Back to Journals » Clinical, Cosmetic and Investigational Dermatology » Volume 9
Water-soluble egg membrane enhances the immunoactivating properties of an Aloe vera-based extract of Nerium oleander leaves
Authors Benson KF, Newman RA ![]() , Jensen GS
, Jensen GS ![]()
Received 7 June 2016
Accepted for publication 10 August 2016
Published 3 November 2016 Volume 2016:9 Pages 393—403
DOI https://doi.org/10.2147/CCID.S114471
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Jeffrey Weinberg
Video abstract presented by Gitte S Jensen
Views: 2501
Kathleen F Benson,1 Robert A Newman,2,3 Gitte S Jensen1
1NIS Labs, Klamath Falls, OR, 2Department of Experimental Therapeutics, University of Texas MD Anderson Cancer Center, Houston, 3Nerium Biotechnology Inc, San Antonio, TX, USA
Objective: To evaluate a blend of two natural ingredients on immune parameters relevant for their current topical use and potential support of microcirculation in skin tissue.
Materials and methods: A blend (BL) of Aloe vera-based Nerium oleander extract (NAE-8i, oleandrin-free) and hydrolyzed water-soluble egg membrane (WSEM) was applied to human whole-blood cultures for 24 hours, with each separate ingredient serving as a control. Immune-cell subsets were analyzed for expression levels of the activation markers CD69 and CD25. Culture supernatants were analyzed for cytokines, chemokines, and immunoregulating peptides.
Results: BL increased CD69 expression on lymphocytes, monocytes, and CD3–CD56+ natural killer cells, and CD25 expression on natural killer cells. The number of CD69+CD25+ lymphocytes increased in cultures treated with BL and the separate ingredients. BL triggered production of multiple cytokines and chemokines, where CC chemokines MIP1α and MIP3α, as well as cytokines involved in wound healing – Groα, Groβ, ENA78, and fractalkine – reached levels manyfold above treatment with either NAE-8i or WSEM alone.
Conclusion: Data on BL showed that WSEM strongly enhanced NAE-8i’s effects on immunoactivation in vitro. This has potential relevance for support of immunity in skin tissue, including antibacterial and antiviral defense mechanisms, wrinkle reduction, and wound care.
Keywords: chemokines, cytokines, leukocyte activation
Introduction
The skin is the largest organ of the human body, and serves as a critical front line of defense with respect to physical, chemical, and biological insults from the external environment. It is composed of well-characterized resident epidermal and dermal cell types, as well as immune cells. The principal way in which skin responds to damage or injury is through an acute inflammatory response. This typically consists of an innate system of cellular and humoral responses in which the body attempts to restore dermal tissue to its preinjury state. This involves a complex orchestration of events, including activation of endothelial cells, adhesive interactions between leukocytes and the vascular endothelium, activation of tissue macrophages, and activation of platelets and complement.1–3 Equally important is the subsequent resolution of the acute inflammatory response. Such a resolution involves the sequential interdependent release of additional vasoactive and chemotactic mediators.4,5
In addition to acute skin injury or that due to disease, the formation of facial wrinkles and tissue aging has also been linked to changes in skin biochemistry (eg, loss of elastic properties in dermal tissue) and immunology. Healthy skin tissue represents a dynamic environment in which immunogenic vigilance and response play a key role in maintaining skin integrity and homeostasis.6,7 While acute inflammatory responses are important to the immune defense against bacterial invaders of dermal tissue, it is suspected that impaired innate immunity is related to chronic inflammatory skin conditions with a bacterial association.8 Acne, a chronic slow-resolving inflammatory disease, is an example of this. Acne development is linked to the combination of genetic and environmental factors, among which a prominent role is played by the follicular colonization of Propionibacterium acne. This induces stimulation of an inflammatory response followed by the secretion of proinflammatory molecules and by activation of innate immunity.9 Increased cutaneous and systemic oxidative stress then serve as factors in the pathogenesis of acne as a chronic inflammatory condition.10 Resolution of acute inflammation is under strict checkpoint control by endogenous proresolution factors that are crucial in preventing excessive tissue injury, autoimmunity, and chronic inflammation.11
