Back to Journals » Clinical Ophthalmology » Volume 13
Visual evoked potentials after panretinal photocoagulation in patients with proliferative diabetic retinopathy
Authors Amini Vishte R, Mirzajani A, Khojasteh H
Received 25 April 2019
Accepted for publication 25 July 2019
Published 28 August 2019 Volume 2019:13 Pages 1635—1640
DOI https://doi.org/10.2147/OPTH.S213448
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Rasoul Amini Vishte,1 Ali Mirzajani,1 Hassan Khojasteh2
1Department of Optometry, School of Rehabilitation Sciences, Iran University of Medical Sciences, Tehran, Iran; 2Department of Ophthalmology, Farabi Eye Research Center, Farabi Eye Hospital, Tehran University of Medical Sciences, Tehran, Iran
Correspondence: Ali Mirzajani
Department of Optometry, School of Rehabilitation Sciences, Iran University of Medical Sciences, No. 24 flat 2, 13th Alley, Behzad Hesari St., Mirdamad Ave., Tehran, Iran
Tel +98 212 290 9608
Fax +98 212 222 0946; +98 212 291 3036
Email [email protected]
Purpose: To evaluate the changes of latency and amplitudes of pattern-reversal visual evoked potentials (PRVEP) in patients with proliferative diabetic retinopathy after panretinal photocoagulation (PRP).
Methods: PRVEP was recorded in 21 eyes of 21 patients with proliferative diabetic retinopathy prior to, 1 week after every sessions of laser therapy and 1.5 months after the final treatment. Results were compared between pre and post laser treatment sessions in the study group and paired t-test was used for statistical analysis.
Results: The P100 amplitude showed a significant difference among all treatment sessions with a decreasing trend in the study group after PRP (P<0.001). Also, P100 latency evaluation showed a significant increase after PRP in the study group in all post-PRP sessions (P<0.05). However, 1.5 months after laser treatment, an increase in amplitudes (P<0.001) and a decrease in latencies (P<0.001) of PRVEP were observed and the magnitudes of the parameters approximately returned to their baseline values.
Conclusion: Although decreasing changes in the amplitude and increasing changes in the latency of PRVEP were observed after laser treatment in proliferative diabetic retinopathy patients, one and a half months after the completion of laser therapy, partial recovery of these parameters values was observed.
Keywords: photocoagulation, visual evoked potential, proliferative diabetic retinopathy
Introduction
According to the World Health Organization, the global prevalence of diabetes among adults over 18 years of age has risen from 4.7% in 1980 to 8.5% in 2014 and by 2030 about 438 million people will be affected by diabetes mellitus; however, diabetes can be treated and its consequences can be prevented or delayed by the use of proper diet, physical activity and regular screening.1,2
Diabetes can have different effects on different parts of the visual system. One of these changes can be visual disturbances due to vascular and metabolic defects that can affect the retina and the visual pathway.3 These abnormalities can cause changes in the ganglionic and preganglionic elements of the retina and macular region. In addition, neural conduction may be delayed along post-retinal central visual pathways.4
Diabetic retinopathy is one of the major complications of diabetes and is the main cause of blindness and visual impairment throughout the world.5 The progression of retinopathy leads to the formation of new vessels in the retina and causes proliferative diabetic retinopathy, so neovascularization occurs in diabetic retinopathy in response to retinal ischemia and new vessels may grow on optical discs or elsewhere. Since the blood vessels are abnormal, they can flow into the retina and affect the patient’s vision. To prevent the growth of abnormal new vessels, bleeding in the retina and other parts of the eye and severe vision loss, a standard treatment method called panretinal photocoagulation (PRP) is used.6 In a 2014 survey, 98% of the retina specialists reported using PRP for initial proliferative diabetic retinopathy management in the absence of diabetic macular edema.7 Also, the usage of intravitreal pharmacologic agents in diabetic macular edema treatment reduces the risk of diabetic retinopathy worsening and increases the chance of improvement.8 Unfortunately, a third of patients have an incomplete response to anti-VEGF therapy, but the best second-line therapy in these patients remains unknown.9 The visual evoked potential is an objective and non-invasive method that expresses a mass bioelectrical response from the visual cortex to a specific visual stimulus, so abnormal visual evoked potential responses observed in diabetic retinopathy indicate a general involvement of the visual system or of one of its composing structures and this test is used to evaluate ocular health from macula to cortex.10,11 Because the visual evoked potential response is mainly due to the macular region, so the changes that occur due to retinopathy and post-PRP in this region will affect the visual evoked potential components. Therefore, the visual evoked potential is a suitable technique for evaluating these changes.
