Back to Journals » Clinical Ophthalmology » Volume 9
Vitrectomy and internal limiting membrane peeling for macular folds secondary to hypotony in myopes
Authors Nadal J, Carreras E, Canut MI, Barraquer RI
Received 12 April 2014
Accepted for publication 12 December 2014
Published 15 May 2015 Volume 2015:9 Pages 859—864
DOI https://doi.org/10.2147/OPTH.S66074
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Scott Fraser
Jeroni Nadal,1–3 Elisa Carreras,2,3 Maria Isabel Canut,1–3 Rafael I Barraquer1–3
1Centro de Oftalmologia Barraquer, 2Universitat Autònoma de Barcelona, 3Instituto Barraquer, Barcelona, Spain
Background: Hypotony maculopathy (HM) changes may persist, and visual acuity remains poor, despite normalization of intraocular pressure (IOP). The aim of this study was to evaluate the visual and anatomical results of pars plana vitrectomy (PPV), internal limiting membrane (ILM) peeling, and 20% SF6 gas tamponade in five myopic patients with HM.
Methods: This retrospective interventional study was conducted at the Barraquer Center of Ophthalmology, a tertiary care center in Barcelona, Spain, and included five eyes from five consecutive patients (aged 55.4±13.1 years) with HM caused by different conditions. All the patients were treated with 23-gauge PPV, ILM peeling, and 20% SF6 gas tamponade. Preoperative and postoperative evaluation was performed using anterior and posterior biomicroscopy and best corrected visual acuity (BCVA) by logMAR charts.
Results: Before surgery, median spherical equivalent was -13.1 (range -7, -19) diopters of myopia. Preoperatively, four cases presented IOP <6.5 mmHg for 3 (range 2–8) weeks. In three of these four cases, IOP >6.5 mmHg was achieved over 16 (range 16–28) weeks, without resolution of HM; increased IOP was not achieved in the remaining case treated 2 weeks after diagnosis of HM. One case presented IOP >6.5 mmHg with HM for 28 weeks before surgery. Preoperative BCVA was 0.7 (range 0.26–2.3) logMAR, and 0.6 (range 0.3–0.7) logMAR and 0.5 (range 0.2–1) logMAR, respectively, at 4 and 12 months after surgery. There was no statistically significant difference between preoperative and postoperative BCVA. Hyperpigmentation lines in the macular area were observed in three cases with hypotony. These lines progressed after surgery despite resolution of the retinal folds in the three cases, and BCVA decreased in parallel in two of these cases.
Conclusion: PPV with ILM peeling followed by gas tamponade is a good alternative for the treatment of HM in myopic patients. However, persistent choroidal folds may compromise BCVA. We therefore recommend initiating treatment as early as possible.
Keywords: hypotony maculopathy, retinal pigment epithelial hyperpigmentation, macular folds, retinal pigment epithelium, vitrectomy
Introduction
Ocular hypotony is defined as a reduction in intraocular pressure (IOP).1 Pederson2 differentiated “statistical” from “clinical” hypotony; the former refers to IOP <6.5 mmHg, which is more than 3 standard deviations below the mean, whereas clinical hypotony is IOP that causes decreased visual acuity. Sustained low IOP can trigger structural and functional changes in the eyeball, including hypotony maculopathy (HM). This was first described in 1954 by Dellaporta,3 who reported four cases that he classified as “papilloedema ex vacuo”. These patients had hypotony, papilledema, vascular tortuosity, and chorioretinal folds. The term “macular hypotony” was introduced in 1972 by Gass,4 who described ten cases with similar funduscopic alterations being responsible for visual loss.
HM is usually preceded by intraocular surgery, most commonly anti-glaucoma filtration, or penetrating trauma, leading to abnormally low IOP. Its incidence is reported as 1.3%–18%.5,6 In physiopathological terms, reduced IOP causes scleral shrinkage leading to redundancy of choroidal and retinal tissue with radial macular fold formation, optic disc edema, venous tortuosity, and a consequent decrease in visual acuity.4 Fluorescence angiography reveals alternating bands of hypofluorescence and hyperfluorescence consistent with the folds. Ocular coherence tomography (OCT) can detect subtle changes showing an irregular contour of the retina and choroid.7
First-line treatment for HM involves identifying the causes and implementing measures to increase IOP to reverse the scleral shrinkage. Sometimes, despite normalization of IOP, changes in the posterior pole may persist and visual acuity remains poor.8–10 Our approach in the present study was based on the hypothesis that pars plana vitrectomy (PPV) with internal limiting membrane (ILM) peeling and gas tamponade can improve visual acuity and fundus findings in patients with unresolved HM.