We recently reported the beneficial antioxidant and anti-inflammatory effects of a novel extract, NAE-8i, extracted from Nerium oleander using Aloe barbadensis (also known as Aloe vera) gel material.12 The extract has now been further modified, such that it no longer contains cardiac glycosides, such as oleandrin, yet retains compounds from both Nerium oleander and Aloe that are known to benefit skin health. Another natural ingredient with noteworthy benefits for skin health is water-soluble egg membrane (WSEM). EM is the thin protective layer of tissue between the egg white and the eggshell and contains an abundance of antimicrobial, immunomodulatory, and other bioactive proteins and peptides.13–18 Proteoglycans in EM have been used successfully in treating nonhealing wounds and burns, due to their biocompatibility, biodegradability, and similarity to macromolecules found in the human body.19,20 Of specific relevance for the work reported here, a hydrolyzed WSEM product triggered upregulation of antioxidant-response elements in human keratinocytes, and significantly reduced production of reactive oxygen species by polymorphonuclear (PMN) cells in vitro. Furthermore, human dermal fibroblasts treated with WSEM in vitro showed increase in the production and secretion of collagen and elastin.21 These properties suggest several modes of action for the reduction of facial wrinkles, seen in a recently completed clinical study, where significant changes in wrinkle contours of facial skin wrinkles were documented after 8 weeks of topical use.21 The potential for synergistic biological effects of the plant-based NAE-8i and the EM-based WSEM is therefore pertinent and the driving reason for the testing reported in this paper.
Materials and methods
Reagents
Phosphate-buffered saline, Roswell Park Memorial Institute 1640 medium, penicillin–streptomycin 100×, interleukin-2 (IL-2), and lipopolysaccharide (LPS) were purchased from Sigma-Aldrich Co. (St Louis, MO, USA). Cal-Lyse™ whole-blood lysing solution was purchased from Thermo Fisher Scientific (Waltham, MA, USA). CD69 fluorescein isothiocyanate, CD56 phycoerythrin, CD3 peridinin chlorophyll protein, CD25 brilliant violet 421 and heparin Vacutainer tubes were purchased from BD (Franklin Lakes, NJ, USA). The Bio-Plex Pro™ human chemokine 40-Plex was purchased from Bio-Rad Laboratories Inc. (Hercules, CA, USA).
Extracts and blend
A liquid A. vera-based N. oleander extract (NAE-8i) was provided by Nerium Biotechnology Inc (San Antonio, TX, USA). Basic details of the extraction procedure are given in the issued patent US8524286. The extract was further modified to remove cardiac glycosides, yet retain other Aloe- and N. oleander-based phytochemicals. Dilutions of extracts were performed using physiological saline. A hydrolyzed WSEM powder was obtained by Nerium Biotechnology from Biova LLC (Johnston, IA, USA). A stock solution was prepared by dispersing 0.5 g into 5 mL physiological saline and allowing the soluble compounds to dissolve under gentle agitation for 1 hour. A small amount of undissolved solid matter was removed by centrifugation at 2,400 rpm for 10 minutes. The liquid was then passed through a 0.22 μm sterile filter in preparation for adding to cell cultures. The NAE-8i–WSEM blend (BL) was then made by combining equal volumes of WSEM at 50 g/L with NAE-8i at 200 mL/L to reflect the relative doses in which this blend is used in topical skin-care formulations.
