Back to Journals » Clinical Ophthalmology » Volume 14
A Comparative Evaluation of Visual, Refractive, and Patient-Reported Outcomes of Three Extended Depth of Focus (EDOF) Intraocular Lenses
Authors Ang RE , Picache GCS, Rivera MCR, Lopez LRL , Cruz EM
Received 24 March 2020
Accepted for publication 3 July 2020
Published 20 August 2020 Volume 2020:14 Pages 2339—2351
DOI https://doi.org/10.2147/OPTH.S255285
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Robert Edward T Ang,1,2 Gian Carlo S Picache,2 Mark Christian R Rivera,1 Lord Ryan L Lopez,1 Emerson M Cruz1
1Asian Eye Institute, Rockwell Center, Makati, Philippines; 2Department of Ophthalmology, Cardinal Santos Medical Center, San Juan, Philippines
Correspondence: Robert Edward T Ang Email [email protected]
Objective: To compare visual, refractive, and patient-reported outcomes of patients implanted with one of three types of extended depth of focus (EDOF) intraocular lenses.
Setting: Asian Eye Institute, Philippines.
Design: Retrospective cohort study.
Methods: Subjects implanted with Symfony (Johnson and Johnson, USA), IC-8 (AcuFocus, USA), and WIOL (Medicem, Czech Republic) EDOF intraocular lenses were recruited. Spherical equivalent, uncorrected and corrected visual acuity, defocus curve, and modulation-transfer function, Strehl ratio, and visual Strehl optical transfer function values, photic phenomena, and questionnaire answers were measured and assessed.
Results: A total of 32 eyes with the Symfony lens, 30 with the IC-8 lens, and 32 with the WIOL lens were included in the study. Mean postoperative spherical equivalent was − 0.24 D for the Symfony, − 0.17 D for the IC-8, and 0.27 D for WIOL. There were no significant differences in postoperative monocular and binocular uncorrected and corrected visual acuity. On a monocular defocus curve, the IC-8 and Symfony showed significantly better vision than WIOL. The Symfony had significantly better modulation-transfer function, Strehl ratio, and visual Strehl optical transfer function. No difference was seen among the three lenses with regard to glare or starburst, while patient satisfaction remained high in all groups for far, intermediate, and near vision.
Conclusion: All eyes implanted with the three EDOF designs achieved excellent far and intermediate vision, with acceptable near vision. The IC-8 and Symfony exhibited a better range of vision on defocus-curve testing. The Symfony showed superior results in quality of vision. Patient satisfaction was high in all three EDOF groups.
Keywords: EDOF, Symfony, IC-8, WIOL, diffractive echelette, small aperture
Introduction
The evolution and advancement of intraocular lens (IOL) technology has led to improved visual outcomes and raised the bar for patient satisfaction.1 Spectacle independence is at the forefront of patient expectations, and if not met may result in refractive disappointment. Ordinarily, monofocal IOLs are targeted for emmetropia, and provide the best-possible distance vision for a patient. Despite excellence in correcting distance vision, monofocal IOLs fall short in correcting intermediate and near distances, because patients remain presbyopic postoperatively, leading to continued need for corrective eyewear for reading small print after cataract surgery.8 The development of multifocal IOLs and extended depth of focus (EDOF) IOLs has greatly increased the probability of spectacle independence and patient satisfaction.2–4
Monofocal IOLs provide the best correction for distance vision, yet intermediate and near vision remains uncorrected. Multifocal IOLs, more specifically trifocal IOLs, were developed to provide better uncorrected distance, intermediate, and near vision by splitting light energy to three focal points. However, there are several drawbacks in trifocal multifocal technology. First, the intermediate vision provided by the trifocal IOLs is not as strong as distance and near vision.2–4 Second, due to diffractive optic design, it is unavoidable that quality of vision is mildly sacrificed, leading to decreased contrast sensitivity.5,6 Third, diffractive optics and ring design still produce photic phenomena, such as glare, starburst, and halos. Even though patients are given more vision and spectacle independence, shortcomings in quality of vision can lead to patient dissatisfaction in a fair amount of patients.7 This led IOL designers to explore a novel concept called EDOF as an innovative way of balancing the improvement in range of vision and lessening the sacrifice in quality of vision.
The concept of EDOF IOL technology is to create a single elongated focal point to enhance range of vision or DOF without splitting light and without lens movement. EDOF technology focuses on improving visual outcomes by providing intermediate-distance correction while preserving good far vision and having decent near vision. However, the success of EDOF IOLs will not only depend on the range of vision they can provide. It is likewise important to explore the effect on photic symptoms and patient-reported outcomes, because these factors affect quality of life after cataract surgery.20 There are three types of EDOF IOLs that we have had experience with: the Symfony (Johnson and Johnson, USA), IC-8 (AcuFocus, USA), and WIOL (Medicem, Czech Republic). These EDOF IOLs use different optic designs to achieve the desired visual outcome. The purpose of this study was to compare the visual performance of eyes that had been implanted with these three types of EDOF IOLs.
