318
15. Hemmat N, Bannazadeh Baghi H. Association of human papillomavirus infection and inflammation in cervical cancer. Pathog Dis. 2019;77(5). doi:10.1093/femspd/ftz048
16. Kim MK, Kim HS, Kim SH, et al. Human papillomavirus type 16 E5 oncoprotein as a new target for cervical cancer treatment. Biochem Pharmacol. 2010;80(12):1930–1935. doi:10.1016/j.bcp.2010.07.013
17. Daina A, Michielin O, Zoete V. SwissTargetPrediction: updated data and new features for efficient prediction of protein targets of small molecules. Nucleic Acids Res. 2019;47(W1):W357–W3664. doi:10.1093/nar/gkz382
18. Zhang Y, Guo Y, Zhou X, Wang X, Wang X. Prognosis for different patterns of distant metastases in patients with uterine cervical cancer: a population-based analysis. J Cancer. 2020;11(6):1532–1541. doi:10.7150/jca.37390
19. Balasubramaniam SD, Balakrishnan V, Oon CE, Kaur G. Key molecular events in cervical cancer development. Med. 2019;55(7). doi:10.3390/medicina55070384
20. Sun B, Zhang Y, Zhou L, et al. The proliferation of cervical cancer is promoted by miRNA-125b through the regulation of the HMGA1. Onco Targets Ther. 2019;12:2767–2776. doi:10.2147/OTT.S197740
21. Li R, Yan Q, Tian P, et al. CBX7 inhibits cell growth and motility and induces apoptosis in cervical cancer cells. Mol Ther. 2019;15:108–116. doi:10.1016/j.omto.2019.09.002
22. Xia C, Liu C, He Z, Cai Y, Chen J. Metformin inhibits cervical cancer cell proliferation by modulating PI3K/Akt-induced major histocompatibility complex class I-related chain A gene expression. J Exp Clin Cancer Res. 2020;39(1). doi:10.1186/s13046-020-01627-6
23. George RF, Kandeel M, El-Ansary DY, El Kerdawy AM. Some 1,3,5-trisubstituted pyrazoline derivatives targeting breast cancer: design, synthesis, cytotoxic activity, EGFR inhibition and molecular docking. Bioorg Chem. 2020;99:103780. doi:10.1016/j.bioorg.2020.103780
24. Fares J, Fares MY, Khachfe HH, Salhab HA, Fares Y. Molecular principles of metastasis: a hallmark of cancer revisited. Signal Transduct Target Ther. 2020;5(1):28. doi:10.1038/s41392-020-0134-x
25. Pachmayr E, Treese C, Stein U. Underlying mechanisms for distant metastasis - molecular biology. Visc Med. 2017;33(1):11–20. doi:10.1159/000454696
26. Sever B, Altıntop MD, Radwan MO, et al. Design, synthesis and biological evaluation of a new series of thiazolyl-pyrazolines as dual EGFR and HER2 inhibitors. Eur J Med Chem. 2019;179:182. doi:10.1016/j.ejmech.2019.111648
27. Alkamaly OM, Altwaijry N, Sabour R, Harras MF. Dual EGFR/VEGFR2 inhibitors and apoptosis inducers: synthesis and antitumor activity of novel pyrazoline derivatives. Arch Pharm. 2021;354(4):2000351. doi:10.1002/ardp.202000351
28. Nawaz F, Alam O, Perwez A, et al. Design, synthesis, molecular docking, and anticancer evaluation of pyrazole linked pyrazoline derivatives with carbothioamide tail as EGFR kinase inhibitors. Anticancer Agents Med Chem. 2020;21(1):42–60. doi:10.2174/1871520620666200727093613
29. Arfaoui A, Kriaa L, Znaidi N, et al. Over-expression of Egfr is closely correlated to poor prognosis in tunisian patients with non-small cell lung Adenocarcinoma. J Immunoass Immunochem. 2014;35(3):256–268. doi:10.1080/15321819.2013.848813
