Back to Journals » Clinical Ophthalmology » Volume 16
A Cost-Effectiveness Analysis of AcrySof IQ Vivity Intraocular Lens (IOL) from Private Health Fund Perspective in Australia
Authors Bala C ![]() , Athanasiov P, Holland J, Dhariwal M
, Athanasiov P, Holland J, Dhariwal M ![]() , Gupta A, Rathi H
, Gupta A, Rathi H
Received 12 April 2022
Accepted for publication 29 June 2022
Published 2 August 2022 Volume 2022:16 Pages 2403—2412
DOI https://doi.org/10.2147/OPTH.S370420
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 5
Editor who approved publication: Dr Scott Fraser
Chandra Bala,1 Paul Athanasiov,2 Jason Holland,3 Mukesh Dhariwal,4 Amit Gupta,5 Hemant Rathi6
1personalEYES Pty Ltd, Sydney, NSW, Australia; 2Eye Surgeons SA, North Adelaide, SA, Australia; 3The Eye Health Centre, Brisbane, QLD, Australia; 4Alcon Vision LLC, Fort Worth, TX, USA; 5Skyward Analytics Pvt Ltd, Gurgaon, India; 6Skyward Analytics Pte Ltd, Singapore
Correspondence: Chandra Bala, personalEYES Pty Ltd, Level 2, 33 York Street, Sydney, NSW, 2000, Australia, Tel +61-2-88337111, Fax +61-2-88337112, Email [email protected]
Purpose: AcrySof IQ Vivity is a unique non-diffractive extended depth of focus intraocular lens with wavefront-shaping X-WAVE technology. This study evaluated the cost-effectiveness of AcrySof IQ Vivity intraocular lens (DFT015) compared with standard aspheric monofocal intraocular lens (SN60WF), from a private health fund perspective in Australia.
Methods: A Markov model was developed using the following health states: well, need for spectacles (near/distance/bifocal/varifocal), very bothersome visual disturbances (glare/haloes/starbursts) – with/without spectacles, and death. Model inputs were sourced from a randomized clinical study (NCT03010254), published literature, prostheses list and clinical opinion. A lifetime horizon (up to 30 years) was considered, and cost and health outcomes were discounted at 5% per annum. Model outcomes included incremental cost–effectiveness ratio defined as incremental cost per quality adjusted life year gain. Sensitivity and scenario analyses were also conducted.
Results: Bilateral implantation of DFT015 intraocular lens provided quality adjusted life year gain of 0.16 at an incremental cost of AU$307 compared to bilateral SN60WF, leading to an incremental cost-effectiveness ratio of AU$1908/quality adjusted life year, well below the cost-effectiveness threshold (Range: AU$45,000-AU$75,000) typically used by Medical Services Advisory Committee in Australia. Results were most sensitive to intraocular lens costs, post-operative spectacle dependence, and disutility due to wearing glasses. Robustness of the results was further confirmed by probabilistic sensitivity analysis and scenario analyses.
Conclusion: AcrySof IQ Vivity intraocular lens is a highly cost-effective treatment strategy with improved vision-related quality of life outcomes for presbyopic cataract surgery patients.
