Back to Journals » International Journal of Women's Health » Volume 6
A pilot study comparing the DuoFertility® monitor with ultrasound in infertile women
Authors Rollason J, Outtrim J, Mathur R
Received 12 December 2013
Accepted for publication 5 March 2014
Published 16 July 2014 Volume 2014:6 Pages 657—662
DOI https://doi.org/10.2147/IJWH.S59080
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 5
Jennie CB Rollason,1 Joanne G Outtrim,2 Raj S Mathur1
1Cambridge IVF, Addenbrooke's NHS Trust, 2University Division of Anaesthesia, University of Cambridge, Addenbrooke's Hospital, Cambridge, UK
Background: The purpose of this study was to assess the accuracy of ovulation detection by the DuoFertility® monitor compared with transvaginal ultrasound in infertile women with regular menstrual cycles.
Methods: Eight infertile patients, aged 27–40 years, with a body mass index of 19–29, regular menses, normal ovaries on pelvic ultrasound scan, and normal early follicular luteinizing hormone (LH), follicle-stimulating hormone, and prolactin were recruited from infertility clinics in primary and secondary care for this pilot, prospective, observational study. The patients were asked to use the DuoFertility monitor for the whole cycle, with investigators and patients blind to DuoFertility data. Daily urine LH monitoring commenced on cycle day 8, with daily transvaginal ultrasound following the first positive LH until ovulation was observed. Ovulation was further confirmed by serum progesterone. The main outcome measure was detection of ovulation by the DuoFertility monitor, and correlation between day of ovulation assessed by DuoFertility and ultrasound.
Results: DuoFertility identified ovulation as having occurred within one day of that determined via ultrasound in all cycles. The sensitivity of ovulation detection was 100% (95% confidence interval 82–100). The specificity could not be concluded from the data.
Conclusion: In infertile women with regular cycles, the DuoFertility monitor appears to accurately identify ovulatory cycles and the day of ovulation.
Keywords: fertility monitor, ovulation, infertility, ultrasound scan, urinary luteinizing hormone
Introduction
Infertility affects 13%–15% of couples worldwide.1 The use of fertility monitors is widespread in this population. However, these monitors are generally tested on a group of fertile couples to assess their accuracy2–6 rather than on a population of infertile couples.
Guidelines from the UK National Institute of Health and Clinical Excellence state that the use of basal body temperature (BBT) charts to confirm ovulation does not reliably predict ovulation and is not recommended for infertile couples.7 Several studies have assessed the accuracy of traditional BBT methods compared with ovulation (luteinizing hormone) tests by comparing them with transvaginal ultrasound. These studies have reported a lack of reliability of traditional BBT measurements.8,9
The DuoFertility® monitor (Cambridge Temperature Concepts Ltd, Cambridge, UK) is a new electronic device that has been developed to help couples identify their most fertile days to plan intercourse, and has been used in the UK since 2009.10 The DuoFertility monitor measures temperature, heat flow, and movement through an axillary sensor worn by the patient, as seen in Figure 1. This allows quality of sleep to be inferred, and therefore the best time of the night to measure resting BBT can be identified. Thousands of measurements are taken every 24 hours and the patient can visualize a summary temperature on their software showing the most representative temperature of the night. It is plausible that continuous temperature monitoring during the night may offer a more representative indication of BBT than a single temperature measurement upon waking. In addition, a continuous body-worn sensor may increase compliance beyond traditional daily oral measurement on waking, as there is no requirement on the part of the user to remember to use the device at any specific time.
  | Figure 1 DuoFertility® sensor. |
The primary aim of this pilot study was to compare the ability of the DuoFertility monitor to detect ovulation with that of the gold standard method of transvaginal ultrasonography, supplemented by serum progesterone estimation, in a population of infertile women with regular menstrual cycles. A secondary aim was to compare the date of ovulation as estimated by the monitor with the date of ovulation as judged on serial transvaginal ultrasound sonography by an experienced operator. The study was not designed to compare this device with other methods of ovulation detection (BBT or ovulation tests) as these are not the gold standard. This was a pilot study examining the value of this test and therefore needed to be compared with the gold standard.
