Back to Journals » Clinical Ophthalmology » Volume 14
A Comparison of Efficacy and Safety of Two Lipid-Based Lubricant Eye Drops for the Management of Evaporative Dry Eye Disease
Authors Jerkins G, Greiner JV , Tong L , Tan J , Tauber J , Mearza A , Srinivasan S
Received 1 April 2020
Accepted for publication 27 May 2020
Published 18 June 2020 Volume 2020:14 Pages 1665—1673
DOI https://doi.org/10.2147/OPTH.S256351
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Scott Fraser
Gary Jerkins,1 Jack V Greiner,2,3 Louis Tong,4– 6 Jacqueline Tan,7 Joseph Tauber,8 Ali Mearza,9,10 Sruthi Srinivasan11
1Advancing Vision Research, LLC, Nashville, TN, USA; 2Clinical Eye Research of Boston, Boston, MA, USA; 3Department of Ophthalmology, Harvard Medical School, Boston, MA, USA; 4Singapore National Eye Center, National University of Singapore, Singapore; 5Singapore Eye Research Institute, Singapore; 6Duke-National University of Singapore Medical School, Singapore; 7University of New South Wales, Sydney, NSW, Australia; 8Tauber Eye Center, Kansas City, MO, USA; 9Imperial College Healthcare NHS Trust, London, UK; 10Ophthalmic Consultants of London, London, UK; 11Alcon Research, LLC, Johns Creek, GA, USA
Correspondence: Gary Jerkins
Advancing Vision Research, LLC, Nashville, TN 37205, USA
Tel +1 615-502-2871
Fax +1 615-915-1825
Email [email protected]
Purpose: The aim of this study was to compare the efficacy of two lipid-based lubricant eye drops in patients with lipid-deficient dry eye.
Methods: This Phase IV, multicenter, prospective, double-masked study enrolled adults (aged ≥ 18 years) who had a tear film breakup time (TFBUT) of ≤ 15 seconds(s), and unanesthetized Schirmer I test of ≥ 3 mm to ≤ 12 mm in at least one eye, at both screening and baseline visits. Eligible patients (n=231) were randomized (1:1) and received either Systane® Balance (SYSB; n=117) or Refresh® Optive Advanced (RFO-Ad, n=114), four-times a day, for 35 days. The primary endpoint was non-inferiority for change from baseline in TFBUT at Day 35 (non-inferiority was established if the lower limit of the 95% confidence interval (CI) for the difference between the treatment groups was > – 1.0 s); secondary endpoints (test of superiority) were change in TFBUT and global ocular discomfort visual analog scale (VAS) score at Day 35. Other endpoints included the impact of dry eye on everyday life (IDEEL) treatment satisfaction scores (inconvenience and effectiveness) and safety.
Results: At Day 35, the mean change from baseline in TFBUT was 0.998 s in the SYSB and 0.868 s in the RFO-Ad groups with a treatment difference: 0.130 s; (95% CI − 0.34, 0.60; P< 0.0001) demonstrating non-inferiority of SYSB to RFO-Ad. The global ocular discomfort VAS scores improved in both groups, with a mean change from baseline of – 9.7 and – 8.8 in SYSB and RFO-Ad groups (treatment difference – 0.8; P=0.62), respectively. No meaningful difference was observed in IDEEL treatment effectiveness and treatment inconvenience scores between SYSB vs RFO-Ad (P> 0.05 for treatment difference). Both treatments were well tolerated.
Conclusion: SYSB lubricant eye drops were non-inferior to RFO-Ad for improvement in TFBUT in patients with lipid-deficient dry eye. Both lubricant eye drops improved TFBUT and ocular discomfort scores in patients with lipid-deficient dry eye.