Patients and methods
We performed a retrospective interventional study of five eyes from five consecutive patients (four males, one female) aged 55.4±13.1 (range 32–62) years and undergoing vitreoretinal surgical treatment of HM. The study was carried out at the Barraquer Ophthalmology Center, Barcelona, Spain, between January 2008 and February 2011. The primary indication for PPV was unresolved HM, despite measures to normalize IOP (successful in some cases and unsuccessful in others).
Preoperative evaluation (one week before surgery) included medical history, best corrected visual acuity (BCVA) by logMAR chart scale, refraction, metamorphopsia test, biomicroscopy of anterior and posterior segments, photographs of the posterior pole with a non-mydriatic retinal camera (CR6-47 NM, Canon Inc, Tokyo, Japan), IOP, etiology of hypotony, stage of glaucoma (based on the new International Classification of Diseases, 9th Revision [ICD-9] staging definitions) and preoperative and postoperative OCT (Spectral domain Cirrus HD-OCT and/or time domain Stratus was used).
Surgery was performed by the same surgeon in all cases. Anesthesia was peribulbar. The technique used was 23-gauge complete PPV of the anterior, middle, and posterior segments with ILM peeling of more than 5,000 μm in diameter, staining with Brilliant Blue 0.25 mg/mL, followed by gas tamponade using 20% SF6. Leaking ports and ports with inappropriate tunnel sclerotomy incisions were sutured with 7/0 Vicryl. No positioning or avoidance of the supine position was indicated.
Postoperatively, one drop of tobramycin 3 mg/mL plus dexamethasone 1 mg/mL (Tobradex® Alcon, El Masnou, Barcelona, Spain) was administered four times a day during the first week, then tapered to one drop per week for one month.
Postoperative evaluation included BCVA, a metamorphopsia test, IOP, biomicroscopy of anterior and posterior segments, glaucoma staging (based on the new ICD-9 definitions), photographs of the posterior pole with a non-mydriatic retinal camera (CR6-47NM, Canon) and OCT at 4 and 12 months. Biomicroscopy of the anterior and posterior segment and IOP were also checked on the day of surgery and one week later.
Statistical analysis of BCVA scores was performed using Statistical Package for the Social Sciences software (SPSS Inc, Chicago, IL, USA) with the Wilcoxon test for repeated measures and 95% confidence intervals. Descriptive statistics are shown for each patient and means with interquartile range in Table 1, together with patient characteristics.
Results
Before surgery, all patients were myopic with a median spherical equivalent of −13.1 (range −7, −19). Preoperatively, four cases presented IOP <6.5 mmHg for a median of 3 (range 2–8) weeks. In three of these four cases, IOP >6.5 mmHg was achieved over 16 (range 16–28) weeks without HM resolution; increased IOP was not achieved in the remaining case, who was treated 2 weeks after HM. One case presented IOP >6.5 mmHg with HM for 7 months before surgery. The causes of hypotony were hyperfunctionality secondary to previous filtering surgery (three cases), hyperfunctional filtering bleb secondary to trauma (one case), and cyclodialysis (one case).
Measures to increase IOP (Table 1) were successful in three cases with statistical hypotony, but anatomofunctional changes were not resolved and they showed clinical hypotony (cases 2, 3, and 5). Of the four cases with hypotony due to hyperfiltration, in three cases the trabeculectomy had five scleral flap sutures, so no more were added postoperatively as IOP was normalized with corticosteroid treatment (two patients) or draining the choroidal detachment (one patient). In the remaining case, the trabeculectomy had three scleral flap sutures and IOP was normalized by adding two complementary sutures to the scleral flap. In case 4, statistical hypotony persisted for 2 weeks until vitrectomy was performed. Preoperative OCT showed chorioretinal folds without epiretinal membranes or ILM thickening in any case. All of these cases had posterior vitreous detachment preoperatively.
