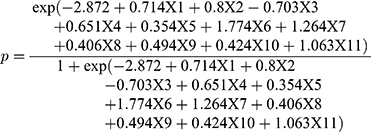Back to Journals » Infection and Drug Resistance » Volume 14
A Comparative Study of Chest Computed Tomography Findings: 1030 Cases of Drug-Sensitive Tuberculosis versus 516 Cases of Drug-Resistant Tuberculosis
Authors Cheng N, Wu S, Luo X, Xu C, Lou Q, Zhu J, You L, Li B
Received 12 January 2021
Accepted for publication 4 March 2021
Published 18 March 2021 Volume 2021:14 Pages 1115—1128
DOI https://doi.org/10.2147/IDR.S300754
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Suresh Antony
Nianlan Cheng, Shuo Wu, Xianli Luo, Chunyan Xu, Qin Lou, Jin Zhu, Lu You, Bangguo Li
Department of Radiology, Affiliated Hospital of Zunyi Medical University, Zunyi, Guizhou Province, People’s Republic of China
Correspondence: Bangguo Li
Department of Radiology, Affiliated Hospital of Zunyi Medical University, Zunyi, Guizhou Province, People’s Republic of China
Tel +86 13511816288
Email [email protected]
Purpose: To investigate the CT features of drug-resistant pulmonary tuberculosis (DR-PTB) and the diagnostic value of CT in DR-PTB diagnosis to provide imaging evidence for the timely detection of drug-resistant Mycobacterium tuberculosis.
Materials and Methods: A total of 1546 cases of pulmonary tuberculosis (PTB) with complete clinical data, chest CT images and defined drug sensitivity testing results were consecutively enrolled; 516 cases of DR-PTB were included in the drug-resistant group, and 1030 cases of drug-sensitive pulmonary tuberculosis (DS-PTB) were included in the drug-sensitivity group. Comparative analyses of clinical symptoms and imaging findings were conducted. Univariate and logistic regression analyses were performed, a regression equation model was developed, and the receiver operating characteristic (ROC) curve was constructed.
Results: In the univariate analysis, some features, including whole-lung involvement, multiple cavities, thick-walled cavities, collapsed lung, disseminated lesions along the bronchi, bronchiectasis, emphysema, atelectasis, calcification, proliferative lesions, encapsulated effusion, etc., were observed more frequently in the DR-PTB group than in the DS-PTB group, and the differences were statistically significant (p< 0.05). Exudative lesions and pneumoconiosis were observed more frequently in the drug-sensitivity group than in the drug-resistant group (p< 0.05). Logistic regression analysis indicated that whole-lung involvement, multiple cavities, thick-walled cavities, disseminated lesions along the bronchi, bronchiectasis, and emphysema were independent risk factors for DR-PTB, and exudative diseases were protective factors. The total prediction accuracy of the regression model was 80.6%, and the area under the ROC curve (AUC) was 82.6%.
Conclusion: Chest CT manifestations of DR-PTB had certain characteristics that significantly indicated the possibility of drug resistance in tuberculosis patients, specifically when multifarious imaging findings, including multiple cavities, thick-walled cavities, disseminated lesions along the bronchi, whole-lung involvement, etc., coexisted simultaneously. These results may provide imaging evidence for timely drug resistance detection in suspected drug-resistant cases and contribute to the early diagnosis of DR-PTB.
Keywords: pulmonary tuberculosis, drug resistance, tomography, X-ray computed, imaging findings, diagnosis, Mycobacterium tuberculosis
Introduction
Drug-resistant tuberculosis (DR-TB) refers to tuberculosis caused by drug-resistant Mycobacterium tuberculosis (MTB). The emergence and rapid propagation of drug-resistant tuberculosis strains, especially multidrug-resistant tuberculosis (MDR-TB) and extensively drug-resistant tuberculosis (XDR-TB) strains, have led to the prolongation of the tuberculosis treatment cycle to a great extent and have been associated with unsatisfying clinical treatment effects and high costs. Drug resistance is one of the greatest challenges in tuberculosis control in the current century.1–3 The early diagnosis and treatment of DR-TB is key in limiting the epidemic and propagation of DR-TB. At present, the most common approaches and gold standards for the clinical detection of MTB resistance are MTB culture and drug sensitivity testing (DST).4,5 They are both time-consuming and are often performed only after empiric therapy failure; thus, a large number of patients with DR-TB do not obtain a timely diagnosis and effective treatment. Chest computed tomography (CT) is the principal method for diagnosis, follow-up and therapeutic efficacy evaluation of pulmonary tuberculosis (PTB). If medical staff can utilize CT signs to identify suspected resistance in patients infected with MTB and thus perform timely DST in the laboratory, the DR-TB diagnosis time can be effectively shortened, and the treatment plan can be adjusted in time to increase therapeutic effect. This study analyzed a large sample of cases of drug-resistant pulmonary tuberculosis (DR-PTB) and drug-sensitive pulmonary tuberculosis (DS-PTB) and analyzed and summarized the clinical and imaging manifestations of DR-PTB, aiming to identify chest CT features associated with DR-PTB.