Keywords: dry eye, lipid-deficient, evaporative dry eye, lubricant eye drops, non-inferiority
Introduction
Tear film instability and tear hyperosmolarity are recognized as underlying causes for all types of dry eye disease (DED), a chronic, multifactorial condition affecting the ocular surface.1 DED is a common ocular condition; prevalence increases with age and varies in different geographical areas.2–4 DED can substantially impair quality of life (QoL), as affected individuals experience ocular discomfort, which includes symptoms such as dryness, burning, stinging, grittiness, foreign body sensation, ocular fatigue, visual function disturbance, and sometimes pain (in severe cases) thereby limiting daily activities and work productivity.1,2,5,6
Evaporative dry eye, the most prevalent subtype of DED, is characterized by alteration/or deficiency in the tear film lipid layer.4 The polar and non-polar lipids are essential components of the outmost layer of the tear film and help to stabilize it by reducing the surface tension and spreading the film during blinking.7 A disruption in the rheology of tear film lipids leads to hyperosmolarity and increases evaporation, subsequently resulting in ocular surface dryness, inflammation, and damage.7,8 Evaporative DED is most commonly caused by Meibomian gland dysfunction (MGD).
Artificial tears or lubricant eye drops are designed to mimic natural tears and form an integral part of the management of DED.9 Several artificial tears are commercially available and are used to provide symptomatic relief in patients with DED.9 The composition varies among different artificial tear formulations and this can potentially have an impact on the beneficial effect that each lubricant eye formulation can offer to patients.
Lipid-containing lubricant eye drops improve ocular signs and symptoms in patients with evaporative DED.10–15 Systane® Balance lubricant eye drops (SYSB; Alcon Laboratories, Fort Worth, TX) containing the viscoelastic agent hydroxypropyl (HP)-guar, a phospholipid (dimyristoyl phosphatidylglycerol), and mineral oil, are formulated to provide tear film lipid layer stabilization and minimize the evaporative loss of tears from the ocular surface.10,16 A carboxymethylcellulose (CMC)-based artificial tear formulation containing castor oil and glycerin, Refresh® Optive Advanced (RFO-Ad) lubricant eye drops (Allergan Inc, Dublin, Ireland) is indicated for temporary relief of burning, irritation, and discomfort due to dryness of the eye or exposure to wind or sun.17 SYSB and RFO-Ad have been shown to alleviate the signs and symptoms of DED.10,12,14,15,18-20
The purpose of this study was to evaluate the clinical benefits of SYSB lubricant eye drops and to assess for the non-inferiority of SYSB to RFO-Ad lubricant eye drops in patients with lipid-deficient DED.
Methods
This was a Phase IV (NCT02776670), multicenter, prospective, randomized, double-masked parallel-group study conducted between July 2016 to November 2017, across 14 centers in 5 countries (Australia, Singapore, Taiwan, the United Kingdom, and the United States). The study protocol employed a non-inferiority trial design that was approved by the Independent Ethics Committee or Institutional Review Board of each participating center. The study was conducted in accordance with the Declaration of Helsinki and Good Clinical Practice guidelines and complied with all federal, regional, and local requirements. All patients provided written informed consent prior to enrollment in the study.
Following screening and examination of signs and symptoms of DED, all eligible patients were instructed to instill preservative-free saline (run-in-phase), one drop in each eye 4 times a day (QID), for ≥7 to ≤14 days prior to baseline measurements (Day 0 visit). After the open-label run-in-phase, patients were re-evaluated for eligibility and were randomized (1:1) to one of the two treatment arms and received either SYSB or RFO-Ad lubricant eye drops (1 drop in each eye) instilled QID (with the last dose at bedtime, before midnight), for a period of 35 days (Figure 1). In the United Kingdom, patients received Optive® Plus lubricant eye drops (Allergan, Inc., Irvine, CA, USA), which has the same formulation composition as RFO-Ad.
 |
Figure 1 Study design. Abbreviations: EOS, end of study; QID, four times daily; RFO-Ad, Refresh Optive Advance/Optive Plus; SYSB, Systane Balance; TFBUT, tear film break-up time. |
Patient randomization codes were generated using an interactive response technology system and were stratified by the study center to ensure a balance of study treatment allocations within each investigational center. The test lubricant eye drops were supplied in commercially approved bottles in identical cartons with approved labeling that included, at a minimum, the protocol and kit identification numbers.
Eligibility
Patients ≥18 years of age (≥20 years and ≥21 years of age as per local regulations in Taiwan and Singapore, respectively) with a sum of 3 measurements of tear film breakup time (TFBUT) of ≤15 s, and an unanesthetized Schirmer I test of ≥3 mm to ≤12 mm in at least 1 eye at both screening and baseline visits; patients with a best-corrected visual acuity (BCVA) of ≥55 Early Treatment Diabetic Retinopathy Study (ETDRS) letters in each eye were eligible for inclusion.
