Back to Journals » OncoTargets and Therapy » Volume 11
125I seed brachytherapy versus external beam radiation therapy for the palliation of painful bone metastases of lung cancer after one cycle of chemotherapy progression
Authors Xiang Z, Wang L, Yan H, Zhong Z, Liu W, Mo Z, Gao F , Zhang F
Received 24 October 2017
Accepted for publication 20 May 2018
Published 27 August 2018 Volume 2018:11 Pages 5183—5193
DOI https://doi.org/10.2147/OTT.S154973
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Jianmin Xu
Zhanwang Xiang,1–3,* Lifei Wang,4,* Huzheng Yan,5 Zhihui Zhong,5 Wangkai Liu,6 Zhiqiang Mo,5 Fei Gao,5 Fujun Zhang5
1Department of Radiology, The Third Affiliated Hospital, Sun Yat-sen University, Guangzhou, China; 2Department of Interventional Radiology, Ling-nan Hospital, Sun Yat-sen University, Guangzhou, China; 3Interventional Radiology Institute, Sun Yat-sen University, Guangzhou, China; Guangzhou, China; 4The Department of Radiology, The Third People’s Hospital of Shenzhen, Shenzhen, China; 5Sun Yat-sen University Cancer Center, State Key Laboratory of Oncology in South China, Collaborative Innovation Center for Cancer Medicine, Guangzhou, China; 6The First Affiliated Hospital, Sun Yat-sen University, Guangzhou, China
*These authors contributed equally to this work
Purpose: This study aimed to compare the outcomes of 125I seed brachytherapy versus external beam radiation therapy (EBRT) for the palliation of painful bone metastases of lung cancer after one cycle of chemotherapy progression.
Materials and methods: We analyzed retrospectively 158 patients with painful bone metastases secondary to lung cancer after one cycle of chemotherapy progression treated between June 2013 and May 2016. Seventy-six patients with 96 lesions received 125I brachytherapy (Group A), whereas 82 patients with 98 metastases received EBRT (Group B). Pain intensity on Brief Pain Inventory, percentage of patients with pain severity, and quality of life were recorded prior to treatment (T0), 2, 4, 6, 8, 12, 16, 20, and 24 weeks (T2, T4, T6, T8, T12, T16, T20, and T24) after treatment during a 24-hour period. Cost-effectiveness and number of treatment appointments were also compared between groups.
Results: One hundred and fifty-eight patients had been treated. Visual analog scale for worst pain in Group A was significantly lower than in Group B at T2, T4, T6, T16, T20, and T24. Group A was superior to group B concerning quality of life scores (T2, T4, T20, and T24), cost-effectiveness, and number of treatment appointments. No significant differences were observed for complications.
Conclusion: Compared with EBRT, 125I seed brachytherapy can be an alternative method for painful bone metastases from lung cancer after one cycle of chemotherapy progression.
Keywords: pain, 125I seed, brachytherapy, EBRT, bone metastases, lung cancer, chemotherapy
Introduction
Bone metastases secondary to lung cancer are frequent, particularly in cases of nonsmall-cell lung cancer. Reportedly, the rate of bone metastasis is as high as 30%–40%, and it continues to show an increasing trend.1,2 Pain is the most common clinical symptom of skeletal metastases. It severely affects the quality of life (QOL) of patients.3,4 Pain remission has become an important part of the comprehensive treatment of lung cancer and plays a key role in improving patient QOL.5
Currently, palliative therapy is regarded as the standard of care for painful bone metastases secondary to lung cancer.6,7 The standard treatments for bone metastases are external beam radiation therapy (EBRT) and surgery. Ablative strategies such as radiofrequency ablation (RFA), ultrasound ablation, microwave ablation, high-intensity focused ultrasound, or vertebral augmentation may also be performed (National Comprehensive Cancer Network guidelines 2017). Reportedly, EBRT is an effective treatment, and 60% of patients achieve pain remission.8,9 A trial conducted by the Radiation Therapy Oncology Group, including 1,016 patients, found that complete pain relief was observed in 53% of patients and partial relief in 83% with a mean duration of response of 12 and 20 weeks in two groups, respectively.10 Some studies also reported that RFA can provide significant pain relief for cancer patients who have failed standard treatments.11 Although EBRT or RFA seems to be effective for pain relief, some limitations are found. First, patients treated with ERBT for bone metastases, specifically spine metastases, may have reached the value of dose tolerance due to previous EBRT treatment. Second, the thermal ablative techniques have some disadvantages, including an increased possibility of pain and intra- or postprocedural injury to critical structures within the ablation zone.