Materials and Methods
Patients
A total of 1546 consecutive patients with PTB from October 2008 to September 2017 were consecutively enrolled based on etiological or pathological results; there were 516 cases of DR-PTB and 1030 cases of DS-PTB (945 men, 601 women, age range 2–97 years, mean age 46.59±18.89 years). The inclusion criteria were as follows: (a) explicit DST results, (b) complete clinical diagnosis and treatment data, and (c) complete chest CT data with an interval between the CT scan and DST of less than 1 month. This study was conducted in accordance with the Declaration of Helsinki. The ethics committee of Affiliated Hospital of Zunyi Medical University had approved the study without requiring informed consent for it was an observational retrospective study, and all patient data were analyzed anonymously.
Research Methods
Laboratory Examinations
The DST results of cases in this study were provided by the respiratory medicine laboratory in our hospital. The clinical MTB specimens were obtained from sputum, bronchial brushing, aspiration, pleuroperitoneal effusion, etc. Sputum culture and DST were carried out in strict accordance with standard laboratory procedures.
Equipment and Parameters
Chest CT was performed using a variety of multidetector row CT scanners, including a 16-slice spiral CT instrument (Somatom Sensation 16; Siemens Medical Systems, Erlangen, Germany), a dual-source CT instrument (Somatom Definition Flash; Siemens Healthcare, Forchheim, Germany), an Optima CT 680 instrument (General Electric Medical Systems; Milwaukee, WI, USA) and a 128-slice CT instrument (Somatom Definition AS 128; Siemens Medical Systems, Erlangen, Germany).
The images were conducted using a slice thickness of 8–10 mm from the raw data, and all the images were displayed at standard window settings that allowed viewing of the lung parenchyma (window level, −500~-600 HU; window width, 1500~1800 HU) and mediastinum (window level, 40–50 HU; window width, 250–350 HU).
Image Analysis
The patients in the drug-resistant group and drug-sensitive group were classified according to the results of DST, and the CT data of patients were retrieved from the Picture Archiving and Communication System (PACS). If there were two or more CT data points, the point for which the most recent examination time was closest to the time of DST was selected for analysis. The images of the two groups were sorted according to the size of the CT number after archiving and read in order.
Two radiologists performed chest imaging diagnosis by reviewing all CT images in a blinded manner. The CT images of diagnosed patients (including DR-TB and DS-TB patients) were selected to preliminarily read before formal reading to reach an agreement on the recording principle and the judgment of lesion manifestations, nature, and extent. If the results of the two reviews were not in accordance, a result was negotiated between the two radiologists or decided by referring to the opinion of a superior physician.
Research Content
Clinical Data
Clinical data included the following: 1) sex; 2) age; 3) diagnostic type: primary PTB, hematogenous PTB (include acute, subacute and chronic types; chest CT features of acute ones consist of profuse tiny, discrete, rounded pulmonary opacities that are generally uniform in size and density and diffusely distributed throughout the lungs), secondary PTB; 4) treatment type: no treatment, retreatment; 5) clinical characteristics: cough and expectoration, hemoptysis, fever, chest distress and (or) chest pain, shortness of breath, etc.; 6) history of intermittent medication.
CT Imaging Data
CT imaging data included the following: 1) extent of lesions: right upper lobe, right middle lobe, right lower lobe, left upper lobe, left lower lobe; 2) total number of lobes involved; 3) tracheobronchial lesion characteristics: emphysema, atelectasis, bronchial stenosis or occlusion, bronchial wall thickening, bronchiectasis, disseminated lesions along bronchi (CT scans mainly manifested as the tree-in-bud pattern which represents centrilobular branching structures that resemble a budding tree. Most lesions locate around the bronchi and form multiple tiny nodules);6 4) pulmonary lesion characteristics: exudative lesions (namely the inflammation of the airspaces and/or interstitium. Patchy, ground-glass or flocculent opacities can be found in bilateral or unilateral lungs on CT scans),6 proliferative lesions (CT scans mainly manifested as single or multiple nodules, masses and (or) large patchy opacities with distinct edges and slowly dynamic changes), fibrotic lesions (consist of fiber texture, and are the healing features of acute and chronic pneumonia, CT scans mainly manifested as the irregular strip or reticulate opacities), calcification, nodules or masses, cavities; 5) cavity-related manifestations: number, location, size, thickness of cavity wall, number of involved lobes, cavities with gas-liquid interface (namely gas, liquid and gas-liquid interface all can be found in cavities), tunnel-like cavities (the main principal manifestation of caseous pneumonia, CT scans mainly manifested as extensive destruction of lung tissues and large patchy opacities with small multiple ecavities), etc.; 6) pleural lesion characteristics: pleural effusion, encapsulated effusion, pneumothorax, hydropneumothorax, pleural thickening and calcification, pleural nodules, etc.; 7) changes in mediastinal and hilar lymph nodes: swelling, calcification; and 8) complications: collapsed lung, fibrosclerosis, pneumoconiosis, fungal ball, etc. If there were two or more CT data points, the outcome (improvement, no obvious change, progression) was recorded.