Methods
This was a retrospective cohort study. The study secured the approval of the Asian Eye Institute Ethics Review Committee and was conducted in accordance with the Declaration of Helsinki. Postoperative patients who had been implanted with an EDOF IOL were recruited and informed consent obtaineds. Subjects had undergone diagnostic testing and asked to answer a questionnaire during one clinic visit. The recruitment period was from January to June 2019.
Criteria for inclusion in the study were patients who had had one of the following IOLs implanted in at least one eye: Symfony, IC-8, and WIOL-CF. Patients must have undergone an uneventful phacoemulsification with in-the-bag lens implantation and a postoperative visit of at least 1 month after surgery. Excluded from the study were those with clinically significant corneal abnormalities, including corneal dystrophy, ocular inflammation, corneal edema, ocular trauma, corneal transplant, retinal conditions, degenerative eye disorders, color-vision deficiencies, glaucoma with significant visual field defects, history of refractive surgery, and complicated cataract surgery. Patients who had other ophthalmic surgeries other than cataract surgery or lens exchange and those whose charts had incomplete data were also excluded.
The Symfony (ZXR00) is a single-piece, ultraviolet-filtering hydrophobic acrylic IOL with an asphericity that causes a spherical aberration of −0.27 μm for the 6 mm corneal aperture. The Symfony’s diffractive echelette surface is a proprietary pupil-independent design that creates an elongated focus without defined focal planes throughout. The IOL has a high Abbe number, thereby correcting chromatic aberration and providing high-contrast sensitivity (Figure 1).12
 |
Figure 1 Johnson and Johnson Tecnis Symfony. |
The IC-8 small-aperture IOL (AcuFocus, USA) is a one-piece hydrophobic acrylic IOL with a centrally located, embedded polyvinylidene difluoride annular mask intended to improve near vision based on the concept of small-aperture optics. The embedded annular mask has an outer diameter of 23 mm with a central aperture of 1.36 mm in diameter, and contains 3,200 microperforations on its annulus (sparing the periphery), of 7–10 µm in diameter and arranged in a pseudorandom fashion. The small-aperture IOL provides increased range of vision from far to near by extending the DOF. It works by eliminating the unfocused peripheral light rays, while the central rays pass unaffected. Reducing the size of the blur circle from unfocused peripheral light improves the image resolution for intermediate and near vision, with minimal to no change in distance vision (Figure 2).14,17
 |
Figure 2 AcuFocus IC-8. |
The WIOL-CF (Wichterle Continuous Focus IOL) is a round disc 7.5 mm in diameter with no haptics that expands to 9.5 mm once implanted to occupy the entire capsular bag. The lens is described as bioanalogic, wherein the design (flat anterior and convex posterior) and material (hydrogel) closely mimic the natural crystalline lens. The mechanism of action is an extension of DOF via polyfocality, where the thickest part of the lens centrally has the highest add power for better reading vision. As the lens get thinner peripherally, the reading add decreases and distant images becomes better focused. This gradual change in refractive power offers a continuous focus from distance to near as the pupil size changes from a larger diameter as the eye looks at distant objects to smaller diameter during accommodation. The design produces an infinite number of focal points that extend the DOF and a large optic that ensures maximum entry of light (Figure 3).15,16
 |
Figure 3 Medicem WIOL. |
Gathered in the study were patients’ demographic data (eye, age, sex, and follow-up in months), preoperative parameters that included visual acuity (VA; uncorrected and corrected distance, intermediate, and near vision), manifest refraction expressed as mean refractive spherical equivalent (MRSE), and biometric data (axial length, mean keratometry, cylinder, anterior-chamber depth, IOL power, and target spherical equivalent). Postoperative uncorrected distance VA (UDVA), intermediate VA (UIVA), and near VA (UNVA), refraction, mesopic contrast-sensitivity testing, and manual defocus curves were also recorded. A standardized ETDRS chart at 6 m, 80 cm, 60 cm, and 40 cm was used to measure VA. Mesopic contrast sensitivity was taken using the functional acuity contrast test. Modulation transfer function (MTF) average height, Strehl ratio, and through-focus visual Strehl optical transfer function (VSOTF) values were obtained using the iTrace aberrometer (Tracey Technologies, Houston, TX, USA).
