Back to Archived Journals » Integrated Blood Pressure Control » Volume 8
Vasomotor sympathetic outflow in the muscle metaboreflex in low birth weight young adults
Authors Chifamba J, Mbangani B, Chimhete C, Gwaunza L, Allen L, Chinyanga H
Received 25 October 2014
Accepted for publication 16 February 2015
Published 27 May 2015 Volume 2015:8 Pages 37—42
DOI https://doi.org/10.2147/IBPC.S76382
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Steven Atlas
Jephat Chifamba,1 Brilliant Mbangani,1 Casper Chimhete,1 Lenon Gwaunza,1 Larry A Allen,2 Herbert Mapfumo Chinyanga1
1Department of Physiology, College of Health Sciences, University of Zimbabwe, Harare, Zimbabwe; 2Section of Advanced Heart Failure and Transplantation, University of Colorado School of Medicine, Aurora, CO, USA
Abstract: A growing body of evidence suggests that low birth weight (LBW) offspring are associated with long-term structural and functional changes in cardiovascular and neuroendocrine systems. We tested the hypothesis that muscle metaboreflex activation produces exaggerated responses in cardiac autonomic tone (represented by heart rate variability ratio) and cutaneous vascular sympathetic tone (represented by plethysmography pulse wave amplitude) in LBW compared to normal birth weight (NBW) young adults. We recruited 23 LBW (18 females and five males) and 23 NBW (14 females and nine males) University of Zimbabwe students with neonatal clinical cards as proof of birth weight at term. Resting electrocardiogram, pulse waves, and blood pressures were recorded. Participants then underwent a static/isometric handgrip exercise until fatigue and a post-exercise circulatory arrest period of 2 minutes. We observed (results mean ± standard deviation) a greater mean increase in heart rate variability ratio from baseline to exercise for LBW compared to NBW individuals (1.015±1.034 versus [vs] 0.119±0.789, respectively; P<0.05). We also observed a greater mean decrease in plethysmography pulse wave amplitude from baseline to exercise (-1.32±1.064 vs -0.735±0.63; P<0.05) and from baseline to post-exercise circulatory arrest (-0.932±0.998 vs -0.389±0.563; P<0.05) for LBW compared to NBW individuals. We conclude that LBW may be associated with an exaggerated sympathetic discharge in response to muscle metaboreflex.
Keywords: blood pressure, heart rate variability, plethysmography pulse
Introduction
A growing body of evidence suggests that low birth weight (LBW) offspring are associated with an increased risk for coronary artery disease,1 hypertension,2 and heart failure.3 Previous studies have also shown that exaggerated vasoconstrictive and neuroendocrine responses are associated with development of hypertension in adults and may function as markers for preclinical or pathophysiologic phases of hypertension.4,5 LBW is associated with an increased sympathetic nerve activity and therefore predisposing LBW individuals to development of hypertension in adulthood.6–8
Static/isometric handgrip exercise evokes an increase in heart rate, mean arterial pressure, muscle and skin sympathetic nervous activity. Forearm blood circulatory arrest just before cessation of the exercise causes blood pressure (BP) to remain above resting levels.6,9 These responses are evoked by two neural mechanisms, components of the muscle metaboreflex, which are: central command (neural signals of central origin) and the exercise pressor reflex (EPR, a reflex arising from exercising muscle).10 The central command has a minor role in cardiovascular regulation during exercise. The EPR is a feedback system arising from thinly myelinated mechanosensitive (group III/Aδ-fibers) and unmyelinated metabosensitive (group IV/C-fibers) afferents in the skeletal muscle.10 When oxygen delivery to active skeletal muscle is insufficient to meet the metabolic demands, metabolites, eg, lactic acid, adenosine, diprotonated phosphate, potassium, H+, and arachidonic acid products among others, accumulate within active muscle and stimulate group III and group IV afferent neurons leading to a reflex sympathetic discharge (the muscle metaboreflex).1,2,10 The EPR is exaggerated in hypertension and related conditions like heart failure, with the over activity of the afferent arms (mechanosensitive and metabosensitive afferents) of this reflex being the cause of the exaggeration.3
Pulse wave amplitude (PWA) is useful in monitoring sympathetic influences on skin blood flow in the finger and heart rate variability (HRV) ratio monitors the cardiac autonomic tone.9,11
Studies have assessed the modulation of the sympathetic outflow by the muscle metaboreflex.9,12 However, no study has compared the modulation of the sympathetic nerve activity to the cardiovascular system by the muscle metaboreflex, between individuals with LBW and normal birth weight (NBW). In this study, we hypothesized that individuals born with LBW would respond with exaggerated sympathetic discharge to the heart and the vascular system during stimulation of the muscle metaboreflex, compared to NBW individuals.