Keywords: cataract surgery, ATIOL, EDoF, presbyopia, ICER, quality of life
Introduction
Cataract surgery is one of the safest and most commonly performed surgical procedures, with approximately 20 million surgeries carried out worldwide every year.1–3 In Australia, a total of 277,597 cataract surgeries were recorded in 2018 alone.4 Additionally, many patients undergoing cataract surgery present with presbyopia, the most common refractive disorder for individuals 40 years and older.5,6
During cataract surgery, standard monofocal intraocular lenses (IOLs) are typically implanted, which have a single focal point for distance vision correction; however, many patients may end up with life-long dependency on glasses for near and/or intermediate vision correction.7 Multifocal IOLs have more than one focal point and provide vision over a range of distances (near, intermediate, and distance focal points), thereby reducing spectacle dependence.8 However, diffractive multifocal IOLs are associated with increased risk of visual disturbances and reduced contrast sensitivity compared to monofocal IOLs.9 More recently, extended depth of focus (EDoF) IOLs have been introduced, which offer an extended range of vision and may lower the risk of visual disturbances.10
AcrySof IQ Vivity IOL (model DFT015; Alcon) is a unique non-diffractive EDoF IOL with the wavefront-shaping X-WAVE technology, which stretches and shifts the wavefront instead of splitting it to provide a continuous focal range while maintaining a monofocal halo profile.9,11,12 In an international, multi-center, randomized clinical trial (RCT, NCT03010254), AcrySof IQ Vivity EDoF IOL provided superior intermediate and near vision and non-inferior distance vision compared with an aspheric monofocal IOL (model SN60WF; Alcon).9 Further, DFT015 demonstrated a visual disturbance profile similar to SN60WF.9
In Australia’s public health system, patients usually receive a standard monofocal IOL implantation during cataract surgery.14 Alternatively, more than half of the Australian population (13.6 million) has private insurance,15 which provides them coverage to seek healthcare in a private clinic with an option to choose a premium presbyopia and/or astigmatism correcting IOL. In Australia, a medical device must be included in the “Prosthesis list” to be eligible for specific benefits provided by private health insurers.16,17 In order to be included in the “Prosthesis list”, a medical device must typically showcase improved or equivalent clinical and/or economic benefits over already listed devices.16
To evaluate the clinical and cost benefits of new technologies, a cost–effectiveness analysis is a useful economic tool that allows decision makers to make informed choices regarding coverage and reimbursement for such technologies.18,19 The preferred measure of effectiveness by the Pharmaceutical Benefits Advisory Committee (PBAC) and Medical Services Advisory Committee (MSAC) is quality adjusted life years (QALYs).20,21 QALY is the academic standard for measuring potential impact of an intervention on patients’ life.22,23 QALY has two components: “quality of life” and “length of life”. QALY is estimated as the length of life multiplied by the utility weight (which represents the quality of life).23 Utility value represents how good an individual is feeling in a particular health state on a scale of 0 to 1, where 0 means “Death” and 1 means “Perfect health”. Utility values for health states are measured using standardised patient reported outcomes (PRO) measures, such as EuroQol – 5 dimension (EQ-5D), Short Form – 36 (SF-36) questionnaire etc. One QALY denotes 1 year of life in perfect health.22,23
Currently, there are no published cost–effectiveness studies of DFT015 IOL in cataract surgery in Australia. Hence, this study was conducted to evaluate the cost–effectiveness of DFT015 IOL compared with an aspheric monofocal IOL (SN60WF) in patients undergoing bilateral cataract surgery from the Australian private health fund perspective.
Methods
Model Structure
The Markov model used in the present study (Figure 1) was adapted from a recently published health economic evaluation study of a trifocal IOL.24 The patient baseline characteristics for the two IOL groups (DFT015 and SN60WF) were sourced from a multi-center, randomized clinical trial (NCT03010254), which aimed to evaluate the effectiveness and safety of these two IOLs.9,25 The median age across both IOL groups (70 years) was used in the analysis.25 Model health states were based on probable outcomes post-cataract surgery and included: well (spectacle-independent patients without any very bothersome visual disturbances like glare/haloes/starburst), spectacle dependent (reading, distance, bifocal, and varifocal glasses), very bothersome visual disturbances (glare/haloes/starbursts) – with/without spectacles, and death (absorbing health state).
