Back to Journals » Patient Preference and Adherence » Volume 12
A new pulmonary rehabilitation maintenance strategy through home-visiting and phone contact in COPD
Authors Li Y , Feng J, Li Y , Jia W, Qian H
Received 2 September 2017
Accepted for publication 8 December 2017
Published 11 January 2018 Volume 2018:12 Pages 97—104
DOI https://doi.org/10.2147/PPA.S150679
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Naifeng Liu
Yi Li,1,2 Jing Feng,3,4 Yuechuan Li,2 Wei Jia,2 Hongyu Qian2
1Graduate School, Tianjin Medical University, 2Department of Respiratory and Critical Care Medicine, Tianjin Chest Hospital, 3Respiratory Department, Tianjin Medical University General Hospital, Tianjin, People’s Republic of China; 4Neuropharmacology Section, Laboratory of Toxicology and Pharmacology, National Institute of Environmental Health Sciences, National Institutes of Health, Research Triangle Park, NC, USA
Background: The benefit of pulmonary rehabilitation (PR) for patients with COPD diminishes over time. We investigated a new strategy involving home-visit and phone contact and compared this to usual care in maintenance of PR benefits.
Methods: A total of 172 stable COPD patients receiving 8-week PR program were recruited for this prospective study. Patients were allocated into usual care group (UC) and PR maintenance group (PRMG) randomly. Patients in PRMG participated in maintenance strategy at home under supervision through home-visit and phone contact. The 6-minute walking test (6MWT), COPD assessment test (CAT), and modified Medical Research Council scale (mMRC) scores were evaluated every 3 months.
Results: Of the total, 151 patients completed 8-week PR program with satisfactory PR results (p<0.001), and 104 patients finished the follow-up. The clinical improvements in 6MWT, CAT, and mMRC scores were maintained (p<0.001) in PRMG. In comparison, the benefit of PR diminished gradually in UC. The differences in 6MWT, CAT, and mMRC scores between groups were observed 6, 9, and 6 months after PR, respectively (p<0.05). Total frequency of exacerbations in PRMG was lower than UC (p=0.021).
Conclusion: Maintenance strategy involving home-visit and phone contact is superior to usual care to preserve PR benefits, and reduces the acute COPD exacerbation rate.
Keywords: COPD, pulmonary rehabilitation, 6-minute walking test, COPD assessment test, maintenance
Introduction
COPD is characterized by restriction of airflow and manifests some features of systematic disease due to its association with extrapulmonary symptoms, such as cardiovascular disease, skeletal muscle atrophy, anxiety, and depression.1 Although pharmacological treatment could relieve symptoms, reduce the exacerbation, and slow down the development of the disease, accumulating evidence has suggested pulmonary rehabilitation (PR) plays a significant role in nonpharmacological treatment and self-management of COPD,2,3 leading to improved exercise capacity and health-related quality of life (HRQoL). However, factors such as duration of rehabilitation program, adherence, and transportation problems could affect the outcomes of rehabilitation.4 Previous studies have demonstrated the effect of longer-duration PR is more beneficial than shorter ones.5,6 Some people suggest that the benefits of a 6- to 12-week PR program could last for 1 year without any maintenance strategy,7 while others demonstrate that these benefits tend to diminish gradually and maintenance strategy could slow down this tendency.2 Therefore, maintenance strategy after initial PR has been recommended.2,8 However, the strategy and frequency of maintenance program are still unclear.9 For example, a lower frequency of maintenance strategy after initial PR did not show any differences in exercise tolerance and symptoms.10 Another study demonstrated that PR maintenance implemented by health care network for 1 year led to greater improvement in 6-minute walking test (6MWT) in moderate-to-severe patients with COPD.11 Recent clinical research reported that there were no significant differences in reduction of COPD exacerbation between home-based maintenance and hospital-based maintenance strategy.12 Additionally, because the effect of maintenance is also different depending on the duration and intensity of maintenance, adherence of patients,13 and especially the location where the strategy is performed,11 a new strategy to maintain the beneficial effects of PR program is needed.