Patients who were hypersensitive to any study product or any excipient, had a history of ocular or intraocular surgery, keratorefractive procedure, corneal transplant, or serious ocular trauma in either eye within 6 months prior to the screening visit, had a punctal plug insertion or diathermy procedure or had participated in any other investigational clinical study within 30 days prior to screening visit, had initiated lid hygiene therapy ≤4 weeks prior to the screening visit (Note: Patients who had been on a consistent lid hygiene therapy [ie, no change to the type of lid hygiene therapy being used or to the frequency of use] for >4 weeks prior to the Screening Visit were not excluded. However, they could neither stop or change this regimen for the duration of the study. In addition, patients who were not using lid hygiene therapy at the time could not start for the duration of the study), were taking systemic medications known to cause dry eye (eg antihistamines, antipsychotics, anti-depressants) for <1 month and had any anticipated change in dosing regimen during the course of the study, unwilling to discontinue artificial tear products other than study treatment during the course of the study, or had used any topical ocular medication preserved with benzalkonium chloride or other products known to be toxic to the tear film lipid layer within 3 months prior to the screening visit or had initiated any topical ocular (over-the-counter or prescribed except artificial tears/lubricant eye drops/gels) medications ≤2 weeks prior to screening visit, had any uncontrolled active systemic disease or active ocular infection were not eligible to participate in the study. Additionally, patients who were not willing to discontinue contact lens wear at least 30 days before screening and for the study duration, female patients who were breastfeeding or pregnant, or who had a positive pregnancy test at screening were not considered for enrollment.
Endpoints
The primary endpoint of non-inferiority was based on the change from baseline in TFBUT (seconds [s]) on Day 35. The secondary endpoints included testing for superiority in terms of change from baseline in TFBUT and in global ocular discomfort visual analog scale (VAS) score at Day 35. Exploratory endpoints included the change from baseline in lid wiper epitheliopathy (LWE) scores and in the impact of dry eye on everyday life (IDEEL) treatment satisfaction scores (both treatment effectiveness and treatment inconvenience scores) at Day 35.21,22 Safety assessments included adverse events (AEs), BCVA assessments, and slit-lamp biomicroscopic examination.
Assessments
The TFBUT, LWE, BCVA and slit-lamp biomicroscopic examinations were assessed at the screening, Day 0 (baseline), Day 15, and Day 35 (exit) study visits. The LWE score assessment was only conducted at five select study centers. The ocular discomfort questionnaire and the IDEEL treatment satisfaction questionnaire were provided at Day 0 (Baseline), Day 15 (Visit 2), and Day 35 (Visit 3 or early exit). Details of the assessments are provided in Supplementary file 1.
All AEs reported before the initiation of study treatment were classified as pretreatment AEs and all AEs with an onset after study treatment initiation and up to discontinuation of study treatment (ie 35±7 days) were classified as treatment-emergent AEs.
Statistical Analysis
With at least 200 evaluable patients, the study had an 80% power to demonstrate the non-inferiority of SYSB to RFO-Ad for the mean change from baseline in TFBUT. Non-inferiority was to be established if the lower limit (LL) of the 95% confidence interval (CI) for the mean difference between treatment groups (SYSB−RFO-Ad) was above −1.0 s. The efficacy endpoints were evaluated using a mixed model repeated-measures analysis including baseline assessments, treatment, visit, and treatment-by-visit interaction.
For the secondary endpoints, a P-value of <0.05 for a positive difference between treatment groups (SYSB−RFO-Ad) was to be considered as an advantage of SYSB lubricant eye drops over RFO-Ad. The secondary hypotheses were tested using the Hochberg testing procedure to control the type 1 error rate. Exploratory endpoints were evaluated descriptively. All analyses were performed using SAS® statistical software (Version 9.4). Estimates of the difference in mean change from baseline between treatments and within-group, and the associated 95% CIs are presented.