The Modified Type Questionnaire was used to measure satisfaction in distance, intermediate, and near vision.20 Questionnaire responses were analyzed for patients with binocular implantation of the same lens. A subset analysis of their binocular VA (uncorrected and best-corrected distance, intermediate [80 and 60 cm], and near [40 cm] VA), glarometer scores (halo and starburst), and defocus curves were also assessed.Using the rule of thumb of Browne et al, a minimum sample size of 30 per group should be used when estimating the sample size for a pilot trial, which is the simplest method to apply.19 The deidentified data sets were recorded in a password-protected Excel spreadsheet. ANOVA was employed to compare data sets among the three groups. Statistical significance was set at p<0.05 using SPSS 20.0.
Results
A total of 94 eyes were enrolled in this study and divided into three groups: 32 in the Symfony group, 30 in the IC-8 group, and 32 in the WIOL group. Mean ages ofsubjects were 65.94±5.44 years in the Symfony group, 60.7±6.4 years in the IC-8 group, and 69.6±8.59 years in the WIOL group. The total mean follow-up period from date of operation to the study visit was 40.01±27.7 months. The Symfony group had a mean follow-up of 3.02±1.57 months, while the IC-8 and WIOL groups had mean follow up of 56.76±8.4 and 63±5.29, respectively (Table 1).
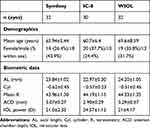 |
Table 1 Subject Demographics and Preoperative Profile (logMAR Visual Acuity, Refraction and Biometric Data) |
Postoperative MRSE was −0.24±0.28 D in the Symfony group, −0.17±0.47 D in the IC-8 group, and 0.27±0.41 D in the WIOL group (Table 2). Postoperative mean monocular and binocular uncorrected distance, intermediate, and near vision are shown in Table 3. Though the UNVA of the IC-8 seemed better monocularly and the UNVA of the IC-8 and Symfony better binocularly, no statistically significant difference in UDVA, UIVA, or UNVA was found across study arms (p=0.645, p=0.736, p=0.314, p=0.427).
 |
Table 2 Postoperative Manifest Refraction on Study Visit |
 |
Table 3 Uncorrected Postoperative Visual Acuity on Study Visit (logMAR) |
With regard to the best corrected DVA, even though distance-corrected NVA (DCNVA; 40 cm) seemed better with the IC-8 and Symfony, there was no statistical difference in distance-corrected vision among the three groups monocularly or binocularly (Table 4). In monocular defocus curve testing, the Symfony and IC-8 had significantly better VA, while the WIOL had poorer performance from manifest refraction up to 3.0 D defocus (Figure 4). In binocular defocus testing, no significant difference was seen up to 2.5 D defocus (Figure 5).
 |
Table 4 Best-corrected Postoperative Visual Acuity on Study Visit (logMAR) |
 |
Figure 4 Monocular defocus curves. |
 |
Figure 5 Binocular defocus curves. |
Wavefront-aberrometry readings revealed an unusually high total higher-order aberration and coma reading for the IC-8 lens. This leads us to suspect that the small aperture may be causing false aberrometry readings. The Symfony had lower total higher-order and coma measurements compared to the WIOL (Table 5). Monocular mesopic contrast sensitivity, both without glare and with glare, showed no significant difference among the three groups (without glare — p=0.13, p=0.12, p=0.16, p=0.05 for cycles per degree [CPD; Figure 6A]; with glare — p=0.18, p=0.34, p=0.80, p=0.26 for CPD [Figure 6B]).
 |
Table 5 Postoperative Aberrations on Study Visit |
 |
Figure 6 Mesopic contrast sensitivity without glare (A) and with glare (B). |
The Symfony had statistically significantly better results in (MTF) analysis (far, p=0; near, p=0.06; Figure 7), Strehl-ratio point-spread function (PSF; far, p=0.05; near, p=0.025; Figure 8) and VSOTF (far, p=0.01; near, p=0; Figure 9) compared to the IC-8 and WIOL. Monocular and binocular glarometer and starburst testing showed no significant differences among the three study arms monocularly (glare, p=0.344; starbursts, p=0.842; Figure 10A) or binocularly (glare, p=0.287; starburst, p=0.479; Figure 10B).
 |
Figure 7 Modulation transfer function (average height). |
 |
Figure 8 Strehl ratio (point-spread function). |
 |
Figure 9 Visual Strehl optical transfer function (VSOTF). |
 |
Figure 10 Monocular glarometer scores (A) and binocular glarometer scores (B). |
Results of the Modified Type Questionnaire showed approximately 40% of patients in the Symfony and IC-8 arms reported symptoms of glare. However, these were only mild, and no difficulty in daily activities was noted. There was no significant difference reported with regard to halos during the night among the three IOL groups (p=0.416) (Table 6). Some subjects in each group reported difficulty in reading small print in newspapers and leaflets. Nevertheless, patient satisfaction remained high in all three groups for DVA, IVA, and NVA (Figure 11).
