Back to Journals » Clinical Ophthalmology » Volume 14
Vitreous Reflux Frequency and Intraocular Pressure After First-Time Intravitreal Aflibercept Injections: Comparison of 30- and 32-Gauge Needles
Received 22 December 2019
Accepted for publication 19 February 2020
Published 3 March 2020 Volume 2020:14 Pages 625—634
DOI https://doi.org/10.2147/OPTH.S243370
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Tetsuya Muto, Shigeki Machida
Department of Ophthalmology, Dokkyo Medical University Saitama Medical Center, Koshigaya, Japan
Correspondence: Tetsuya Muto
Department of Ophthalmology, Dokkyo Medical UniversitySaitama Medical Center, 2-1-50 Minamikoshiagya, Koshigaya 343-8555, Japan
Tel +81 48 965 1111
Fax +81 48 965 1127
Email [email protected]
Purpose: To compare the effects of 30-gauge versus 32-gauge needles on vitreous reflux (VR) frequency and intraocular pressure (IOP) following first-time intravitreal aflibercept injections.
Materials and Methods: Overall, 116 patients (116 eyes) who received intravitreal injections using 30-gauge needles and 104 patients (104 eyes) who received the same injection using 32-gauge needles were reviewed. The medical records of 116 patients who each received an intravitreal injection using a 30-gauge needle (median age: 67.5 ± 13.9 years) and 104 patients who each received the same injection using a 32-gauge needle (median age: 66. 3 ± 10.6 years) from January 2015 to June 2019 were compared.
Results: No significant difference in the frequency of VR was observed between patients injected using 30-gauge needles (38/116) and patients injected using 32-gauge needles (31/104, P = 0.64). There were no significant differences in the VR rates of patients with phakic and pseudophakic eyes between those injected using 30-gauge (P = 0.94) or 32-gauge needles (P = 0.77). Axial length did not significantly differ between patients with and without VR when injected using 30-gauge (P = 0.89) and with 32-gauge needles (P = 0.69). IOP immediately after injection was significantly higher in patients injected using 30-gauge needles than in patients injected using 32-gauge needles (P < 0.01).
Conclusion: VR frequency was not correlated with needle size, lens status, or axial length. Patients receiving injections using 30-gauge needles had higher IOP immediately after intravitreal injection.
Keywords: aflibercept, intravitreal injection, vitreous reflux, intraocular pressure
Introduction
Intravitreal injection is one of the treatment methods in ophthalmology. The effectiveness of the intravitreal injection of an anti–vascular endothelial growth factor (anti–VEGF) antibody has been recognized as essential drugs for treatment of various posterior segment diseases lately, and the use of this treatment is expected to increase. However, a severe complication of intravitreal injection is infectious endophthalmitis. Chen et al reported that vitreous reflux (VR) might be a cause of endophthalmitis after intravitreal injection.1
Occasionally, ophthalmologists encounter VR after intravitreal injection in practice. This VR resembles a subconjuctival bleb. Pang et al reported that the rate of VR after the intravitreal injection of ranibizumab and aflibercept was 53% when a 30-gauge needle was used and 13% when a 32-gauge needle was used.2 Usman Saeed et al reported that the rate of VR after the intravitreal injection of ranibizumab and bevacizumab was 31.6%.3 VR may affect the total amount or total concentration of anti–VEGF antibodies after intravitreal injection.
Another frequent complication of intravitreal injection is a significant short-term elevation of intraocular pressure (IOP). IOP is affected by many factors, but VR is the most important determinant of IOP elevation after intravitreal injection.2,4–7 Some authors have concerns that a momentary IOP elevation may further damage the ganglion cells in the optic nerve, especially in patients who suffer from glaucoma or optic nerve damage.7 Pang et al reported that IOP immediately after intravitreal injection was significantly higher when a 32-gauge needle was used as opposed to when a 30-gauge needle was used (41.4 vs 30.8 mmHg, respectively).2 Similarly, Sasajima et al reported that IOP immediately after intravitreal injection was significantly higher when a 34-gauge needle was used than when a 30-gauge needle was used (42.7 vs 38.8 mmHg, respectively).8 Overall, smaller-gauge needles may cause elevated IOP immediately after intravitreal injection.
Multiple intravitreal injections may affect the rate of VR. Uyar et al reported that VR and its amount decreased with increasing total number of intravitreal injections.4 To the best of our knowledge, there are no reports about VR associated with first-time intravitreal injection of aflibercept (Eylea®; Regeneron, Tarrytown, NY, USA) only. The aim of this study is to investigate how VR rates differ when 30- and 32-gauge needles are used to administer first-time intravitreal aflibercept injections. We also investigated the effect of axial length and lens status on VR frequency and IOP.