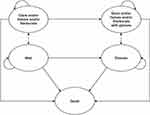 |
Figure 1 Structure of Markov Model. |
Model Inputs
Clinical Inputs
The movement of patients between health states is defined by transition probabilities. All patients start in the “Well” health state. The overall spectacle dependence with bilateral implantation of DFT015 IOL and SN60WF IOL was 69.8% and 92.5%, respectively,25 and the proportion of patients experiencing very bothersome visual disturbances (glares/haloes/starbursts) were 3.8% and 5.0% for DFT015 IOL and SN60WF IOL, respectively.9,25 The rate of resolution of these very bothersome visual disturbances (glares/haloes/starbursts) was assumed to be 81% based on Hu et al study.26 The rate of development of very bothersome visual disturbances (glares/haloes/starbursts) and the subsequent resolution were assumed to be similar for both spectacle dependent and spectacle-independent patients. Based on Hu et al, it was assumed that patients not developing any very bothersome visual disturbances (glares/haloes/starbursts) within the first year of cataract surgery would not experience them afterwards.26 The transition probabilities for different model health states are described in Table 1. Patient distribution based on the type of vision correction needed for post-cataract surgery (near, intermediate, and/or distance) in the two IOL groups (DFT015 and SN60WF) was sourced as reported in the Vivity RCT study (Table 2).25
 |
Table 1 Transition Probabilities Post-Cataract Surgery |
 |
Table 2 Distribution of Patients Wearing Each Type of Spectacle Post-Cataract Surgery Among Those Patients Requiring Spectaclesa |
In this model, death is an absorbing state as it is possible that a patient could die due to any cause in the post-cataract surgery period. Mortality rates were obtained from the Australian Bureau of Statistics life tables (2017–2019)27 and were applied to the proportion of alive patients in each health state in each cycle. The probability of death was assumed to be the same for DFT015 IOL and SN60WF IOL groups.
Cost Inputs
Model costs included DFT015 IOL and SN60WF IOL costs, cataract surgery costs (including surgeon’s fee and hospital fee), and cost of glasses (reading glasses, distance glasses, bifocal glasses, varifocal glasses). Cataract surgery fees at the hospital typically include surgeon’s fee, anaesthetist’s fee, and hospital facility fee. However, the anaesthetist’s fee is not covered by private health funds and assumed to be borne by the patient/government; therefore, it was not included in cataract surgery costs to the private health fund. With regard to the surgeon’s fee, the total rebate is $791.45 under Medicare Benefits schedule item code 42702.28 Based on local practice, it was assumed that the private health funds would only pay 25% of the scheduled fee (ie $197.86), and the rest is patient out-of-pocket. The cost of glasses was sourced from a private health fund in Australia (CBHS Corporate Health Insurance).29 The replacement frequency of glasses was assumed to be one year based on clinical experience. The detailed cost inputs are summarized in Table 3.
 |
Table 3 Cost Inputs |
(Dis-)Utilities
In the model, the “Well” health state represents perfect health (spectacle-independent patients not experiencing any visual disturbance). Hence, the utility of patients in this health state was assumed to be 1. Disutility due to very bothersome visual disturbances (glare/haloes/starbursts) (−0.18) and wearing glasses (−0.06) were sourced from Brown et al30 and Dobrez and Calhoun,31 respectively. Patients were assumed to experience very bothersome visual disturbances (glares/haloes/starbursts) for approximately 4 hours per day, mostly during the evening, which equates to approximately 2 months (60.90 days) in a year.
Base-Case Analysis
The primary analysis was conducted with a yearly cycle length and a lifetime horizon (30 years). The 30-year time horizon was chosen to simulate patients’ remaining lifetime post-cataract surgery. The starting age of the patients in the economic model was 70 years based on the median age of patients in the Vivity clinical trial. The economic model estimates the conditional survival given that the patients were alive at 70 years. Based on the Australian Bureau of Statistics life tables,27 the model predicts that approximately 40% and 16% of patients are alive at the age of 90 and 95, respectively. Therefore, a 30-year time horizon was used to fully capture the improvement in quality of life of these patients.
A half cycle correction was used to account for any model transitions within a cycle length. The primary cost–effectiveness outcomes included total costs, QALYs, economically justifiable price (EJP) and incremental cost–effectiveness ratio (ICER). The EJP is defined as the maximum price until which the use of product may be considered cost-effective at the pre-defined willingness-to-pay (WTP) threshold.32 ICER is a standard measure used in cost-effectiveness analysis to assess the value of an intervention and allows policymakers to inform treatment choices for efficient use of healthcare resources.33,34 The ICER is calculated as a ratio of the difference in total costs to total QALYs for the two IOL groups (DFT015 and SN60WF). An intervention is considered to be cost-effective if the ICER is below the health technology assessment (HTA) agency's recommended ICER threshold.