The primary efficacy analysis was based on the per-protocol set (PPS) population and the secondary efficacy assessments were performed on the full analysis set (FAS) population. The PPS consisted of patients in the FAS who satisfied all inclusion/exclusion criteria and who had no major protocol deviations. The FAS included all randomized patients who had at least one post-baseline primary endpoint (ie, TFBUT) assessment. Safety analyses were conducted using the safety analysis set that included all patients who were exposed to the study treatment, post-randomization.
For efficacy analysis, the study eye was the worst eye for each baseline parameter. If the baseline values were equivalent, the right eye was selected. For safety analyses, the study eye was selected as the eye with the worst change from baseline to any visit (scheduled or unscheduled). If both eyes had the same level of worsening, the right eye was selected.
Results
Of the 308 patients screened, 231 were randomized and received the study treatment (SYSB, n=117; RFO-Ad, n=114). All patients in the SYSB group (n=117) and 108 patients in the RFO-Ad completed the study; 6 patients in the RFO-Ad group discontinued the study (due to: AEs [n=2], protocol violation [n=2], and withdrawal of consent [n=1], and other [n=1, due to surgery]).
The FAS and PPS population included 231 and 228 patients, respectively. Three patients were excluded from the PPS due to protocol deviation (1 patient in the SYSB group) and treatment with incorrect randomized study drug (1 patient in each treatment group). The treatment groups were balanced for demographic and baseline characteristics; mean age of the study cohort was 56.2 years, and 78.8% of patients were female (Table 1).
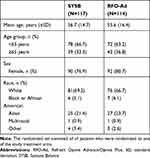 |
Table 1 Demographic Characteristics of Patients by Treatment Groups–Randomized Set |
Primary Outcome
At Day 35, the mean (±standard deviation [SD]) TFBUT showed an increase from baseline of 2.93±1.14 to 3.93±2.26 s in the SYSB group and from 2.90±1.15 s to 3.76±2.09 s in the RFO-Ad group, respectively. The least-square (LS) mean difference between the treatment groups was 0.13 s (95% CI of −0.34, 0.60; P<0.0001) thereby demonstrating the non-inferiority of SYSB to RFO-Ad (Figure 2).
Secondary Outcomes
On Day 35, although the LS mean difference in treatment groups for the change from baseline in TFBUT was numerically in favor of SYSB vs RFO-Ad (treatment difference: 0.12 s; 95% CI −0.35 to 0.59; one-sided P=0.31); however, superiority was not demonstrated, as the P-value was not significant (Table 2).
 |
Table 2 Summary of TFBUT, Global Ocular Discomfort VAS Score, and IDEEL Treatment-Satisfaction Scores (Effectiveness and Inconvenience) by Treatment Group at Each Study Visit- FAS Population |
Both treatment groups showed an improvement in global ocular discomfort VAS score compared with baseline at Day 35 (Table 2). However, the superiority of SYSB over RFO-Ad was not demonstrated as the difference between the groups was not significant (treatment difference: −0.8; 95% CI −6.4 to 4.7; one-sided P=0.62).
Exploratory Outcomes
At baseline and Day 35, the mean±SD LWE scores were 1.3±0.99 and 1.1±0.96 in the SYSB group (n=59), and 1.3±1.0 and 1.0±1.03 in the RFO-Ad group (n=57), respectively. The LS mean (standard error) change from baseline in the LWE score was –0.20 (0.1) in the SYSB group and –0.34 (0.1) in the RFO-Ad (treatment difference = 0.14; 95% CI –0.13, 0.41; P=0.15).
An increase in the mean±SD IDEEL scores for treatment effectiveness was observed in both groups at Day 35 vs baseline (Table 2). The difference between the groups for the LS mean change from baseline in the treatment effectiveness score at Day 35 was 1.3 units (P=0.35), with a nominal trend in favor of SYSB (Figure 3). The IDEEL scores for treatment inconvenience showed a small decrease (ie increased inconvenience) in both groups at Day 35 vs baseline; treatment inconvenience scores were numerically higher (ie less inconvenience) for the SYSB group (79.1±16.83) vs the RFO-Ad group (75.8±17.50; Table 2). The difference between the groups for the LS mean change from baseline in the treatment inconvenience score was 2.3 units (P=0.154), with nominal trend in favor of SYSB (Figure 3).