Materials and Methods
Written informed consent was obtained from all patients. The study adhered to the tenets of the Declaration of Helsinki, and its protocol was approved by the Institutional Review Board at Dokkyo Medical University Saitama Medical Center. The patients of one physician (T.M.) who received intravitreal injections of aflibercept (2 mg/0.05 mL) for the first time were included in this study. The immediate preinjection IOP, immediate postinjection IOP, and 24 h postinjection IOP of each patient were measured by using an icare PRO® (Tiolat, Helsinki, Finland) in sitting position. Immediate postinjection IOP was consistently measured within one minute after the intravitreal injection. VR was the flow from the vitreous body through the punctured conjunctiva and sclera layers, and the bleb formation due to the conjunctiva closure while the sclera hole remains open. Immediate postinjection VR from the injection site was assessed by the injecting surgeon. The axial length of all patients was measured by using an IOLMaster 700 (Carl Zeiss Meditec AG, Jena, Germany). Patients who used anti-glaucoma eye solutions, had a past history of intravitreal injection or vitrectomy, or had undergone glaucoma surgery were excluded from this study. Pseudophakia patients who had undergone uncomplicated cataract surgery >3 months previously, had a past history of retinal photocoagulation, or had undergone capsulotomy were included in this study. None of the patients had used a Honan IOP reducer before receiving the intravitreal injection.
A 4% lidocaine solution (Xylocaine® solution 4%; Aspen Japan, Tokyo, Japan) was topically applied to anesthetize the conjunctiva. The eyelids and ocular surface were disinfected with a 0.027% iodine solution (PA·IODO® Ophthalmic and Eye washing solution; Nitten Pharmaceutical Co., Ltd., Nagoya, Japan). In an operating room, aflibercept was injected into either the superotemporal or superonasal quadrant via the pars plana and into the vitreous cavity 3–4 mm posterior to the limbus by using a 30-gauge needle (Dentronics 30 G®; Dentronics, Tokyo, Japan) or a 32-gauge needle (Dentronics 32 G; Dentronics, Tokyo, Japan). The outside diameter of the 30-gauge needle was 0.30 mm and that of the 32-gauge needle was 0.26 mm, while the inner diameter of the 30-gauge needle was 0.12 mm and that of the 32-gauge needle was 0.08 mm. Both the 30 and 32-gauge needles were 12 mm long. Postinjection light perception was then assessed. No eyes received anterior chamber paracenteses. A topical 0.5% levofloxacin solution (0.5% Cravit® ophthalmic solution; Santen Pharmaceutical, Osaka, Japan) was applied four times daily for three days before and after each intravitreal injection.
All data were expressed as the mean ± SD. The differences in the variables corresponding to the use of 30-gauge needles and 32-gauge needles were compared by performing unpaired t-tests. The discrete variables between the use of 30-gauge needles and 32- gauge needles were compared by performing a chi-square test or Fisher’s exact probability test. The statistical analysis was performed using StatMate version V for Macintosh (ATMS, Tokyo, Japan). A P value of <0.05 was indicated to be indicative of statistical significance.
Results
Eyes of 220 patients with neovascular age-related macular degeneration, cystoid macular edema due to retinal vein occlusion, or diabetic macular edema were studied. One hundred and sixteen eyes received intravitreal aflibercept injections with 30-gauge needles and 104 eyes received injections with 32-gauge needles.