Methodology
Participants
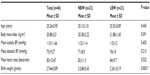
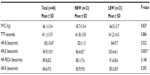
The study was conducted in the Exercise Laboratory in the Department of Physiology, University of Zimbabwe. Forty-six healthy voluntary participants were drawn from students at the University of Zimbabwe. Before the actual day of study, all participants were provided with the full details of the study and we familiarized each subject with the protocol. Informed consent was sought and obtained from the participants. The study participants were screened using a self-administered questionnaire. Participants were recruited on the basis of documented proof of birth weight in the form of a Ministry of Health, Child Health Card. The exclusion criteria were 1) age <18 or >25 years; 2) a history of cardiovascular disease or hypertension (defined as the current use of antihypertensive medication or resting BP ≥140/90 mmHg); 3) previous endurance training; 4) pre-term birth; 5) body mass index (BMI) more than 24.5 kg/m2. The participants were assigned into two groups, LBW and NBW. LBW was defined as birth weight <2,500 g at term. Participants in the two groups were matched for BMI. Ethical permission was granted by Joint Parirenyatwa Hospital and College of Health Sciences Research Ethics Committee and the Medical Research Council of Zimbabwe.
Anthropometric and resting measurements
Weight and height were measured with the participants putting on light clothing and without shoes. BMI for each participant was calculated (kg/m2). Resting BPs were measured. Resting computerized electrocardiogram (ECG) recording was obtained from lead II and resting pulse wave recordings were obtained from the finger pulse plethysmography for 5 minutes, using iWorx research system hardware (IWX/214) (iWorx Sytems, Inc., Dover, NH, USA) and the Labscribe 2 software.
Exercise protocol
In the 24 hours before exercise, participants were asked to abstain from taking alcohol, energy drinks, and caffeinated products. Participants underwent a static/isometric handgrip exercise performed with the dominant hand using a handgrip dynamometer (Lab BSL 3.7.7; BIOPAC, Goleta, CA, USA) at 40% maximum voluntary contraction (MVC) until fatigue to ensure that they have all reached a common metabolic end point. This level of force was chosen since it has been previously shown that handgrips sustained at 40% and 60% of MVC elicited a comparable increase in muscle sympathetic nerve activity.12 Once the exerted force declined to <80% of the desired force (40% MVC) for >2 seconds, a 2-minute post-exercise circulatory arrest (PECA) phase began using an arm cuff inflated to a pressure >200 mmHg. During the static handgrip exercise and PECA participants were instructed to avoid breath holding. After PECA period, participants were given a 5-minute recovery period. ECG and pulse wave recordings were obtained during the period of exercise, PECA, and recovery.