Statistical Analysis
All data was analyzed with SPSS 18.0 statistical software (IBM, Armonk, New York), and p<0.05 was considered statistically significant. Single-factor analysis was performed with the chi-square test or t-test according to the data type. Then, the independent risk factors leading to drug resistance were screened by performing a logistic regression analysis in which the factors with significant differences (p<0.01) were considered independent variables, the drug-resistance status was the dependent variable, and the regression equation model was achieved. Finally, the receiver operating characteristic (ROC) curve and the area under the curve (AUC) were used to assess the predictive efficacy of the regression equation model and calculate the optimal cutoff value for predicting drug resistance; calibration was verified by the Hosmer–Lemeshow test.
Results
Clinical Data
The clinical data of patients in the drug-resistant group and drug-sensitive group are summarized in Table 1. In this study, there was no significant difference in age (p>0.05) and sex ratio (p>0.05) between the drug-resistant group and the drug-sensitive group. The diagnostic types and clinical characteristics did not differ significantly between the two groups (p>0.05), but retreated patients and the patients with an intermittent medication history were more frequently observed in the drug-resistant group than in the drug-sensitive group, and the differences were statistically significant (p<0.05) (Table 1).
 |
Table 1 Comparison of Clinical Data Between the Drug-Resistant Group and the Drug-Sensitive Group |
CT Imaging Data
A summary of the CT findings in the drug-resistant group and drug-sensitive group is provided in Tables 2–5. The extent of lesions were classified according to the lung lobe unit in this study, and there was no significant difference in the extent of lesions between the drug-resistant group and the drug-sensitive group (p>0.05). Regarding the total number of lobes involved, whole-lung involvement was more commonly observed in the drug-resistant group than in the drug-sensitive group (p<0.01), whereas involvement in two lung lobes and four lung lobes was more frequently observed in the drug-sensitive group (p<0.05) (Table 2).
 |
Table 2 Comparison of Imaging Data (Extent of Lesions and Total Number of Lobes Involved) Between the Drug-Resistant Group and Drug-Sensitive Group |
 |
Table 3 Comparison of Imaging Data (Tracheobronchial Lesions, Pulmonary Lesions and Pleural Lesions) Between the Drug-Resistant Group and Drug-Sensitive Group |
 |
Table 4 Comparison of Imaging Data (Changes in Mediastinal and Hilar Lymph Nodes, Complications and Outcome) Between the Drug-Resistant Group and Drug-Sensitive Group |
 |
Table 5 Comparison of Cavity-Related Manifestations Between the Drug-Resistant Group and the Drug-Sensitive Group |
There was a significant association between the drug-resistance status and emphysema (p<0.01), atelectasis (p<0.05), bronchiectasis (p<0.01) and disseminated tracheobronchial lesions along the bronchi (p<0.01). Nevertheless, the distributions of patients with bronchial stenosis or occlusion and bronchial wall thickening (p>0.05) were similar among those with a drug-resistant state and a drug-sensitive state. Regarding pulmonary lesions, proliferative lesions (p<0.01), calcification (p<0.01) and cavities (p<0.01) were more frequently observed in the drug-resistant group than in the drug-sensitive group, while exudative lesions were more common in the drug-sensitive group, and the difference was statistically significant (p<0.01). Moreover, there were no significant differences in the distributions of patients with fibrotic lesions and nodules or masses (p>0.05). Regarding pleural lesions, the distributions of patients with encapsulated effusion (p<0.01) and pleural thickening and calcification (p<0.05) were significantly greater in the drug-resistant group than in the drug-sensitive group. However, no significant differences were detected between the two groups in the distribution of patients with pleural effusion, pneumothorax, hydropneumothorax and pleural nodules (p>0.05) (Table 3).
The rates of calcification and swelling of mediastinal and hilar lymph nodes did not differ significantly between the two groups (p>0.05). Regarding complications, a collapsed lung (p<0.01) was more frequently observed in the drug-resistant group than in the drug-sensitive group, and pneumoconiosis (p<0.05) was more common in the drug-sensitive group, with a statistically significant difference. In contrast, there were no significant differences in the distributions of patients with fungal ball and fibrosclerosis between the two groups (p>0.05). Regarding outcomes, the number of cases for comparison was 275 and 443 in the drug-resistant group and drug-sensitive group, respectively. Progression (p<0.01) and no obvious change (p<0.05) in illness were observed more frequently in the drug-resistant group than in the drug-sensitive group, whereas the distribution of improved cases in the drug-sensitive group was significantly higher than that in the drug-resistant group (p<0.01) (Table 4).