In our previous study, we have shown that 125I brachytherapy, a new indication of iodine brachytherapy, was a safe and effective method for palliation of painful bone metastases secondary to lung cancer.12 Patients had significantly lower scores in the pain visual analog scale (VAS) after treatment with 125I brachytherapy. The QOL of patients was also obviously improved. No previous reports have compared computed tomography (CT)-guided 125I brachytherapy and EBRT for pain palliation. Thus, this study aimed to analyze the outcomes of 125I seed brachytherapy versus EBRT and evaluate whether 125I brachytherapy can be an alternative method to manage painful bone metastases secondary to lung cancer.
Materials and methods
Patients
This retrospective study was approved by the institutional review board at our hospital (Trail number: B2016-014-01). All individual participants agreed to participate in the study and signed informed consent. One hundred fifty-eight patients with one or two painful bone metastatic lesions secondary to lung cancer after one cycle of chemotherapy progression (score of ≥4 in worst pain over the past 24 hours according to the Brief Pain Inventory [BPI] Short Form) were enrolled in this study between June 2013 and May 2016. Then, 76 patients with 96 bone lesions received percutaneous CT-guided 125I seed implantation (Group A) and 82 patients with 98 bone metastases received EBRT (Group B).
All enrolled patients met the following inclusion criteria: 1) bone metastases secondary to lung cancer histologically or cytologically proven and confirmed by imaging examinations; 2) no more than two bone lesions sized less than 6 cm each; 3) BPI-Short Form score ≥4 for worst pain over the past 24 hours; 4) tumor progression after one cycle of chemotherapy; 5) expected survival time ≥3 months; 6) Eastern Cooperative Oncology Group performance status ≤2; 7) white blood cell ≥3×109/L and heemoglobin ≥90 g/dL; 8) absence of coagulopathy (prothrombin activity >40% or platelet count >50,000/μL); and 9) absence of impending fracture. All individual participants agreed the study and signed informed consent.
125I brachytherapy
The 125I seed type (Yunke Pharmaceutical Limited Liability Company, Chengdu, China) was 6,711, with a 0.8 mm diameter and 4.5 mm length. The central source of the particles was a 125I radionuclide silver rod. The matched peripheral dose (MPD) was 120 Gy (110–140 Gy), and the average energy was 27–32 KeV. Each deposit had an initial activity of 0.8 mCi and a half-life of 59.6 days, with a continuous release of low-dose γ-rays and soft X-rays (5% of 35 keV and 95% of 28 keV, respectively). About 93%–97% of dose activity was delivered in the first 8–10 months after the implant.
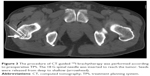
Before 125I brachytherapy, all patients underwent detailed tumor volume evaluation, using enhanced CT scans with a 5 mm thickness within 1 week before the procedure (Figure 1). Treatment was mapped and dosed for each patient using a computerized treatment planning system (TPS) (RT-RSI; Beijing Atom and High Technique Industries Inc, Beijing, China). The clinical target volume (CTV) was defined as a 1.0 cm expansion external to the gross tumor volume (GTV). The planned target volume (PTV) was defined as a 0.5 cm expansion external to the CTV. The prescribed dose was averaged at 120 Gy (range 100–140 Gy). Based on three orthogonal diameters within the target tumor, the prescribed MPD average was 120 Gy. The TPS generated dose–volume histogram, isodose curves of different percentages, and calculated the position of the brachytherapy applicator as well as the dose and number of implanted seeds (Figure 2). The PTV edge was covered by 70%–90% of the isodose curve. The MPD on vital organs and tissues was reduced according to the following criteria: large vessels <80 Gy, heart 45–50 Gy, oesophagus <60 Gy, trachea <50 Gy, spinal cord 45–50 Gy, breast <50 Gy, thyroid <45 Gy, kindey <20 Gy, and skin <50 Gy.