Materials and methods
The study (Clinical trial registration number NCT01360684, A091932, 10/H0308/35) was approved by the Cambridgeshire Central Research Ethics Committee. Female partners of couples presenting with infertility were recruited from the Fertility Clinic at the Rosie Hospital, Cambridge and the Saffron Walden Community Hospital, Saffron Walden (both secondary care), and the Newnham Walk Surgery, Cambridge (primary care). The inclusion criteria were as follows: trying to conceive for at least 12 months; female aged 18–44 years; female body mass index 19–29; and regular menstrual cycles ranging from 21 to 35 days with less than 7 days variation between cycles in the last 12 months.
Patients who were considered likely to be eligible for the study were provided with preliminary information after their consultation and asked to contact the investigators if they wished to consider participation in the study. Patients who established contact with the investigators were given further information and underwent a screening assessment (pelvic ultrasound scan and early follicular phase follicle-stimulating hormone, luteinizing hormone, and prolactin). The following exclusion criteria were applied: any diagnosed systemic illnesses, including but not restricted to thyroid disease, diabetes, or inflammatory diseases; polycystic ovarian syndrome; endometriosis or other pelvic pathology (including proven tubal disease); taking steroids, including oral contraceptives or anti-inflammatory drugs; ovaries not clearly visualized on transvaginal ultrasound; and unwilling to undergo transvaginal ultrasound scan or to wear the DuoFertility monitor.
A total of eight patients were recruited for the study. Written consent was obtained from all participants in accordance with the approval from the research ethics committee. Women were advised to start testing their first morning sample of urine using luteinizing hormone tests (ClearBlue® ovulation test; Swiss Precision Diagnostics GmbH, Geneva, Switzerland) on day 8 of their cycle and to come for an ultrasound scan on the day of the first positive ovulation test. Transvaginal ultrasound scanning was repeated daily until evidence of ovulation was obtained in the form of collapse of a previously seen follicle. A maximum of four ultrasound scans per cycle was performed on each patient. The presence or absence of free fluid in the pouch of Douglas was noted. Serum progesterone was measured 3–10 days after the presumed date of ovulation. All but three scans of a total of 40 scans were performed by the same operator, which should limit the subjectivity of interpretation that has been reported in several studies.11,12 Transvaginal scans were performed on an ultrasound machine used for monitoring ovarian response in assisted conception cycles (CoreVision Pro model number SSA-350A; Toshiba, Tokyo, Japan).
Patients were asked to wear the DuoFertility monitor continuously from day 1 of their menstrual cycle. The monitor could be removed for bathing/swimming if the patient wished to do so, but this was not required. Data from the monitor were downloaded to the DuoFertility handheld monitor and the data were automatically transferred to the DuoFertility servers. The DuoFertility monitor uses proprietary algorithms to automatically identify the date of ovulation for a particular cycle and patient. Although the DuoFertility monitor can accept additional user input such as home luteinizing hormone tests or cervical mucus, no data beyond the temperature and movement data collected by the sensor were used in this study. The date of ovulation generated by the monitor was used as the date of ovulation identified by DuoFertility.
Patients and investigators performing the ultrasound scans were blind to the results obtained by the DuoFertility monitor until the end of the study. Interpreters of the DuoFertility data were blind to the ultrasound and progesterone results. Following unblinding, the following comparisons were made: ovulation identified by DuoFertility versus ovulation identified by serial transvaginal ultrasound supplemented by serum progesterone measurement; and date of ovulation identified by DuoFertility versus date of ovulation identified by serial transvaginal ultrasound.