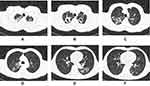
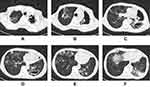
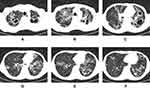
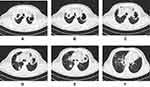
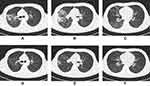
As shown in Table 5, among the cavity-related manifestations, there were significant associations between the drug-resistant state and gas-liquid interface cavities (p<0.01), thick-walled cavities (p<0.01), multiple cavities (p<0.01) and multilobe cavities (p<0.01). In addition, the distribution of patients with tunnel-like cavities was not significantly different between the two groups (p>0.05) (Table 5, Figures 1 and 2).
Logistics Regression and ROC Curve Analysis
The logistic regression analysis results are presented in Table 6. Whole-lung involvement (OR, 2.042; 95% CI, 1.544∼2.699; p<0.001), emphysema (OR, 2.225; 95% CI, 1.572∼3.150; p<0.001), encapsulated effusion (OR, 1.918; 95% CI, 1.112~3.306; p=0.019), bronchiectasis (OR, 1.425; 95% CI, 1.049~1.935; p=0.024), disseminated lesions along the bronchi (OR, 5.896; 95% CI, 4.356~7.979; p<0.001), proliferative lesions (OR, 3.541; 95% CI, 2.328~5.385; p<0.001), calcification (OR, 1.501; 95% CI, 1.015~2.220; p<0.001), multiple cavities (OR, 1.639; 95% CI, 1.103~2.435; p=0.015), thick-walled cavities (OR, 1.528; 95% CI, 1.118~2.088; p=0.008), and disease progression (OR, 2.896; 95% CI, 1.883~4.453; p<0.001) were significant risk factors, and exudative lesions (OR, 0.495; 95% CI, 0.357~0.686; p<0.001) was a protective factor for DR-PTB (Table 6).
 |
Table 6 Logistic Regression Analysis Results |
X1: Whole-lung involvement, X2: Emphysema, X3: Exudative lesions, X4: Encapsulated effusion, X5: Bronchiectasis, X6: Disseminated lesions along the bronchi, X7: Proliferative lesions, X8: Calcification, X9: Multiple cavities, X10: Thick-walled cavities, X11: Progression.
The prediction model was established with the above regression equation, with a total accurate prediction rate of 80.6%. The false-positive and false-negative rates were 9.4% and 39.3%, respectively, and the prediction specificity and sensitivity were 90.6% and 60.7%, respectively. In addition, the Hosmer–Lemeshow test indicated that there were no significant differences between the model predictions and the actual observations (p=0.054). To further verify and evaluate the prediction efficiency of the regression model, the predicted values of the drug-resistance probability calculated by the regression model were analyzed by ROC curves (Figure 3). The AUC used to distinguish DR-PTB from DS-PTB was 0.826 (95% CI, 0.803∼0.849, p<0.001), and the optimal cutoff value was 0.3542567 (Figures 4–6).
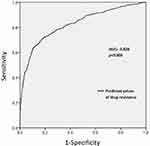 |
Figure 3 ROC curve of predicted values of drug-resistance probability in the regression model. |
Discussion
The DR-PTB is the PTB, which has been proven to be resistant to one or more anti-tuberculosis drugs. The main cause of developing DR-PTB are non-standard anti-tuberculous treatment and the spread of resistant strains. The diagnosis of DR-PTB mainly relies on laboratory including traditional bacteriological testing and rapid molecular detection technology, however, the main detection methods and gold standards are still the time-consuming MTB culture and DST7; thus a large number of DR-PTB patients fail to get the best treatment. Therefore, reducing the diagnosis time of DR-PTB through an effective method such as CT scans is of great significance to improving prognosis and decreasing the mortality of DR-PTB patients.
Clinical Features of DR-PTB
It was reported that young and middle-aged males have the highest rates of DR-PTB, and it is especially prevalent in migrant workers who are often out of treatment supervision and do not complete standard treatment; thus, PTB easily develops into DR-PTB.8 In terms of clinical characteristics, previous authors have suggested that the clinical manifestations are affected by different pathological bases, stages of lesion development and individual differences, etc., and the coexistence of multiple symptoms is becoming increasingly common. There are no specific identifying characteristics compared with those of DS-PTB, but the severity of the disease is greater and the duration of symptoms is longer.