  | Figure 1 Prebrachytherapy shows one lesion in the right iliac bone (arrowhead). |
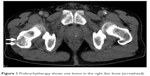
At the beginning of the operation, the patient was positioned in the CT gantry and the treatment site was localized. Then the CT scan, with a 5 mm thickness, was obtained to delineate the upper and lower borders of the tumor according to the preoperative TPS. After local infiltration anesthesia with 5–15 mL of 1% lidocaine (Liduokayin; Yimin, Yichang, China), an 18-G spinal needle (Yunke Pharmaceutical Limited Liability Company) was inserted to reach the farthest tumor edge, but was kept at approximately or less than 5 mm of the border (Figure 3). The seeds were released from deep to shallow while retracting the needle and keeping adjacent particles at a distance of 5–15 mm. To avoid intraprocedural complications, all of the spinal needles were retained until the implantation was completed and then removed simultaneously. A CT scan was performed after completion of the procedure to assess postoperative complications, such as bleeding. The last scan image was also reviewed to verify the position and intensity of 125I seeds according to TPS (Figure 4).
External beam radiation therapy
Before treatment, the physician and radiation physicist discussed and made an initial treatment plan based on the patient’s medical history, location of the lesion, and laboratory and imaging data. The procedure for all patients was preplanned on the CT simulator and was then administered by three-dimensional conformal radiation therapy or intensity-modulated radiotherapy.
Patients were taken into the simulator room, and the radiation physicist and physician selected the appropriate position according to the circumstances of each patient. To ensure the accuracy and repeatability of radiotherapy and reduce the impact of patient position changes, patients were made as comfortable as possible and a fixed mould was made for each patient. After a 5-mm thickness CT scan, image data were transmitted to the preoperative radiotherapy TPS to account for the entire data of bone metastases and surrounding tissues.
The physician and radiation physicist determined the target region and dose based on the obtained CT images. The GTV was delineated by fusing bone metastases volume on CT images, and CTV was defined as a 1.0 cm expansion external to GTV. The PTV included an additional margin of 10 mm in the cranio-caudal direction and 5 mm in the axial direction and was also defined as a 0.5 cm expansion external to the CTV. The prescribed dose average was 30 Gy (range 20–40 Gy). Normal tissue doses were calculated for the following structures: mean total lung dose, mean contralateral lung dose, maximum spinal cord dose (<18 Gy), maximum oesophagus dose (<27 Gy), maximum heart dose (<30 Gy), maximum brachial plexus dose (<24 Gy), and maximum trachea or mainstem bronchi dose (<30 Gy). Then, the radiotherapy procedure was completed according to the preoperative TPS. The dose of EBRT was given in 2 or 3 Gy per fraction, 5 days a week at a dose of 30 or 40 Gy in this study.
Follow-up
Efficacy assessment
A dynamic enhanced CT was performed every 2 weeks for the first 2 months, and then once every month thereafter (Figure 5). We assessed and compared pain intensity and need of pain medication between groups in a 24-hour period at 2, 4, 6, 8, 12, 16, 20, and 24 weeks (T2, T4, T6, T8, T12, T16, T20, and T24) by example. The worst pain and average pain with responses from 0 to 10 (0, no pain; 10, maximum pain intensity) were recorded to compare the pain controlling effects between both groups. Pain relief by pain treatments or medications was scored on a 0%–100% scale in 24 hours (0%, no relief; 100%, complete relief) in both groups.
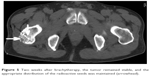  | Figure 5 Two weeks after brachytherapy, the tumor remained stable, and the appropriate distribution of the radioactive seeds was maintained (arrowhead). |
To further evaluate the analgesic results, pain scores of 0–3 were categorized as mild, 4–6 as moderate, and 7–10 as severe. The percentage distribution of patients whose worst pain score fell in each category was compared during various periods between Groups A and B.