Data analysis
Frequency domain measures of ECG R-R variability were used to estimate the high frequency (HF, 0.15–0.4 Hz) and low frequency (LF, 0.04–0.15 Hz) power. The HF power peak represents modulation of efferent parasympathetic (vagal) activity by ventilation, but only in the presence of sinus rhythm. The LF power peak represents combined modulation of efferent parasympathetic and efferent sympathetic nervous system activity by baroreflex activity. Overall the amplitudes of LF and HF power reflect modulation of sinus node firing rate and the LF/HF ratio (HRV ratio) has been proposed as an index of sympathovagal balance.9,13 However, very LF indices could not be used since they require power spectra that are more than 5 minutes long, which was not in keeping with our experimental protocol. The IWORX Labscribe 2 software which is part of the physiological system was used to analyze these variables (iWorx Sytems, Inc.). SPSS software version 16.0 (SPSS Inc., Chicago, IL, USA) was used to analyze the data. Independent samples two-tailed Student’s t-test at 5% level of significance was used to compare the various means of the cardiovascular parameters between NBW and LBW individuals (P<0.05 was considered significant). The data are represented as mean ± standard error of the mean on figures and as mean and standard deviation (SD) in tables.
Results
A total of 23 LBW (18 females and five males) and 23 NBW (14 females and nine males) black adults, with a median age of 20 years (18–25 years), successfully completed the experiment. The baseline characteristics and mean MVC, time to fatigue as well as the heart rate changes are shown in Tables 1 and 2, respectively.
HRV ratio
The trends of mean HRV ratios for LBW and NBW individuals during the experimental protocol are shown in Figure 1. The mean increase in HRV from baseline to exercise was greater for LBW compared to NBW individuals (1.015±1.034 versus [vs] 0.119±0.789, respectively; P<0.05). However, the mean increase in HRV ratio from baseline to PECA and from baseline to recovery was not significantly different between LBW and NBW individuals (Table 3).
PWA
The trends of the mean PWA for LBW and NBW individuals during the experimental protocol are shown in Figure 2. The mean decrease in PWA from baseline to exercise was greater for LBW compared to NBW individuals (−1.32±1.064 vs −0.735±0.63, respectively; P<0.05). The mean decrease in PWA from baseline to PECA was also greater for LBW compared to NBW individuals (−0.932±0.998 vs −0.389±0.563, respectively; P<0.05). However, the mean change in PWA from baseline to PECA was not significantly different between LBW and NBW individuals (Table 3).
Discussion
HRV ratio and PWA responses to activation of the muscle metaboreflex are exaggerated in LBW compared to NBW individuals. This suggests that the EPR, which is involved in tight regulation of the cardiovascular response to exercise, is persistently dysregulated into early adulthood for LBW individuals.10,14
HRV ratio for LBW was modestly elevated at rest, although there was no significant difference between the HRV ratios of the two groups. This may be due to LBW individuals being prenatally programmed to increase sympathetic nerve activity secondary to the adverse intra-uterine conditions that the fetus will be subjected to.14 Studies have shown that the EPR is exaggerated in hypertensive rats, in part due to over activity of the afferents.3,13 We investigated the efferent component of the EPR; the afferent pathway has not been investigated in LBW subjects. In hypertensive subjects, exercise evokes an excessive increase in BP from a chronically elevated resting value.14 During exercise, compared to baseline, LBW individuals experienced an exaggerated sympathetic response to the static handgrip. This was indicated by a significant mean increase in HRV ratio (increased HRV ratio reflects an increase in sympathetic tone compared to the vagal tone) as well as a significant mean decrease in PWA (Table 3). LBW and NBW individuals had an increased sympathetic discharge to the heart and the peripheral vessels, although that of LBW individuals was exaggerated. The increased sympathetic discharge we observed in both groups is in keeping with the findings by Jarvis et al12 and Ichinose et al2 who described an increase in sympathetic discharge in normal individuals. However, in these two studies birth weight was not considered as a variable while in our study it was.9,12 The mean increase in HRV ratio from baseline to PECA decreased but was not significantly different between the two groups (Table 3), whilst the mean decrease in PWA showed a significant difference between the two groups (LBW and NBW). This indicated a potentiated sympathetic discharge in response to accretion of metabolites in the once active muscle, with the differences in the sympathetic discharge to the heart and the peripheral vessels during PECA being probably due to the fact that there is organ specific sympathetic nerve activity and regional differences in this nerve activity as is experienced particularly in cardiovascular diseases.12,15,16 Animal models in which there was ganglionic and α-adrenergic blockage using hexamethonium and phentolamine, respectively, showed an abolishment of the exaggerated pressor response to exercise, suggesting that these exaggerated hemodynamic responses were mediated, in part, by the abnormally large EPR-induced sympathetic nerve activity.14 This might be the same reason why the LBW individuals in our study had an exaggerated response to the EPR. Exaggerated neuroendocrine and vasoconstrictive responses have been shown to predict the development of hypertension in adults.4 Therefore, the exaggerated EPR response, in our study, may function as a marker for the pathophysiologic phases of hypertension.