Most of the patients with DR-PTB must be retreated because of disease complexity and poor compliance; they often practice intermittent treatment because of protopathic diseases, drug side effects and other reasons. Intermittent treatment is the main reason for the development of DR-PTB, followed by unreasonable chemotherapy regimens.9,10 Therefore, patients with a history of intermittent treatment are prone to drug resistance. In addition, the clinical prognosis of DR-PTB is poor, and the disease, especially MDR-TB and XDR-TB, progresses easily due to a lack of effective drugs.11–13 In the present study, drug resistance occurred more frequently in patients with retreatment, a history of intermittent treatment and disease progression, and disease progression was an independent risk factor for drug resistance, consistent with previous reports.
Imaging Features of DR-PTB
The imaging features of DR-PTB were summarized in this research through a comparison and analysis of chest imaging findings in patients with DR-PTB and DS-PTB, and it was indicated that patients with DR-PTB were more likely than those with DS-PTB to exhibit the following imaging signs:
An Extensive Range of Involvement
In our study, the distribution of cases with whole-lung involvement in the drug-resistant group was significantly greater than that in the drug-sensitive group. Meanwhile, previous studies suggested that the distribution of cases involving more than 3 lobes was significantly greater in the DR-PTB than in the DS-PTB,14 which was consistent with this study. Lesions were classified according to the lung lobe unit in this study, and there was no significant difference in lesion location between the two groups, but previous study has suggested that uncommon sites, such as the anterior segments of bilateral superior lobes and basal segments of lower lobes, were also easily invaded in DR-PTB patients in addition to the common sites of TB.15
Thick-Walled Cavities and Multiple Cavities
Tuberculosis cavities are considered not only important routes in transmitting MTB but also the biological basis of drug resistance in PTB.16 DR-PTB patients, especially those with tuberculosis cavities, tend to have a higher sputum smear-positivity rate and MTB load than DS-PTB patients. Cavity walls, particularly thick walls with surrounding vascular rarefication, serve as favorable barriers; consequently, it is difficult to attain an effective drug concentration in the cavities, resulting in the repeated and continuous excretion of bacteria and a high bacterial load, which may increase the possibility of spontaneous gene mutations. Ultimately, drug resistance was developed by the establishment of drug-resistant bacterial groups.17–19
Previous literatures showed that matrix metalloproteinases (MMPs), especially MMP-9, are key factors in the formation of cavities at the molecular level. MMPs are capable of destroying and degrading the extracellular matrix and eroding the airway to further accelerate the formation of cavities.20 The activity and expression level of MMPs in PTB are related to the extent of infection.21 DR-PTB can effectively exert a strong ability to degrade extracellular matrix, which promotes the increase in and enlargement of cavities due to severe infection and high-level expression of MMPs. It can be seen that there is a greater correlation between cavities and DR-TB.
Cavities were more frequently observed in the drug-resistant group than in the drug-sensitive group in the present study; moreover, multiple cavities and thick-walled cavities were independent risk factors for DR-PTB. Multiple cavities and thick-walled cavities are closely associated with the occurrence of DR-PTB, especially MDR-TB and XDR-TB. As such, multiple cavities can be considered highly specific signs in imaging for MDR-TB, particularly when the number of cavities is ≥3 and the diameter of cavities is ≥30 mm.17 Cavities in XDR-TB patients may have an even thicker wall and larger size than those in MDR-TB patients, and the cavity is a key factor in transforming MDR-TB into XDR-TB during treatment.22–24 Thus, the tuberculosis cavity is perhaps an important sign that PTB patients may have developed resistance, and it is one of the most pivotal factors in drug resistance, especially multiple cavities and thick-walled cavities.
Bronchial Disseminated Lesions
DR-PTB is often accompanied by a high MTB load; thus, it progresses easily and forms bronchial disseminated lesions with marked activity.17 The probability of disseminated lesions, such as acinar nodules in DR-PTB patients, especially MDR-TB and XDR-TB patients, was significantly higher than that in DS-PTB patients.24 In this study, disseminated lesions along the bronchi were observed more frequently in the drug-resistant group than in the drug-sensitive group and became an independent risk factor for drug resistance, indicating that bronchial disseminated lesions, which are one of the most significant imaging signs of active PTB, may be more common in DR-PTB patients than in DS-PTB patients. In addition, the transmission of MTB depends on its ability to damage lung tissues to a large extent.25 Consequently, DR-MTB with substantial lung tissue damage is likely to cause the extensive spread of TB lesions.
Bronchiectasis
The results of this study suggested that bronchiectasis was observed more frequently in DR-PTB patients and was an independent risk factor for drug resistance. Tuberculosis, especially DR-PTB with high virulence, causes bronchial wall damage and inflammatory changes by invading airway mucosa, potentially leading to severe bronchial injury and chronic invasion. Extensive fibrous tissue hyperplasia during a long disease course results in tracheobronchial stenosis, distortion and inadequate drainage due to traction and compression that ultimately promote bronchiectasis. Levofloxacin, streptomycin and other medicines are commonly used to relieve bronchiectasis symptoms,26,27 and their frequent application further aggravates the risk of drug resistance.