Expression of CD69 and CD25 on leukocyte subsets
Upon written informed consent from subjects, and approval by the Sky Lakes Medical Center Institutional Review Board, Federalwide Assurance 2603, peripheral whole blood was drawn from three human donors and used to establish cultures where blood (40 μL) was combined with Roswell Park Memorial Institute 1640 medium (140 μL) containing penicillin–streptomycin. Serial dilutions of products or LPS (10 ng/mL) were added to cultures at a volume of 20 μL, and cultures were then incubated at 37°C, 5% CO2 for 24 hours. The highly inflammatory bacterial LPS from Escherichia coli was used as a positive control for immune-cell activation. IL-2 was used as a positive control for natural killer (NK)-cell activation. Untreated controls consisted of cells exposed to phosphate-buffered saline in the absence of test products. All treatments, including each dose of test product and each positive and negative control, were tested in triplicate. After 24 hours, blood cells were isolated from each culture well and stained for 15 minutes with labeled antibodies at the recommended concentration. Cells were then fixed and red blood cells lysed using Cal-Lyse whole-blood lysing solution following the manufacturer’s instructions. Flow cytometry was performed with an Attune acoustic-focusing flow cytometer (Thermo Fisher Scientific). Data analysis utilized gating on forward/side scatter to evaluate CD69 expression on lymphocyte monocyte/macrophage- and PMN-cell subsets. The lymphocyte subpopulation was further analyzed for CD25 and CD69 expression on CD3–CD56+ NK cells, and to determine the percentage of CD69+CD25+ double-positive lymphocytes.
Cytokine profiles from whole-blood cultures
Supernatants were harvested from human whole blood (three donors) 24-hour cultures, and concentrations of 40 cytokines and chemokines were analyzed. IL-1β, IL-2, IL-4, IL-6, IL-8 (CXCL8), IL-10, IL-16, interferon gamma (IFNγ), tumor necrosis factor alpha (TNFα), MIF, granulocyte-macrophage colony-stimulating factor (GM-CSF), I-309 (CCL1), eotaxin (CCL11), eotaxin 2 (CCL24), eotaxin 3 (CCL26), MIP1 (CCL3), MIP3α (CCL19), MIP3β (CCL20), MIP5 (CCL15), MCP1 (CCL2), MCP2 (CCL8), MCP3 (CCL7), MCP4 (CCL13), TARC (CCL17), 6Ckine (CCL21), MDC (CCL22), MPIF1 (CCL23), TECK (CCL25), CTACK (CCL27), Groα (CXCL1), Groβ (CXCL2), ENA-78 (CXCL5), GCP-2 (CXCL6), MIG (CXCL9), IP-10 (CXCL10), I-TAC (CXCL11), SDF1α (CXCL12), BCA1 (CXCL13), SCYB16 (CXCL16), and fractalkine (CX3CL1) were quantified using Bio-Plex protein arrays (Bio-Rad Laboratories Inc.) and utilizing xMAP technology (Luminex, Austin, TX, USA).
Statistical analysis
Average and standard deviation calculations for each data set were made using Microsoft Excel. Statistical analysis of in vitro data was performed using the two-tailed, independent t-test. Statistical significance was set at P<0.05, and a high level of significance at P<0.01.
Results
Activation of leukocyte subsets
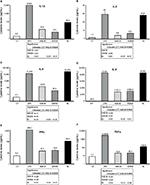
Immune-cell activation was determined by measuring cell-surface expression of the activation markers CD69 and CD25. The use of fluorescently labeled antibodies to CD3, CD56, CD69, and CD25 allowed the monitoring of changes to leukocyte subsets, including lymphocytes, monocytes/macrophages, NK cells, and PMN cells. Treatment of whole-blood cultures with either the Aloe-based Nerium extract NAE-8i or the WSEM for 24 hours led to dose-dependent activation of lymphocytes and monocytes (Figure 1A and B), with BL inducing the highest level of activation. The effects on PMN-cell activation were more complex: the two highest doses of WSEM and the intermediate doses of NAE-8i and BL led to decreased CD69 expression on PMN cells (Figure 1C), while the highest dose of BL triggered an increase in CD69 expression.