Sensitivity Analysis
Three types of sensitivity analyses were conducted to evaluate the impact of variation in model input parameters on the base-case results. In a one-way sensitivity analysis (OWSA), each model input parameter was varied independently to assess its impact on model results and to identify key parameters driving the model results. All parameters were varied within published or estimated 95% confidence intervals (CIs). Mean, standard deviation, standard error, and/or the number of observations were used to estimate 95% CIs for input parameters where 95% CIs were not reported. In probabilistic sensitivity analysis (PSA), all model input parameters were assigned certain distributions based on the type of parameters and were simultaneously varied in 1000 simulations. In each simulation, a value was drawn for each input parameter by random sampling based on their respective distributions. Additionally, the following scenario analyses were conducted: time horizon (5, 10, 15, 20, and 25 years), discount rates (0% and 3.50%), and patients’ age (50, 55, 60, and 65 years old).
Results
Base-Case Results
In the base-case analysis, total costs for DFT015 IOL and SN60WF IOL groups were AU$7528 and AU$7221, respectively, over the lifetime horizon. Total accrued lifetime quality adjusted life years (QALY) were 10.845 and 10.684 for bilateral DFT015 IOL and SN60WF IOL treatment groups, respectively. Bilateral implantation of DFT015 IOL provided an incremental QALY gain of 0.161 at an incremental cost of AU$307 compared to bilateral SN60WF IOL groups, resulting in an ICER per QALY gain of AU$1908. The economically justifiable price (EJP) for bilateral DFT015 IOL would be AU$8240 and AU$13,069 at typically used WTP threshold of AU$45,000 and AU$75,000, respectively.35 The disaggregated base-case results are presented in Table 4.
 |
Table 4 Disaggregated Base-Case Resultsa |
Sensitivity Analyses
In the one-way sensitivity analysis, it was found that the ICER was most sensitive to IOL costs, post-operative spectacle dependence, and disutility due to wearing glasses (Figure 2).
 |
Figure 2 Tornado diagram summarizing the OWSA results. Abbreviations: IOL, intraocular lens; ICER, incremental cost–effectiveness ratio; OWSA, one-way sensitivity analysis. |
Probabilistic sensitivity analysis (PSA) confirmed the robustness of the base-case results. From 1000 simulations, an average ICER of AU$1822 was obtained, which was similar to base-case ICER of AU$1908. At a willingness-to-pay (WTP) threshold of AU$15,000 per QALY gain and above, bilateral implantation of DFT015 IOL would be 100% cost-effective compared to SN60WF IOL (Figure 3).
 |
Figure 3 Cost-effectiveness acceptability curve. Abbreviation: IOL, intraocular lens. |
Scenario analyses demonstrated that the ICER was most affected by change in time horizon to 5 and 10 years, followed by discount rate at 0% and patients’ age at 50 years. Scenario analyses results are presented in Figure 4.
 |
Figure 4 Scenario analysis results. Abbreviation: ICER, incremental cost–effectiveness ratio. |
Discussion
The present study is a health economic evaluation of a novel non-diffractive EDoF IOL (AcrySof IQ Vivity). In the analysis, the incremental cost–effectiveness ratio (ICER) per QALY gain was estimated to be AU$1908. The HTA authorities in Australia, such as PBAC and MSAC, do not explicitly recommend a cost-effectiveness threshold, however, it has been observed that ICERs in the range of AU$45,000-AU$75,000 are typically considered cost-effective.35 Therefore, bilateral implantation of DFT015 IOL is a highly cost-effective treatment strategy for presbyopic cataract patients. At a willingness-to-pay (WTP) threshold of AU$45,000 and AU$75,000, the economically justifiable price (EJP) of bilateral DFT015 IOL would be AU$8240 and AU$13,069, respectively, ie, DFT015 IOL could be within the suggested cost-effectiveness thresholds even if its prosthesis list price increases by 6 to 10 times.