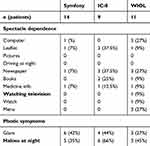 |
Table 6 Spectacle Dependence and Photic Symptoms (Questionnaire) |
 |
Figure 11 Visual satisfaction scores (far, intermediate, and near vision). |
Discussion
A new category of IOLs — EDOF lenses — emerged out of the need to address the main shortfall of monofocal lenses, which is poor intermediate and near vision, and the two shortfalls of multifocal IOLs, which are photic phenomena and quality of farintermediate vision. By definition, EDOF means a continuous range of vision from far to near, in contrast to multifocal IOLs, wherein there are dips in vision between the distinct focal points of far, intermediate, and near. EDOF IOLs have found a place between monofocal and multifocal IOLs and have been gaining popularity in recent years, because they are promoted as having fewer photic symptoms than multifocal IOLs and better vision than monofocal IOLs. Patient satisfaction remains high in those implanted with EDOF lenses, despite some photic phenomena reported and some degree of postsurgical presbyopia.9–18
Extending the DOF can be accomplished via different mechanisms in different IOLs. The Symfony IOL has an echellete or diffractive grating mechanism whereby focal points are very close to each other, forming a continuous range of vision. The IC-8 IOL uses small-aperture or pinhole optics to achieve an extended range of vision, whereas the WIOL uses polyfocality, resulting in modulation of spherical aberration, created by the thickest portion of the lens centrally to achieve its extended depth of field.
The Symfony was the first to create this niche category, and is the most popular among the EDOF lenses. In a paper by Kohnen et al,27 their results had median UDVA of 0.01 logMAR and DCVA of −0.05 logMAR, which is comparable to our study, wherein we had mean UDVA of 0.04 and DCVA of 0.01 logMAR. Attia et al reported that distance vision with the Symfony IOL is comparable to multifocal lenses, such as the AT Lisa (Zeiss, Germany) and PanOptix (Alcon, USA).24 Monocular and binocular uncorrected intermediate (80 and 60 cm) vision in our study of 0.04 logMAR (80 cm) and 0.08 logMAR (60 cm) was similar to Kohnen et al’s who had UIVA of 0.05 logMAR for 80 cm and 0.07 logMAR for 60 cm. Near vision (40 cm) was slightly better in our study, but was not statistically significant. Near-vision results using the Symfony lens were comparable to other recent studies. Pedrotti et al showed a mean UNVA of 0.18 logMAR,25 while the Concerto study had a mean 0.17 logMAR.13 This was similar to our study finding of 0.16 logMAR. Our study shows that acceptable DCNVA can be achieved with a mean of 0.10 logMAR compared to the results of Pedrotti et al (0.21 logMAR) and Ganesh et al (0.216 logMAR).21
Ruiz-Mesa et al found excellent defocus of the Symfony lens from manifest refraction to −2.0 D.26 Kohnen et al showed a defocus of <0.02 logMAR at manifest refraction to −1.5 D, with a dip in mean VA to 0.14 logMAR at −2.5 D.27 In our study, the Symfony achieved excellent results from manifest refraction to −1.5 D at a mean of 0.1 logMAR, with a dip to 0.18 logMAR at −2.0 D. In terms of contrast sensitivity, our findings of normal sensitivity in mesopic conditions are similar to the postoperative contrast–sensitivity studies of Kohnen et al,27 Ganesh et al,21 Ruiz-Mesa et al,26 and Cochener et al.13
MTF) and PSF measure quality of image resolution, and are expressed in average height and Strehl ratio. The closer the MTF and PSF are to 1, the better the quality of vision and contrast sensitivity seen through the IOL. DOF can be measured using VSOTF, which is one of the best indicators of visual performance. Measurements are performed using wavefront aberrometers, and have been proven to correlate strongly with subjective VA.16,23,24 To our knowledge, no study has compared MTF, PSF, and VSOTF in the literature of Symfony lenses to other EDOF lenses. The Symfony was found to have good MTF, with mean average height of 0.4, PSF of 0.36, and VSOTF of 0.42. In the Symfony arm, six of 14 patients (42%) reported some glare and five of 14 (35%) mild halos at night. Of those implanted with the Symfony, patient satisfaction remained high, and 100% of participants were spectacle-free for distance and intermediate vision. Four of 14 patients (28%) reported having to use reading glasses for computers, leaflets, newspapers, and medicine information. All patients recommended undergoing the same procedure.