All three products triggered an increase in CD69 expression on CD3–CD56+ NK cells, where cells treated with BL showed the highest level of CD69 expression on NK cells (Figure 2A). BL also increased CD25 expression on NK cells at the highest dose of 20 mg/mL concentration (Figure 2B). Immunophenotypic analysis also allowed evaluation of a rare population of lymphocytes that express both CD69 and CD25. CD69+CD25+ double-positive lymphocytes represent a rare subpopulation of cells whose function is poorly understood, but that appear to be a subset of CD8+ NK cells.22 A dose-dependent increase in the number of lymphocytes expressing both the CD69- and CD25-activation markers was seen following treatment of cultures with all three products (Figure 2C). The greatest increases in the number of CD69+CD25+ lymphocytes were seen in cultures treated with BL followed by cultures treated with NAE-8i.
Production of cytokines and chemokines in human whole-blood cultures
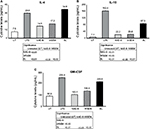
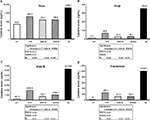
Supernatants from the whole-blood cultures exposed to products for 24 hours were simultaneously assayed for the levels of 40 different cytokines and chemokines using a magnetic bead-based array and Luminex xMAP technology. Robust increases in the number of cytokines with various regulating properties were seen. This included upregulation of certain proinflammatory cytokines, including IL-1β, IL-2, IL-6, IL-8, IFNγ, and TNFα (Figure 3). Increases were also seen in the anti-inflammatory cytokines IL-4 and IL-10 and the growth factor GM-CSF (Figure 4). Increases were seen in cytokines playing a role in wound healing (Figure 5), and included the CX3CR1-receptor ligand fractalkine and the CXCR2-receptor ligands Groα, Groβ, and ENA78. Cytokines involved in NK-cell activation and antiviral immunodefense activity were also upregulated and included MIP1α, MIP3α, SDF1α, and IP10 (Figure 6), as well as MIP3β (data not shown) and fractalkine (Figure 5D).
Discussion
Despite increased knowledge of the molecular mechanisms involved in maintaining skin health, few studies exist on the potential role of natural products for topical skin agents and their influence on immunologic processes of relevance for dermal health. The goal of the present research was to investigate a blend of two natural ingredients – an oleandrin-free version of an A. vera-based N. oleander extract (NAE-8i) – combined with hydrolyzed WSEM, used for topical applications in antiaging skin products. We have previously shown that an Aloe-based Nerium extract possesses potent antioxidant and anti-inflammatory properties, while also inducing specific cytokines in dermal fibroblasts of relevance to wound healing. We also reported that topical application of WSEM is associated with significant wrinkle reduction, likely due to a combination of its effect on dermal fibroblast production of collagen and elastin in combination with induction of antioxidant-response elements downstream of NRF2. In order to study the immunomodulating effects of BL of these two natural ingredients, human whole-blood cultures were used, as this culture model allows cross talk between relevant cell types in an orchestrated manner and pertains directly to microvascular blood perfusion in skin tissue.
The data reported here showed that BL induced biological responses beyond simple additive effects contributed by each ingredient. Due to the likelihood of engaging different cell-surface receptors by active compounds in A. vera, N. oleander, and EM, synergy was expected. That said, the magnitude of the synergistic effects was more robust than anticipated, as the amplification of the responses to BL was above and beyond the additive effects of each ingredient. NK cells were activated in response to BL, as seen by increased CD69 and CD25 expression. Since CD69+ NK cells are associated with increased cytotoxicity, while CD25+ NK cells show increased proliferation,23 the data suggest that BL supports innate immunity, particularly toward virally infected or transformed cells. Increased production of the MIP1α and MIP3α chemokines, involved in NK-cell activation and antiviral protection, was seen following treatment of whole-blood cultures with BL, far beyond a simple additive effect contributed by each of the two natural ingredients. The activation of NK cells and monocytes in whole-blood cultures implies that similar events take place in skin tissue exposed to BL in a topical application.