To the best of the authors’ knowledge, this is the first cost-effectiveness analysis of a novel non-diffractive EDoF IOL (DFT015 IOL) from an Australian health fund perspective. Furthermore, it is important to highlight that findings from this study not only apply to the private health fund in Australia but also can be applicable to other countries with similar healthcare systems. Several factors contribute to the overall strength of the analysis. The present analysis followed the recommended guidelines and methodology proposed by MSAC, Australia.21 Markov model methodology was used in this analysis, which is the most widely used medical decision analytic modelling methodology. The US Institute of Clinical and Economic Review (ICER) and PBAC in Australia recommends using Markov models to capture the changing health states over a long-time horizon.20 Additionally, the key efficacy inputs such as rates of overall spectacle dependence post-cataract surgery and very bothersome visual disturbances (glare/haloes/starbursts) were sourced from the AcrySof IQ Vivity EDoF IOL randomized clinical trial in which validated patient-reported questionnaires were used to measure these outcomes.9,25 The robustness of the base-case results was further validated with sensitivity analyses. The average results of probabilistic sensitivity analysis were found to be similar to the deterministic base-case results. Other sensitivity analyses, such as one-way sensitivity analysis and scenario analysis in which various model settings and/or inputs were varied, showed that even with shorter time horizons, greater discount rates, or older age at time of surgery, DFT015 IOL was a consistently cost-effective technology. It is important to note that some post-cataract health outcomes (lens explantation, YAG capsulotomy to correct posterior capsular opacification) and resource utilization (intraocular medications, optometrist visits) that are not covered under private health funds in Australia were excluded from this analysis. They are typically covered by the Medicare Benefits Schedule (MBS) with patient co-pay.
This model-based analysis has some limitations. First, there is a lack of long-term clinical efficacy data, and the present analysis was conducted for the patients’ remaining lifetime (30 years). This is not uncommon in health economic evaluations as the clinical trials are often of relatively short duration compared to the overall life expectancy of patients. Second, patient (dis-)utilities were not directly assessed in the study setting (Australia) or the trial population; however, it is an acceptable practice to source evidence from independent publications. Finally, the cost of glasses was estimated using publicly available coverage information from an Australian private health fund.29 The authors recognize this could be an under/-overestimation as each health plan offers multiple options to patients. To account for the variance, the cost of glasses by the type of vision correction (readers, progressives, distance, bifocal) was varied by ±20% in one-way sensitivity analysis, and it did not impact the model outcome.
For future studies, an economic analysis evaluating the cost-effectiveness of DFT015 IOL with other EDOF lens options for the correction of presbyopia could be considered. Additionally, quantifying the impact these IOLs may have on orthopedic hazards could be another interesting outcome to consider for future research.
Conclusion
In conclusion, this analysis demonstrates that AcrySof IQ Vivity EDoF IOL is a highly cost-effective treatment strategy at the currently approved “Prostheses List” price in Australia and provides greater improvement in vision-related quality of life outcomes for presbyopic cataract patients compared to standard monofocal IOLs.
Abbreviations
IOL, intraocular lens; EDoF, extended depth of focus; PBAC, Pharmaceutical Benefits Advisory Committee; MSAC, Medical Services Advisory Committee; QALY, quality adjusted life years; PRO, patient reported outcomes; EJP, economically justifiable price; ICER, incremental cost–effectiveness ratio; OWSA, one-way sensitivity analysis; CI, confidence intervals; PSA, probabilistic sensitivity analysis; WTP, willingness-to-pay; MBS, Medicare Benefits Schedule; HTA, health technology assessment.
Acknowledgments
The authors would like to thank Ritu Gupta (Skyward Analytics Pvt. Ltd., India) for support in the model development. The authors would also like to thank Shantanu Jawla (Skyward Analytics Pvt. Ltd., India) and Kayla Mills (Alcon Vision LLC, USA) for managing and providing writing assistance towards the development of this manuscript.
Author Contributions
All authors contributed towards study design, data analysis, and critically revising the manuscript and agreed to be accountable for all aspects of the work.
Funding
The study was sponsored by Alcon Vision LLC, Fort Worth, Texas, USA.
