Back to Journals » Journal of Pain Research » Volume 16
A Pain That is Easily Overlooked: Referred Pain Caused by OVCF
Authors Lu W , Teng Z, Chen J, Shi R , Zou T, Gao M, Li W, Peng Z, Wang L, Zhao Y , Lu S
Received 3 June 2022
Accepted for publication 25 February 2023
Published 17 March 2023 Volume 2023:16 Pages 961—971
DOI https://doi.org/10.2147/JPR.S375966
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Erika Petersen
Wei Lu,1– 3 Zhaowei Teng,1,2 Jiayu Chen,1– 3 Rongmao Shi,4 Tiannan Zou,1– 3 Ming Gao,1– 3 Weichao Li,1– 3 Zhi Peng,1– 3 Long Wang,1– 3 Yonghui Zhao,1– 3 Sheng Lu1– 3
1Yunnan Key Laboratory of Digital Orthopedics, the First People’s Hospital of Yunnan Province, Kunming, People’s Republic of China; 2Department of Orthopedics, the First People’s Hospital of Yunnan Province, Kunming, People’s Republic of China; 3Department of Orthopedics, the Affiliated Hospital of Kunming University of Science and Technology, Kunming, 650032, People’s Republic of China; 4Department of Orthopedics, Clinical College of the 920th Hospital of Joint Logistics Support Force of Kunming Medical University, Kunming, People’s Republic of China
Correspondence: Sheng Lu, Department of Orthopedics, the First People’s Hospital of Yunnan Province, No. 157 Jinbi Road, Kunming, 650032, People’s Republic of China, Tel +86-0871-63633071, Fax +86-0871-63633071, Email [email protected]
Purpose: The objective of this study was to analyze the clinical characteristics and the therapeutic effects of treatment at our spinal center in OVCF patients associated with referred pain. The underlying goals were to deepen the understanding of referred pain caused by OVCFs, improve the currently low early diagnosis rate of OVCFs, and improve the effectiveness of treatment.
Methods: The patients who had referred pain from OVCFs and met the inclusion criteria were retrospectively analyzed. All patients were treated with percutaneous kyphoplasty (PKP). Visual analog scale (VAS) scores and Oswestry Disability Index (ODI) were used to evaluate the therapeutic effect at different time points.
Results: There were 11 males (19.6%) and 45 females (80.4%). Their corresponding mean bone mineral density (BMD) value was − 3.3 ± 0.4. The regression coefficient of BMD in the linear regression equation was − 4.51 (P< 0.001). According to the classification system for referred pain in OVCFs, there were 27 cases of type A (48.2%), 12 cases of type B (21.2%), 8 cases of type C (14.3%), 3 cases of type D (5.4%), and 6 cases of type E (10.7%). All patients were followed up for at least 6 months, and both VAS scores and ODI were found to be significantly better postoperatively than preoperatively (P< 0.001). There was no significant difference in VAS scores and ODI between different types preoperatively or 6 months postoperatively (P > 0.05). Within each type, there were significant differences in VAS scores and ODI between the pre- and postoperative timepoints (P < 0.05).
Conclusion: Attention should be paid to referred pain in OVCF patients, which is not uncommon in clinical practice. Our summary of the characteristics of referred pain caused by OVCFs can improve the early diagnosis rate of OVCFs patients and provide a reference for their prognosis after PKP.
Keywords: referred pain, radicular pain, vertebral fracture, percutaneous kyphoplasty, osteoporosis, spinal fracture
Introduction
With the global advent of an aging society, nearly 200 million osteoporosis patients are diagnosed every year, and nearly 9 million osteoporotic fractures occur worldwide, depriving patients of independence and the ability to carry out activities of daily living. These injuries also place a burden on public health systems.1,2
Osteoporotic vertebral compression fractures (OVCFs) have a lower early diagnosis rate than hip or wrist fractures, with only one-third of OVCF patients receiving a clinical diagnosis in a timely manner.3,4 The most likely reason is that some cases of OVCF do not coincide with a significant history of trauma and may be associated with spinal degenerative disease instead; in those cases, patients may fail to seek medical treatment and may not undergo timely imaging examination. A considerable number of patients with OVCFs often complain of lumbosacral, buttock, iliac crest and even leg pain despite a lack of obvious nerve compression on imaging.