In this study, we designed a well-supervised and home-based strategy with 12-month follow-up after initial 8 weeks of standard PR program. The 6MWT, COPD assessment test (CAT), and modified Medical Research Council scale (mMRC) scores were evaluated every 3 months.
Methods
Study population
This study was performed at Tianjin Chest Hospital (Tianjin, People’s Republic of China) from June 2014 to April 2016 and was a prospective research of 1-year follow-up duration after standard 8-week PR. Patients with COPD were enrolled if they were eligible. We included patients who were 1) having a diagnosis of COPD according to GOLD guideline,14 2) clinically stable in the first 4 weeks, 3) able to complete the PR program and questionnaire survey successfully and independently, 4) willing to participate in the PR and maintenance program, and 5) willing to sign informed consent. The exclusion criteria included the following: 1) having asthma, 2) obstructive sleep apnea syndrome, or 3) underdiagnosis of cancer; 4) diagnosed with Alzheimer’s disease or depression and anxious disorder; 5) unavailable for exercise even during the stable stage; 6) having severe dysfunction of the heart, liver, or kidney; and 7) suffering emotional trauma in the previous 6 months such as relative death and divorce. The exit criteria included 1) exacerbation during PR and maintenance, 2) frequent accident events during PR and maintenance, and 3) death. This study was approved by the Ethics Committee of Tianjin Chest Hospital. All patients provided written informed consent.
Interventions
Usual care
Usual care included health education, smoking cessation, oxygen therapy when patients suffered respiratory failure, as well as dietary and nutrition consultation along with necessary pharmacological therapy without systemic corticosteroids.
PR program at outpatient department
Aerobic training
Firstly, all patients were evaluated by treadmill test to identify the maximum walking speed and maximum heart rate (HR) for 3 times in 1 week at outpatient department. After routine warm-up and being given general information about PR, patients started the test at a speed of 1 km/h, and increased the speed by 0.2 km/h every 10 minutes until complaining of breathlessness or muscle weakness. Both maximum HR and maximum speed were recorded during test. Then, the target HR was suggested to 70%–80% of maximum HR. The duration of aerobic training was suggested to 5 minutes from the onset, and then accumulate to 20 minutes. Patients adjusted speed according to their symptoms and real-time HR. The signs to stop aerobic training were stethalgia, breathlessness, profuse sweating, systolic pressure higher than 180 mmHg, and oxyhemoglobin saturation lower than 85%.
Upper resistance training
Briefly, patients performed upper resistance training under 0.5 kg-weight load. Patients were requested to hold on for 5 seconds at the end of inspiration and then expire slowly. This training was repeated 3 times a week for 2 months.
Respiratory training
Half-closed-lip abdominal respiratory training and sputum drainage was performed once every day for 2 months. This section was recommended for 30 minutes.
PR maintenance program at home
Three parts of PR described above were performed at home at less frequency as maintenance strategy with the same duration of each session. For example, the frequency of aerobic training was reduced to once per week, upper resistance training was also decreased to once per week, and respiratory training was reduced to 3 times per week. Additionally, patients adjusted speed or exercise intensity according to maximum HR and symptoms when they were ready for aerobic training.
Outcomes measurement
Pulmonary function parameters, such as forced expiratory volume in 1 second (FEV1), FEV1%pred, forced vital capacity (FVC), and FEV1/FVC%, were measured. Pulmonary function was tested at the baseline and post-inhaling bronchodilator. Meanwhile, bronchial dilation test was also performed to exclude asthma.
- 6MWT: According to the protocol of the American Thoracic Society,15 patients were asked to walk as far as they can in 6 minutes in a 30-m straight corridor without any interruption. At the end of the test, the distance walked by the patients and symptom of apnea were recorded.
- CAT: This16 was used to evaluate the HRQoL in the follow-up. This was performed in an interview format.
- mMRC: This scale17 was applied to assess the severity of breathlessness. The mMRC is an instrument to evaluate apnea under different active conditions that are considered critical to COPD patients.
- Beck Depression Inventory (BDI) and State–Trait Anxiety Inventory (STAI):18 The content of BAI includes 21 items including evaluation of nervousness, dizziness, and inability to relax, while STAI is aimed at measuring severity of current anxiety and tendency to be anxious.
