Back to Journals » International Medical Case Reports Journal » Volume 9
A case of optic-nerve hypoplasia and anterior segment abnormality associated with facial cleft
Authors Miyake T, Kojima S, Sugiyama T, Ueki M, Sugasawa J, Oku H, Tajiri K, Shigemura Y, Ueda K, Harada A, Yamasaki M, Yamanaka T, Utsunomiya H, Ikeda T
Received 27 October 2015
Accepted for publication 5 March 2016
Published 22 July 2016 Volume 2016:9 Pages 207—212
DOI https://doi.org/10.2147/IMCRJ.S99237
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Tomoko Miyake,1 Shota Kojima,1 Tetsuya Sugiyama,2 Mari Ueki,1 Jun Sugasawa,1 Hidehiro Oku,1 Kensuke Tajiri,1 Yuka Shigemura,3 Koichi Ueda,3 Atsuko Harada,4 Mami Yamasaki,4 Takumi Yamanaka,4 Hidetsuna Utsunomiya,5 Tsunehiko Ikeda1
1Department of Ophthalmology, Osaka Medical College, Takatsuki City, 2Nakano Eye Clinic of Kyoto Medical Co-operative, Kyoto, 3Department of Plastic and Reconstructive Surgery, Osaka Medical College, Takatsuki City, 4Department of Pediatric Neurosurgery, Takatsuki General Hospital, Takatsuki City, 5Department of Radiological Science, International University of Health and Welfare, Graduate School, Fukuoka, Japan
Introduction: The incidence of facial cleft is rare and ranges between 1.43 and 4.85 per 100,000 births. To date, there have been few reports of detailed ophthalmologic examinations performed in cases of facial cleft. Here, we report a case of optic-nerve hypoplasia and anterior segment abnormality associated with facial cleft.
Case report: A 9-day-old female infant was delivered by cesarian section at 34 weeks of gestational age (the second baby of twins) and weighed 2,276 g upon presentation. She had a facial cleft and ectrodactyly at birth. Right eye-dominant blepharophimosis was obvious. Examination of the right eye revealed inferior corneal opacity with vascularization, downward corectopia, and optic-nerve hypoplasia. The corneal diameter was 8 mm in both eyes, and tonometry by use of a Tono-Pen® XL (Reichert Technologies, Depew, NY, USA) handheld applanation tonometer revealed that her intraocular pressure was 11–22 mmHg (Oculus Dexter) and 8 mmHg (Oculus Sinister). B-mode echo revealed no differences in axial length between her right and left eyes. When she was 15–16 months old, we attempted to examine her eyes before she underwent plastic surgery under general anesthesia. She had a small optic disc in both eyes and the right-eye disc was tilted. After undergoing canthotomy, gonioscopy and ultrasound biomicroscopy revealed that almost all directions were open except for the peripheral anterior synechia. Since magnetic resonance imaging revealed ventriculomegaly associated with an interhemispheric cyst at birth, a ventriculoperitoneal shunt was inserted at 12 days of age. At 25 months of age, her condition suddenly deteriorated due to occlusion of the ventricular shunt catheter, and she died 5 days later. In this patient, amniotic band syndrome was presumed to be the primary cause due to the clinical findings.
Conclusion: We experienced a case of optic-nerve hypoplasia and anterior segment abnormality that occurred with facial cleft. The cause of these abnormalities is unclear, yet amniotic band syndrome is a possible candidate.
Keywords: facial cleft, optic-nerve hypoplasia, amniotic band syndrome
Introduction
Facial clefts are among the most disfiguring of all facial anomalies. The incidence of facial cleft is rare and ranges between 1.43 and 4.85 per 100,000 births.1 Because of their rarity, classification of these clefts has been difficult. A great advancement in the study of these rare clefts has been the Tessier classification, first reported in 1976.2 It occurs with lid closure failure, blepharophimosis, blepharocoloboma, lacrimal passage disorder, microphthalmos, anophthalmos, corneal opacity, corneal staphyloma, and coloboma iridis.1,3–6 To date, there have been few reports of detailed ophthalmologic examinations performed in cases of facial cleft. Here, we report a case of optic-nerve hypoplasia and anterior segment disorders that occurred with facial cleft.