Safety
Overall, 12% and 14% of patients in the SYSB and RFO-Ad groups, respectively, experienced at least 1 AE (Table 3). Ocular AEs were reported in 7.7% of patients in the SYSB group and 9.6% of patients in the RFO-Ad group. The most common ocular AEs were eye irritation and vital dye staining cornea present in the SYSB group. In the RFO-Ad group, the most common ocular AEs reported were conjunctival hyperemia and eye pain. The corneal dystrophy in five of the patients (1 in SYSB group and 4 in RFO-Ad group) was observed as corneal gutatta, a preexisting condition; the condition was bilateral in two patients, unilateral in the other three, all reported by one investigative site. All ocular AEs were mild in severity.
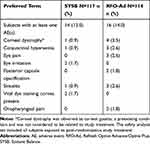 |
Table 3 Proportion of Patients with Common Adverse Events (≥1.0% Incidence) by Preferred Term-Safety Analysis Set |
The ocular AEs were assessed by the investigator as related to the study treatment in 3 (2.6%) patients in the SYSB group (vision blurred and eye irritation in 1 patient, vital dye staining cornea present in 1 patient, and eye irritation in 1 patient) and in 1 patient (0.9%) in the RFO-Ad group (vision blurred, conjunctival disorder, and eye pain).
Non-ocular AEs were reported in 6.0% and 6.1% of patients in the SYSB and RFO-Ad groups, respectively. One non-ocular AE, rhinorrhea in the RFO-Ad group was assessed to be treatment-related. Only 1 (0.9%), non-ocular SAE (abscess in the neck) in the RFO-Ad group was reported in the study; there were no serious AEs in the SYSB group.
Overall, two patients discontinued the study due to ocular AEs as a result of study treatment; one patient experienced eye irritation (note this patient was randomized to the RFO-Ad group but received SYSB) and one patient in the RFO-Ad group experienced vision blurred, conjunctival disorder, and eye pain that led to study discontinuation. No deaths were reported during the study.
Discussion
This study compared the efficacy and safety of two commercially available lipid-containing lubricant eye drops for improvement in TFBUT in lipid-deficient dry eye. The study met its primary objective and demonstrated that SYSB lubricant eye drops are non-inferior to RFO-Ad lubricant eye drops for improvement of TFBUT in patients with lipid-deficient DED. Following QID dosing, patients treated with SYSB and RFO-Ad had a gradual increase in TFBUT and reported a corresponding improvement in ocular discomfort scores from baseline to Day 15 and Day 35. Ocular discomfort is a manifestation of an unstable tear film and its alleviation is an important aspect of DED management to improve the QoL of affected individuals.1,5,8 Taken together these findings support the benefit of the use of SYSB and RFO-Ad lubricant eye drops for immediate relief in signs and symptoms in DED patients. Consistent with the present findings, another comparative study reported improvement in non-invasive TFBUT up to 45-mins post-instillation with RFO-Ad and SYSB in patients with mild evaporative DED.23
Although the secondary endpoint of the superiority of SYSB over RFO-Ad was not demonstrated, the difference observed between the groups with respect to change from baseline in TFBUT showed a nominal numerical trend in favor of SYSB. Previously, SYSB has been shown to significantly extend the TFBUT in MGD patients compared with another mineral oil-containing lubricant eye drops.24 A higher TFBUT is indicative of better tear function and protection of the ocular surface.25 It has been shown that 73% of DED patients report ocular discomfort within 1 s of TFBUT.26 SYSB belongs to the Systane® family of lubricant eye drops which contain the gellable HP-guar that forms a cross-linked matrix in the presence of borate ions on the ocular surface, prolonging retention of the lubricant. SYSB also contains a unique microemulsion of two lipids (LipiTech™ system): an anionic phospholipid, dimyristoylphosphatidyl glycerol, and a non-polar mineral oil.9,16 The anionic polar phospholipids in the lubricant drops act as surfactants and provide a stable interface between neutral non-polar lipids and the aqueous layer thereby replenishing and stabilizing the lipid layer, decreasing tear film evaporation. Studies have shown that the SYSB lubricant eye drops improve lipid layer thickness, ocular surface disease index (OSDI) score, total ocular surface staining scores, goblet cell density, meibomian gland functionality, reduce symptoms, and ocular discomfort in patients with dry eye, MGD, and contact lens users.18,27,28 The RFO-Ad lubricant eye drop formulation contains castor oil (a non-polar lipid that spreads across the aqueous layer of the tear film) which reduces evaporation, CMC that provides lubrication, and glycerin to protect the ocular surface from hypertonic stress. The RFO-Ad drops have been shown to improve TFBUT, reduce dryness symptom scores, OSDI scores, and corneal staining in patients with DED.12,15,20,23