Some articles have identified OVCFs as the cause of this pain phenomenon,5–9 which is also called nonmidline pain by some researchers.5 Additionally, some articles have reported that low back pain and somatic referred pain are common, while radicular pain is uncommon. Radicular pain, strictly defined, has an incidence of only 12% or less.10 This also suggests that the actual clinical diagnosis and treatment may not clearly distinguish between referred pain and radicular pain. Therefore, the existing research data may not be completely accurate.
To improve the unsatisfactory early diagnosis rate of OVCFs, we should deepen the current understanding of its referred pain symptoms to improve the effects of treatment. Therefore, in this study, we retrospectively analyzed the clinical characteristics and treatment outcomes of OVCF patients with referred pain admitted to our spinal center.
Materials and Methods
This study was approved by the Medical Ethics Committee of the First People’s Hospital of Yunnan Province and conformed to the principles of the Declaration of Helsinki (KHLL2020-KY041). The participants were asked to provide written informed consent before study commencement. A total of 173 patients with OVCFs from June 2015 to October 2019 were included in this study. Among them, 56 (32.37%) were OVCF patients with somatic referred pain. These subjects underwent X-ray, magnetic resonance imaging (MRI) and computerized tomography (CT) examination of the spine; bone mineral density (BMD) was also measured using dual-energy X-ray absorptiometry (DXA). Finally, all patients were carefully asked about their disease history and underwent a physical examination by a spine specialist. The results of the imaging and physical examinations were combined to diagnose OVCF and determine which vertebral body was causing pain.
Inclusion Criteria
1. Age over 50 years for postmenopausal females or 60 years for males; 2. a definite diagnosis of OVCF (low-energy injury; BMD: T value less than −2.5), with obviously attributable somatic referred pain in addition to pain on physical percussion of the responsible vertebral body; 3. clearly painful single-segment vertebral fracture on MRI or nonunion on CT or MRI with intact posterior vertebral walls; 4. detailed medical history indicating that conservative treatment was ineffective for back pain; and 5. hospitalization due to referred pain and a physical condition permitting surgery.
Exclusion Criteria
1. History of previous spinal surgery; 2. tumor, infection, or symptomatic degenerative disease of the spine, such as spinal stenosis or lumbar spondylolisthesis; 3. clear symptoms of spinal canal compression or radicular irritation; 4. high-energy injury, pathological fracture, or multisegmental fracture; 5. involvement of pain caused by other diseases, such as psychological or psychiatric disorders; and 6. inability complete MRI due to conditions such as claustrophobia or artificial pacemaker implantation.
Classification of Somatic Referred Pain in OVCF
Patients who met the inclusion criteria reported the location of their pain by drawing. Somatic referred pain can be divided into 5 types based on location. The criteria are as follows:
Type A: located in the lumbosacral and buttock regions;
Type B: located on the side or front of the waist (abdomen, iliac crest or groin region);
Type C: located on the anterolateral or posterior part of the thigh;
Type D: beyond the knee joint, on the posterior or anterolateral part of the calf;
Type E: any two or more of types A-C, excluding type D (Figure 1).
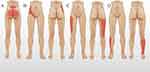 |
Figure 1 Schematic diagram of type A-D referred pain. |
Operation and Outcome Measures
All confirmed patients underwent percutaneous kyphoplasty (PKP) for treatment. The patient was in the prone position, and the abdomen was suspended. After the operation site was marked, a sterile drape was applied, and local anesthesia was administered. After locating the vertebral body and puncture point by fluoroscopy with a C-arm machine, the surgeon established a transpedicular working channel under fluoroscopic guidance and confirmed it by unilateral puncture. For kyphoplasty, a balloon was used to expand the vertebral body; the process was observed closely until the height of the vertebral body was satisfactory. Then, the balloon was removed, and polymethyl methacrylate (PMMA) with a toothpaste-like consistency was injected into the space. During this process, care was taken to avoid leakage. After the above process was completed, the patient remained on the operating table until the cement hardened.
Patients’ visual analog scale (VAS) scores and Oswestry Disability Indexes (ODIs) were analyzed at several different timepoints, namely, the preoperative (Pre), immediately postoperative (PO), 1-month postoperative (PO1M), 3-month postoperative (PO3M) and 6-month postoperative (PO6M) points.