Case report
A 9-day-old female infant (the second baby of twins) was referred from the Pediatrics Department to the Department of Ophthalmology in Osaka Medical College for ophthalmological examination. The obstetric history the mother revealed was that human chorionic gonadotropin administration and the water flow method were used to establish the pregnancy due to tubal occlusion. The children were dichorionic diamniotic twins. The mother was hospitalized with preterm labor at the time of 30 weeks. Family history revealed no systemic abnormal findings or malformations with the first child, and she was delivered by cesarian section at 34 weeks of gestational age and weighed 2,276 g upon presentation. At late pregnancy, she was diagnosed as having severe ventriculomegaly and a cleft lip palate. Other abnormal echo findings in the course of pregnancy had not been reported.
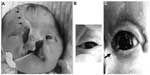
The infant’s face had a complete cleft on the left side that extended from the lip and undermined the alar base (Tessier No. 3); a bony cleft extended from the left orbital roof (Tessier No. 11) and the cleft of the left upper lip (Tessier No. 5). In addition, she had a complete cleft of the right side of the face that extended from the lip to the forehead near the eyebrow. There was an amniotic-like band from the cranial cleft end to the hair line of the forehead (Tessier Nos. 2 and 12) (Figure 1A).
The first eye examination revealed severe blepharophimosis in the right eye (Figure 1A and B) and a notch in the left lower eyelid (Figure 1C); however, there was no eyelid closure disorder in both eyes. Her right-eye cornea had inferior opacity, pseudopterygium, downward corectopia, and anterior synechia. The cornea of her left eye was clear. Fluorescein staining testing revealed no corneal or conjunctival epithelial disorders in both eyes. The anterior chamber and lens were normal in her left eye. Upon ophthalmoscopic examination, optic-nerve hypoplasia without mydriasis was suspected in her right eye. The corneal diameter was 8 mm in both eyes. Intraocular pressure (IOP) was 11–22 mmHg in her right eye and 8 mmHg in her left eye (Tono-Pen® XL; Reichert Technologies, Depew, NY, USA). B-mode echo without graduation on the scale revealed no difference in axial length between her right and left eyes, and her right eye showed no reaction to light. However, light produced a “dazzle” effect in her left eye. She also had ectrodactyly in the left hand and foot. There were no abnormalities of the blood test and visceral malformation.
Magnetic resonance imaging revealed an interhemispheric cyst associated with ventriculomegaly and agenesis of the corpus callosum (Figure 2A and B). A ventriculoperitoneal shunt was inserted at 12 days of age. Computed tomography (CT) scan images revealed no apparent differences of diameter or density between the right and left optic nerve.
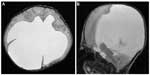  | Figure 2 Axial (A) and sagittal (B) T2-weighted magnetic resonance images reveal a huge interhemispheric cyst-associated ventriculomegaly and agenesis of the corpus callosum. |
We focused on the IOP, especially in the infant’s right eye, because the value was inaccurate due to blepharophimosis which inhibited the tip of the tonometer from touching the cornea correctly in that eye. Furthermore, we could not rule out the possibility of glaucoma in the right eye because the Tono-Pen® XL and Perkins Tonometer® (HAAG-STREIT, Koeniz, Switzerland) always showed that the IOP in the right eye, which had optic-nerve hypoplasia, was higher than that in the left eye. We measured IOP several times under sedation by Tono-Pen® XL and Perkins Tonometer® from 6 to 13 months of age, and IOP was found to be 20–43 mmHg in the right eye (Tono-Pen® XL). In the infant’s left eye, the IOP was 14–23 mmHg (Tono-Pen® XL) and 12–14 mmHg (Perkins Tonometer®). It was difficult to accurately measure the IOP of the right eye by Perkins Tonometer® due to blepharophimosis.