LWE is caused by an unstable tear film that results in friction and inflammation of the marginal conjunctiva of the lid wiper region. The severity of LWE is graded on a scale of 0–3 based on the extent of lid margin staining.22 In the present study, a slight decrease in the severity of LWE scores compared with baseline was observed in both treatment groups but the difference between SYSB and RFO-Ad groups at Day 35 was not meaningful. One possible reason for this could be due to the relatively small number of patients, as this assessment was only performed at selected study sites. The SYSB lubricant drops have been shown to significantly reduce LWE in contact lens users.28,29
Further, consistent with the main findings, patient-reported IDEEL scores for treatment effectiveness showed an improvement on Day 35. IDEEL is a specific questionnaire designed to assess the impact of dry eye on patients’ everyday life, treatment satisfaction, and symptom bother.21 This study used the IDEEL treatment satisfaction module that encompasses treatment effectiveness and inconvenience. A small increase in IDEEL treatment inconvenience was seen with both treatments and a slightly greater inconvenience was reported with RFO-Ad than with SYSB. Though the exact reason for this is not known, it could be due to the viscosity of the lipid-based lubricant eye drops that may cause slight vision blur on instillation or the frequency of dosage that was mandated by the protocol.9 Dry eye patients reported greater inconvenience with QID dosing vs pro-re-nata in a previous study.30 Vision blur was also reported by only one patient in each treatment group in this study. Both formulations were well tolerated and the incidence of ocular AEs were low. No ocular serious AEs were reported and one patient in each treatment group discontinued the study due to an AE. The reported AEs were consistent with the known safety profile of each lubricant drop.
Potential limitations of this study include the absence of long-term follow-up and lack of other objective clinical measures of DED which would have been useful to identify differences between the two treatments and should be considered in future studies. The study did not include objective MGD evaluation which may affect the symptoms. The invasive TFBUT technique is known to be influenced by the volume of the fluorescein dye, residual tear volume, and environmental conditions such as temperature, humidity, and air circulation and is therefore considered less reproducible than non-invasive measurements.25,31 Further, the absence of a “gold standard” of dry eye assessment, the great degree of variability in symptoms displayed by DED patients, as well as a lack of correlation between signs and symptoms, makes it challenging to compare the relative efficacy of two lubricant eye drops.31,32
In conclusion, the SYSB lubricant eye drops were non-inferior to the RFO-Ad lubricant eye drops for improvement in TFBUT. Overall, both treatments improved TFBUT, reduced ocular discomfort, and were well tolerated, in patients with lipid-deficient DED.
Data Sharing Statement
The study results are available on clinicaltrials.gov. https://clinicaltrials.gov/ct2/show/results/NCT02776670. Due to varying rights of individuals and contractual rights of parties involved, Alcon does not make a practice of sharing datasets.
Ethics Approval and Consent to Participate
The study protocol was approved by the Institutional Review Board of each participating country (Bellberry Limited, Adelaide, South Australia; Human Research Ethics Committee, The University of New South Wales, Australia; Singhealth Centralised Institutional Board Singapore Health Services Pte Ltd; Singapore; Research Ethics Review Committee, Far Eastern Memorial Hospital, Taiwan; South West Ethics Research Committee, Bristol HRA Centre, UK; Advarra – Advancing Better Research; Columbia, USA). The study was conducted in accordance with the Declaration of Helsinki and Good Clinical Practice (GCP) and was in compliance with all federal, local, or regional requirements. All participants provided written informed consent before entering the study.
Acknowledgment
Manuscript writing support was provided by Shivani Vadapalli (Novartis Healthcare Pvt. Ltd., Hyderabad, India).