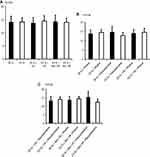
No significant differences in age, axial length, lens status, the axial length of pseudophakic eyes, or the axial length of phakic eyes were observed in the eyes before the intravitreal injections were administered (Table 1). There were no significant differences pertaining to preinjection IOP between all patients injected using 30-gauge needles (15.6 ± 4.2 mmHg) and those injected using 32-gauge needles (15.5 ± 2.9 mmHg; P = 0.77). More specifically, there were no significant differences in preinjection IOP among patients with VR injected using 30-gauge needles (15.5 ± 4.5 mmHg) and those injected using 32-gauge needles (15.5 ± 3.4 mmHg; P = 0.96), while there were also no significant differences in preinjection IOP among patients without VR injected using 30-gauge needles (15.6 ± 4.1 mmHg) and those injected using 32-gauge needles (15.5 ± 2.6 mmHg; P = 0.75) (Figure 1A). Further, there were no significant differences in preinjection IOP among phakic patients injected using 30-gauge needles (15.8 ± 3.9 mmHg) and those injected using 32-gauge needles (15.7 ± 2.8 mmHg; P = 0.79). There were no significant differences in preinjection IOP among pseudophakic patients injected using 30-gauge needles (15.1 ± 5.0 mmHg) and those injected using 32-gauge needles (14.8 ± 2.9 mmHg; P = 0.76). In addition, there were no significant variations in preinjection IOP among phakic patients with VR injected using 30-gauge needles (16.0 ± 3.9 mmHg) and those injected using 32-gauge needles (15.6 ± 3.7 mmHg; P = 0.75) (Figure 1B). There were also no significant differences in preinjection IOP among pseudophakic patients with VR injected using 30-gauge needles (14.5 ± 6.3 mmHg) and those injected using 32-gauge needles (15.0 ± 2.1 mmHg; P = 0.82). No significant differences were observed in preinjection IOP among pseudophakic patients without VR injected using 30-gauge needles (15.7 ± 4.0 mmHg) and those injected using 32-gauge needles (15.7 ± 2.4 mmHg; P = 0.94). There were no significant differences apparent in preinjection IOP among pseudophakic patients without VR injected using 30-gauge needles (15.4 ± 4.5 mmHg) and in those injected using 32-gauge needles (14.7 ± 3.2 mmHg; P = 0.58) (Figure 1C). Although none of the patients who received an injection with a 30-gauge needle had undergone capsulotomy, one patient injected with a 32-gauge needle had undergone capsulotomy.
 |
Table 1 Comparison of Eyes Prior to Receiving Intravitreal Injections Using Either 30-Gauge or 32-Gauge Needles |
No significant differences in VR, VR in pseudophakia, VR in phakia, the axial length with VR eyes, or the axial length without VR eyes were observed in the eyes after the intravitreal injections were administered (Table 2). The IOP values recorded in the total population immediately after intravitreal aflibercept injection (53.8 ± 14.7 mmHg vs 50.0 ± 10.0 mmHg; P < 0.01), immediately after intravitreal aflibercept injection in eyes without VR (60.0 ± 10.6 mmHg vs 51.0 ± 10.0 mmHg; P < 0.001), and immediately after intravitreal aflibercept injection in pseudophakic eyes (56.2 ± 15.0 mmHg vs 45.1 ± 9.2 mmHg; P < 0.01) were significantly higher among patients injected using 30-gauge needles than those injected using 32-gauge needles. There were no significant differences reported in IOP values recorded immediately after intravitreal aflibercept injection among patients with VR injected using 30-gauge needles (40.3 ± 13.1 mmHg) and those injected using 32-gauge needles (44.2 ± 8.5 mmHg; P = 0.14) (Figure 2A). Meanwhile, there were also no significant differences in IOP values immediately after intravitreal aflibercept injection in phakic patients injected using 30-gauge needles (52.9 ± 14.5 mmHg) and those injected using 32-gauge needles (50.0 ± 10.0 mmHg; P = 0.14). There were no significant differences in IOP values noted immediately after intravitreal aflibercept injection among phakic patients with VR injected using 30-gauge needles (39.9 ± 13.6 mmHg) and those injected using 32-gauge needles (44.4 ± 8.3 mmHg; P = 0.14) (Figure 2B). There were also no significant differences in IOP values immediately after intravitreal aflibercept injection among pseudophakic patients with VR injected using 30-gauge needles (44.1 ± 13.5 mmHg) and those injected using 32-gauge needles (43.3 ± 10.0 mmHg; P = 0.90) (Figure 2C).