Before surgery, median BCVA was 0.7 (range 0.26–2.3) logMAR, and postoperatively was 0.6 (range 0.3–0.7) logMAR at 4 months and 0.5 (range 0.2–1) logMAR at 12 months. There was no statistically significant difference between preoperative and postoperative BCVA or between BCVA at 4 and 12 months. All five patients had preoperative metamorphopsia, which was still present (albeit but less intense) at one year after surgery. Median IOP at 12 months after surgery was 12 (range 11–18) mmHg, without the need for hypotensive drops or anti-glaucoma surgery in any of these patients and stable visual fields. The stage of glaucoma persisted as moderate in all cases (based on the new ICD-9 staging definitions). No postoperative complications were observed, and no cases presented hypotony at 24 hours or at follow-up 7 days later.
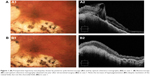
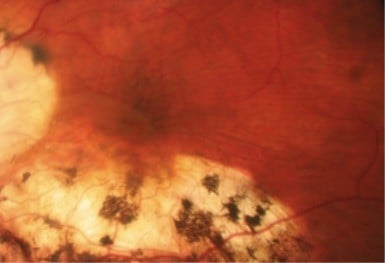
Hyperpigmentation lines were observed in the troughs of folds in the macular area in 3 cases at 30, 32, and 56 weeks after diagnosis of hypotony, corresponding to patients with the most prolonged clinical and statistical hypotony (24–28 weeks). Hyperpigmentation lines in the postoperative period progressed despite resolution of retinal folds, because choroidal folds persisted (Figure 1). This progression occurred in parallel with decreasing BCVA in two cases, while in the remaining patient BCVA remained stable.
Discussion
Ocular hypotony can lead to complications including HM, keratopathy, cataract, choroidal effusion, optic nerve edema, visual loss, and phthisis bulbi. However, many eyes with hypotony do not develop any of these complications.6 Therefore, hypotony per se is not the causal agent but rather a predisposing factor. In our sample, all patients had myopia. This is a known risk factor for development of HM, as the thin sclera in these eyes offers no resistance to scleral collapse during hypotony. In addition, vitreoretinal characteristics in patients with high myopia may hinder determination of funduscopic changes in HM. However, medical history together with IOP and OCT values help to make the diagnosis. Besides this, there are other risk factors, such as topical application of anti-metabolites, young age, primary filtering surgery, and elevated preoperative IOP.6,8
The primary objective before treatment was to identify the etiology of HM in each case and then implement measures to increase the IOP as soon as possible in order to reverse scleral contraction. Various treatments have been described for the management of hyperfiltration due to aqueous humor leakage through the filtration bleb, including contact lenses, simulating bleb fibrosis (using concentrates of fibrinogen or blood, gentamicin, YAG laser therapy, cryotherapy, diathermy, trichloroacetic acid), supplementary sutures, bleb excision, and use of a scleral patch graft.2 Three cases had primary hyperfiltration; one patient (case 1 with IOP >6.5 mmHg) received topical corticosteroid and another (case 3) was treated with supplementary scleral sutures, with good results; and in the patient with primary hyperfiltration plus non-self-limiting choroidal detachment (case 2), drainage via the sclerotomy was performed, but IOP did not normalize until introduction of steroids. The patient with HM secondary to trauma (case 4) received subtenonian corticosteroid therapy in an attempt to reduce inflammation of the ciliary body and restore production of aqueous humor, without success. In our series, the patient with cyclodialysis (case 5) was unsuccessfully treated with subtenonian steroids to raise IOP, so cryocoagulation was then performed with application of intraocular SF6 gas, with good results.
Measures adopted to increase IOP should be chosen according to the etiology of the hypotony. However, the sample size in our series was too small to be able to draw conclusions about the success rate of the measures adopted to increase IOP in each disease.