In this study, the amplitude and latency of P100 wave of visual evoked potential were compared between pre- and post-PRP in the study group to find the retinal and visual pathway functional changes due to retinopathy and photocoagulation.
Materials and methods
Subjects
Twenty-one proliferative diabetic retinopathy patients according to guidelines for DR issued by the American Academy of Ophthalmology12 with the mean value of HbA1C of 9.58±0.80 (range from 8.30 to 11.40) and with approximately the same intensity in retinal involvement were selected according to high resolution spectral-domain optical coherence tomography (SD-OCT, Spectralis, Heidelberg Engineering, Heidelberg, Germany) as a study group.
All patients underwent a complete ophthalmological examination including measurement of best-corrected visual acuity in 6 m with a Snellen E chart (LED visual chart projector, LC-13, MEDIZ Inc., City, Korea), slit-lamp biomicroscopy (BM 900, Haag-Streit, Bern, Switzerland) using a Volk 90D lens (Volk Optical, Mentor, Ohio, USA), fundus photography with high-resolution spectral-domain optical coherence tomography (SD-OCT, Spectralis, Heidelberg Engineering, Heidelberg, Germany) and fluorescein angiography (HRA II Heidelberg, Heidelberg, Germany) for evaluating the DR.
Subjects with any history of previous treatments for diabetic retinopathy including surgery and laser therapy, BCVA of 20/200 and worse, any type of significant opacity, any ocular disease other than diabetic retinopathy that may change the visual evoked potential responses, pre- and post-PRP significant macular edema (all sessions of treatment) and any history of systemic diseases other than diabetes were excluded. It is necessary to note that in this study, the screening process for evaluation of diabetic macular edema in 1 week after each laser therapy session with high-resolution SD-OCT (Spectralis, Heidelberg Engineering, Heidelberg, Germany) and fluorescein angiography (HRA II Heidelberg, Heidelberg, Germany) was performed and patients with macular edema were excluded to avoid a possible change in visual evoked potential components.
The study was undertaken in compliance with the Helsinki Declaration and a written informed consent from patients was obtained before enrollment.
Panretinal photocoagulation
PRP was carried out by Ellex Integre Duo Photocoagulator (Mawson Lakes, SA, Australia) by one of the authors (Hassan Khojasteh). Thirty minutes before the start of the laser therapy, a 1% tropicamide drop was applied 3 times at a time interval of 5 mins for pupil dilation. Also, 15 mins before the start of laser therapy, a 0.5% tetracaine drop was applied 2 times at a time interval of 5 mins for corneal anesthesia. After the pupil dilation and corneal anesthesia, a wide field contact lens (Volk SuperQuad 160 Panfundus lens, Volk Co., Mentor, OH, USA) with 1% methylcellulose gel, to prevent possible damage, was applied to the cornea.
All patients received PRP with a power setting of 250 to 500 mW, the spot size of 500 μm and a pulse duration of 0.1 s in three steps with a 1-week time interval. Also, an average of 350 to 400 spots was used in every session of laser therapy in each eye.