QOL is defined as the effect of area of pain on the life state of patients according to Short Form-36 questionnaire (SF-36). QOL factors, including sleep, appetite, mental state, and fatigue, were rated by patients at T2, T4, T6, T8, T12, T16, T20, and T24 by using a five-point categorical scale (1=worst, 2=bad, 3=mild, 4=normal, and 5=very good). The differences in scores during these time periods were calculated and compared.
Cost-effectiveness and number of treatment appointments
The cost-effectiveness analysis is defined as the total cost of treatment (associated with 125I brachytherapy or EBRT process) paid by himself or herself in our country, not including medical insurance and donations. The cost-effectiveness included comparisons of mean treatment costs in Chinese yuan and dollars between Groups A and B. The number of treatment appointments was defined as the average number of appointments after brachytherapy or EBRT between doctor and patient during the entire study course.
Safety assessment
According to the National Cancer Institute Common Terminology Criteria for Adverse Events,13 the percentage of complications was analyzed for each group and compared.
Statistical analysis
The statistical software package SPSS, version 10.0 was used for statistical analyses. Values are presented as the mean ±SD for continuous variables with normal distribution, as median (interquartile range) for those with nonnormal distribution, and number of patients and percentages for categorical variables. A P-value of less than 0.05 was considered as an indicator of a statistically significant difference. BPI pain and QOL scores were compared between groups and analyzed by using Wilcoxon’s test. The percentage distribution of patients in each pain category, cost-effectiveness, and the number of treatment appointments in the two groups were analyzed with Pearson’s χ2 test.
Results
Patient characteristics
The characteristics of individual patients, tumors, and treatments are summarized in Table 1. When comparing the number of metastases, a single lesion was treated in 56 patients, whereas two lesions were treated in 20 patients, for a total of 96 tumors treated in Group A; a single lesion in 66 patients and two lesions in 16 patients, for a total of 98 tumors treated in Group B. In Group A, the total number of implanted seeds was 2,208, with an average of 24±4.6 seeds per lesion.
  | Table 1 Characteristics of patients and tumors |
Comparison of pain response between groups
The effectiveness of pain palliation in both groups is summarized in Table 2. VAS for worst pain in Group A was significantly lower than in Group B at T2 (P<0.01), T4 (P<0.01), T6 (P<0.01), T16 (P<0.01), T20 (P<0.01), and T24 (P<0.01). Although the pain relief for average pain was significantly improved from T0 to T24 in Group A, there was a significant difference in scores at T2, T4, T20, and T24 between the two groups.
Further comparison of the distribution of pain severity showed that the proportions of mild and moderate pain were significantly higher after treatment than prior to treatment. When Groups A and B were compared, the percentage of patients with pain was significantly different at T2 (P<0.01), T4 (P<0.01), T6 (P<0.01), T8 (P<0.01), T20 (P<0.01), and T24 (P<0.01), whereas there was no difference in scores at T12 (P=0.36) and T16 (P=0.51) (Table 3).
Differences in of QOL scores between groups
Comparison of QOL scores at T2 and T4 showed that recovery of quality of sleep, appetite, spirit, and fatigue in Group A was both significantly better than in Group B (P<0.01). When comparing Groups A and B at T6, T8, T12, and T16, there was no significant difference in the quality of sleep (P=0.31, 0.22, 0.36, 0.46), appetite (P=0.44, 0.52, 0.23, 0.33), mental state (P=0.33, 0.75, 0.41, 0.36), and fatigue (P=0.76, 0.26, 0.38, 0.56). However, the QOL scores decreased at T20 and T24 in Group B, and there were significant differences between both groups (P<0.01) (Table 4).
Cost-effectiveness and the number of treatment appointments
Regarding the cost-effectiveness and number of treatments appointments, lower costs and fewer treatments were observed for Group A. The difference between groups was statistically significant (P<0.01) (Table 5).
Local tumor control and complications between groups
All local bone tumors in Groups A and B were stable after 2 weeks of treatment according to the modified Response Evaluation Criteria in Solid Tumors (m-RECIST) on CT or positron emission tomography-CT images, and the range of tumor necrosis gradually increased with the prolonging of follow-up.