 |
Table 2 Comparison of Eyes After Receiving Intravitreal Injections Using Either 30-Gauge or 32-Gauge Needles |
There were no significant differences in IOP values at 24 h postinjection among all patients injected using 30-gauge needles (14.1 ± 4.5 mmHg) and those injected using 32-gauge needles (14.1 ± 3.4 mmHg; P = 0.99). Further, there were no significant differences in IOP values at 24 h postinjection in patients with VR injected using 30-gauge needles (13.8 ± 4.1 mmHg) and those injected using 32-gauge needles (14.5 ± 4.0 mmHg; P = 0.44). Similarly, there were no significant differences in IOP values at 24 h postinjection in patients without VR injected using 30-gauge needles (14.3 ± 4.8 mmHg) and those injected using 32-gauge needles (14.0 ± 3.1 mmHg; P = 0.67) (Figure 3A). Separately, there were no significant differences in IOP values at 24 h postinjection IOP in phakic patients injected using 30-gauge needles (13.9 ± 3.8 mmHg) and those injected using 32-gauge needles (14.5 ± 3.2 mmHg; P = 0.29) as well as no significant differences in IOP values at 24 h postinjection in pseudophakic patients injected using 30-gauge needles (14.7 ± 6.1 mmHg) and those injected using 32-gauge needles (12.8 ± 3.5 mmHg; P = 0.15). Also, there were no significant differences in IOP values at 24 h postinjection in phakic patients with VR injected using 30-gauge needles (14.1 ± 3.9 mmHg) and those injected using 32-gauge needles (14.6 ± 4.2 mmHg; P = 0.63) (Figure 3B) and there were no significant differences in IOP values at 24 h postinjection in pseudophakic patients with VR injected using 30-gauge needles (13.3 ± 4.8 mmHg) and those injected using 32-gauge needles (14.0 ± 3.1 mmHg; P = 0.98). There were no significant differences in IOP values at 24 h postinjection IOP phakic patients without VR injected using 30-gauge needles (13.8 ± 3.8 mmHg) and those injected using 32-gauge needles (14.4 ± 2.8 mmHg; P = 0.32). Lastly, there were no significant differences in IOP values at 24 h postinjection in pseudophakic patients without VR injected using 30-gauge needles (15.4 ± 6.7 mmHg) and those injected using 32-gauge needles (12.4 ± 3.6 mmHg; P = 0.084) (Figure 3C).
The patients who received intravitreal aflibercept injections using 30-gauge needles were divided into two groups: patients with and without VR. The IOP values immediately after intravitreal aflibercept injection (P < 0.001) and immediately after intravitreal aflibercept injection in both pseudophakic (P < 0.001) and phakic eyes (P < 0.001) were significantly higher in patients without VR than in those with VR. No significant differences in other factors between the groups were observed (Table 3).
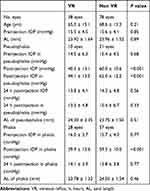 |
Table 3 Comparison of Eyes’ Vitreous Reflux and Non-Vitreous Reflux Before and After Receiving Intravitreal Injections Using 30-Gauge Needles |
The patients who received intravitreal aflibercept injections using 32-gauge needles were divided into two groups: patients with and without VR. The IOP values immediately after intravitreal aflibercept injection (P < 0.01) and immediately after intravitreal aflibercept injection in phakic eyes (P < 0.001) were significantly higher in patients without VR. No significant differences were observed in other factors (Table 4).
 |
Table 4 Comparison of Eyes’ Vitreous Reflux and Non-Vitreous Reflux Before and After Receiving Intravitreal Injections Using 32-Gauge Needles |
All patient who received intravitreal aflibercept injections were divided into two groups: those with pseudophakic eyes and those with phakic eyes. No significant differences were observed in the immediate preinjection IOP, immediate postinjection IOP, or 24 h postinjection IOP values of patients injected using 30-gauge needles. Although no significant differences were observed in the immediate preinjection IOP values, we found that when a 32-gauge needle was used, the immediate postinjection IOP and 24 h postinjection IOP values were significantly lower in patients with pseudophakic eyes than in those with phakic eyes (Table 5).