Visual evoked potentials
The pattern-reversal visual evoked potential (PRVEP) test was performed using the Metrovision MonPack 3 vision monitoring system (Metrovision, Lille, France), before and after the laser treatment. At first, the patient was seated at a distance of 1 m from the pattern-shift screen. The patterned checkerboard stimuli with contrast 80% and mean luminance 50 cd/m2 in a reversal rate of two reversals per second were used to elicit the standard PRVEP. In order to assess the function of each eye, according to the International Society for Clinical Electrophysiology of Vision standard, the visual stimuli were presented monocularly.
Before placing the electrodes, the areas of the patient’s scalp were completely cleaned to put the electrodes. Then, the gold cup electrodes were placed on the related areas according to the international 10/20 electrode system, ie, the active electrode on the scalp over the visual cortex at Oz, the reference electrode at Fz and the ground electrode on the left ear, were placed.
Once the electrode was correctly positioned and the patient was properly positioned from the screen, the test was started. The monitor displayed pattern reversal checkerboard with check size of 1 degree (60 mins of arc), amplification 2000 times and band-pass filters of 1–100 Hz. The values of the P100 amplitude and latency of visual evoked potential were studied at angular dimension 60´ of the stimulus. These results were compared among the three sessions of laser therapy and before and after PRP.
Data analysis
Statistical analysis was performed using software SPSS 21.0 for Windows (SPSS Inc., Chicago, IL, USA). Paired t-test was used to compare the P100 amplitude and latency among three sessions of laser therapy, pre- and post-PRP and pre and 1.5 months after final treatment.
All data were expressed as mean difference±SD and accepted as statistically significant if P<0.05.
Results
Twenty-one patients with proliferative diabetic retinopathy (13 males and 8 females) with an average age of 57.6±10.3 years were selected from participating patients in the study.
Table 1 shows the mean and standard deviation of amplitudes and latencies of the P100 components of the PRVEP before and after PRP in 21 proliferative diabetic retinopathy eyes in the form of the study group.
 |
Table 1 The mean and standard deviation of PRVEP before and after the treatment of panretinal photocoagulation (PRP) |
According to the results summarized in Table 1, amplitude changes in the study group at different sessions compared to the pre-treatment session and also each session compared to the previous one showed a decrease change, in a way that all of these changes were statistically significant (P<0.001). In addition, the results of P100 latencies showed a statistically significant increase during laser therapy sessions (from the first session to the third session of laser therapy) in the affected eye (P<0.001). However, 1.5 months after the completion of PRP treatment, an increase in amplitude and a decrease in latency of the P100 component of the PRVEP were observed (P<0.001).
In addition, the mean percentage difference of different sessions with each other is shown in Table 2, indicating a decrease in the amplitude and an increase in latency of P100 component of PRVEP during the treatment sessions (from the first session to the third session of laser therapy). The changes in the parameters of VEP after one and a half months after the final treatment showed an increase in amplitude and a reduction in latency of the P100 wave (P<0.001).
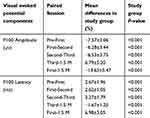 |
Table 2 % mean differences of PRVEP components groups in different paired sessions |
Discussion
Based on the results of the present study, the P100 amplitudes after the first laser treatment session showed an approximate decrease of 7.6%, which continued with a decrease of approximately 6.3% and 6.5% after the second and third sessions of laser therapy, respectively. However, an increase in the P100 wave amplitude with an approximate magnitude of 6.8% was observed in one and a half months after the final laser therapy session, indicating the partial recovery of the visual pathway and primary visual cortex functions.