Emphysema
Emphysema can cause compression of the alveolar wall capillaries and disturbance to the blood supply to the alveolar wall. Chronic inflammatory stimulation and PTB recurrence can accelerate the progression of emphysema.28 Therefore, PTB patients with comorbid emphysema repeatedly achieve an unsatisfactory curative effect due to poor blood supply, resulting in drug resistance due to the repeated excretion of bacteria from the lesions. In this study, emphysema was observed more frequently in the drug-resistant group than in the drug-sensitive group and was an independent risk factor for drug resistance, indicating that emphysema may increase the risk of MTB drug resistance to a certain extent. In addition, emphysema is the most basic and main manifestation of chronic obstructive pulmonary disease (COPD). Second-line antituberculous drugs, such as fluoroquinolones, which are the core of the DR-PTB therapeutic regimen, are often used as symptomatic therapy for COPD during acute exacerbation.29 Their intermittent use is an important risk factor for the development of DR-PTB.
Proliferative Lesions and Calcification
In the current study, proliferative lesions occurred more frequently in the drug-resistant group, indicating that proliferative lesions may be regarded as common imaging signs of DR-PTB. Previous authors have suggested that the proliferative lesions are not only one of the main manifestations of protracted and recurrent illness but also particularly common manifestations with a wide extent of involvement in DR-PTB, which is manifested mainly as multiple large nodules and clustered small nodules. Multiple proliferative nodules are a pulmonary abnormality in patients with MDR-/XDR-TB.30
In this study, the cases with intrapulmonary calcification were more frequently observed in the drug-resistant group, and calcification was an independent risk factor for drug resistance. Calcification is usually observed in necrotic tissue, especially the caseous focus of TB. Intrapulmonary calcification indicates a long PTB course. Accordingly, the risk of drug resistance caused by MTB gene mutation is increased to a certain extent; thus, calcification is observed more frequently in cases of DR-PTB.31
The Value of CT Imaging in the Diagnosis of DR-PTB
Etiological examination has some limitations. There is often a certain deviation in the etiological detection results when testing for some special types of TB (such as culture-negative TB and TB coinfected with HIV), some special patient groups (such as pregnant women and children), and latent TB.32 CT imaging becomes an indispensable supplement and can provide critical information for clinical practice when etiological detection is unsatisfactory in evaluating the condition and severity of TB patients. For instance, in PTB patients with thick-walled cavities, the accuracy of sputum detection is low during treatment, and the key to successful treatment is detecting the cavities and tracking their changes on CT imaging instead of only focusing on indicators such as the quantity of bacteria in sputum. If the permeability and absorption of antituberculosis drugs are seriously inhibited due to large cavities or thick cavity walls, alternative treatment, such as surgery, should be administered.32
Although CT scans do not have the price advantage compared with CXR, CT scans are far superior to CXR in finding occult lesions and showing tiny lesions and lesions in the overlapping locations. CT scans bring greater auxiliary value of clinical diagnosis and efficacy judgment so that they get a wide range of applications. Besides, although CT imaging should not become the basis for a definitive diagnosis nor the gold standard for DR-PTB, along with etiology, pathology, and molecular biology methods, CT imaging is an important auxiliary examination and evaluation method for DR-PTB. It can be of great significance in the early diagnosis of DR-PTB, this study found that “multiple cavities”, “thick-walled cavities”, “whole-lung involvement”, etc. were all independent risk factors for PTB, meaning that the possibility of drug resistance in patients with PTB should be seriously considered when the above imaging signs exist simultaneously. At this time, clinicians can evaluate the possibility of drug resistance through the aforementioned logistics regression equation. If the calculated predicted values of the drug-resistance probability is higher than the critical value (0.354), then the case will be predicted to be resistant, otherwise, it will be predicted to be sensitive. The specificity and sensitivity of the tool were 90.6% and 60.7%, respectively. And the prediction correction rate was 80.6%. A timely DST will be necessary in cases predicted to be resistant and so effectively shorten the DR-TB diagnosis time and improve the clinical diagnostic efficacy, and the treatment plan can be adjusted in time to increase therapeutic effect.
Our study had several limitations. First, the results and prediction model were established on the basis of data obtained from a single center, which was a tertiary referral hospital, and as such, the study had inherent flaws relating to selection and referral bias. Second, the clinical and imaging manifestations of different types of DR-PTB and DS-PTB and each drug-resistant type were not compared and analyzed, respectively, and the difference in imaging findings among different types of DR-PTB patients could not be reflected; this requires further research. Furthermore, although we selected patients for quite a long time, we still enrolled only a small number of cases with XDR-TB, and the small sample size of XDR-TB patients in the drug-resistance group may have resulted in selection bias.