30. Lee HJ, Seo AN, Kim EJ, et al. Prognostic and predictive values of EGFR overexpression and EGFR copy number alteration in HER2-positive breast cancer. Br J Cancer. 2015;112(1):103–111. doi:10.1038/bjc.2014.556
31. Tian WJ, Huang ML, Qin QF, Chen Q, Fang K, Wang PL. Prognostic impact of epidermal growth factor receptor overexpression in patients with cervical cancer: a meta-analysis. PLoS One. 2016;11(7):e0158787. doi:10.1371/journal.pone.0158787
32. Noordhuis MG, Eijsink JJH, Ten Hoor KA, et al. Expression of epidermal growth factor receptor (EGFR) and activated EGFR predict poor response to (Chemo)radiation and survival in cervical cancer. Clin Cancer Res. 2009;15(23):7389–7397. doi:10.1158/1078-0432.CCR-09-1149
33. Ha GH, Kim JL, Breuer EKY. TACC3 is essential for EGF-mediated EMT in cervical cancer. PLoS One. 2013;8(8). doi:10.1371/journal.pone.0070353
34. Schelch K, Vogel L, Schneller A, et al. EGF induces migration independent of EMT or invasion in A549 lung adenocarcinoma cells. Front Cell Dev Biol. 2021:9:484. DOI:10.3389/fcell.2021.634371
35. Kim J, Kong J, Chang H, Kim H, Kim A. EGF induces epithelial-mesenchymal transition through phospho-Smad2/3-Snail signaling pathway in breast cancer cells. Oncotarget. 2016;7(51):85021–85032. doi:10.18632/oncotarget.13116
36. Robichaud N, del Rincon SV, Huor B, et al. Phosphorylation of eIF4E promotes EMT and metastasis via translational control of SNAIL and MMP-3. Oncogene. 2014;34(16):2032–2042. doi:10.1038/onc.2014.146
37. Wang S, Pang T, Gao M, et al. HPV E6 induces eIF4E transcription to promote the proliferation and migration of cervical cancer. FEBS Lett. 2013;587(6):690–697. doi:10.1016/j.febslet.2013.01.042
38. Shen MR, Hsu YM, Hsu KF, Chen YF, Tang MJ, Chou CY. Insulin-like growth factor 1 is a potent stimulator of cervical cancer cell invasiveness and proliferation that is modulated by αvβ3 integrin signaling. Carcinogenesis. 2006;27(5):962–971. doi:10.1093/carcin/bgi336
39. Javed S, Bhattacharyya S, Bagga R, Srinivasan R. Insulin growth factor-1 pathway in cervical carcinoma cancer stem cells. Mol Cell Biochem. 2020;473(1–2):51–62. doi:10.1007/s11010-020-03807-6
40. Peng J, Qi S, Wang P, Li W, Liu C, Li F. Diagnosis and prognostic significance of c-met in cervical cancer: a meta-analysis. Dis Markers. 2016;2016:1–9. doi:10.1155/2016/6594016
41. Lv Y, Cang W, Li Q, et al. Erlotinib overcomes paclitaxel-resistant cancer stem cells by blocking the EGFR-CREB/GRβ-IL-6 axis in MUC1-positive cervical cancer. Oncogenesis. 2019;8(12). doi:10.1038/s41389-019-0179-2
42. Alanazi IO, Khan Z. Understanding EGFR signaling in breast cancer and breast cancer stem cells: overexpression and therapeutic implications. Asian Pacific J Cancer Prev. 2016;17(2):445–453. doi:10.7314/APJCP.2016.17.2.445
43. Codony-Servat J, Codony-Servat C, Cardona AF, et al. Cancer stem cell biomarkers in EGFR-mutation–positive non–small-cell lung cancer. Clin Lung Cancer. 2019;20(3):167–177. doi:10.1016/j.cllc.2019.02.005
44. Abhold EL, Kiang A, Rahimy E, et al. EGFR kinase promotes acquisition of stem cell-like properties: a potential therapeutic target in head and neck squamous cell carcinoma stem cells. PLoS One. 2012;7(2):e32459. doi:10.1371/journal.pone.0032459
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.