Statistical analysis was performed using the R statistical package.13 As recommended by Brown et al14 for the challenging case of small n and P~1, the Wilson score interval15 was used to estimate the confidence interval for sensitivity in this study. It should be noted that, in such analysis, each menstrual cycle is considered independently, which may not be correct in cases where women contributed more than one cycle to the study.
Results
The inclusion of patients for this study is detailed in Figure 2. Ten patients gave consent, of whom eight eventually took part in the study. One of the two patients who consented but did not take part in the study conceived in the cycle before she was due to start and the other withdrew for personal reasons. One of the participants contributed to one included cycle (she became pregnant in the first cycle), three participants contributed to three included cycles, and two participants contributed to four included cycles.
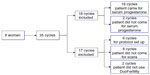  | Figure 2 Patient inclusion. |
A total of 35 cycles was collected, of which 17 were excluded from analysis and 18 cycles were included. The reasons for excluding these cycles were as follows: six cycles were used to set up the protocol and were not included in the analysis because no data were collected on the DuoFertility monitor. Eight cycles could not be included because the patients did not come in for ultrasound scans at the required time. Two cycles were not included because the patients did not use the DuoFertility monitor.
For all cycles included in this study, at least one scan was performed prior to ovulation, and ovulation was documented by follicular collapse on subsequent measurement. Free fluid in the pouch of Douglas was documented in nine cycles. For 16 of the cycles, a blood test 7 days following ovulation was performed to measure the progesterone level, with all results above 19.8 nmol/L confirming ovulation. The other two patients did not come for their blood test.
In every cycle for which ultrasound identified ovulation, DuoFertility also identified that ovulation had occurred. Although the data imply 100% sensitivity (ie, a 0% false-negative rate), it is possible to calculate the expected worst-case sensitivity from the observed data. The lower bound of a Wilson score15 95% confidence interval for the sensitivity as a binomial parameter can be calculated as 82%, implying a worst-case sensitivity of 82%. This assumes statistical independence of cycles, which may not be correct as some women contributed multiple cycles to the study. There were no anovulatory cycles observed in this data set, as patients who do not ovulate are not the target user group of the DuoFertility monitor, so a measure of specificity (ie, false-positive rate) is not possible from the data collected in this pilot study.
Figure 3 shows the temporal correlation between the day of ovulation as identified by ultrasound scanning and the DuoFertility monitor. In Figure 3, “−1” indicates that the DuoFertility monitor has identified ovulation on the day between the last scan showing the dominant follicle and the scan showing collapse of the follicle. “0” indicates that the DuoFertility monitor identified ovulation on the day of the follicle collapse. Finally “+1” indicates that the DuoFertility monitor identified ovulation as occurring between the day of collapse of the follicle and the following day. The result shows a 100% correlation of the results at ±24 hours, which is the best resolution that could be archived using our experimental design.
  | Figure 3 Correlation between ultrasound scan and DuoFertility®. |
Discussion
The aim of this study was to compare the accuracy of ovulation detection by the DuoFertility monitor with that of transvaginal ultrasonography. Ovulation was further confirmed by serum progesterone measurements. This appears to be the first study looking at the accuracy of a home fertility monitor performed in a group of infertile patients. The authors believe that this is an important distinction because patients using fertility monitors are (in the vast majority of cases) infertile, and thus may not be well represented by the general (fertile) population.
The data suggest that such a monitoring technique may be useful for identifying ovulation in infertile women with regular cycles. However, the use of fertility monitors in general should not be a substitute for regular intercourse as recommended by the UK National Institute of Health and Clinical Excellence guidelines.
In this study, we used collapse of the follicle as seen on transvaginal ultrasound imaging as a marker of ovulation, and use this as a reference point to compare the ovulation date provided by the DuoFertility monitor. Daily ultrasound imaging is routinely used in assisted reproductive technology cycles to monitor growth of the follicles, and can also be used in natural cycles to identify the occurrence of ovulation. Interpretation of the images can be subjective, and when possible should be performed by the same operator.16 The use of transvaginal ultrasonography is recommended for greater accuracy and reliability than is possible with abdominal ultrasonography.17 This pilot study shows that the results for the DuoFertility, which are based on high-resolution temperature measurements, appear to identify ovulation as accurately as a series of transvaginal ultrasound scans.