In the P100 latency analysis, one of the most diagnostic visual evoked potential parameters, P100 wave latency changes showed an increasing trend after laser therapy in the study groups so that after first, second and third laser therapy sessions, an approximate increase of 2.7%, 2.6% and 3.3% of the corresponding wave in latency was occurred, respectively. Also, the evaluation of the amplitudes and latencies of P100 component of PRVEP test in the pre-treatment and 1.5 months after the final treatment showed that the lower difference between the pre-treatment and the 1.5-month post-treatment period in the latency compared to the amplitude parameter. In fact, the amplitudes as well as latency following PRP recovered, but there was greater recovery with latency. This can be because of that in the clinical practice, latency delay and interocular differences in latency of the P100 wave are seen as more sensitive and reliable indicators of dysfunction in the visual pathway than amplitude changes.13
Due to diffuse peripheral polyneuropathy, metabolic and vascular factors affecting peripheral nerves in diabetic retinopathy,14,15 the purposeful destruction of a significant fraction of the photoreceptors, as well as other more superficial retinal layers and retinal scaring following PRP,16,17 the possible neural conduction delay in the visual pathway can cause a delay in the visual evoked potential latencies as observed in results of the present study. However, after one and a half months of completion of laser therapy, a decrease of approximately 1.7% in the latency of P100 component of PRVEP test was observed. The argument that cortical “plasticity” or “reorganization” occurs after focal retinal laser lesions hinge on the crucial observation that the cortex is initially silent and then regains responsiveness. In fact, delayed cortical recovery after laser lesions is related to retinal healing18 as it was seen in the current study after 1.5 months of the completion of laser therapy.
Few studies have investigated the effect of PRP laser in proliferative diabetic retinopathy patients on visual evoked potential parameters. Shenoy et al19 assessed PRVEP parameters prior to and 4 weeks after PRP in patients with uncontrolled diabetes mellitus and reported a significant decrease in VEP amplitudes in 48% and increase in latency in 75% of eyes. Consistent with Shenoy’s19 study, reduction in P100 amplitude and delay in P100 latency was observed in the patients with proliferative diabetic retinopathy in the current study. However, the relative recovery of P100 amplitude and latency were observed after 1.5 months of final laser therapy. Perhaps the reason for the differences in the results of these two studies is related to the number of patients, the method of measuring VEP parameters, the laser treatment method and the duration of the evaluation after the PRP. In fact, most studies have assessed the changes of visual evoked potential components in diabetes without any medical intervention in eye structures or even prior to the onset of vascular abnormalities. The results of various studies about changes in visual evoked potential components due to diabetes pointed to a delay in latency, especially P100 wave, in patients with different types of diabetes, as well as with or without retinopathy.20–25 In fact, the high variability of P100 latency changes ranging from 9% to 77% were found in these studies. However, in the present study, changes in PRVEP parameters after laser therapy were evaluated and, contrary to the mentioned studies, changes were not only considered in diabetes but in all of the patient’s therapeutic procedures including three laser therapy sessions.
In a study conducted by Gottlob et al,26 a significant decrease in the PRVEP amplitude was observed 1 day after laser therapy, although latency changes were not significant. While the results of the present study indicated a decrease in the amplitude of the PRVEP after PRP and in this respect, it was consistent with the results of the Gottlob’s26 study, but after laser therapy, also an increase in latency was observed, which was not found in the Gottlob’s26 study results. Probably the reason for the differences is related to the different duration of evaluation of VEP parameters in the two studies, which was evaluated 1 week and 1 day after the treatment in the present and Gottlob’s23 studies, respectively. In addition, Öner et al27 found no significant reduction in the amplitudes and latencies of PVER test in the first-month recordings. Contrary to the results of Öner’s27 study, the results of the current study reported a decrease in amplitude and an increase in latency of P100 component of PRVEP during the laser treatment process. This unaffected results of PRVEP might be due to the timing of the tests or due to the technique of the PRP application which might minimize the damage to the retina.
Some studies have suggested that these changes are due to neural damage in ganglion cells and nerve fiber layers of the retina, especially in patients with proliferative diabetic retinopathy.14,15 In addition, the PRP plays an important role in the visual evoked potential components changes. Matsubara et al15 showed that the effects of PRP on the levels of cytochrome oxidase (CO), Zif268, synaptophysin and growth-associated protein 43 (GAP-43) in the primary visual cortex of adult monkeys could result in metabolic activity changes and redistribution of neurochemicals in the visual cortex. These changes result in an anomalous visual functional loss that alters the visual evoked potential components and can also explain the degradation of a currently damaged visual system that was mentioned in the present study. It should also be taken into account that PRP by using argon lasers can further stabilize peripheral retinal lesions, and, therefore, the central retinal changes after PRP may also continue, which in turn can cause changes in visual evoked potential components.28
Conclusion
PRP in diabetic retinopathy patients may effect on the function of the visual cortex and cause changes in PRVEP parameters. However, over time, retinal healing after laser therapy can occur and ultimately the partial recovery of visual pathway can be possible.