Inflammation is an important aspect of immunity, specifically protection of the host from pathogens and maintenance of the skin barrier, as well as playing a role in wound healing. Data from studies in knockout mice,24,25 as well as human diseases, including diabetes, and glucocorticoid treatment26 show that lack of an appropriate proinflammatory response early on leads to delayed or aberrant wound healing. The data reported here showed that several proinflammatory cytokines were robustly induced by BL, including IL-1β, TNFα, and IFNγ. This may also suggest improved innate immunodefense activity, since in atopic dermatitis TNFα, IFNγ, and IL-1β are reduced and patients have an increased susceptibility to microorganisms.27 Known to increase cellular responsiveness to growth factors and induce signaling pathways that lead to proliferation, TNFα is associated with such conditions as psoriasis,28 yet in normal skin epithelium, low levels of TNFα are generally localized within the epidermal basal cell layer29 and likely play a key role in maintenance of the permeability barrier. IL-6 is also correlated with psoriasis at elevated levels, and is induced after epithelial disruption, yet both IL-6 and TNFα promote normal keratinocytic growth, epithelial barrier integrity, and collagen deposition.30
The increase in cytokine production in response to BL may potentially help support reparative functions in skin, since the IL-1 family of cytokines signals keratinocyte proliferation, and reduced signaling of the IL-1 cytokine family in aging skin tissue has been associated with an altered epidermal permeability barrier.31 The induction of IL-1β and TNFα is known to also trigger hyaluronic acid synthases in skin tissue,32 thus contributing to intrinsic hyaluronic acid production, associated with improved skin hydration and function. Potential wrinkle-reducing effects of topical use of BL may be associated with a combination of the intrinsic effects of WSEM directly on matrix deposition by skin cells21 and the induction of cytokines by skin-residing immune cells.
The immunoactivating events mediated by BL were selective and did not apply uniformly to all cell types. Reduced CD69 expression of inflammatory PMN cells by BL is seen in the context of previously published results on each ingredient’s capacity to reduce formation of reactive oxygen species.12,21 This anti-inflammatory property was further substantiated by the induction of IL-4 and IL-10 production in the cultures.
The complex cytokine responses induced by BL are interesting in light of previous documentation showing that topical application of a combination of growth factors and cytokines was associated with reduction in wrinkles and general signs of photoaging of human facial skin.33 Rather than application of extrinsic growth factors, the topical use of the natural BL has the potential to induce intrinsic production of a similarly complex array of cytokines. This in combination with the known antioxidant and anti-inflammatory properties of each ingredient makes BL a promising natural and noninvasive alternative for cosmetic and overall skin health-related purposes.
A different set of observations reported here includes the induction of a set of chemokines involved in wound healing, specifically acting through the CXCR1/CXCR2 receptors. Very large increases in the production of Groα, Groβ, ENA78, and fractalkine were seen, as well as IL-8. These chemokines belong to a group of angiogenic chemokines that contain an ELR amino acid motif.34,35 IL-8, a proinflammatory cytokine with sentinel function, is rapidly induced from blood and skin cells in response to various sensitizers, and then triggers chemotaxis for T lymphocytes, basophils, and NK cells.36 However, since IL-8 also transactivates EGFR, it could facilitate epitheliogenesis after or along with inflammatory chemotaxis.37 Under normal conditions, it is found at only mild levels in the normal epidermis and is absent in the dermis.38 Dermokine, a gene upregulated in inflammatory diseases, has been shown to interfere with expression of ELR+ chemokines and result in delayed wound healing.39,40 Therefore, further work is needed on BL, such as three-dimensional cell-culture models for wound healing and repair.