Statistical Analysis
Statistical Package for the Social Sciences version 22.0 was used for statistical analysis (SPSS, Inc., Chicago, IL, USA). All measurement data conformed to a normal distribution, as verified by the Shapiro‒Wilk test, and the variance was homogeneous. The VAS scores and ODIs at different time points and in different groups are described as the mean and standard deviation (X ± SD). Linear regression analysis was used to determine the relationship between BMD and the time interval from injury to referred pain. The VAS scores and ODIs were compared among different groups and time points by repeated-measures analysis of variance (ANOVA). If the assumption of sphericity held true according to Mauchly’s test (P > 0.05), no correction was needed. If the assumption of sphericity was not met (P < 0.05), a Greenhouse‒Geisser, Huynh-Feldt or lower-bound correction was applied. If there was no significant interaction between time and type (P > 0.05), the main effects of the VAS score and ODI between different times and types were analyzed (P < 0.05). If the interaction was statistically significant (P < 0.05), the individual effects of the VAS score and ODI were compared within times and groups. Comparisons were made between groups and between timepoints using Bonferroni-corrected significance thresholds.
Results
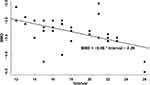
The demographic characteristics of the patients included in this study are presented in Table 1. The patients comprised 11 males (19.6%) and 45 females (80.4%). The age of these subjects ranged from 55 to 91 years (mean: 73.2±7.2 years), and the duration of symptoms ranged from 3 to 60 days (mean: 23.1±14.2 days). Among the 38 patients (67.9%) with a definite history of injury, the interval between injury and referred pain was 12–26 days (mean: 18.1 ± 3.8 days), and the corresponding average BMD was −3.3 ± 0.4. The coefficient of BMD in the linear regression equation was −4.51 (P<0.001) (Figure 2).
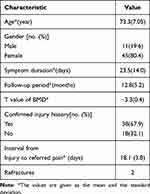 |
Table 1 Data of Patients Participating in the Study (n=56) |
The levels of major vertebral fractures causing referred pain were L1 (n = 18), followed by T11 (n =7), L3 (n = 7), T10 (n = 6), L2 (n = 6), T9 (n = 2) and L4 (n=1). According to the classification of referred pain in OVCFs, there were 27 cases of type A (48.2%), 12 cases of type B (21.2%), 8 cases of type C (14.3%), 3 cases of type D (5.4%), and 6 cases of type E (10.7%) pain (Figure 3).
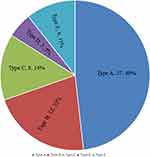 |
Figure 3 Diagram demonstrating the distribution of OVCFs associated with referred pain in this study. |
All patients were treated with PKP. There were no serious intraoperative complications, although there were 6 cases of slight bone cement leakage. Follow-up lasted for at least 6 months, ranging from 6–33 months (mean: 12.8 ± 5.2 months).
The trends in the VAS score and ODI over time by are shown in Table 2. The VAS score and ODI at different stages postoperatively were significantly improved compared with the preoperative values (Table 3, P<0.001).
 |
Table 2 Baseline Data of VAS and ODI at Different Time of Different Types (X ± SD) |
 |
Table 3 When Time is Fixed, Two Comparisons are Used for VAS and ODI Effects of Different Types |
A comparison of different types within time points by repeated-measures ANOVA is shown in Table 4. There was no significant difference in the VAS score among different types at the Pre or PO6M timepoint (P > 0.05); type D was significantly different from types A, B and C at the PO timepoint, with P values of 0.023, 0.026, and 0.026, respectively. There was a significant difference between type D and type C at the PO3M timepoint (P = 0.045).
 |
Table 4 When the Type is Fixed, Two Comparisons are Used for the Vas and ODI Effects of Different Types |
In addition, at the Pre and PO6M timepoints, there was no significant difference in the ODI between different types (P > 0.05); at the PO timepoint, there was a significant difference between type D and types A, B and C, with P values of 0.011, 0.009, and 0.04, respectively. At the PO1M timepoint, there was a significant difference between type D and types A and B, with P values of 0.011 and 0.015, respectively. At the PO3M timepoint, type D was significantly different from types A, B, C and E, with P values of 0.001, 0.002, 0.003, and 0.038, respectively.
Within types, the individual effects at different times were compared, as shown in Table 3. Across different types, there were significant differences in the VAS score and ODI between the Pre and PO timepoints (P < 0.05). Additionally, in type D, the ODI was significantly different between the PO3M and PO6M timepoints (P = 0.013); there was no significant difference between these timepoints in the other groups (P > 0.05). During the follow-up period, two patients suffering from refracture were treated with PKP.