Disclosure
Dr Louis Tong reports research funding, advisory boards, travel sponsorship from Alcon, Novartis, Santen, Allergan, Bausch and Lomb. Dr Jacqueline Tan reports grants from Allergan, Azura Ophthalmics, and Stiltec, outside the submitted work. Dr Joseph Tauber reports grants from Alcon, during the conduct of the study. Dr Gary Jerkins is affiliated with Advancing Vision Research, LLC. Dr Sruthi Srinivasan is an employee of Alcon Research, LLC. The authors report no other conflicts of interest in this work.
References
1. Craig JP, Nelson JD, Azar DT, et al. TFOS DEWS II report executive summary. Ocul Surf. 2017;15(4):802–812. doi:10.1016/j.jtos.2017.08.003
2. Lemp MA, Crews LA, Bron AJ, Foulks GN, Sullivan BD. Distribution of aqueous-deficient and evaporative dry eye in a clinic-based patient cohort: a retrospective study. Cornea. 2012;31(5):472–478. doi:10.1097/ICO.0b013e318225415a
3. Dana R, Bradley JL, Guerin A, et al. Estimated prevalence and incidence of dry eye disease based on coding analysis of a large, all-age United States health care system. Am J Ophthalmol. 2019;202:47–54. doi:10.1016/j.ajo.2019.01.026
4. Stapleton F, Alves M, Bunya VY, et al. TFOS DEWS II epidemiology report. Ocul Surf. 2017;15(3):334–365. doi:10.1016/j.jtos.2017.05.003
5. Uchino M, Schaumberg DA. Dry eye disease: impact on quality of life and vision. Curr Ophthalmol Rep. 2013;1(2):51–57. doi:10.1007/s40135-013-0009-1
6. Yu J, Asche CV, Fairchild CJ. The economic burden of dry eye disease in the United States: a decision tree analysis. Cornea. 2011;30(4):379–387. doi:10.1097/ICO.0b013e3181f7f363
7. Cwiklik L. Tear film lipid layer: a molecular level view. Biochim Biophys Acta. 2016;1858(10):2421–2430. doi:10.1016/j.bbamem.2016.02.020
8. Milner MS, Beckman KA, Luchs JI, et al. Dysfunctional tear syndrome: dry eye disease and associated tear film disorders - new strategies for diagnosis and treatment. Curr Opin Ophthalmol. 2017;27(Suppl 1):3–47. doi:10.1097/01.icu.0000512373.81749.b7
9. Jones L, Downie LE, Korb D, et al. TFOS DEWS II management and therapy report. Ocul Surf. 2017;15(3):575–628. doi:10.1016/j.jtos.2017.05.006
10. Aguilar AJ, Marquez MI, Albera PA, Tredicce JL, Berra A. Effects of systane((R)) balance on noninvasive tear film break-up time in patients with lipid-deficient dry eye. Clin Ophthalmol. 2014;8:2365–2372. doi:10.2147/OPTH.S70623
11. Fogt JS, Kowalski MJ, King-Smith PE, et al. Tear lipid layer thickness with eye drops in meibomian gland dysfunction. Clin Ophthalmol. 2016;10:2237–2243. doi:10.2147/OPTH.S120158
12. Simmons PA, Carlisle-Wilcox C, Vehige JG. Comparison of novel lipid-based eye drops with aqueous eye drops for dry eye: a multicenter, randomized controlled trial. Clin Ophthalmol. 2015;9:657–664. doi:10.2147/OPTH.S74849
13. Simmons PA, Liu H, Carlisle-Wilcox C, Vehige JG. Efficacy and safety of two new formulations of artificial tears in subjects with dry eye disease: a 3-month, multicenter, active-controlled, randomized trial. Clin Ophthalmol. 2015;9:665–675. doi:10.2147/OPTH.S78184
14. Sindt CW, Foulks GN. Efficacy of an artificial tear emulsion in patients with dry eye associated with meibomian gland dysfunction. Clin Ophthalmol. 2013;7:1713–1722. doi:10.2147/OPTH.S35833
15. Kaercher T, Thelen U, Brief G, Morgan-Warren RJ, Leaback R. A prospective, multicenter, noninterventional study of optive plus((R)) in the treatment of patients with dry eye: the prolipid study. Clin Ophthalmol. 2014;8:1147–1155. doi:10.2147/OPTH.S58464
16. Benelli U. Systane lubricant eye drops in the management of ocular dryness. Clin Ophthalmol. 2011;5:783–790. doi:10.2147/OPTH.S13773
17. Refresh Optive® Lubricant eye drops. Available from: https://dailymed.nlm.nih.gov/dailymed/fda/fdaDrugXsl.cfm?setid=7f8d88af-fd3b-4138-af61-f24c59ccebd7&type=display.