The presence of various immunoregulatory cells in the skin, and the programming of these cells toward pro- versus anti-inflammatory activities, has known profound effects on skin health.41 Furthermore, cross talk between immune cells and skin cells is important in up- or downregulation of inflammatory conditions in the skin, as well as the initiation of repair mechanisms.42,43 Macrophages and fibroblasts are two main players in skin-tissue health and homeostasis and play crucial roles in tissue repair. The cross talk between macrophages and dermal fibroblasts directs the overall processes of repair due to damage caused by external stressors within the microenvironment of the skin. The proinflammatory cytokines IL-1β, IL-6, and TNFα have been shown to strongly induce dermal fibroblasts to produce keratinocyte growth factor in culture.44
The data presented here suggest that the benefits of skin-care products containing NAE-8i and WSEM on skin health and maintenance may directly result from enhancement of early immune events within the skin. These effects may support immune responses in skin tissue to daily environmental insults and induce a more rapid return to homeostasis. The blend of two natural ingredients triggered robust immunoactivating events, including NK-cell activation of relevance for immunoprotection of skin tissue, both with respect to antiviral protection, such as from Herpes simplex, and protection from precancerous events associated with ultraviolet exposure.45 Activation of monocytes is of relevance for antigen presentation and immunoregulation, which in combination with the complex cytokine effects is of interest in the resolution of acne, as well as support of wound healing. The underlying in vivo mechanisms of action of BL are likely to include not only direct effects on dermal fibroblasts but also direct effects on skin-residing macrophages, as well as leukocytes recruited to the skin. Further studies will need to address more specifically the effects of BL in situations pertaining to antiviral effects (H. simplex), antibacterial effects (Propionibacterium acnes), ultraviolet-radiation insults, wrinkle reduction, and wound repair.
Acknowledgments
The study was conducted at NIS Labs, an independent contract-research laboratory specializing in natural product research. The study was sponsored by Nerium Biotechnology Inc.
Disclosure
KFB and GSJ are employed by NIS Labs, an independent contract-research laboratory specializing in natural product research. RAN serves as chief science officer for Nerium Biotechnology Inc, the sponsor of this study. The authors report no other conflicts of interest in this work.
References
Thelan M, Stein JV. How chemokines invite leukocytes to dance. Nat Immunol. 2008;9(9):953–959. | ||
Kupper TS, Fuhlbrigge RC. Immune surveillance in the skin: mechanisms and clinical consequences. Nat Rev Immunol. 2004;4(3):211–222. | ||
Tan SY, Roediger B, Weninger W. The role of chemokines in cutaneous immunosurveillance. Immunol Cell Biol. 2015;93(4):337–346. | ||
Serhan CN. Resolution phase of inflammation: novel endogenous anti-inflammatory and proresolving lipid mediators and pathways. Annu Rev Immunol. 2007;25:101–137. | ||
Ward PA. Acute and chronic inflammation. In: Serhan CN, Ward PA, Gilroy DW, editors. Fundamentals of Inflammation. 2010. New York: Cambridge University Press; 1–16. | ||
Pasparakis M, Haase I, Nestle FO. Mechanisms regulating skin immunity and inflammation. Nat Rev Immunol. 2014;14(5):289–301. | ||
Cooper PR, Takahashi Y, Graham LW, Simon S, Imazato S, Smith AJ. Inflammation-regeneration interplay in the dentine-pulp complex. J Dent. 2010;38(9):687–697. | ||