Disclosure
Chandra Bala is an employee of personalEYES Pty Ltd and reports personal fees and non-financial support from Alcon Vision LLC, during the conduct of the study, and grants and non-financial support from Alcon Vision LLC and Johnson & Johnson, outside the submitted work. Paul Athanasiov reports personal fees from Alcon, during the conduct of the study and being managing director of Surgical Order, outside the submitted work. Dr. Chandra Bala, Dr. Paul Athanasiov, and Jason Holland received consulting fees from Alcon. Dr. Mukesh Dhariwal is an employee of Alcon Vision LLC (the study sponsor). Amit Gupta and Hemant Rathi are employees of Skyward Analytics Pvt. Ltd. and Skyward Analytics Pte. Ltd., respectively, and received consulting fees from Alcon to conduct this study. The authors report no other conflicts of interest related to this work.
References
1. Nath M, Khodifad AM, Odayappan A, Christy JS, Manoharan S. Intraocular lens-sling technique: a safe approach for lens implantation in complicated cataract surgery and secondary intraocular lens implantation. Indian J Ophthalmol. 2020;68:632–635.
2. Dhariwal M, Bouchet C, Jawla S. Comparing the long-term impact on health care resources utilization and costs due to various single-piece acrylic monofocal intraocular lens implantation during cataract surgery: a cost-consequence analysis for the United Kingdom, Italy, and Denmark. Clin Ophthalmol. 2019;13:169–176.
3. Binetter R. Your complete guide to cataract surgery. Available from: https://binettereyecentre.com.au/2019/04/08/cataract-surgery-guide/.
4. OECD. Healthcare utilisations cataract surgery procedure. Available from: https://stats.oecd.org/index.aspx?queryid=30167.
5. Davidson RS, Dhaliwal D, Hamilton DR, et al.; Subcommittee ARCS. Surgical correction of presbyopia. J Cataract Refract Surg. 2016;42:920–930.
6. Berdahl J, Bala C, Dhariwal M, Lemp-Hull J, Thakker D, Patient JS. Economic burden of presbyopia: a systematic literature review. Clin Ophthalmol. 2020;14:3439–3450.
7. Liu J, Dong Y, Wang Y. Efficacy and safety of extended depth of focus intraocular lenses in cataract surgery: a systematic review and meta-analysis. BMC Ophthalmol. 2019;19:198.
8. Modi S, Lehmann R, Maxwell A, et al. Visual and patient-reported outcomes of a diffractive trifocal intraocular lens compared with those of a monofocal intraocular lens. Ophthalmology. 2021;128:197–207.
9. Bala C, Poyales F, Guarro M, et al. Multi-country clinical outcomes of a new nondiffractive presbyopia-correcting intraocular lens. J Cataract Refract Surg. 2021; 48(2):136–143.
10. Kondylis G, Klavdianou O, Palioura S. Multifocal and extended depth of focus intraocular lenses. Annals of Eye Science. 2019;4:5.
11. Kohnen T. Nondiffractive wavefront-shaping extended range-of-vision intraocular lens. J Cataract Refract Surg. 2020;46:1312–1313.
12. Alcon. Alcon announces launch of AcrySof IQ Vivity, the first and only non-diffractive extended depth of focus intraocular lens in the U.S. (Media Release); 2021. Available from: https://www.alcon.com/media-release/alcon-announces-launch-acrysof-iq-vivity-first-and-only-non-diffractive-extended.
13. Envision Eye Centre. Navigating your cataract surgery options. Available from: https://www.envisioneye.com.au/eye-conditions/cataracts/navigating-your-cataract-surgery-options/.
14. Australian Government Department of Health. Private health insurance reforms. Available from: https://www.health.gov.au/health-topics/private-health-insurance/private-health-insurance-reforms.
15. Australian Government Department of Health. Prosthesis list-guide to listing. Available from: https://www.health.gov.au/sites/default/files/documents/2020/06/prostheses-list-guide.pdf.