 |
Table 5 Comparison of Vitreous Reflux and Intraocular Pressure of Pseudophakic Eyes with Those of Phakic Eyes in the 30-Gauge and 32-Gauge Needle Groups |
Discussion
Previous studies have reported that smaller-gauge needles had reduced frequency and total amount of VR.2,8,9 Thus, we initially expected 32-gauge needles to cause less VR. However, our results showed that there was no significant difference in the frequency of VR when a 30-gauge needle was used versus when a 32-gague needle was used. Pang et al and Sasajima et al injected both ranibizumab and aflibercept,2,8 while Lorenz et al injected bevacizumab.9 In their research, they did not concern about number of prior injections.2,8,9 Because we included first-time intravitreal aflibercept injection cases only, our results are different from those of previous studies.2,8,9 A simple comparison would not be ideal with different drugs. Uyar et al hypothesized that recurrent injections in the same quadrant may decrease the total amount of VR because of recurrent scleral trauma.4 We hypothesized that recurrent injections would cause several VR events and consequently reduce the liquefied vitreous body. First-time intravitreal injection patients have a sufficient liquefied vitreous body, so VR occurs easily. However, needle size did not correlate with the frequency of VR in such cases. In a previous cadaveric study, more VR was found after using 32-gauge needles than after using 30-gauge needles.10
Additionally, immediate postinjection IOP was higher when 30-gauge needles were used than when 32-gauge needles were used, which were contrary to our prediction. We guessed the difference in the total amount of injected aflibercept to be a reason for this finding. Because the inner diameter of a 32-gauge needle is small and aflibercept is sticky, the amount of aflibercept that attached to the inner wall of the needle might be larger than that in the 30-gauge needle. Although there was no significant difference, the VR rate in pseudophakic cases was slightly higher when 30-gauge needles were used than when 32-gauge needles were used (31/116 vs 22/104, respectively). The difference in immediate postinjection IOP in pseudophakic eyes (56.2 ± 15.0 vs 45.1 ± 9.2 mmHg, respectively) might have directly affected this result. Although Uyar et al reported that VR and its amount were important determinants for immediate postinjection IOP,4 we only measured the VR rate. In the near future, we will try to determine why immediate postinjection IOP was higher when 30-gauge needles were used than when using 32-gauge needles were used. Gómez-Mariscal et al reported that the repeated intravitreal injection of anti–VEGF antibodies could lead to irreversible changes in optic nerve head structures.11 Furthermore, transient IOP spikes after intravitreal injection may lead to additional adverse effects.12,13 Our results demonstrated that immediate postinjection IOP was low in patients with VR. However, the occurrence of VR during intravitreal injection might decrease drug efficacy and increase infectious endophthalmitis.
Because long axial lengths are associated with a large vitreous volume, the VR rate after intravitreal injection may be low. However, Pang et al reported that axial length was not correlated with VR.2 Additionally, pseudophakic eyes appear to have a larger intravitreal volume than that of phakic eyes. Ohtsuka et al reported that the VR rate was significantly higher in phakic eyes than in pseudophakic eyes.14 Kerimoglu et al reported that IOP decreased to safe levels more quickly in pseudophakic eyes than in phakic eyes after the intravitreal injection of triamcinolone acetonide, but they excluded VR cases.15 However, Usman Saeed et al3 and Uyar et al4 reported that lens status was not correlated with VR. No significant correlation was observed between VR and axial length or between VR and lens status when using either a 30-gauge or 32-gauge needle. Ohtsuka et al reported that the VR rate after first-time intravitreal ranibizumab injections were administered using a 30-gauge needle was significantly higher in phakic eyes than in pseudophakic eyes.14 Although their study used first-time intravitreal injection patients, they injected ranibizumab, and their sample totaled 52 cases,14 so comparing our results with theirs are not ideal. Our immediate postinjection IOP and 24 h postinjection IOP values were significantly lower in pseudophakic eyes than in phakic eyes only when 32-gauge needles were used. Demirel et al reported that patients with pseudophakic eyes had lower mean IOP values than petients with phakic eyes 30 min after intravitreal ranibizumab injections were administered using 30-gauge needles.16 However, Uyar et al reported that lens status did not affect immediate postinjection IOP when using 30-gauge needles.4 Similar results were obtained in a study reported by Arikan et al.17 They also reported that lens status did not affect immediate postinjection IOP.17 They used 26-gauge needles to administer intravitreal injections of triamcinolone acetonide and 30-gauge needles to administer ranibizumab injections.17
Our study has several limitations. The diameter and height of the conjunctival bleb, which might have affected the exact volume of VR, were not measured. We also could not determine why the immediate postinjection IOP was higher when using the 30-gauge needles than when using 32-gauge needles. Because pseudophakia and phakia differ in the degree of angle opening, we investigated both pseudophakia and phakia, respectively. However, our study has certain advantages. Because we limited our study to first-time intravitreal aflibercept injections, and a single surgeon administered all injections, we could more easily compare the effect of needle size.
Conclusion
In summary, we found that the frequency of VR after first-time intravitreal aflibercept injections did not correlate with needle size. Furthermore, VR did not correlate with lens status or axial length. Immediate postinjection IOP was higher when using a 30-gauge needle than when using a 32-gauge needle. When a 32-gauge needle was used for intravitreal injection, immediate postinjection IOP and 24 h postinjection IOP were higher in patients with phakic eyes than in patients with pseudophakic eyes. Although both 30- and 32-gauge needles may be comparable means for intravitreal injection with regard to VR, use of a 32-gauge needle may be better for managing IOP.