Conclusion
The chest CT manifestations of DR-PTB had certain characteristics that significantly indicated the possibility of drug resistance in patients with PTB when there were multifarious simultaneous image findings, including multiple cavities, thick-walled cavities, bronchial disseminated lesions, whole-lung involvement, etc. Clinicians can comprehensively analyze the imaging manifestations of PTB and evaluate the possibility of drug resistance through prediction models, thus providing imaging evidence for timely DST in patients with suspected DR-PTB and improving the therapeutic effect and prognosis. Sustainable development goals (SDGs) 3 proposed by WHO includes a target to end the TB epidemic, and the key to achieving the goal is to reduce the TB incidence, realize the early diagnosis of TB and DR-TB and reduce mortality. It is believed that CT imaging is conducive to the early diagnosis of DR-TB and the early realization of SDGs 3 to some extent.
Abbreviations
AUC, area under the curve; CXR, chest X-ray; DR-PTB, drug-resistant pulmonary tuberculosis; DR-TB, drug-resistant tuberculosis; DS-PTB, drug-sensitive pulmonary tuberculosis; DST, drug sensitivity testing; MDR-TB, multidrug-resistant tuberculosis; MMP, matrix metalloproteinase; MTB, Mycobacterium tuberculosis; PTB, pulmonary tuberculosis; ROC, receiver operating characteristic; SDGs, Sustainable development goals; XDR-TB, extensively drug-resistant tuberculosis.
Acknowledgments
We would like to thank professor Chen Ling (Respiratory Medicine Laboratory of Zunyi Medical University) for cases providing of drug-resistant tuberculosis and professor Li Hong (Public Health School of Zunyi Medical University) for her assistance in statistical analysis.
Disclosure
All authors declared no conflicts of interest in this work.
References
1. World Health Organization. Global Tuberculosis Report 2020. Geneva: World Health Organization; 2020.
2. Pontali E, Raviglione MC, Migliori GB, et al. Regimens to treat multidrug-resistant tuberculosis: past, present and future perspectives. Eur Respir Rev. 2019;28(152):190035. doi:10.1183/16000617.0035-2019
3. Lange C, Dheda K, Chesov D, et al. Management of drug-resistant tuberculosis. Lancet. 2019;394(10202):953–966. doi:10.1016/S0140-6736(19)31882-3
4. Rasool G, Khan AM, Mohy-Ud-Din R, et al. Detection of Mycobacterium tuberculosis in AFB smear-negative sputum specimens through MTB culture and GeneXpert® MTB/RIF assay. Int J Immunopathol Pharmacol. 2019;33:1–6. doi:10.1177/2058738419827174
5. Suárez I, Fünger SM, Kröger S, et al. The diagnosis and treatment of tuberculosis. Dtsch Arztebl Int. 2019;116(43):729–735. doi:10.3238/arztebl.2019.0729
6. Hansell DM, Bankier AA, MacMahon H, et al. Fleischner society: glossary of terms for thoracic imaging. Radiology. 2008;246(3):697–722. doi:10.1148/radiol.2462070712
7. Zürcher K, Ballif M, Fenner L, et al. Drug susceptibility testing and mortality in patients treated for tuberculosis in high-burden countries: a multicentre cohort study. Lancet Infect Dis. 2019;19(3):298–307. doi:10.1016/S1473-3099(18)30673-X
8. Nguyen QH, Contamin L, Nguyen TVA, et al. Insights into the processes that drive the evolution of drug resistance in Mycobacterium tuberculosis. Evol Appl. 2018;11(9):1498–1511. doi:10.1111/eva.12654
9. Elduma AH, Mansournia MA, Foroushani AR, et al. Assessment of the risk factors associated with multidrug-resistant tuberculosis in Sudan: a case-control study. Epidemiol Health. 2019;41:e2019014. doi:10.4178/epih.e2019014
10. Yang Y, Wu J. Significance of the differential peptidome in multidrug-resistant tuberculosis. Biomed Res Int. 2019;2019:5653424.
11. Lee EH, Yong SH, Leem AY, et al. Improved fluoroquinolone-resistant and extensively drug-resistant tuberculosis treatment outcomes. Open Forum Infect Dis. 2019;6(4):ofz118. doi:10.1093/ofid/ofz118
12. Basit A, Ahmad N, Khan AH, et al. Predictors of two months culture conversion in multidrug-resistant tuberculosis: findings from a retrospective cohort study. PLoS One. 2014;9(4):e93206. doi:10.1371/journal.pone.0093206
13. Huang Q, Yin Y, Kuai S, et al. The value of initial cavitation to predict re-treatment with pulmonary tuberculosis. Eur J Med Res. 2016;21(1):20. doi:10.1186/s40001-016-0214-0
14. Chuchottaworn C, Thanachartwet V, Sangsayunh P, et al. Risk factors for multidrug-resistant tuberculosis among patients with pulmonary tuberculosis at the central chest institute of Thailand. PLoS One. 2015;10(10):e0139986. doi:10.1371/journal.pone.0139986
15. Wong KS, Huang YC, Lai SH, et al. Validity of symptoms and radiographic features in predicting positive AFB smears in adolescents with tuberculosis. Int J Tuberc Lung Dis. 2010;14(2):155–159.