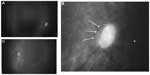
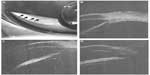
At 15 months of age, the patient underwent a detailed ophthalmological examination under general anesthesia before undergoing plastic surgery. IOP was 11–14 mmHg OD and 5–11 mmHg OS by Tono-Pen® XL and 11–14 mmHg OD and 5–6 mmHg OS by Perkins Tonometer®. The right-eye cornea had opacity (subepithelial to deep stroma) from the inferior to central area with vascularization in the corneal stroma, pseudopterygium, downward corectopia, and anterior synechia (Figure 3). Abnormalities were not observed with fluorescein staining. Fundus examination revealed a tilted disc and a double-ring sign in the right eye, and the disc macula distance/ disc diameter (DM/DD) ratio (ie, the ratio of the distance between the center of the optic disc and the fovea centralis to the diameter of the optic disc7) was ~4.0 in the right eye (Figure 4A and B). The DM/DD ratio was ~2.8 in the left eye (Figure 4C). After a canthotomy was performed, gonioscopy revealed that almost all directions were open except for the peripheral anterior synechia in the right eye (Figure 5A). Ultrasound biomicroscopy revealed that almost all directions were open except for the peripheral anterior synechia in the right eye (Figure 5B–D). Gonioscopy and ultrasound biomicroscopy revealed that all directions had no abnormalities in the left eye.
At 25 months of age, the patient presented to an emergency department because of vomiting and consciousness disturbance. A CT scan revealed dilatation of the ventricles, and she was diagnosed with shunt malfunction. Although emergency ventricular drainage was performed, the process ultimately led to brain death and the patient was pronounced dead 5 days later.
Discussion
There are various presumed mechanisms that induce facial cleft, such as dysraphisms of facial processes, invagination of the amniotic band, and insufficient blood flow in utero. In such cases, amniotic band syndrome is presumed to be the primary cause due to the clinical findings.8,9 Amniotic band syndrome refers to the collection of fetal malformations (limbs, head, and face) caused by the entanglement or attachment of amniotic remnants after rupture of the amniotic sac.10 Moreover, various complications, such as central nervous system abnormalities (ie, cerebral hypoplasia, corpus callosum dysplasia, and hydrocephalus), may occur in early pregnancy amniotic rupture.11–13
Miller et al10 reported the ophthalmic findings of nine cases of amniotic band syndrome. Five of those cases showed corneal opacity, like in our patient. Patient age at the time of their ophthalmologic examination ranged from 2 months to 13 years (the age in one case was unknown). One case also showed the cornea being covered with conjunctiva. In this study, they speculated that the cause of the corneal opacity was eyelid defects or poor eyelid closure due to the opacities being located adjacent to the eyelid defects and facial clefts.
In the case shown in this present study, corneal opacity was observed with no corneal erosion at 9 days of age. Unlike the findings in the report by Miller et al, no obvious eyelid closure failure or eyelid defects were found in the patient’s right eye. Furthermore, corneal opacity was from the subepithelial to deep stroma with vascularization in the corneal stroma, pseudopterygium, downward corectopia, and anterior synechia. We presumed that the corneal opacity that occurred developed in the embryonic stage, not after the birth, because there was no obvious corneal erosion at the first ophthalmic examination. However, the exact mechanism remains unclear.
The case in this present study showed optic-nerve hypoplasia in the right eye. The DM/DD ratio of 4.0 and the double-ring sign found in this case was helpful for the diagnosis of optic-nerve hypoplasia.14 In the CT scan images, no apparent differences were found between the right and left optic nerves. However, the dysplasia might not have been severe enough to detect the abnormality in the CT scan images. The mechanism of optic-nerve hypoplasia in our case is unclear. Furthermore, the relationship among the optic-nerve malformation, malformation of the brain (such as interhemispheric cyst and agenesis of the corpus callosum), and amniotic band syndrome remains unknown. To the best of our knowledge, this is the first report of a case of optic-nerve hypoplasia that occurred with facial cleft.