Discussion
Referred pain caused by OVCFs has not been seriously considered by clinicians and can easily be missed or misdiagnosed. There is little evidence to directly categorize the symptoms and analyze the effects of treatment to help surgeons in their decision-making process. To address this deficiency, the current study included 56 OVCF patients who had associated referred pain and had completed at least 6 months of follow-up.
In this study, we observed that approximately 95% of patients had pain that did not cross from the knee joint to the calf. Among them, there were cases of type A (48%), B (22%), C (14%) and E (11%) pain, with types A and B alone accounting for 70%.
Similarly, a few studies have noted similar phenomena before. Friedrich et al7 investigated the pain of 51 patients with OVCFs from T8 to L2. Of these patients, 88.2% suffered from lumbosacral and buttock pain, and 31.4% suffered from lumbosacral or buttock pain after thoracic fracture.7 In 2010, a cohort study of 504 postmenopausal women examined pain in the lateral waist area after vertebral fracture and found it to be associated with a 4.5-fold increase in the risk of vertebral fracture.11 Therefore, the lumbosacral, buttock, iliac crest and groin regions are common areas for the occurrence of referred pain caused by OVCFs.
It is generally believed that sensory innervation is the basis for pain. The source of bone pain is the periosteum. The periosteum of the vertebral body is innervated by sympathetic and sinus vertebral nerves. Some studies have also confirmed the presence of nerves in the vertebrae.12–15 However, the presence of sensory innervation in the cancellous bone of the vertebral body remains controversial. On the one hand, Buonocore et al16 agreed that there were only a small number of nerves in the vertebral body, and most of them were sympathetic nerves. Therefore, the role of nerves within bones in low back pain is questionable. On the other hand, Fagan et al17 showed that basivertebral nerves are CGRP positive and belong to the category of nociceptors. In vertebroplasty, nerve ablation plays a part in relieving pain.18 Even vertebral perforation can relieve the pain caused by vertebral compression fractures.19 Meanwhile, basivertebral nerve ablation can significantly relieve the symptoms of chronic low back pain.20,21 These findings also substantiate the presence of sensory nerves (SNs) in the vertebral body. According to the convergence-projection hypothesis, the afferent nerve fibers from two different regions converge into the same secondary neurons in the spinal cord, leading to the central nervous system’s mislocalization of the source of pain.22–24 The SN fibers of lumbar vertebrae are dominated by the dorsal root ganglia of upper multisegmental levels. In nonsegmental innervation, SN fibers enter the paraspinal sympathetic trunk and reach the L2 dorsal root ganglion.15,25,26 Therefore, the convergent projection theory can be used to explain the pain phenomenon observed in our study.
In addition, type D pain accounts for 5% and has rarely been reported in the literature. It is generally believed that the pain will not extend beyond the knee joint. While stimulation of the sciatic nerve roots should occur somewhere, the MRI results of this type of case in this study cannot confirm stimulation of the nerve roots. As early as 2002, O’Neill et al27 found that noxious stimulation of the intervertebral disc by intradiscal electrothermal annuloplasty (IDET) resulted in the same observed pattern of referred pain as noxious stimulation of other somatic structures of the spine. Interestingly, the range of referred pain may be related to the intensity of stimulation. Within a certain range, as the stimulation time and temperature increase, the distance of pain referral can increase as well. Moreover, the neural mechanisms of referred pain have not yet been completely clarified.27,28 Therefore, some researchers have proposed the concept of dichotomizing axons.15 However, there is still a lack of corresponding experimental techniques to directly observe and demonstrate the concept.