Consent for publication
Written informed consents was obtained to publish the details from each participant.
Ethics approval and informed consent
The study protocol adhered to the tenets of the Declaration of Helsinki and was approved by the institutional review board of the Iran University of Medical Sciences, the Steering Committees of the School of Rehabilitation Sciences and Farabi Eye Hospital of the Tehran University of Medical Sciences. All patients signed a written informed consent form to allow the evaluation of their clinical data.
Acknowledgment
The authors have no financial interest in any of the products mentioned in the manuscript. The authors would like to extend their sincere appreciation to Frabai eye hospital staff for their assistance and co‐operation in carrying out the electrophysiological tests by MonPack3 system. This study was part of a Masters thesis supported by the Tehran University of Medical Sciences (grant No:91/01/6931).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Sarwar N, Gao P, Seshasai SRK, et al. Diabetes mellitus, fasting blood glucose concentration, and risk of vascular disease: a collaborative meta-analysis of 102 prospective studies. Lancet (London, England). 2010;375(9733):2215–2222. doi:10.1016/S0140-6736(10)60484-9
2. Lieth E, Gardner TW, Barber AJ, Antonetti DA. Retinal neurodegeneration: early pathology in diabetes. Clin Exp Ophthalmol. 2000;28(1):3–8.
3. Heravian J, Ehyaei A, Shoeibi N, et al. Pattern visual evoked potentials in patients with type II diabetes mellitus. J Ophthalmic Vis Res. 2012;7(3):225–230.
4. Cohen SR, Gardner TW. Diabetic retinopathy and diabetic macular edema. Dev Ophthalmol. 2015;55:137–146.
5. Wu G. Diabetic Retinopathy-The Essentials.
6. Celesia GG, Polcyn RD, Holden JE, Nickles RJ, Gatley JS, Koeppe RA. Visual evoked potentials and positron emission tomographic mapping of regional cerebral blood flow and cerebral metabolism: can the neuronal potential generators be visualized? Electroencephalogr Clin Neurophysiol. 1982;54(3):243–256. doi:10.1016/0013-4694(82)90174-2
7. American Society of Retina Specialists (ASRS) global trends in retina survey results; 2015. Available from: https://www.asrs.org/content/documents/2014_global_trends_comprehensivepostmtg.pdf.
8. Elman MJ, Aiello LP, Beck RW, et al. Randomized trial evaluating ranibizumab plus prompt or deferred laser or triamcinolone plus prompt laser for diabetic macular edema. Ophthalmology. 2010;117(6):1064–1077. doi:10.1016/j.ophtha.2010.02.031
9. Stewart MW. Treatment of diabetic retinopathy: recent advances and unresolved challenges. World J Diabetes. 2016;7(16):333–341. doi:10.4239/wjd.v7.i16.333
10. Parisi V, Uccioli L. Visual electrophysiological responses in persons with type 1 diabetes. Diabetes Metab Res Rev. 2001;17(1):12–18. doi:10.1002/(ISSN)1520-7560
11. Horton JC, Hoyt WF. The representation of the visual field in the human striate cortex. Arch Ophthalmol. 1991;109:816–824. doi:10.1001/archopht.1991.01080060080030
12. American Academy of Ophthalmology Retina/Vitreous Panel Preferred Practice Pattern Guidelines. Diabetic Retinopathy. San Francisco, CA: American Academy of Ophthalmology; November 2016. Available from: http://www.aao.org/ppp.