In this study, we examined the patient’s eyes frequently because we could not rule out the possibility of glaucoma in the right eye due to the repeated high IOP and the disc with optic-nerve hypoplasia. However, the IOP value was ultimately found to be normal. We theorize that there are two possibilities for the high IOP values, the first being that the tip of the Tono-Pen® XL may have touched the eyelid, thus causing an overestimation of the IOP. The second is that the patient’s eyelids were spread excessively by the eyelid device, thus adding more pressure on the eyeball. However, the problems were ultimately resolved and more accurate IOP measurements were obtained when canthotomy was performed before plastic surgery.
In conclusion, we report a rare case of optic-nerve hypoplasia and blepharophimosis that occurred with facial cleft. As for the anterior segment symptoms, similarities and differences were observed in comparison to those previously reported. Some of those symptoms point toward amniotic band syndrome involving malformation of the anterior segment of the eye. Thus, this report may be helpful in considering and estimating the impact of this syndrome on the eye when in the womb. Since facial cleft is rare, further studies with an additional accumulation of data are needed.
This case study was approved by the Ethics Committee of the Osaka Medical College. Parents or guardians provided written consent for the use of the patients’ image.
Acknowledgment
The authors thank Mr John Bush for reviewing the manuscript.
Disclosure
The authors report no conflicts of interest in this work.
References
Kawamoto HK Jr. The kaleidoscopic world of rare craniofacial clefts: order out of chaos (Tessier classification). Clin Plast Surg. 1976;3(4):529–572. | |
Tessier P. Anatomical classification of facial, cranio-facial and latero-facial clefts. J Maxillofac Surg.1976;4(2):69–92. | |
Coruh A, Gunay GK. A surgical conundrum: Tessier number 4 cleft. Cleft Palate Craniofac J. 2005;42(1):102–106. | |
David DJ, Moore MH, Cooter RD. Tessier clefts revisited with a third dimension. Cleft Palate J. 1989;26(3):163–185. | |
Whitaker LA, Katowitz JA, Randall P. The nasolacrimal apparatus in congenital facial anomalies. J Maxillofac Surg.1974;2(2–3):59–63. | |
Portier-Marret N, Hohlfeld J, Hamedani M, de Buys Roessingh AS. Complete bilateral facial cleft (Teissier4) with corneal staphyloma: a rare association. J Pediatr Surg. 2008;43(10):E15–E18. | |
Wakakura M, Alvarez E. A simple clinical method of assessing patients with optic nerve hypoplasia. The disc-macula distance to disc diameter ratio (DM/DD). Acta Ophthalmol (Copenh). 1987;65(5):612–617. | |
van der Meulen JC. Oblique facial clefts caused by amniotic bands. Plast Reconstr Surg. 1982;70(2):269–270. | |
Eppley BL, David L, Li M, Moore CA, Sadove AM. Amniotic band facies. J Craniofac Surg.1998;9(4):360–365. | |
Miller MT, Deutsch TA, Cronin C, Keys CL. Amniotic bands as a cause of ocular anomalies. Am J Ophthalmol.1987;104(3):270–279. | |
Stanek J, de Courten-Myers G, Spaulding AG, Strub W, Hopkin RJ. Case of complex craniofacial anomalies, bilateral nasal proboscides, palatal pituitary, upper limbs reduction, and amnion rupture sequence: disorganization phenotype? Pediatr Dev Pathol. 2001;4(2):192–202. | |
Tarantal AF, Hendrickx AG. Amniotic band syndrome in a rhesus monkey: a case report. J Med Primatol. 1987;16(5):291–299. | |
Donnenfeld AE, Dunn LK, Rose NC. Discordant amniotic band sequence in monozygotic twins. Am J Med Genet.1985;20(4):685–694. | |
Zeki SM, Dudgeon J, Dutton GN. Reappraisal of the ratio of disc to macula/disc diameter in optic nerve hypoplasia. Br J Ophthalmol. 1991; 75(9):538–541. |
 © 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.