This study challenges the received wisdom that body temperature measurement is not useful for monitoring ovulation. Even though previous studies have not found BBT to be a reliable method of ovulation detection, technological advances as represented by the DuoFertility monitor may mean that this parameter should be re-examined for its clinical value. It is important to note that use of fertility monitors has been implicated as a potential cause of emotional stress.18 However, this relates to methods based on manual recording of temperature or urinary luteinizing hormone by the patient,19,20 rather than this novel method in which data are recorded automatically while the patient is asleep.
The authors acknowledge that there are several limitations to this study design. Firstly, the study was restricted to women with regular cycles and no anovulatory cycles were observed, as a result of which it was not possible to judge the specificity of ovulation detection (false-positive rate) by DuoFertility. Secondly, both the daily ultrasound measurements and the DuoFertility monitor have a resolution of 24 hours, so the resolution of the results cannot be more precise than ±24 hours. Finally, the number of patients included in the study was small. This is partly due to the nature of the population of patients from which the subjects were recruited, as many eligible patients in the hospital fertility clinic chose to move to in vitro fertilization rather than participate in the study, and the fact that a series of daily ultrasound scans can be relatively invasive. As this was a pilot study, we did not perform a formal sample size calculation.
Despite these limitations, this study suggests that the DuoFertility monitor, possibly as a result of increased frequency of temperature measurement, appears to be an accurate method for ovulation detection in infertile women with regular cycles, and comparable with the gold standard of transvaginal ultrasound scanning supplemented with serum progesterone. The use of new technology to continuously monitor temperature in a convenient manner (DuoFertility is a small sensor worn under the arm on a small adhesive pad) allows accurate identification of the date of ovulation with minimal invasiveness. Further investigations to overcome the limitations of this pilot study, in particular relating to the size of the study, number of cycles per woman monitored, inclusion of women with anovulatory cycles, and inclusion of women with irregular cycles, should be considered. We are currently planning a further trial of this device in women with irregular cycles who may or may not be ovulatory.
DuoFertility could provide an alternative means of monitoring ovulation in some particular cases. It has been suggested by the American Society for Reproductive Medicine practice committee21 that couples who have infrequent intercourse may in particular benefit from the use of devices that can predict or determine the time of ovulation. Because DuoFertility is a noninvasive method for the patient to use at home, with data transmitted over the Internet, we speculate that some patients may find it at least as, if not more, acceptable than a more clinical method such as transvaginal ultrasound monitoring, with potential benefits for patient compliance and data collection.
Acknowledgments
The authors are grateful to Cambridge Temperature Concepts Ltd, Cambridge, UK, for help with the DuoFertility information. Administrative support for the study was provided by Amie Ashley. Kate Waldock, Ann Loudon, and Ericka Spooner are acknowledged for performing some of the ultrasound scans. Statistical support was provided by Richard Samworth, Statistical Laboratory, Centre for Mathematical Sciences, University of Cambridge. Dr Pauline Brimblecombe of Newnham Walk Surgery, Cambridge, and Dr Imogen Shaw of Freshwell Health Centre, Braintree, are thanked for their referral of patients.
Disclosure
The authors report no conflicts of interest in this work, and alone are responsible for the content and writing of the paper.