Regarding the safety evaluation, the proportion of patients who had complications in Groups A and B, respectively, were as follows: local skin reaction (10.5%, 19.5%; P=0.13); fever (3.9%, 2.4%; P=0.67), granulocytopenia (2.6%, 7.3%; P=0.28), a small amount of subcutaneous hemorrhage (19.7%; 0), seed displacement (2.6%; 0), radiation pneumonia (3.7%), and hydropneumothorax (1.2%). There was no statistically significant difference between the groups during the entire course of treatment. The mild-to-moderate complications were resolved after expectant management, and severe complications, such as pathological fracture and massive bleeding, were not seen in either group (Figure 6).
Discussion
Multiple minimally invasive image-guided ablation strategies (RFA, MWA, cryoablation) have been studied and received good results as treatments for painful bone metastases.14 Goetz et al reported score for worst pain was 7.9 prior to treatment, and the scores reduced to 5.8, 4.5, 3.0, and 1.4, respectively, at 1, 4, 8, and 24 weeks following RFA.15 Matthew et al found that score for worst pain using the BPI was 7.1 before treatment, and reduced to 5.1, 4.0, 3.6, and 1.4 at 1, 4, 8, and 24 weeks, respectively.16 However, these studies also found that a majority of patients would experience increased pain.
The present study results showed that 125I brachytherapy can be an alternative method for palliation of painful bone metastases secondary to lung cancer. VAS pain scores, the proportion of patients by pain severity, QOL, cost-effectiveness, and adverse events were all significantly improved in the brachytherapy group compared with the EBRT group. In fact, owing to variable individual tolerability and other factors, 125I brachytherapy may be an alternative method for patients with painful bone metastases secondary to lung cancer.17 At a low-dose-rate brachytherapy, the 125I seed emits continuous γ rays that inhibit tumor cell mitosis and decrease the resistance of hypoxic cells to radiation.18,19 Because the radiation from 125I seed decreases with increasing distance, much less radiation is delivered to the neighboring organs;20 thus, we considered the possibility that 125I brachytherapy might be an alternative treatment. To the best of our knowledge, our study is the first to compare 125I brachytherapy with EBRT and show that 125I brachytherapy can be useful to control painful bone metastases secondary to lung cancer.
Another relevant finding of this study is that 125I brachytherapy achieved pain palliation faster, with less pain recurrence in comparison with EBRT. VAS for worst pain in Group A was significantly lower than in Group B at T2 (P<0.01), T4 (P<0.01), T6 (P<0.01), T16 (P<0.01), T20 (P<0.01), and T24 (P<0.01). The proportion of patients according to the severity of pain was also significantly different between Groups A and B at T2 (P<0.01), T4 (P<0.01), T6 (P<0.01), T8 (P<0.01), T20 (P<0.01), and T24 (P<0.01). These results are consistent with those of previous studies on 125I brachytherapy for the management of painful bone metastases secondary to lung or liver cancer.21 A potential reason for the successful pain palliation achieved with 125I brachytherapy might be the radiation dose. First, large radiation doses are emitted “from the inside out” of the bone tumor; then, these undergo a rapid decrease as the radioactive energy is inversely correlated with the square of the radius, meaning that the normal tissue is less affected.22 Second, sustained low-dose radiation decreases the repair rates of sublethal damaged tumor cells.23
For patients with painful bone metastases, QOL is the main health-related factor that is affected.24 According to the comparison of QOL scores (including quality of sleep, appetite, mental state, and fatigue), there was a significant difference between 125I brachytherapy and EBRT at T2 and T4 (P<0.01). Additionally, these scores decreased at T20 and T24 in Group B. These results are consistent with the VAS scores reported by patients and further illustrate the faster pain palliation and less pain recurrence achieved with 125I brachytherapy.25 Additionally, we found that 125I brachytherapy was relatively superior to EBRT in terms of lower treatment costs (9.5±0.3 vs 46.3±0.5) and fewer number of post-treatment appointments (9.2±0.6 vs 12.6±1.4).26
The American Brachytherapy Society’s “dual 90” guideline states that to achieve prostate cancer cure, 90% of the tumor volume needs to receive at least 90% of the prescribed dose.27 Our study showed that TPS can contribute to the achievement of the peripheral tumor doses (MPD of 100–140 Gy) and ensure that more than 95% of the tumor receives 100% of the prescribed dose.28,29 Using CT guidance, all patients with bone metastases secondary to lung cancer were successfully treated. Fifteen patients (19.7%) presented minor subcutaneous hemorrhages, probably because of injury to subcutaneous vessels, but all recovered after expectant treatment. Eight (10.5%) and 16 (19.5%) patients presented local skin reactions in Groups A and B, respectively, but there was no significant difference between groups. All patients recovered after local treatment. No serious complications were observed in either group. These findings further illustrate that 125I brachytherapy is a safe and feasible treatment for pain palliation.