- Activities of daily living (ADLs): These include activities and tasks that people routinely perform in their daily life inside/outside their homes. Measuring ADLs is one of the best ways to evaluate the level of health,19 assess the progress of the disease, and evaluate the efficacy of rehabilitation or other treatments in patients with COPD.
- The adherence of PR description:20 To monitor the compliance of patients with PR and maintenance strategy, we applied a definition that completion of more than 50% of scheduled maintenance training session is considered adherence.
- Once any accident events of tachycardia (higher than 85% of target HR), hypoxia (pulse oxygen saturation [SaO2] lower than 10% of baseline), hypertension (blood pressure higher than 200/100 mmHg), and syncope were observed during exercise, patients were forbidden to continue training and given therapy immediately.
Process
After first baseline assessment (pre-PR), all stable eligible COPD patients underwent PR program for 8 weeks. At the end of PR (post-PR), patients were ruled out if they dropped out, were poorly adherent, or had frequent occurrence of accident events during program. Participants who successfully completed the program were allocated into 2 groups including usual care group (UC) and PR maintenance group (PRMG). All patients were reevaluated before follow-up observation. In UC, patients were treated by usual care for 1 year which was consistent with guideline,2 while patients in PRMG attended a home-based PR maintenance strategy under professional supervision and consultation regularly. All patients were requested to go back to hospital for regular review. During 1-year maintenance observation, we adopted 3 evolutionary steps to supervise patients’ performance and adherence to maintenance exercise.
Step 1: |
Home-visit once every 2 weeks to conduct rehabilitation exercise strategy and provide health education for 2 months. |
Step 2: |
Home-visit every 4 weeks and phone contact once a week for 4 months. |
Step 3: |
Phone contact once a week for 6 months. |
The primary outcomes of this study were exercise tolerance, symptoms of breathlessness, as well as CAT scores at the end of follow-up. The second outcome was frequencies of exacerbation in 2 groups.
Groups
All the 172 eligible patients who were enrolled at the baseline assessment underwent PR program for 2 months. Of these patients, 5 dropped out, 10 reported bad adherence, 2 were unable to persist in training, 3 were excluded because of transportation problems, and 1 did not show concern about benefits of PR. A total of 151 patients were allocated into 2 groups randomly, 69 patients in UC and 82 patients in PRMG. The demographic characteristics are shown in Table 1. During 1-year observation, 7 patients dropped out, 16 (23.2%) experienced exacerbation, and 3 died due to cardiovascular disease in UC. In the PRMG, 5 patients quitted maintenance program during follow-up, and the aggravation and death rate in PRMG were 8.5% (7/82) and 2.4% (2/82), respectively (Figure 1).
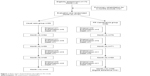  | Figure 1 Flow chart of participants throughout the study. |
Statistical analysis
Descriptive statistics and chi-squared test were used to demonstrate the sample characteristics of population and socialization. Post hoc paired t-test analysis was used to compare all pairs of variables in each group. Unpaired Student’s t-test was to assess and compare all independent variables in 2 groups both at pre-PR and post-PR. We applied repeated-measure analysis of variance (ANOVA) and multivariate ANOVA to test the differences over time in 6MWT, CAT, mMRC, BDI, State Anxiety Inventory (SAI), and Trait Anxiety Inventory (TAI) between groups. We analyzed data via Statistical Package for the Social Sciences (SPSS) version 17.0 software. A p-value of <0.05 was considered statistically significant.
Results
Adherence assessment
At the end of follow-up, 7 patients were excluded due to completion of only less than 50% of scheduled maintenance program according to adherence standards described above. In maintenance group, 61 patients (89.7%) were considered adherent, and the mean adherence was 64%.
Effect of PR
As analyzed by post hoc paired t-tests (Table 2), 6MWT, CAT, mMRC, BDI, SAI, and TAI showed statistically significant improvement in both groups (p<0.001) after PR. Neither pre-PR nor post-PR, the differences in each outcome measure between 2 groups were statistically significant as evaluated using unpaired Student’s t-test.