References
World Health Organization. Report of the Meeting on the Prevention of Infertility at the Primary Health Care Level. Geneva, Switzerland: World Health Organization; 1983. | |
Behre HM, Kuhlage J, Gassner C, et al. Prediction of ovulation by urinary hormone measurements with the home use ClearPlan Fertility Monitor: comparison with transvaginal ultrasound scans and serum hormone measurements. Hum Reprod. 2000;15(12):2478–2482. | |
Schmutzler RK, Diedrich K, Krebs D. [Value of the Clearplan ovulation test in sterility treatment]. Geburtshilfe Frauenheilkd. 1995;55(5):266–269. German. | |
Freundl G, Bremme M, Frank-Herrmann P, Baur S, Godehardt E, Sottong U. The CUE Fertility Monitor compared with ultrasound and LH peak measurements for fertile time ovulation detection. Adv Contracept. 1996;12(2):111–121. | |
Freundl G, Frank-Herrmann P, Bremme M. Results of an efficacy-finding study (EFS) with the computer-thermometer Cyclotest 2 plus containing 207 cycles. Adv Contracept. 1998;14(4):201–207. | |
Freundl G, Frank-Herrmann P, Godehardt E, et al. Retrospective clinical trial of contraceptive effectiveness of the electronic fertility indicator Ladycomp/Babycomp. Adv Contracept. 1998;14(2):97–108. | |
National Collaborating Centre for Women’s and Children’s Health 2004. Available from: http://www.nice.org.uk/guidance/cg156/resources/guidance-fertility-pdf. Accessed July 7, 2014. | |
Leader A, Wiseman D, Taylor PJ. The prediction of ovulation: a comparison of the basal body temperature graph, cervical mucus score, and real-time pelvic ultrasonography. Fertil Steril. 1985;43(3):385–388. | |
Guida M, Tommaselli GA, Palomba S, et al. Efficacy of methods for determining ovulation in a natural family planning program. Fertil Steril. 1999;72(5):900–904. | |
Chausiaux O, Hayes J, Long C, et al. Pregnancy prognosis in infertile couples on the DuoFertility® programme compared with IVF/ICSI. Eur J Obstet Gynaecol. 2011;6(2):92–94. | |
Metcalfe SC, Evans JA. A study of the relationship between routine ultrasound quality assurance parameters and subjective operator image assessment. Br J Radiol. 1992;65(775):570–575. | |
Catalano MF, Sivak MV, Bedford RA, et al. Observer variation and reproducibility of endoscopic ultrasonography. Gastrointest Endosc. 1995;41(2):115–120. | |
R Development Core Team. A Language and Environment for Statistical Computing. Vienna, Austria: R Foundation for Statistical Computing; 2009. | |
Brown LD, Cai TT, DasGupta A. Interval estimation for a binomial proportion. Stat Sci. 2001;16(2):2101–2133. | |
Wilson EB. Probable inference, the law of succession, and statistical inference. J Am Statist Assoc. 1927;22(158):209–212. | |
Gerris J, De Sutter P. Self-operated endovaginal telemonitoring (SOET): a step towards more patient-centred ART? Hum Reprod. 2010;25(3):562–568. | |
Raine-Fenning N, Jayaprakasan K, Clewes J, et al. SonoAVC: a novel method of automatic volume calculation. Ultrasound Obstet Gynecol. 2008;31(6):691–696. | |
Lenzi A, Lombardo F, Salacone P, Gandini L, Jannini EA. Stress, sexual dysfunctions, and male infertility. J Endocrinol Invest. 2003; 26(Suppl 3):72–76. | |
Severy LJ, Robinson J, Findley-Klein C, McNulty J. Acceptability of a home monitor used to aid in conception: psychosocial factors and couple dynamics. Contraception. 2006;73(1):65–71. | |
Eichner SF, Timpe EM. Urinary-based ovulation and pregnancy: point-of-care testing. Ann Pharmacother. 2004;38(2):325–331. | |
Practice Committee of the American Society for Reproductive Medicine. Optimizing natural fertility: a committee opinion. Available from: http://www.sart.org/uploadedFiles/ASRM_Content/News_and_Publications/Practice_Guidelines/Committee_Opinions/optimizing_natural_fertility(1).pdf. Accessed June 10, 2014. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