16. Ihms EA, Urbanowski ME, Bishai WR. Diverse cavity types and evidence that mechanical action on the necrotic granuloma drives tuberculous cavitation. Am J Pathol. 2018;188(7):1666–1675. doi:10.1016/j.ajpath.2018.04.006
17. Wáng YXJ, Chung MJ, Skrahin A, Rosenthal A, Gabrielian A, Tartakovsky M. Radiological signs associated with pulmonary multi-drug resistant tuberculosis: an analysis of published evidences. Quant Imaging Med Surg. 2018;8(2):161–173. doi:10.21037/qims.2018.03.06
18. Li D, He W, Chen B, et al. Primary multidrug-resistant tuberculosis versus drug-sensitive tuberculosis in non-HIV-infected patients: comparisons of CT findings. PLoS One. 2017;12(6):e0176354. doi:10.1371/journal.pone.0176354
19. Metcalfe JZ, Makumbirofa S, Makamure B, et al. Drug-resistant tuberculosis in high-risk groups, Zimbabwe. Emerg Infect Dis. 2014;20(1):135–137. doi:10.3201/eid2001.130732
20. Walker NF, Stek C, Wasserman S, et al. The tuberculosis-associated immune reconstitution inflammatory syndrome: recent advances in clinical and pathogenesis research. Curr Opin HIV AIDS. 2018;13(6):512–521. doi:10.1097/COH.0000000000000502
21. Ordonez AA, Tasneen R, Pokkali S, et al. Mouse model of pulmonary cavitary tuberculosis and expression of matrix metalloproteinase-9. Dis Model Mech. 2016;9(7):779–788. doi:10.1242/dmm.025643
22. Cheon H. Comparison of CT findings of between MDR-TB and XDR-TB: a propensity score matching study. Imaging Med. 2017;9:125–129.
23. Shin SS, Keshavjee S, Gelmanova IY, et al. Development of extensively drug-resistant tuberculosis during multidrug-resistant tuberculosis treatment. Am J Respir Crit Care Med. 2010;182(3):426–432. doi:10.1164/rccm.200911-1768OC
24. Jeong YJ, Lee KS. Pulmonary tuberculosis: up-to-date imaging and management. AJR Am J Roentgenol. 2008;191(3):834–844. doi:10.2214/AJR.07.3896
25. Woodman M, Haeusler IL, Grandjean L. Tuberculosis genetic epidemiology: a Latin American perspective. Genes (Basel). 2019;10(1):53. doi:10.3390/genes10010053
26. Chandrasekaran R, Mac AM, Chalmers JD, et al. Geographic variation in the aetiology, epidemiology and microbiology of bronchiectasis. BMC Pulm Med. 2018;18(1):83. doi:10.1186/s12890-018-0638-0
27. Schäfer J, Griese M, Chandrasekaran R, et al. Pathogenesis, imaging and clinical characteristics of CF and non-CF bronchiectasis. BMC Pulm Med. 2018;18(1):79. doi:10.1186/s12890-018-0630-8
28. Aggarwal D, Gupta A, Janmeja AK, et al. Evaluation of tuberculosis-associated chronic obstructive pulmonary disease at a tertiary care hospital: a case-control study. Lung India. 2017;34(5):415–419. doi:10.4103/lungindia.lungindia_522_16
29. Pettigrew MM, Tsuji BT, Gent JF, et al. Effect of fluoroquinolones and macrolides on eradication and resistance of Haemophilus influenzae in chronic obstructive pulmonary disease. Antimicrob Agents Chemother. 2016;60(7):4151–4158. doi:10.1128/AAC.00301-16
30. Ben Saad-Baouab S, Hantous S, Daghfous H, et al. Contribution of imaging in the management of resistant tuberculosis. Tunis Med. 2019;97(3):445–454.
31. Song Q, Zhang G, Jiang H, et al. Imaging features of pulmonary CT in type 2 diabetic patients with multidrug-resistant tuberculosis. PLoS One. 2016;11(3):e0152507. doi:10.1371/journal.pone.0152507
32. Nahid P, Mase SR, Migliori GB, et al. Treatment of drug-resistant tuberculosis. An official ATS/CDC/ERS/IDSA clinical practice guideline. Am J Respir Crit Care Med. 2019;200(10):e93–e142. doi:10.1164/rccm.201909-1874ST
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.