Effect of PR maintenance program
In PRMG, the scores of 6MWT increased gradually from month 3 to month 12 compared to month 0 (447±71, p<0.001; 459±70, p<0.001; 461±68, p<0.001; 466±69, p<0.001). In contrast, compared to month 0, the results of 6MWT in UC showed initial increase from month 3 to month 6, and then a decline below the level of month 0 over time (430±44, p<0.001; 429±45, p<0.001; 423±49, p=0.071; 397±49, p<0.001). The mean differences in scores of 6MWT between groups were observed in months 3–6, months 6–9, and months 9–12 (p=0.017, p=0.002, and p<0.001, respectively) (Figure 2).
Compared to month 0, the scores of CAT showed significant decrease from month 3 to month 9 (12.8±6.8, p<0.001; 9.9±5.6, p<0.001; 8.5±5.0, p<0.001). At month 12, the scores of CAT remained the same as prior time point and were lower than the level of month 0 in PRMG (8.5±4.9, p<0.001). However, in UC, the scores of CAT decreased gradually from month 3 to month 6 in comparison with month 0 (13.0±7.0, p<0.001; 11.5±6.2, p<0.001), and increased over time from month 6 to month 12 (13.2±6.1, p<0.001; 15.1±6.2, p=0.915). At the end of month 12, the scores returned to the level of month 0. The mean differences in CAT scores between groups were observed in months 6–9 and months 9–12 (both p<0.001) (Figure 2).
Compared to month 0, the results of mMRC showed significant decreases from the starting point and were maintained at the same level over time in PRMG (1.3±0.9, p<0.001; 1.3±0.9, p<0.001; 1.1±0.8, p<0.001; 1.2±0.7, p<0.001). However, after initial decrease, the scores of mMRC in UC were increased significantly over time from month 3 to month 12, and even higher than the level of month 0 (1.6±1.0, p<0.001; 2.1±0.9, p=0.044; 2.8±0.9, p<0.001; 3.3±0.7, p<0.001). Similarly, the mean differences in mMRC scores between groups were observed in months 3–6, months 6–9, and months 9–12 (all p<0.001) (Figure 2).
During follow-up, the measurements of BDI (pUC=0.217, pPRMG=0.160), SAI (pUC=0.227, pPRMG=0.151), and TAI (pUC=0.370, pPRMG=0.136) showed no changes over time compared with the values at month 0 in both groups. Likewise, there were also no mean differences between groups in BDI (pmonths 0–3=0.551, pmonths 3–6=0.358, pmonths 6–9=0.628, pmonths 9–12=0.626), SAI (pmonths 0–3=0.245, pmonths 3–6=0.186, pmonths 6–9=0.254, pmonths 9–12=0.228), and TAI (pmonths 0–3=0.962, pmonths 3–6=0.976, pmonths 6–9=0.864, pmonths 9–12=0.878) (Table 3).
Exacerbation
At the end of 1-year follow-up, the total frequency of exacerbation in UC was 23.2% (16/69). The patients in PRMG had 8.5% (7/82) exacerbation. The differences in exacerbation rate between 2 groups were statistically significant (p=0.021).
Discussion
This study showed that patients with stable COPD achieved significant clinical improvement from the initial 8-week PR program, consistent with previous research.21 During follow-up after completion of standard PR, we found that maintenance strategy maintained the benefits of initial PR, and even improved the performance of exercise tolerance and HRQoL compared with post-PR. However, these effects were not seen in UC during the observation, consistent with previous conclusions.22 Notably, there were also differences in 6MWT, CAT, and mMRC scores between groups at later time points from month 6 to month 12. Additionally, maintenance strategy also reduced the frequency of exacerbation compared with UC.