Despite our important findings, our study had some limitations. First, assessments of pain scores and QOL were based on patient-answered questionnaires. Patients, for the most part, did not have experience with the rating systems, and their assessments could have varied during the treatment course. Second, although pathology subtypes of lung cancer metastases were clarified, it is not clear which pathology subtype of metastasis is more sensitive to 125I brachytherapy or EBRT. Third, the injection of local anesthesia might have contributed to pain relief experience in brachytherapy. Last, many researchers would consider the use of less fractionated treatments and variable treatment schedules in EBRT might improve QOL; thus, a method for planning the most appropriate dose for bone metastases is the future direction of study.
Conclusion
Our results showed that CT-guided 125I brachytherapy can be a justifiable alternative method for palliation of painful bone metastases secondary to lung cancer after one cycle of chemotherapy progression. Compared with EBRT, 125I brachytherapy resulted in faster pain palliation, better QOL, less pain recurrence, less cost-effectiveness, and similar complications. Studies, in larger cohorts, could help substantiate this claim and might help broaden application of this therapy.
Acknowledgments
This study was funded by National Natural Science Foundation of China (Grant No 81371654 to FZ and 81571780 to FG) and Natural Science Foundation of Guangdong Province (Grant No 2014A030310006 to FG). The authors extend their thanks to Professor Lian Xiong Yuan for providing statistical advice for the data from the inception of the study. This work was supported by the institutional review board at our hospital (the Sun Yat-Sen University Cancer Center, Trial number: B2016-014-01, Date of registration: 9 August 2013).
Disclosure
The authors report no conflicts of interest in this work.
References
Mundy GR. Metastasis to bone: causes, consequences and therapeutic opportunities. Nat Rev Cancer. 2002;2:584–593. | ||
Oliveira MB, Mello FC, Paschoal ME. The relationship between lung cancer histology and the clinicopathological characteristics of bone metastases. Lung Cancer. 2016;96:19–24. | ||
Jacox A, Carr DB, Payne R. New clinical-practice guidelines for the management of pain in patients with cancer. N Engl J Med. 1994;330:651–655. | ||
Portenoy RK, Miransky J, Thaler HT, et al. Pain in ambulatory patients with lung or colon cancer. prevalence, characteristics, and effect. Cancer. 1992;70:1616–1624. | ||
Tang Y, Qu J, Wu J, et al. Effect of surgery on quality of life of patients with spinal metastasis from non-small-cell lung cancer. J Bone Joint Surg Am. 2016;98:396–402. | ||
Smith HS. Painful osseous metastases. Pain Physician. 2011;14:E373–E403. | ||
Callstrom MR, Atwell TD, Charboneau JW, et al. Painful metastases involving bone: percutaneous image-guided cryoablation-prospective trial interim analysis. Radiology. 2006;241:572–580. | ||
Sze WM, Shelley M, Held I, Mason M. Palliation of metastatic bone pain: single fraction versus multifraction radiotherapy: a systematic review of the randomized trials. Cochrane Database Syst Rev. 2004;2:CD004721. | ||
Dupuy DE, Liu D, Hartfeil D, et al. Percutaneous radiofrequency ablation of painful osseous metastases: a multicenter American College of Radiology Imaging Network trial. Cancer. 2010;116:989–997. | ||
Tong D, Gillick L, Hendrickson FR. The palliation of symptomatic osseous metastases: final results of the Study by the Radiation Therapy Oncology Group. Cancer. 1982;50:893–899. | ||