18. Baudouin C, Galarreta DJ, Mrukwa-Kominek E, et al. Clinical evaluation of an oil-based lubricant eyedrop in dry eye patients with lipid deficiency. Eur J Ophthalmol. 2017;27(2):122–128. doi:10.5301/ejo.5000883
19. Foulks GN, Sindt C, Griffin J. Efficacy evaluation of a novel emulsion based, anionic phospholipid containing artificial tear in meibomian gland dysfunction (MGD) subjects.
20. Simmons PA, Carlisle-Wilcox C, Chen R, Liu H, Vehige JG. Efficacy, safety, and acceptability of a lipid-based artificial tear formulation: a randomized, controlled, multicenter clinical trial. Clin Ther. 2015;37(4):858–868. doi:10.1016/j.clinthera.2015.01.001
21. Abetz L, Rajagopalan K, Mertzanis P, et al. Development and validation of the impact of dry eye on everyday life (IDEEL) questionnaire, a patient-reported outcomes (PRO) measure for the assessment of the burden of dry eye on patients. Health Qual Life Outcomes. 2011;9(1):111. doi:10.1186/1477-7525-9-111
22. Korb DR, Herman JP, Greiner JV, et al. Lid wiper epitheliopathy and dry eye symptoms. Eye Contact Lens. 2005;31(1):2–8. doi:10.1097/01.ICL.0000140910.03095.FA
23. Connor C, Ottenbreit R, Schroede RL, Rabin J, Narayanan S. Comparison of three commercially available tear substitutes designed for evaporative dry eye treatment.
24. Korb DR, Blackie CA, Meadows D, Christensen MT, Tudor MR. Evaluation of extended tear stability by two emulsion based artificial tears.
25. McMonnies CW. Tear instability importance, mechanisms, validity and reliability of assessment. J Optom. 2018;11(4):203–210. doi:10.1016/j.optom.2017.11.004
26. Nally L, Ousler GW, Abelson MB. Ocular discomfort and tear film break-up time in dry eye patients: a correlation. IOVS. 2000;41(4(ARVO Abstract)):1436.
27. Korb DR, Blackie CA, Finnemore VM, Douglass T. Effect of using a combination of lid wipes, eye drops, and omega-3 supplements on meibomian gland functionality in patients with lipid deficient/evaporative dry eye. Cornea. 2015;34(4):407–412. doi:10.1097/ICO.0000000000000366
28. Guthrie SE, Jones L, Blackie CA, Korb DR. A comparative study between an oil-in-water emulsion and nonlipid eye drops used for rewetting contact lenses. Eye Contact Lens. 2015;41(6):373–377. doi:10.1097/ICL.0000000000000138
29. Elder K, Mickles C, Azhar J, Belloli A, Hans S. The effect of artificial tear treatment on lid wiper epitheliopathy and ocular surface staining in contact lens wearers.
30. Asbell P, Vingrys AJ, Tan J, et al. Clinical outcomes of fixed versus as-needed use of artificial tears in dry eye disease: a 6-week, observer-masked phase 4 clinical trial. Invest Ophthalmol Vis Sci. 2018;59(6):2275–2280. doi:10.1167/iovs.17-23733
31. Wolffsohn JS, Arita R, Chalmers R, et al. TFOS DEWS II diagnostic methodology report. Ocul Surf. 2017;15(3):539–574. doi:10.1016/j.jtos.2017.05.001
32. Alves M, Fonseca EC, Alves MF, et al. Dry eye disease treatment: a systematic review of published trials and a critical appraisal of therapeutic strategies. Ocul Surf. 2013;11(3):181–192. doi:10.1016/j.jtos.2013.02.002
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.