There were still controversies regarding the effect of PR maintenance program on COPD patients. A study on a maintenance exercise with a frequency of 2 hours in every 3 months for 1 year showed that patients with COPD showed no significant improvement in exercise capacity, domains of chronic respiratory questionnaire, as well as exacerbation.10 The data we showed here suggest otherwise. Because we applied a maintenance exercise with a frequency of once a week for 1 year, the capacity of exercise and the clinical symptoms of breathlessness improved significantly. Three other studies including 204 patients with COPD demonstrated that lower-supervised exercise preserved the exercise tolerance, peak exercise capacity,23 and HRQoL24 compared with post-PR, but without statistically significant differences compared with control group. In contrast, in the current study, the clinical improvements in 6MWT, mMRC, and CAT scores also showed significant differences between the groups. A systematic review recommended that supervised maintenance strategy with less frequency is insufficient to maintain the gain of PR.9 Meanwhile, the effect of reduced intensity or frequency of maintenance strategy (eg, once a month) is unclear. Indeed, the present study performed the same PR program once weekly for 1 year and found that it was effective in keeping and extending the clinical improvement after initial PR during 1-year period, in contrast to previous studies.25,26 In addition, the frequency of home-visit or phone contact also played critical roles in maintaining benefits of PR, especially at the early stage of follow-up.
Similar to initial PR, in terms of the location of maintenance strategy application, the distance and/or the travel time are important for people with COPD to attend and complete the maintenance program.4 Although recent research verified the feasibility and safety of community-based maintenance program,27 the shortcoming of this strategy was still the transportation for patients living in the suburb to come to hospital; therefore, they might have less chance to maintain their improvement of PR. In our study, the results of benefit maintenance were partially attributed to regular home-visit combined with phone call supervision, to overcome the distance obstacle for patients with longer than 30-minute travel.4 We speculated that this convenient pattern increased the participant rate of PR maintenance program and protected the benefits of PR from diminishing over time. Meanwhile, home-visit and phone contact help in early recognition of deterioration symptoms, reducing the accident of exacerbation and emergency visiting or frequency of hospitalization.28 This may be partial reasons for the reduced rate of exacerbation in PRMG.
In the current study, the mean adherence rate was 64% which is higher than the average adherence rate of a long-term therapy of chronic diseases.29 We speculate reasons from patients’ perspectives for this phenomenon: during 12-month maintenance, most of the patients recognized their health condition much better, enhanced self-worth consciousness, changed habits and life style, avoided exacerbation,28 as well as showed concern about benefits of PR. Meanwhile, direct professional consultation and technical support offered during our study also played a crucial role in increasing the motivation of attendance and completion which otherwise have a trend of decline over time.30
Emotion is an important component of psychology. Most of the COPD patients face the challenge of anxiety and depression.31 In our study, we found the emotional dysfunction was relieved after PR. Because mood disorders are associated with dyspnea and active tolerance,32 our exercise training ameliorated the symptoms of dyspnea and increased the exercise tolerance by increasing coordination of neuromuscles and respiratory efficiency.33 Therefore, such explanation was partially possible reasons for emotional improvements in our study. During the follow-up, although the improvement of emotional function after PR was not changed over time compared with post-PR in both groups, our home-based maintenance could also provide family support and professional health education to increase the confidence of patients to overcome depression and anxiety, as described in previous research.34
The limitations of the present study are the following: 1) although CAT is a clear and convenience questionnaire used in comparison with St George’s Respiratory Questionnaire, it is necessary but insufficient to identify all domains of HRQoL in COPD patients; 2) due to sex distribution of COPD, the relief from anxiety and depression obtained from PR reported in our study cannot be extrapolated to female patients; and 3) we supervised the patients only through phone contact without home-visit during the last 6 months, so it is unclear whether the patients received sufficient exercise during this period. Recent research35 demonstrated that the effect of long-term PR maintenance on 6MWT declined from the 12th month post-PR, and vanished at the end of the second year. To better identify the effect of home-based PR maintenance strategy, larger, longer-term, and multicenter controlled trials are warranted.
Conclusion
Collectively, this new home-based PR maintenance strategy maintains the clinical improvements in 6MWT, mMRC, and CAT, and reduces their decline. This strategy involving home-visit and phone contact is superior to usual care and feasible to preserve the benefits of PR, and reduces the acute COPD exacerbation rate.
Acknowledgment
This study was supported by the grants from the National Natural Science Foundation of China (No. 81270144, 81570084, 30800507), and the Science and Technology Plan Project of Tianjin (13ZCZDSY02000).
Disclosure
The authors report no conflicts of interest in this work.