Disclosure
The authors report no conflicts of interest in this study.
References
1. Chen SD, Mohammed Q, Bowling B, et al. Vitreous wick syndrome–a potential cause of endophthalmitis after intravitreal injection of triamcinolone through the pars plana. Am J Ophthalmol. 2004;137:1159–1160. doi:10.1016/j.ajo.2004.01.028
2. Pang CE, Mrejen S, Hoang QV, et al. Association between needle size, postinjection reflux, and intraocular pressure spikes after intravitreal injections. Retina. 2015;35:1401–1406. doi:10.1097/IAE.0000000000000476
3. Usman Saeed M, Batra R, Qureshi F, et al. Reflux of drug during intra-vitreal anti-VEGF therapies. Semin Ophthalmol. 2011;26:357–360. doi:10.3109/08820538.2011.588648
4. Uyar E, Ulas F, Sahin S, et al. Major factors affecting intraocular pressure spike after intravitreal ranibizumab injection: vitreousreflux and its amount. Eur J Ophthalmol. 2019;29:361–367. doi:10.1177/1120672119836613
5. Hoang QV, Tsuang AJ, Gelman R, et al. Clinical predictors of sustained intraocular pressure elevation due to intravitreal antivascular endothelial growth factor therapy. Retina. 2013;33:179–187. doi:10.1097/IAE.0b013e318261a6f7
6. Benz MS, Albini TA, Holz ER, et al. Short-term course of intraocular pressure after intravitreal injection of triamcinolone acetonide. Ophthalmology. 2006;113:1174–1178. doi:10.1016/j.ophtha.2005.10.061
7. Bakri SJ, Pulido JS, McCannel CA, et al. Immediate intraocular pressure changes following intravitreal injections of triamcinolone, pegaptanib, and bevacizumab. Eye. 2009;23:181–185. doi:10.1038/sj.eye.6702938
8. Sasajima H, Tsuboi K, Ono H, et al. A randomized trial of a short 34-gauge needle for intravitreal injections. Ophthalmology. 2018;125:947–948. doi:10.1016/j.ophtha.2018.01.038
9. Lorenz K, Zwiener I, Mirshahi A. Subconjunctival reflux and need for paracentesis after intravitreal injection of 0.1 mL bevacizumab: comparison between 27-gauge and 30-gauge needle. Graefes Arch Clin Exp Ophthalmol. 2010;248:1573–1577. doi:10.1007/s00417-010-1490-y
10. Hubschman JP, Coffee RE, Bourges JL, et al. Experimental model of intravitreal injection techniques. Retina. 2010;30:167–173. doi:10.1097/IAE.0b013e3181b094cf
11. Gómez-Mariscal M, Puerto B, Muñoz-Negrete FJ, et al. Acute and chronic nerve head biomechanics and intraocular pressure changes in patients receiving multiple intravitreal injections of anti-VEGF. Graefes Arch Clin Exp Ophthalmol. 2019;257:2221–2231. doi:10.1007/s00417-019-04354-7
12. Lemos V, Cabugueira A, Noronha M, et al. Intraocular pressure in eyes receiving intravitreal antivascular endothelial growth factor injections. Ophthalmologica. 2015;233:162–168. doi:10.1159/000369478
13. Brodie FL, Ruggiero J, Ghodasra DH, et al. Volume and composition of reflux after intravitreal injection. Retina. 2014;34:1473–1476. doi:10.1097/IAE.0000000000000098
14. Ohtsuka Y, Hashida M, Yamamoto Y, et al. Frequency of vitreous reflux in intravitreal injection of anti-VEGF antibody. Atarashii Ganka. 2011;28:727–729.
15. Kerimoglu H, Ozturk BT, Bozkurt B, et al. Does lens status affect the course of early intraocular pressure and anterior chamber changes after intravitreal injection? Acta Ophthalmol. 2011;89:138–142. doi:10.1111/j.1755-3768.2009.01656.x
16. Demirel S, Yanik O, Batioglu F, et al. Intraocular pressure changes related to intravitreal injections of ranibizumab: analysis of pseudophakia and glaucoma subgroup. Int Ophthalmol. 2015;35:541–547. doi:10.1007/s10792-014-9981-1
17. Arikan G, Osman Saatci A, Oner FH. Immediate intraocular pressure rise after intravitreal injection of ranibizumab and two doses of triamcinolone acetonide. Int J Ophthalmol. 2011;4:402–405. doi:10.3980/j.issn.2222-3959.2011.04.16
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.