Nikolakis G, Join-Lambert O, Karagiannidis I, Guet-Revillet H, Zouboulis CC, Nassif A. Bacteriology of hidradenitis suppurativa/acne inversa: a review. J Am Acad Dermatol. 2015;73(5 Suppl 1):S12–S18. | ||
Antiga E, Verdelli A, Bonciani V, Caproni M, Fabbri P. Acne: a new model of immune-mediated chronic inflammatory skin disease. G Ital Dermatol Venereol. 2015;150(2):247–254. | ||
Eichenfield LF, Del Rosso JQ, Mancini AJ, et al. Evolving perspectives on the etiology and pathogenesis of acne vulgaris. J Drugs Dermatol. 2015;14(3):263–272. | ||
Gilroy DW, Lawrence T, Peretti M, Rossi AG. Inflammatory resolution: new opportunities for drug discovery. Nat Rev Drug Discov. 2004;3(5):401–416. | ||
Benson KF, Newman RA, Jensen GS. Antioxidant, anti-inflammatory, anti-apoptotic, and skin regenerative properties of an Aloe vera-based extract of Nerium oleander leaves (Nae-8®). Clin Cosmet Investig Dermatol. 2015;8:239–248. | ||
Ahlborn G, Sheldon BW. Identifying the components in eggshell membrane responsible for reducing the heat resistance of bacterial pathogens. J Food Prot. 2006;69(4):729–738. | ||
Mageed AM, Isobe N, Yoshimura Y. Immunolocalization of avian β-defensins in the hen oviduct and their changes in the uterus during egg formation. Reproduction. 2009;138(6):971–978. | ||
Makkar S, Liyanage R, Kannan L, Packialakshmi B, Lay JO Jr, Rath NC. Chicken egg shell membrane associated proteins and peptides. J Agric Food Chem. 2015;63(44):9888–9898. | ||
Kaweewong K, Garnjanagoonchorn W, Jirapakkul W, Roytrakul S. Solubilization and identification of hen eggshell membrane proteins during different times of chicken embryo development using the proteomic approach. Protein J. 2013;32(4):297–308. | ||
Cordeiro CM, Esmaili H, Ansah G, Hincke MT. Ovocalyxin-36 is a pattern recognition protein in chicken eggshell membranes. PLoS One. 2013;8(12):e84112. | ||
Kovacs-Nolan J, Cordeiro C, Young D, Mine Y, Hincke M. Ovocalyxin-36 is an effector protein modulating the production of proinflammatory mediators. Vet Immunol Immunopathol. 2014;160(1–2):1–11. | ||
Mogosanu GD, Grumezescu AM. Natural and synthetic polymers for wounds and burns dressing. Int J Pharm. 2014;463(2):127–136. | ||
Ohto-Fujita E, Konno T, Shimizu M, et al. Hydrolyzed eggshell membrane immobilized on phosphorylcholine polymer supplies extracellular matrix environment for human dermal fibroblasts. Cell Tissue Res. 2011;345(1):177–190. | ||
Jensen GS, Shah B, Holtz R, Patel A, Lo DC. Reduction of facial wrinkles by hydrolyzed water soluble egg membrane peptides: suggested mechanisms include reduction of free radical stress and support of matrix production by dermal fibroblasts. Clin Cosmet Investig Dermatol. In press 2016. | ||
Wang J, Urbanowicz RA, Tighe PJ, Todd I, Corne JM, Fairclough LC. Differential activation of killer cells in the circulation and the lung: a study of current smoking status and chronic obstructive pulmonary disease (COPD). PLoS One. 2013;8(3):e58556. | ||
Clausen J, Vergeiner B, Enk M, Petzer AL, Gastl G, Gunsilius E. Functional significance of the activation-associated receptors CD25 and CD69 on human NK-cells and NK-like T-cells. Immunobiology. 2003;207(2):85–93. | ||
Lin Q, Fang D, Fang J, et al. Impaired wound healing with defective expression of chemokines and recruitment of myeloid cells in TLR3-deficient mice. J Immunol. 2011;186(6):3710–3717. | ||
Lin ZQ, Kondo T, Ishida Y, Takayasu T, Mukaida N. Essential involvement of IL-6 in the skin wound-healing process as evidenced by delayed wound healing in IL-6-deficient mice. J Leukoc Biol. 2003;73(6):713–721. | ||
Hübner G, Brauchle M, Smola H, Madlener M, Fässler R, Werner S. Differential regulation of pro-inflammatory cytokines during wound healing in normal and glucocorticoid-treated mice. Cytokine. 1996;8(7):548–556. | ||