References
Vogelmeier CF, Criner GJ, Martínez FJ, et al. Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Lung Disease 2017 report: GOLD executive summary. Eur Respir J. 2017;49(3):pii 1700214. | ||
Ries AL, Bauldoff GS, Carlin BW, et al. Pulmonary rehabilitation: joint ACCP/AACVPR evidence-based clinical practice guidelines. Chest. 2007;131(5 Suppl):4S–42S. | ||
Spruit MA, Singh SJ, Garvey C, et al; ATS/ERS Task Force on Pulmonary Rehabilitation. An official American Thoracic Society/European Respiratory Society statement: key concepts and advances in pulmonary rehabilitation. Am J Respir Crit Care Med. 2013;188(8):e13–e64. | ||
Rochester CL, Vogiatzis I, Holland AE, et al; ATS/ERS Task Force on Policy in Pulmonary Rehabilitation. An official American Thoracic Society/European Respiratory Society policy statement: enhancing implementation, use, and delivery of pulmonary rehabilitation. Am J Respir Crit Care Med. 2015;192(11):1373–1386. | ||
Solanes I, Güell R, Casan P, et al. Duration of pulmonary rehabilitation to achieve a plateau in quality of life and walk test in COPD. Respir Med. 2009;103(5):722–728. | ||
Sewell L, Singh SJ, Williams JE, Collier R, Morgan MD. How long should outpatient pulmonary rehabilitation be? A randomised controlled trial of 4 weeks versus 7 weeks. Thorax. 2006;61(9):767–771. | ||
Troosters T, Gosselink R, Decramer M. Short- and long-term effects of outpatient rehabilitation in patients with chronic obstructive pulmonary disease: a randomized trial. Am J Med. 2000;109(3):207–212. | ||
Bolton CE, Bevan-Smith EF, Blakey JD, et al. British Thoracic Society guideline on pulmonary rehabilitation in adults. Thorax. 2013;68 Suppl 2:ii1–ii30. | ||
Alison JA, McKeough ZJ, Johnston K, et al. Australian and New Zealand pulmonary rehabilitation guidelines. Respirology. 2017;22(4):800–819. | ||
Wilson AM, Browne P, Olive S, et al. The effects of maintenance schedules following pulmonary rehabilitation in patients with chronic obstructive pulmonary disease: a randomised controlled trial. BMJ Open. 2015;5(3):e005921. | ||
Moullec G, Ninot G, Varray A, Desplan J, Hayot M, Prefaut C. An innovative maintenance follow-up program after a first inpatient pulmonary rehabilitation. Respir Med. 2008;102(4):556–566. | ||
Vasilopoulou M, Papaioannou AI, Kaltsakas G, et al. Home-based maintenance tele-rehabilitation reduces the risk for acute exacerbations of COPD, hospitalisations and emergency department visits. Eur Respir J. 2017;49(5):pii 1602129. | ||
Moy ML, Wayne PM, Litrownik D, et al. Long-term Exercise After Pulmonary Rehabilitation (LEAP): design and rationale of a randomized controlled trial of Tai Chi. Contemp Clin Trials. 2015;45(Pt B):458–467. | ||
Qaseem A, Wilt TJ, Weinberger SE, et al; American College of Physicians; American College of Chest Physicians; American Thoracic Society; European Respiratory Society. Diagnosis and management of stable chronic obstructive pulmonary disease: a clinical practice guideline update from the American College of Physicians, American College of Chest Physicians, American Thoracic Society, and European Respiratory Society. Ann Intern Med. 2011;155(3):179–191. | ||
ATS Committee on Proficiency Standards for Clinical Pulmonary Function Laboratories. ATS statement: guidelines for the six-minute walk test. Am J Respir Crit Care Med. 2002;166(1):111–117. | ||
Gupta N, Pinto LM, Morogan A, Bourbeau J. The COPD assessment test: a systematic review. Eur Respir J. 2014;44(4):873–884. | ||
Mahler DA, Wells CK. Evaluation of clinical methods for rating dyspnea. Chest. 1988;93(3):580–586. | ||
Julian LJ. Measures of anxiety: State-Trait Anxiety Inventory (STAI), Beck Anxiety Inventory (BAI), and Hospital Anxiety and Depression Scale-Anxiety (HADS-A). Arthritis Care Res (Hoboken). 2011;63 Suppl 11:S467–S472. | ||