In our study, osteoporosis was found in all patients, most of whom were female. The findings also suggest that there is a significant relationship between referred pain and osteoporosis. SNs play an important role in the regulation of bone homeostasis and regeneration as well as nociceptors. Burt-Pichat et al29 found that SN innervation and bone mass2 were reduced after ovariectomy in rats. However, SN activity increases during fracture healing.30 In the process of repair after fracture, nerves regulate the dynamic rearrangement of bone tissue in a manner similar to that during the development process. Following fracture, before vascularization and ossification, fibers sprout in the cartilage formation area. Around the hematoma and periosteum, many regenerating GAP43+ axons sprout.31 Numerous CGRP+ and SP+ sensory fibers appear from the deep periosteum, terminate at the end of the thin varicose vein in the cartilage callus, and penetrate the newly formed woven bone around the fracture site.32–34 Usually, as healing progresses, SN fibers withdraw. The patients included in this study had osteoporosis and a decreased SN fiber distribution. The sensitivity of osteoblasts to mechanical signals decreased, as did angiogenesis and the abundance of mesenchymal stem cells, and there was local mechanical instability in the vertebral body where fracture occurred.35 The patients in our study had no characteristic symptoms in the early stage of injury and had significant pain 2–3 weeks later. Our study also found a relationship between the interval from injury to referred pain and the corresponding BMD. The regression coefficient of BMD was −4.51, confirming the relationship between the above basic research conclusions and the interval from injury to referred pain. In other words, symptoms occur at almost exactly the same stage as callus formation, seemingly fulfilling the adage, “no pain, no gain”. Delayed referred pain is a characteristic symptom in OVCF patients. However, this view still needs further support by relevant basic research.
Based on patient histories, we summarized the key points that distinguish referred pain caused by OVCFs: a) The referred pain usually begins later than the local pain of the fractured vertebral body, seldom appearing at the same time. b) The pain is alleviated when the patient lies flat, and it is aggravated by turning over to stand up or by walking. c) Percussion fracture of the vertebral body may induce referred pain, and the range of referred pain may be related to percussion strength. d) The referred pain area is inconsistent with the dermatome map. e) There is no numbness or hypoesthesia in the referred pain area. Therefore, it is very important to fully understand the types of pain in OVCF patients and to inquire carefully about each patient’s history to understand the changes in pain over time and improve the initial OVCF diagnosis rate.
Currently, vertebroplasty is the most widely used method to treat OVCFs. As a filling agent, bone cement can provide immediate stability through microinterlock formation, volume filling and bulk filling mechanisms to reduce the mechanical stimulation of nerves. In addition, heat produced in the polymerization process can cause nerve tissue necrosis.36 In a retrospective study by Gibson et al,5 206 patients with nonmidline pain were treated with vertebroplasty, and 83% of them experienced relief. Yang et al9 reported that 11 OVCF patients with distal pain were treated with PKP, and the VAS score and ODI were significantly improved after the operation. Therefore, in our study, patients achieved good results. However, at the PO, PO1M, and PO3M timepoints, there were some differences in the VAS score and ODI between type D and other types (Table 4). This also suggests that the prognosis of this type of patient is worse than that of other types of patients.
There are several limitations to this study. There is a certain relationship between the amount and distribution of bone cement and the efficacy. Because these indicators were not included in the statistics in this study, there may be bias or influence on the comparison of the VAS score and ODI. Only some bone metabolic indexes were monitored in these patients, and these data could not further explain the association between referred pain and osteoporosis. In addition, this study was a retrospective study. There may have been selection bias in the inclusion of patients, and the sample size was limited, especially for types D and E.
Conclusions
Attention should be paid to referred pain in OVCF patients, which is not uncommon in clinical practice. Our summary of the characteristics of referred pain caused by OVCFs can improve the early diagnosis rate of OVCFs and provide a reference for the prognosis of PKP after treatment. In the future, we will expand the sample size; pursue more detailed observation indicators and a higher level of evidence; continue to improve the classification; and use tissue transparency technology to study the distribution of vertebral nerves and other peripheral nerves to further explore the mechanism of referred pain caused by OVCFs.
Acknowledgments
We appreciate the contribution of all patients, their families, the investigators, and the medical staff. We are grateful for the contributions of all authors.
Funding
This study was supported by the Joint Special Fund of Applied Fundamental Research of Kunming Medical University granted by the Science and Technology Office of Yunnan, grant no. 202001AY070001-172.
Disclosure
The authors declare that they have no conflicts of interest.