The first published report of the IC-8 small-aperture IOL was in 2015 by Grabner et al.14 Several other studies have since been done showing the visual performance of the IC-8. Dick et al reported 6-month postoperative UDVA of 0.06±0.15 logMAR, UIVA of 0.08±0.12 logMAR, UNVA of 0.18±0.14 logMAR.17 Our study showed comparable postoperative results: UDVA of 0.04±0.08 logMAR, UIVA 1 (80 cm) 0.06±0.11 logMAR, and UNVA (40 cm) 0.18±0.21 logMAR. In terms of defocus curve, Dick et al reported vision of 0.3 logMAR or better in 100% of his subjects at the defocus range of 0.50 D to −1.50 D, whereas our data showed a wider range of vision, with subjects maintaining vision of 0.3 logMAR or better in the range of 1.0 D to −2.5 D.17 Mesopic contrast sensitivity was found to be within normal limits, similarly to other previous studies. Higher-order aberrations, MTF, PSF, and VSOTF values were erratic, probably because the small aperture made it difficult for aberrometry readings to be taken. While four of nine patients (44%) reported symptoms of glare, three graded this as mild, with no difficulty in vision. Six of the nine patients (66%) reported some halos at night, but found it to be mild and not bothersome. This finding is consistent with Son et al reporting minimal photic phenomena using the IC-8.28 Of those implanted with the IC-8, patient satisfaction remained high. All participants were spectacle-free for distance and intermediate vision. Two of nine required near-vision correction for reading books and one needed glasses for reading medicine information. All patients recommended undergoing the same procedure.
Very few studies have been published about the WIOL. In a study by Studeny et al using the WIOL, postoperative vision testing at 6 months showed UDVA of 0.07±0.11 logMAR, UIVA 0.18±0.12 logMAR, and UNVA 0.33±0.15 logMAR. With correction, they reported DCVA of 0.05±0.12 logMAR, DCIVA of 0.19±0.14 logMAR, and DCNVA of 0.34±0.13 logMAR.15 Our results for UDVA of 0.05±0.07 logMAR, UIVA 1 (80 cm) 0.06±0.11 logMAR, UNVA (40 cm) 0.12±0.28 log MAR, BDVA 0.05±0.06 log MAR, DCIVA (80 cm) 0.11±0.11 log MAR, and and DCNVA (40 cm) 0.16±0.15 log MAR were comparable. Monocular defocus curves seemed to show lower visual function at plano to −3.0D defocus, but when tested binocularly, the WIOL exhibited a similar range of vision compared to the Symfony and IC-8. Siatiri et al obtained MTF (average height) values of 0.139±0.04 for far and 0.14±0.04 for near vision compared to our study of 0.20±0.08 for far and 0.25±0.07 for near vision.16 In a patient-satisfaction survey of 48 patients by Studeny, 18 patients (37.5%) were very satisfied, eleven (22.9%) satisfied, 15 (31.2%) rather satisfied, four (8.3%) rather dissatisfied, and none dissatisfied. In sum, 23 of their patients (47.9%) require reading glasses postoperatively, while 19 (39.6%) used reading glasses occasionally for reading small fonts and six (12.5%) used reading glasses regularly. Half their study population experienced no halos or glare, while 8.3% experienced no glare with mild halos. Mild glare and halos were seen in 20.8% of their study patients. One patient reported disturbing halos and two disturbing glare.15 Our study found that three of eleven patients (27%) experienced mild glare and five ofleven (45.4%) mild halos. Overall, photic phenomena appeared to be mild and well tolerated. We concur with Studeny et al that despite need for near-vision correction for fine print, those implanted with the WIOL were generally satisfied with their vision.
This study reinforces the impression that EDOF IOLs perform at a level between monofocal and multifocal IOLs, because they are able to provide good distance and intermediate and decent reading vision. All three lenses belong to the EDOF category, because their defocus curves follow the gentle sloping of vision from distance to intermediate to near vision, in contrast to multifocal IOLs, which show a dip in intermediate-vision defocus testing. When comparing the three lenses, monocular distance and intermediate- and near-vision defocus up to 3.0 D was superior with the Symfony and IC-8 compared to the WIOL.
The refractive outcome of mild myopia with the Symfony and IC-8 became advantageous in providing more uncorrected near vision while not sacrificing uncorrected distance vision. Upon analysis of best distance-corrected vision, wherein the influence of refraction was removed, the IC-8 seemed to give better near vision than other two lenses, although the difference was not statistically significant. Refractive outcome will definitely play a role in the visual performance of the lenses. When computing IOL power, we recommend targeting −0.25 to −0.50 D MRSE for the Symfony and WIOL to give some additional near vision with minimal sacrifice in distance vision. For the IC-8, a refractive target of −0.75 D is recommended, because the pinhole optics are able to correct the residual myopia and maintain good distance vision and at the same time take advantage of the −0.75 D myopia to boost near vision.