Groenemeyer DH, Schirp S, Gevargez A. Image-guided percutaneous thermal ablation of bone tumors. Acad Radiol. 2002;9:467–477. | ||
Xiang Z, Mo Z, Li G, et al. 125I brachytherapy in the palliation of painful bone metastases from lung cancer after failure or rejection of conventional treatments. Oncotarget. 2016;7:18384–18393. | ||
Kluetz PG, Chingos DT, Basch EM, Mitchell SA. Patient-reported outcomes in cancer clinical trials: measuring symptomatic adverse events with the National Cancer Institute’s Patient-Reported Outcomes Version of the Common Terminology Criteria for Adverse Events (PRO-CTCAE). Am Soc Clin Oncol Educ Book. 2016;35:67–73. | ||
Liberman B, Gianfelice D, Inbar Y, et al. Pain palliation in patients with bone metastases using MR-guided focused ultrasound surgery: a multicenter study. Ann Surg Oncol. 2009;16:140–146. | ||
Goetz MP, Callstrom MR, Charboneau JW, et al. Percutaneous imageguided radiofrequency ablation of painful metastases involving bone: a multicenter study. J Clin Oncol. 2004;22:300–306. | ||
Callstrom MR, Dupuy DE, Solomon SB, et al. Percutaneous image-guided cryoablation of painful metastases involving bone: multicenter trial. Cancer. 2013;119:1033–1041. | ||
Feng S, Wang L, Xiao Z, et al. 125I seed implant brachytherapy for painful bone metastases after failure of external beam radiation therapy. Medicine. 2015;94:e1253. | ||
Deweese TL, Shipman JM, Dillehay LE, Nelson WG. Sensitivity of human prostatic carcinoma cell lines to low dose rate radiation exposure. J Urol. 1998;159:591–598. | ||
Koritzinsky M, Wouters BG, Amellem O, Pettersen EO. Cell cycle progression and radiation survival following prolonged hypoxia and re-oxygenation. Int J Radiat Biol. 2001;77:319–328. | ||
Zhang T, Lu M, Peng S, et al. CT-guide implantation of radioactive 125I seed in advanced non-small-cell lung cancer after failure of first-line chemotherapy. J Cancer Res Clin Oncol. 2014;140:1383–1390. | ||
Jiao D, Wu G, Ren J, Han X. Radiofrequency ablation versus 125I-seed brachytherapy for painful metastases involving the bone. Oncotarget. 2016;7:87523–87531. | ||
Deweese TL, Shipman JM, Dillehay LE, Nelson WG. Sensitivity of human prostatic carcinoma cell lines to low dose rate radiation exposure. J Urol. 1998;159:591–598. | ||
Sgouros G, Knox SJ, Joiner MC, Morgan WF, Kassis AI. MIRD continuing education: bystander and low dose-rate effects: are these relevant to radionuclide therapy? J Nucl Med. 2007;48:1683–1691. | ||
Kougioumtzopoulou A, Zygogianni A, Liakouli Z, Kypraiou E, Kouloulias V. The role of radiotherapy in bone metastases: a critical review of current literature. Eur J Cancer Care (Engl). 2017;26:e12724. | ||
Mendez LC, Raman S, Wan BA, et al. Quality of life in responders after palliative radiation therapy for painful bone metastases using EORTC QLQ-C30 and EORTC QLQ-BM22: results of a Brazilian cohort. Ann Palliat Med. 2017;6:S65–S70. | ||
Zhou B, Wang J, Yan Z, Shi P, Kan Z. Liver cancer: effects, safety, and cost effectiveness of controlled-release oxycodone for pain control after TACE. Radiology. 2012;262:1014–1021. | ||
Nag S. Brachytherapy for prostate cancer: summary of American Brachytherapy Society recommendations. Semin Urol Oncol. 2000;18:133–136. | ||
Pignol JP, Rakovitch E, Keller BM, Sankreacha R, Chartier C. Tolerance and acceptance results of a palladium-103 permanent breast seed implant phase I/II study. Int J Radiat Oncol Biol Phys. 2009;73:1482–1488. | ||
Xiang Z, Li G, Liu Z, et al. 125I Brachytherapy in locally advanced nonsmall cell lung cancer after progression of concurrent radiochemotherapy. Medicine (Baltimore). 2015;94:e2249. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.