References
1. Feron JM, Mauprivez R. Fracture repair: general aspects and influence of osteoporosis and anti-osteoporosis treatment. Injury. 2016;47(Suppl 1):S10–S14. doi:10.1016/S0020-1383(16)30003-1
2. Singer A, Exuzides A, Spangler L, et al. Burden of illness for osteoporotic fractures compared with other serious diseases among postmenopausal women in the United States. Mayo Clin Proc. 2015;90(1):53–62. doi:10.1016/j.mayocp.2014.09.011
3. Ross PD. Clinical consequences of vertebral fractures. Am J Med. 1997;103(2A):
4. Lyles KW, Gold DT, Shipp KM, Pieper CF, Martinez S, Mulhausen PL. Association of osteoporotic vertebral compression fractures with impaired functional status. Am J Med. 1993;94(6):595–601. doi:10.1016/0002-9343(93)90210-G
5. Gibson JE, Pilgram TK, Gilula LA. Response of nonmidline pain to percutaneous vertebroplasty. AJR Am J Roentgenol. 2006;187(4):869–872. doi:10.2214/AJR.05.0084
6. Patel U, Skingle S, Campbell GA, Crisp AJ, Boyle IT. Clinical profile of acute vertebral compression fractures in osteoporosis. Br J Rheumatol. 1991;30(6):418–421. doi:10.1093/rheumatology/30.6.418
7. Friedrich M, Gittler G, Pieler-Bruha E. Misleading history of pain location in 51 patients with osteoporotic vertebral fractures. Eur Spine J. 2006;15(12):1797–1800. doi:10.1007/s00586-006-0065-4
8. Doo TH, Shin DA, Kim HI, et al. Clinical relevance of pain patterns in osteoporotic vertebral compression fractures. J Korean Med Sci. 2008;23(6):1005–1010. doi:10.3346/jkms.2008.23.6.1005
9. Yang YM, Ren ZW, Ma W, Jha RK. Kyphoplasty for the treatment of pain distant to osteoporotic thoracolumbar compressive fractures. Cell Biochem Biophys. 2014;68(3):523–527. doi:10.1007/s12013-013-9732-3
10. Bogduk N. On the definitions and physiology of back pain, referred pain, and radicular pain. Pain. 2009;147(1–3):17–19. doi:10.1016/j.pain.2009.08.020
11. Clark EM, Hutchinson AP, McCloskey EV, et al. Lateral back pain identifies prevalent vertebral fractures in postmenopausal women: cross-sectional analysis of a primary care-based cohort. Rheumatology. 2010;49(3):505–512. doi:10.1093/rheumatology/kep414
12. Antonacci MD, Mody DR, Rutz K, Weilbaecher D, Heggeness MH. A histologic study of fractured human vertebral bodies. J Spinal Disord Tech. 2002;15(2):118–126. doi:10.1097/00024720-200204000-00005
13. Bailey JF, Liebenberg E, Degmetich S, Lotz JC. Innervation patterns of PGP 9.5-positive nerve fibers within the human lumbar vertebra. J Anat. 2011;218(3):263–270. doi:10.1111/j.1469-7580.2010.01332.x
14. Degmetich S, Bailey JF, Liebenberg E, Lotz JC. Neural innervation patterns in the sacral vertebral body. Eur Spine J. 2016;25(6):1932–1938. doi:10.1007/s00586-015-4037-4
15. Fujii T, Sakuma Y, Orita S, et al. Dichotomizing sensory nerve fibers innervating both the lumbar vertebral body and the area surrounding the iliac crest: a possible mechanism of referred lateral back pain from lumbar vertebral body. Spine. 2013;38(25):E1571–E1574. doi:10.1097/BRS.0b013e3182a879cd
16. Buonocore M, Aloisi AM, Barbieri M, Gatti AM, Bonezzi C. Vertebral body innervation: implications for pain. J Cell Physiol. 2010;222(3):488–491. doi:10.1002/jcp.21996
17. Fagan A, Moore R, Vernon RB, Blumbergs P, Fraser R. ISSLS prize winner: the innervation of the intervertebral disc: a quantitative analysis. Spine. 2003;28(23):2570–2576. doi:10.1097/01.BRS.0000096942.29660.B1
18. Niv D, Gofeld M, Devor M. Causes of pain in degenerative bone and joint disease: a lesson from vertebroplasty. Pain. 2003;105(3):387–392. doi:10.1016/S0304-3959(03)00277-X
19. Yokoyama K, Kawanishi M, Yamada M, et al. Validity of intervertebral bone cement infusion for painful vertebral compression fractures based on the presence of vertebral mobility. AJNR Am J Neuroradiol. 2013;34(1):228–232. doi:10.3174/ajnr.A3160
20. Wong DA. Basivertebral nerve ablation: does the path followed suggest this technology is ready for adoption into clinical practice?: COMMENTARY ON: khalil J et al. A prospective, randomized, multicenter study of intraosseous basivertebral nerve ablation for the treatment of chronic low back pain. Spine J. 2019;10:1620–1632. doi:10.1016/j.spinee.2019.05.598
21. Khalil JG, Smuck M, Koreckij T, et al. A prospective, randomized, multicenter study of intraosseous basivertebral nerve ablation for the treatment of chronic low back pain. Spine J. 2019;19(10):1620–1632. doi:10.1016/j.spinee.2019.05.598