The different optics design allows for interesting comparisons. Distance vision is best in the Symfony, because of the diffractive echelette optics and chromatic aberration compensation, but the IC-8 can compete well with the Symfony, because small-aperture optics allow only focused light to enter and can filter out unwanted aberrations and even residual astigmatism. The spherical aberration effect of the WIOL gives it a mild disadvantage in sharpness of distance vision, and this is supported by the lower MTF, PSF, and VSOTF compared to the Symfony, but this negative spherical aberration gives the WIOL its EDOF capability. It is unfortunate that aberrometry measurements with the IC-8 were too low and had to be interpreted as erroneous because iTrace readings are usually obtained between the 3–5mm optical zone size, but the central-aperture diameter of the IC-8 is only 1.36 mm. There is no other way of measuring the optics of the IC-8 objectively. Only with subjective metrics, such as contrast sensitivity, defocus-curve performance, glare/halos and questionnaires are we able to compare the small-aperture lens to other EDOF lenses.
Contrast sensitivity offers another interesting comparison among these three EDOF lenses, because of their different optic designs. We presumed that the Symfony would show significantly higher contrast scores because of its chromatic aberration–compensation feature and higher MTF values, the IC-8 would show the worst because the small aperture would presumably allow less light to reach the retina, and the WIOL would be somewhere in between. Ganesh et al,21 Choi et al,22 and de Medeiros et al23 et al all reported normal contrast sensitivity for the Symfony lens. This is in contrast to the findings of Yamauchi et al, who showed lower contrast sensitivity with the Symfony, due to a decrease in luminous energy and increase in higher-order aberrations, with an increase in multifocality.5 According to Dick et al, eyes implanted with monofocal IOLs had better contrast sensitivity than those implanted with a small aperture lens like the IC-8, but when plotted, contrast sensitivity remained within normal limits.17 Siatiri et al showed normal contrast sensitivity in the WIOL supported by MTF using the iTrace.16 Our study found that contrast-sensitivity scores were within normal limits, and no statistical difference was found among the three groups in mesopic-with-glare or mesopic-without-glare conditions at 3, 6, 12, or 18 CPD. It is worthwhile mentioning that while there is a presumption that the small-aperture IOL would give patients dim vision, contrast-sensitivity tests showed that this is hardly affected, suggesting that the 1.36 mm aperture size is sufficient to allow enough lighting for good photopic and mesopic vision.
Patient-reported outcomes, which include photic phenomena and satisfaction, are crucial in evaluating the success of treatment. Many studies, including Leyland et al, have shown us that patients who have been implanted with a multifocal lens are 3.5 times more likely to experience photic symptoms than those mplanted with monofocal IOLs.2 EDOF IOLs are presumed to have more photic phenomena than monofocal lenses, but less than multifocal IOLs. All three groups reported mild– moderate glare and starbursts, but that these were not significantly bothersome. There was no significant difference among the groups regarding one group of subjects reporting more significant photic symptoms.
Patients reported similarly high satisfaction scores on distance, intermediate, and near vision in all groups of IOLs. However, it is important to recognize that a few subjects in each group reported difficulty in reading small print in newspapers and leaflets. This highlights one of the shortcomings of EDOF lenses. Near vision with EDOF IOLs is not as good as multifocal IOLs; therefore, total spectacle independence cannot be promised. However, strategies to improve near vision include targeting refractive outcomes to mild myopia or a minimonovision approach whereby one eye, typically the nondominant eye, is intentionally targeted to −0.25 to −0.75 to widen the DOF to improve near vision binocularly.
We encountered several limitations in our study. The Symfony had only recently become available; therefore, the interval between surgery and study visit was different among the groups. We hope that the shorter follow-up for the Symfony did not affect responses to the questionnaires. The IC-8 and WIOL gave us long-term outcomes, which are useful in evaluating the performance of such medical devices as IOLs. The iTrace test was not an ideal device to obtain aberration or quality of vision metrics in a small-aperture type of lens, such as the IC-8, so we could not compare the groups objectively. We recommend that patients implanted with the Symfony lens have longer-term follow-up for better comparison with other study arms. A randomized controlled trial wherein patients are randomly assigned to receive EDOF IOLs bilaterally can also be performed to better assess the visual and refractive outcomes of these lenses. There are other types of EDOF lenses, so perhaps a different set of EDOF lenses can be selected and compared.