16. Australian Government Department of Health. Prosthesis list part-A. Available from: https://www.health.gov.au/sites/default/files/documents/2021/06/prostheses-list-part-a-prostheses-list.pdf.
17. World Health Organization. Cost-effectiveness analysis for health interventions. Available from: https://www.who.int/heli/economics/costeffanalysis/en/.
18. Jamison DT, Breman JG, Measham AR, et al. editor. Chapter 3. Cost-Effectiveness Analysis. Priorities in Health. In: The International Bank for Reconstruction and Development. Washington (DC): The World Bank; 2006. Available from. https://www.ncbi.nlm.nih.gov/books/NBK10253/.
19. PBAC. Guidelines for preparing a submission to the pharmaceutical benefits advisory committee. Available from: https://pbac.pbs.gov.au/content/information/files/pbac-guidelines-version-5.pdf.
20. Australian Government Department of Health. Technical guidelines for preparing assessment reports for the medical services advisory committee – service type: investigative. Available from: https://consultations.health.gov.au/mbd/msac-investigative-technical-guidelines/supporting_documents/MSAC_Investigative_Guidelines2015%20final%20for%20consultationaccessible%20150415.docx.
21. Institute for Clinical and Economic Review. Cost-effectiveness, the QALY, and the evLYG. Available from: https://icer.org/our-approach/methods-process/cost-effectiveness-the-qaly-and-the-evlyg/.
22. NICE Glossary. Available from: https://www.nice.org.uk/Glossary?letter=Q.
23. Berdahl JP, Bala C, Dhariwal M, Rathi H, Gupta R. A cost-benefit analysis comparing trifocal Intraocular Lens (IOL) with monofocal IOL from patient perspective in the USA.
24. Alcon. Data on File. Clinical Study Report TDOC-0055575; 2019.
25. Hu JQ, Sarkar R, Sella R, Murphy JD, Afshari NA. Cost-effectiveness analysis of multifocal intraocular lenses compared to monofocal intraocular lenses in cataract surgery. American Journal of Ophthalmology. 2019;208:305–312.
26. Australian Bureau of Statistics. 3302055001DO001_20172019 life tables, 2017–2019.
27. MBS schedule. Available from: http://www.mbsonline.gov.au/internet/mbsonline/publishing.nsf/Content/FBE6CC5B217AC8DACA25859E0016F5A3/$File/mbsbook-july2021m.pdf.
28. CBHS corporate health insurance. Available from: https://www.cbhscorporatehealth.com.au/for-individuals/extras-cover/classic-extras.
29. Brown MM, Brown GC, Brown HC, Peet J, Roth Z. Value-based medicine, comparative effectiveness, and cost-effectiveness analysis of topical cyclosporine for the treatment of dry eye syndrome. Arch Ophthalmol. 2009;127:146–152.
30. Dobrez DG, Calhoun EA. Testing subject comprehension of utility questionnaires. Qual Life Res. 2004;13:369–376.
31. YHEC. Economically Justifiable Price. Available from: https://yhec.co.uk/glossary/economically-justifiable-price/.
32. YHEC. Incremental Cost-Effectiveness Ratio (ICER). Available from: https://yhec.co.uk/glossary/incremental-cost-effectiveness-ratio-icer/.
33. Cohen DJ, Reynolds MR. Interpreting the results of cost-effectiveness studies. J Am Coll Cardiol. 2008;52:2119–2126.
34. PBS. Olaparib, 50 mg capsule, Lynparza® AstraZeneca Pty Ltd. Available from: https://www.pbs.gov.au/industry/listing/elements/pbac-meetings/psd/2016-03/files/olaparib-psd-march-2016.pdf.
35. ICER’s reference case for economic evaluations: principles and rationale. Available from: https://icer.org/wp-content/uploads/2020/10/ICER_Reference_Case_013120.pdf.
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
Recommended articles
Clinical Outcomes of a New Non-Diffractive Extended Depth-of-Focus Intraocular Lens Targeted for Mini-Monovision
Tomagova N, Elahi S, Vandekerckhove K
Clinical Ophthalmology 2023, 17:981-990
Published Date: 25 March 2023