Monjazebi F, Dalvandi A, Ebadi A, Khankeh HR, Rahgozar M, Richter J. Functional status assessment of COPD based on ability to perform daily living activities: a systematic review of paper and pencil instruments. Global J Health Sci. 2015;8(3):210–223. | ||
Bourbeau J, Bartlett SJ. Patient adherence in COPD. Thorax. 2008;63(9):831–838. | ||
Güell R, Casan P, Belda J, et al. Long-term effects of outpatient rehabilitation of COPD: a randomized trial. Chest. 2000;117(4):976–983. | ||
Griffiths TL, Burr ML, Campbell IA, et al. Results at 1 year of outpatient multidisciplinary pulmonary rehabilitation: a randomized controlled trial. Lancet. 2000;355(9201):362–368. | ||
Spencer LM, Alison JA, McKeough ZJ. Maintaining benefits following pulmonary rehabilitation: a randomised controlled trial. Eur Respir J. 2010;35(3):571–577. | ||
Ringbaek T, Brondum E, Martinez G, et al. Long term effects of 1-year maintenance training on physical functioning and health status in patients with COPD: a randomised controlled study. J Cardiopulm Rehabil Prev. 2010;30(1):47–52. | ||
Brooks D, Krip B, Mangovski-Alzamora S, Goldstein RS. The effect of postrehabilitation programmes among individuals with chronic obstructive pulmonary disease. Eur Respir J. 2002;20(1):20–29. | ||
Bestall JC, Paul EA, Garrod R, Garnham R, Jones RW, Wedzicha AJ. Longitudinal trends in exercise capacity and health status after pulmonary rehabilitation in patients with COPD. Respir Med. 2003;97(2):173–180. | ||
Beauchamp MK, Francella S, Romano JM, Goldstein RS, Brooks D. A novel approach to long-term respiratory care: results of a community-based post-rehabilitation maintenance program in COPD. Respir Med. 2013;107(8):1210–1216. | ||
Hoaas H, Andreassen HK, Lien LA, Hjalmarsen A, Zanaboni P. Adherence and factors affecting satisfaction in long-term telerehabilitation for patients with chronic obstructive pulmonary disease: a mixed methods study. BMC Med Inform Decis Mak. 2016;16:26. | ||
Sabaté E. Adherence to Long-Term Therapies: Evidence for Action. Geneva: World Health Organization; 2003. | ||
Hogg L, Grant A, Garrod R, Fiddler H. People with COPD perceive ongoing, structured and socially supportive exercise opportunities to be important for maintaining an active lifestyle following pulmonary rehabilitation: a qualitative study. J Physiother. 2012;58(3):189–195. | ||
Yohannes AM, Alexopoulos GS. Depression and anxiety in patients with COPD. Eur Respir Rev. 2014;23(133):345–349. | ||
de Voogd JN, Sanderman R, Postema K, van Sonderen E, Wempe JB. Relationship between anxiety and dyspnea on exertion in patients with chronic obstructive pulmonary disease. Anxiety Stress Coping. 2011;24(4):439–449. | ||
Regiane Resqueti V, Gorostiza A, Gáldiz JB, López de Santa María E, Casan Clarà P, Güell Rous R. Beneficios de un programa de rehabilitación respiratoria domiciliaria en pacientes con EPOC grave [Benefits of a home-based pulmonary rehabilitation program for patients with severe chronic obstructive pulmonary disease]. Arch Bronconeumol. 2007;43(11):599–604. Spanish [with English abstract]. | ||
Witcher CS, McGannon KR, Hernandez P, et al. A qualitative exploration of exercise among pulmonary rehabilitation participants: insight from multiple sources of social influence. Respir Care. 2015;60(11):1624–1634. | ||
Güell MR, Cejudo P, Ortega F, et al. Benefits of long-term pulmonary rehabilitation maintenance program in patients with severe chronic obstructive pulmonary disease. Three-year follow-up. Am J Respir Crit Care Med. 2017;195(5):622–629. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.