Conclusion
EDOF IOLs have a place in the spectrum of IOL options. Distance and intermediate vision is excellent and near vision acceptable. Defocus-curve testing allows us to confirm which lenses are in this category, despite different optic designs and mechanism of EDOF lenses. Refractive targeting in patients implanted with EDOF IOLs is extremely important to bring out its full potential, and it is recommended to target mild myopia in these eyes. Careful screening and patient education are key to achieving good patient satisfaction. All three EDOF IOLs implanted achieved excellent results in distance and intermediate vision and acceptable near vision. The Symfony and IC-8 exhibited better monocular defocus curve–test results. No significant difference in contrast sensitivity was observed. The Symfony IOL showed superior quality of vision on MTF, PSF, and VSOTF. The iTrace aberrometer does not provide reliable measurements in the eyes with the IC-8 lens. Patient satisfaction was high in all three groups.
Disclosure
Robert Edward Ang is a clinical investigator for AcuFocus, Bausch & Lomb, Johnson & Johnson, Physiol, and Medicem, and reports grants from AcuFocus and Johnson & Johnson outside the submitted work. The authors report no conflicts of interest in this work.
References
1. Tielsch JM, Steinberg EP, Cassard SD, et al. Preoperative functional expectations and postoperative outcomes among patients undergoing first eye cataract surgery. Arch Ophthalmol. 1995;113(10):1312–1318. doi:10.1001/archopht.1995.01100100100038
2. Leyland M, Pringle E. Multifocal versus monofocal intraocular lenses after cataract extraction. Cochrane Database Syst Rev. 2006;4. doi:10.1002/14651858.CD003169.pub2
3. Shah S, Peris-Martinez C, Reinhard T, Vinciguerra P. Visual Outcomes After Cataract Surgery: multifocal Versus Monofocal Intraocular Lenses. J Refract Surg. 2015;31(10):658–666. doi:10.3928/1081597X-20150611-01
4. Greenstein S, Pineda R. The Quest for Spectacle Independence: A Comparison of Multifocal Intraocular Lens Implants and Pseudophakic Monovision for Patients with Presbyopia. Semin Ophthalmol. 2017;32(1):111–115. doi:10.1080/08820538.2016.128400.
5. Yamauchi T, Tabuchi H, Takase K, Ohsugi H, Ohara Z, Kiuchi Y. Comparison of Visual Performance of Multifocal Intraocular Lenses with Same Material Monofocal Intraocular Lenses. PLoS One. 2013;8(6):e68236. doi:10.1371/journal.pone.0068236
6. Kim CY, Chung SH, Kim TI, Cho YJ, Yoon G, Seo KY. Comparison of Higher-Order Aberration and Contrast Sensitivity in Monofocal and Multifocal Intraocular Lenses. Yonsei Med J. 2007;48(4):27–633. doi:10.3349/ymj.2007.48.4.627
7. Hawker MJ, Madge SN, Baddeley PA, Perry SR. Refractive expectations of patients having cataract surgery. J Cataract Refract Surg. 2005;31(10):1970–1975. doi:10.1016/j.jcrs.2005.03.065
8. Medical Advisory Secretariat. Intraocular lenses for treatment of age related cataracts: an evidence based analysis. Ont Health Technol Assess Ser. 2009;9(15):1–62. Epub 2009 Oct 1
9. Millán MS, Vega F. Extended Depth of Focus Intraocular lens: chromatic performance. Biomed Opt Express. 2017;8(9):4294–4309. doi:10.1364/BOE.8.004294
10. Breyer DRH, Kaymak H, Ax T, Kretz FTA, Auffarth GU, Hagen PR. Multifocal Intraocular Lenses and Extended Depth of Focus Intraocular Lenses. Asia Pac J Ophthalmol. 2017;6(4):339–349. doi:10.2608/APO.2017186
11. Gatinel D, Pagoulle C, Gobin L, Loicq J. Comparison of 3 Diffractive IOLs in 3 Wavelengths: bifocal/EDOF/Trifocal. Paper presented at: american Society of Cataract and Refractive Surgery; May 10; 2016; New Orleans, Louisiana12 Gatinel D, Loicq J. Clinically Relevant Optical Properties of Bifocal, Trifocal and Extended Depth of Focus Intraocular Lenses. J Refract Surg. 2016;32(4):273–280. doi:10.3928/1081597X-20160121-07
12. Gatinel D, Loicq J. Clinically Relevant Optical Properties of Bifocal, Trifocal and Extended Depth of Focus Intraocular Lenses. J Refract Surg. 2016;32(4):273–280. doi:10.3928/1081597X-20160121-07
13. Cochener B; Concerto Study Group. Clinical outcomes of a new extended range of vision intraocular lens: international Multicenter Concerto Study. J Cataract Refract Surg. 2016;42(9):1268–1275. doi:10.1016/j.jcrs.2016.06.033
14. Grabner G, Ang RE, Vilupuru S. The Small-Aperture IC-8 Intraocular Lens: A New Concept for Added Depth of Focus in Cataract Patients. Am J Ophthalmol. 2015;160(6):1176–1184.e1. doi:10.1016/j.ajo.2015.08.017.