Macular folds secondary to hypotony are sometimes irreversible despite normalization of IOP and may be secondary to ILM thickening. Duker et al were the first into propose vitreoretinal surgery to treat this structural change.9 They described a case of HM with cataract extraction and PPV with posterior hyaloid peeling (but not ILM peeling) and perfluorocarbon liquid, which resulted in an increase of IOP to 50 mmHg over 10 minutes. The choroidal folds resolved and visual acuity improved, but stellate retinal folds persisted. However, we must take into account the concomitant cataract extraction and the fact that these levels of IOP are not desirable in patients with glaucoma. We believe the reason for the persistent stellate retinal folds in that case was related to the absence of ILM peeling. In accordance with Benson et al11 and Mutoh et al12 who reported excellent visual and functional results, along with others, our surgical technique aimed to eliminate the ILM around the posterior pole in order to achieve relaxation of the retinal surface, freeing it from traction and allowing reapplication of the folds by means of non-expandable gas tamponade using 20% SF6. This treatment is based on the hypothesis that persistent chorioretinal folds can remain irreversible, possibly due to ILM fibrocellular proliferation caused by incorrect retinal positioning, leading to irregular fixation of the retina and choroid.11,13,14 However, fibrocellular proliferation of the ILM was not revealed by preoperative OCT, possibly because it involved a single layer of cells that would be difficult to detect with biomicroscopy or OCT.14 After ILM peeling, the retinal folds disappeared in all cases. On the other hand, although the retinal structures are well preserved after ILM peeling, the vitreoretinal interface may be damaged, thus affecting the final prognosis.15–17 Studies comparing PPV with or without ILM peeling would be necessary to clarify the benefit of peeling in these patients.
Gass described how the retinal pigment epithelium became stacked and compressed in the troughs of the chorioretinal folds, forming lines of hyperpigmentation that persisted despite chorioretinal flattening.4 In our series, three patients with prolonged hypotony (6–7 months) presented such lines of hyperpigmentation in the macula one year before treatment. The hyperpigmentation progressed despite postoperative retinal flattening, probably due to persistent folds at the choroidal level, detected by OCT (Figure 1). The exact time to appearance of these lines is difficult to calculate; in our series, it was between 7.5 and 14 months after appearance of choroidal folds (unresolved with vitrectomy). However, at 12-month follow up, one of the cases with persistent choroidal folds had still not presented macroscopic hyperpigmentation lines. We do not rule out appearance of these lines during a longer follow-up period.
After treatment, both retinal and choroidal folds disappeared in the case with the shortest duration of hypotony <6.5 mmHg (2 weeks), and it was this patient who presented best visual results. In the other cases with a longer duration of hypotony (4.5–7 months), choroidal folds persisted despite the vitrectomy, and it was these patients (with good glaucoma control) who presented the worst visual results. According to Gass, hyperpigmentation leads to a decrease in visual acuity because of impairment of photoreceptors outer segments, which generates residual changes in retinal pigment epithelium.4 In our series, two of the three cases with hyperpigmentation showed decreased BCVA in parallel with progression of hyperpigmentation lines. However, OCT failed to show defects in the photoreceptors due to their masking by the chorioretinal atrophy produced in high myopia (Figure 1).18 For these reasons, we wonder how authors such as Oyakhire et al18 or Delgado et al19 were able to achieve good functional and visual results without describing retinal pigment epithelium changes in the resolution of HM at 5 and 7 years of evolution, respectively. We have in fact observed photoreceptor impairment in a patient not included in the present study: he had retinal folds after retinal detachment surgery, and postoperatively we observed that the hyperpigmentation coincided with the area of atrophy of the union of photoreceptors outer and inner segments, with progressive loss of visual acuity.
Thus, we believe that the main predictors of successful HM surgery are duration of preoperative hypotony and residual changes in the retinal pigment epithelium generated by hyperpigmentation which negatively affect the photoreceptors. For this reason, we believe that macular folds should be treated as soon as possible to improve the visual prognosis, identifying the cause and normalizing IOP. In cases where this approach is unsuccessful, vitreoretinal surgery should be considered, especially before hyperpigmentation lines appear. However, the appropriate time to perform such surgery should be evaluated in a greater number of patients.
In conclusion, macular changes secondary to hypotony may persist despite normalization of IOP. A good treatment option involves surgery to resolve the structural retinal changes with ILM peeling and subsequent non-expandable gas tamponade. However, BCVA may fail to improve in cases with long-standing macular folds due to irreversible changes at the retinal level. We therefore recommend treating HM as early as possible.