Nomura I, Goleva E, Howell MD, et al. Cytokine milieu of atopic dermatitis, as compared to psoriasis, skin prevents induction of innate immune response genes. J Immunol. 2003;171(6):3262–3269. | ||
Donetti E, Mastroianni N, Cornaghi L, Arnaboldi F, Landoni F, Prignano F. Modulation of epidermal proliferation and terminal differentiation in a promising ex vivo human skin model mimicking a psoriatic microenvironment. Ital J Anat Embryol. 2015;120 Suppl 1:104. | ||
Kristensen M, Chu CQ, Eedy DJ, Feldmann M, Brennan FM, Breathnach SM. Localization of tumour necrosis factor-alpha (TNF-α) and its receptors in normal and psoriatic skin: epidermal cells express the 55-kD but not the 75-kD TNF receptor. Clin Exp Immunol. 1993;94(2):354–362. | ||
Hanel KH, Cornelissen C, Luscher B, Baron JM. Cytokines and the skin barrier. Int J Mol Sci. 2013;14(4):6720-6745. | ||
Ye J, Garg A, Calhoun C, Feingold KR, Elias PM, Ghadially R. Alterations in cytokine regulation in aged epidermis: implications for permeability barrier homeostasis and inflammation. I. IL-1 gene family. Exp Dermatol. 2002;11(3):209–216. | ||
Papakonstantinou E, Roth M, Karakiulakis G. Hyaluronic acid: a key molecule in skin aging. Dermatoendocrinol. 2012;4(3):253–258. | ||
Mehta RC, Smith SR, Grove GL,et al. Reduction in facial photodamage by a topical growth factor product. J Drugs Dermatol. 2008;7(9):864–871. | ||
Belperio JA, Keane MP, Arenberg DA, et al. CXC chemokines in angiogenesis. J Leukoc Biol. 2000;68(1):1–8. | ||
Addison CL, Daniel TO, Burdick MD, et al. The CXC chemokine receptor 2, CXCR2, is the putative receptor for ELR+ CXC chemokine-induced angiogenic activity. J Immunol. 2000;165(9):5269–5277. | ||
Takahashi T, Kimura Y, Saito R, et al. An in vitro test to screen skin sensitizers using a stable THP-1-derived IL-8 reporter line, THP-G8. Toxicol Sci. 2011;124(2):359–369. | ||
Venkatakrishnan G, Salgia R, Groopman JE. Chemokine receptors CXCR1/2 activate mitogen-activated protein kinase via the epidermal growth factor receptor in ovarian cancer cells. J Biol Chem. 2000;275(10):6868–6875. | ||
El All HS, Shoukry NS, El Maged RA, Ayada MM. Immunohistochemical expression of IL8 in skin biopsies from patients with inflammatory acne vulgaris. Diagn Pathol. 2007;2:4. | ||
Hasegawa M, Higashi K, Matsushita T, et al. Dermokine inhibits ELR(+) CXC chemokine expression and delays early skin wound healing. J Dermatol Sci. 2013;70(1):34–41. | ||
Hasegawa M, Higashi K, Yokoyama C, et al. Altered expression of dermokine in skin disorders. J Eur Acad Dermatol Venereol. 2013;27(7):867–875. | ||
Ploeger DT, Hosper NA, Schipper M, Koerts JA, de Rond S, Bank RA. Cell plasticity in wound healing: paracrine factors of M1/M2 polarized macrophages influence the phenotypical state of dermal fibroblasts. Cell Commun Signal. 2013;11(1):29. | ||
Almine JF, Wise SG, Hiob M, et al. Elastin sequences trigger transient proinflammatory responses by human dermal fibroblasts. FASEB J. 2013;27(9):3455–3465. | ||
Gaspar K, Kukova G, Bunemann E, et al. The chemokine receptor CCR3 participates in tissue remodeling during atopic skin inflammation. J Dermatol Sci. 2013;71(1):12–21. | ||
Brauchle M, Angermeyer K, Hübner G, Werner S. Large induction of keratinocyte growth factor expression by serum growth factors and pro-inflammatory cytokines in cultured fibroblasts. Oncogene. 1994;9(11):3199–3204. | ||
Pillai S, Oresajo C, Hayward J. Ultraviolet radiation and skin aging: roles of reactive oxygen species, inflammation and protease activation, and strategies for prevention of inflammation-induced matrix degradation – a review. Int J Cosmet Sci. 2005;27(1):17–34. |
 © 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.