13. Kolappan M, Henderson APD, Jenkins TM, et al. Assessing structure and function of the afferent visual pathway in multiple sclerosis and associated optic neuritis. J Neurol. 2009;256(3):305–319. doi:10.1007/s00415-009-5123-5
14. Mooradian AD. Pathophysiology of central nervous system complications in diabetes mellitus. Clin Neurosci. 1997;4(6):322–326.
15. Matsubara JA, Lam DY, Kalil RE, et al. The effects of panretinal photocoagulation on the primary visual cortex of the adult monkey. Trans Am Ophthalmol Soc. 2001;99:33.
16. Little HL, Zweng HC, Peabody RR. Argon laser slit-lamp retinal photocoagulation. Trans Am Acad Ophthalmol Otolaryngol. 1970;74(1):85–97.
17. Fong DS, Girach A, Boney A. Visual side effects of successful scatter laser photocoagulation surgery for proliferative diabetic retinopathy: a literature review. Retina. 2007;27(7):816–824.
18. Horton JC, Hocking DR. Monocular core zones and binocular border strips in primate striate cortex revealed by the contrasting effects of enucleation, eyelid suture, and retinal laser lesions on cytochrome oxidase activity. J Neurosci. 1998;18(14):5433–5455. doi:10.1523/JNEUROSCI.18-14-05433.1998
19. Shenoy R, Al-Belushi H, Al-Ajmi S, Al-Nabhani SM, Ganguly SS, Bialasiewicz AA. Visually evoked potentials after panretinal photocoagulation in omani patients with uncontrolled diabetes mellitus. Middle East Afr J Ophthalmol. 2008;15(2):51–56. doi:10.4103/0974-9233.51992
20. Gregori B, Galié E, Pro S, Clementi A, Accornero N. Luminance and chromatic visual evoked potentials in type I and type II diabetes: relationships with peripheral neuropathy. Neurol Sci. 2006;27(5):323–327. doi:10.1007/s10072-006-0704-x
21. Cirillo D, Gonfiantini E, De Grandis D, Bongiovanni L, Robert JJ, Pinelli L. Visual evoked potentials in diabetic children and adolescents. Diabetes Care. 1984;7(3):273–275.
22. Mariani E, Moreo G, Colucci GB. Study of visual evoked potentials in diabetics without retinopathy: correlations with clinical findings and polyneuropathy. Acta Neurol Scand. 1990;81(4):337–340. doi:10.1111/j.1600-0404.1990.tb01566.x
23. Puvanendran K, Devathasan G, Wong PK. Visual evoked responses in diabetes. J Neurol Neurosurg Psychiatry. 1983;46(7):643–647. doi:10.1136/jnnp.46.7.643
24. Gupta S, Gupta G, Deshpande VK. Visual evoked potential changes in patients with diabetes mellitus without retinopathy. Int J Res Med Sci. 2017;3(12):3591–3598.
25. Lopes de Faria JM, Katsumi O, Cagliero E, Nathan D, Hirose T. Neurovisual abnormalities preceding the retinopathy in patients with long-term type 1 diabetes mellitus. Graefe’s Arch Clin Exp Ophthalmol. 2001;239(9):643–648. doi:10.1007/s004170100268
26. Gottlob I, Prskavec FH, Stelzer N, Hienert I, Weghaupt H, Radda TM. Reversible changes of visual acuity and pattern-electroretinograms after blue-green argon laser photocoagulation of diabetic patients. Doc Ophthalmol. 1989;72(2):105–113. doi:10.1007/BF00156700
27. ÖNER A, AKARSU C, ARDA H, GÜMÜŞ K, KARAKÜÇÜK S, MİRZA E. Electrophysiologic changes after panretinal argon laser photocoagulation in patients with diabetic retinopathy. J Retin. 2015;23:1.
28. Kaufman SC, Ferris FL, Seigel DG, Davis MD, DeMets DL. Factors associated with visual outcome after photocoagulation for diabetic retinopathy. Diabetic retinopathy study report #13. Invest Ophthalmol Vis Sci. 1989;30(1):23–28.
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