Disclosure
The authors report no conflicts of interest in this work, in particular, no financial interest in any of the materials mentioned in this paper.
References
Schubert HD. Postsurgical hypotony: relationship to fistulization, inflammation, chorioretinal lesions, and the vitreous. Surv Ophthalmol. 1996;41:97–125. | ||
Pederson JE. Ocular hypotony. In: Ritch R, Shields MB, Krupin T, editors. The Glaucomas. 2nd ed. St Louis, MO, USA: Mosby; 1996. | ||
Dellaporta A. Fundus changes in postoperative hypotony. Am J Ophthalmol. 1955;40:781–785. | ||
Gass JDM. Hypotony maculopathy. In: Bellows JG, editor. Contemporary Ophthalmology Honouring Sir Stewart Duke-Elder. Baltimore, MD, USA: Williams and Wilkins; 1972. | ||
Suñer IJ, Greenfield DS, Miller MP, Nicolela MT, Palmberg PF. Hypotony maculopathy after filtering surgery with mitomycin C. Ophthalmology. 1997;104:207–216. | ||
Fannin LA, Shchiffman JC, Budenz DL. Risk factor for hypotony maculopathy. Ophthalmology. 2003;110:1185–1191. | ||
Goodkin ML, Grewal DS, Greenfield DS. Three-dimensional high-speed optical coherence tomography for diagnosis of hypotony maculopathy after glaucoma filtration surgery. J Glaucoma. 2010;19:349–355. | ||
Costa VP, Wilson RP, Moster MR, Schmidt CM, Grandham S. Hypotony maculopathy following the use of topical mitomycin C in glaucoma filtration surgery. Ophthalmic Surg. 1993;24:389–394. | ||
Dukers JS, Schuman JS. Successful surgical treatment of hypotony maculopathy following trabeculectomy with topical mitomycin C. Ophthalmic Surg. 1994;25:463–465. | ||
Nuyts RM, Greve EL, Geijssen HC, Langerhorst CT. Treatment of hypotony maculopathy after trabeculectomy with mitomycin C. Am J Ophthalmol. 1994;118:322–331. | ||
Benson SE, Barton K, Gregor ZJ. Vitrectomy for a persisting macular fold in a case of resolved hypotony maculopathy. Am J Ophthalmol. 2004;138:487–489. | ||
Mutoh T, Matsumoto Y, Chikuda M. Four cases of traumatic hypotony maculopathy treated by various methods. Clin Ophthalmol. 2011;5:1223–1226. | ||
Jampel HD, Pasquale LR, Dibernardo C. Hypotony maculopathy following trabeculectomy with mitomycin C. Arch Ophthalmol. 1992;110:1049–1050. | ||
Gandofer A, Rohleder M, Kampik A. Epiretinal pathology of vitreomacular traction syndrome. Br J Ophthalmol. 2002;86:902–909. | ||
Hisatomi T, Notomi S, Tachibana T, et al. Ultrastructural changes of the vitreoretinal interface during long-term follow-up after removal of the internal limiting membrane. Am J Ophthalmol. 2014;158:550–556. e1. | ||
Pichi F, Lembo A, Morara M, et al. Early and late inner retinal changes after inner limiting membrane peeling. Int Ophthalmol. 2014;34:437–446. | ||
Mitamura Y, Ohtsuka K. Relationship of dissociated optic nerve fiber layer appearance to internal limiting membrane peeling. Ophthalmology. 2005;112:1766–1770. | ||
Oyakhire JO, Moroi SE. Clinical and anatomical reversal of long-term hypotony maculopathy. Am J Ophthalmol. 2004;137:953–955. | ||
Delgado MF, Daniels S, Pascal S, Dickens JD. Hypotony maculopathy: improvement of visual acuity after 7 years. Am J Ophthalmol. 2001;132:931–933. | ||
Sayanagi K, Ikuno Y, Soga K, Tano Y. Photoreceptor inner and outer segment defects in myopic foveoschisis. Am J Ophthalmol. 2008;145:902–908. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.