15. Studeny P, Krizova D, Urminsky J. Clinical experience with the WIOL-CF accommodative bioanalogic intraocular lens: czech national observational registry. Eur J Ophthalmol. 2016;26(3):230–235. doi:10.5301/ejo.5000653.
16. Siatiri H, Mohammadpour M, Gholami A, Ashrafi E, Siatiri N, Mirshahi R. Optical aberrations, accommodation, and visual acuity with a bioanalogic continuous focus intraocular lens after cataract surgery. J Curr Ophthalmol. 2017;29(4):274–281. doi:10.1016/j.joco.2017.06.007
17. Dick HB, Piovella M, Vukich J, Vilupuru S, Lin L. Prospective multicenter trial of a small-aperture intraocular lens in cataract surgery. J Cataract Refract Surg. 2017;43(7):956–968. doi:10.1016/j.jcrs.2017.04.038
18. Sachdev GS, Ramamurthy S, Sharma U, Dandapani R. 2018 Visual outcomes of patients bilaterally implanted with the extended range of vision intraocular lens: A prospective study. Indian J Ophthalmol. 2018;66(3):407–410. doi:10.4103/ijo.IJO_813_17
19. Browne RH. On the use of a pilot sample for sample size determination. Stat Med. 1995;14(17):1933–1940. doi:10.1002/sim.4780141709
20. Jevitt JC, Jacobson G, Schittman RM. Validity and reliability of the Cataract TyPE Spec: an instrument for measuring outcomes of cataract extraction. Am J Ophthalmol. 2003;136(2):285. doi:10.1016/S0002-9394(03)00204-6
21. Ganesh S, Brar S, Pawar A, Relekar KJ. Visual and Refractive Outcomes following Bilateral Implantation of Extended Range of Vision Intraocular Lens with Micromonovision. J Ophthalmol. 2018;2018:7321794. doi:10.1155/2018/7321794
22. Choi SH, Lee HK, Yoon CH, Kim MK. Visual Performance after a Unilateral or Bilateral Implantation of Enlarged Depth-of-Focus Intraocular Lens in Patients with Cataract: A Prospective Clinical Trial. J Ophthalmol. 2019;2019:2163809. doi:10.1155/2019/2163809
23. de Medeiros AL, de Araújo Rolim AG, Motta AFP, et al. Comparison of visual outcomes after bilateral implantation of a diffractive trifocal intraocular lens and blended implantation of an extended depth of focus intraocular lens with a diffractive bifocal intraocular lens. Clin Ophthalmol. 2017;11:1911–1916. doi:10.2147/OPTH.S145945
24. Attia MSA, Auffarth GU, Kretz FTA, et al. Clinical evaluation of an extended depth of focus intraocular lens with the salzburg reading desk. J Refract Surg. 2017;33(10):664–669. doi:10.3928/1081597X-20170621-08
25. Pedrotti E, Bruni E, Bonacci E, Badalamenti R, Mastropasqua R, Marchini G. Comparative analysis of the clinical outcomes with a monofocal and an extended range of vision intraocular lens. J Refract Surg. 2016;32(7):436–442. doi:10.3928/1081597X-20160428-06
26. Ruiz-Mesa R, Abengozar-Vela A, Aramburu A, Ruiz-Santos M. Comparison of visual outcomes after bilateral implantation of extended range of vision and trifocal intraocular lenses. Eur J Ophthalmol. 2017;27(4):460–465. doi:10.5301/ejo.5000935.
27. Kohnen T, Böhm M, Hemkeppler E, et al. Visual performance of an extended depth of focus intraocular lens for treatment selection. Eye. 2019;33(10):1556–1563. doi:10.1038/s41433-019-0443-x
28. Son HS, Khoramnia R, Yildirim TM, Baur I, Labuz G, Auffarth GU. Functional Outcomes and Reading Performance After Combined Implantation of a Small-Aperture Lens and a Segmental Refractive Bifocal Lens. J Refract Surg. 2019;35(9):551–558. doi:10.3928/1081597X-20190806-02
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