22. Gebhart GF, Bielefeldt K. Physiology of Visceral Pain. Compr Physiol. 2016;6(4):1609–1633.
23. Rucker HK, Holloway JA. Viscerosomatic convergence onto spinothalamic tract neurons in the cat. Brain Res. 1982;243(1):155–157. doi:10.1016/0006-8993(82)91130-1
24. Milne RJ, Foreman RD, Giesler GJ, Willis WD. Convergence of cutaneous and pelvic visceral nociceptive inputs onto primate spinothalamic neurons. Pain. 1981;11(2):163–183. doi:10.1016/0304-3959(81)90003-8
25. Ohtori S, Inoue G, Koshi T, et al. Sensory innervation of lumbar vertebral bodies in rats. Spine. 2007;32(14):1498–1502. doi:10.1097/BRS.0b013e318067dbf8
26. Chen J, Hou S, Peng B, et al. Effect of the L2 ramus communicans on the nociceptive pathway in lumbar intervertebral discs in rats. Eur J Pain. 2008;12(6):798–803. doi:10.1016/j.ejpain.2007.12.001
27. O’Neill CW, Kurgansky ME, Derby R, Ryan DP. Disc stimulation and patterns of referred pain. Spine. 2002;27(24):2776–2781. doi:10.1097/00007632-200212150-00007
28. Domenech-Garcia V, Skuli PT, Boudreau SA, Herrero P, Graven-Nielsen T. Pressure-induced referred pain areas are more expansive in individuals with a recovered fracture. Pain. 2018;159(10):1972–1979. doi:10.1097/j.pain.0000000000001234
29. Burt-Pichat B, Lafage-Proust MH, Duboeuf F, et al. Dramatic decrease of innervation density in bone after ovariectomy. Endocrinology. 2005;146(1):503–510. doi:10.1210/en.2004-0884
30. Alves CJ, Alencastre IS, Neto E, et al. Bone Injury and Repair Trigger Central and Peripheral NPY Neuronal Pathways. PLoS One. 2016;11(11):e165465. doi:10.1371/journal.pone.0165465
31. Li J, Ahmad T, Spetea M, Ahmed M, Kreicbergs A. Bone reinnervation after fracture: a study in the rat. J Bone Miner Res. 2001;16(8):1505–1510. doi:10.1359/jbmr.2001.16.8.1505
32. Hukkanen M, Konttinen YT, Santavirta S, et al. Rapid proliferation of calcitonin gene-related peptide-immunoreactive nerves during healing of rat tibial fracture suggests neural involvement in bone growth and remodeling. Neuroscience. 1993;54(4):969–979. doi:10.1016/0306-4522(93)90588-7
33. Li J, Kreicbergs A, Bergstrom J, Stark A, Ahmed M. Site-specific CGRP innervation coincides with bone formation during fracture healing and modeling: a study in rat angulated tibia. J Orthop Res. 2007;25(9):1204–1212. doi:10.1002/jor.20406
34. Li J, Ahmed M, Bergstrom J, Ackermann P, Stark A, Kreicbergs A. Occurrence of substance P in bone repair under different load comparison of straight and angulated fracture in rat tibia. J Orthop Res. 2010;28(12):1643–1650. doi:10.1002/jor.21169
35. Cheung WH, Miclau T, Chow SK, Yang FF, Alt V. Fracture healing in osteoporotic bone. Injury. 2016;47(Suppl 2):S21–S26. doi:10.1016/S0020-1383(16)47004-X
36. Buchbinder R, Osborne RH, Ebeling PR, et al. A randomized trial of vertebroplasty for painful osteoporotic vertebral fractures. N Engl J Med. 2009;361(6):557–568. doi:10.1056/NEJMoa0900429
 © 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.