The insignificant difference in mean increase in HRV ratio and significant difference in mean decrease in PWA during PECA may also suggest an increase in cardiac parasympathetic tone caused by buffering by the arterial baroreflex.9 Insensitivity of baroreceptors, which buffer BP increases during exercise, has been reported in hypertensive individuals and individuals with heart failure.13 This might also be present in LBW individuals since they are predisposed to these conditions; therefore, future studies to investigate the baroreceptor sensitivity in LBW individuals are warranted.
Overall, the totality of these findings support the hypothesis that LBW young adults have an exaggerated EPR response that is measureable long before progression to the cardiovascular disorders that LBW individuals are prenatally programmed to develop. The EPR is dysfunctional in cardiovascular disorders such as heart failure and hypertension. This leads to an increased sympathetic discharge underlying the exaggerated increase in BP, heart rate, and peripheral resistance during acute physical activity.3,10,13
Conclusion
We concluded that LBW may be associated with an exaggerated vasomotor sympathetic outflow during the muscle metaboreflex activation during exercise in normotensive young adults. LBW individuals may have an exaggerated EPR response.
Acknowledgments
We are greatly indebted to E Nhandara and RT Mbanje for technical assistance and V Chikwasha for statistical analysis. Authors declare sole responsibility for the scientific content of this research paper. The study was funded by National Institute of Neurological Disorders and Stroke, Office of the Director-National Institutes of Health and National Heart, Lung and Blood Institutes through Medical Education Partnership Initiative Grants to the University of Zimbabwe, College of Health Sciences (1 R24 TW008881 and 1 R24 TW008905).
Disclosure
The authors report no conflicts of interest related to this paper.
References
Adreani CM, Kaufman MP. Effect of arterial occlusion on responses of group III and IV afferents to dynamic exercise. J Appl Physiol (1985). 1998;84(6):1827–1833. | |
Ichinose M, Delliaux S, Watanabe K, Fujii N, Nishiyasu T. Evaluation of muscle metaboreflex function through graded reduction in forearm blood flow during rhythmic handgrip exercise in humans. Am J Physiol Heart Circ Physiol. 2011;301(2):H609–H616. | |
Leal AK, Williams MA, Garry MG, Mitchell JH, Smith SA. Evidence for functional alterations in the skeletal muscle mechanoreflex and metaboreflex in hypertensive rats. Am J Physiol Heart Circ Physiol. 2008;295(4):H1429–H1438. | |
McCormick Covelli M. The relationship of low birth weight to blood pressure, cortisol levels, and reactivity in African American adolescents: a pilot study. Issues Compr Pediatr Nurs. 2006;29(3):173–187. | |
Falkner B, Hulman S, Kushner H. Birth weight versus childhood growth as determinants of adult blood pressure. Hypertension. 1998; 31(1):145–150. | |
IJzerman RG, Stehouwer CD, de Geus EJ, van Weissenbruch MM, Delemarre-van de Waal HA, Boomsma DI. Low birth weight is associated with increased sympathetic activity: dependence on genetic factors. Circulation. 2003;108(5):566–571. | |
Hausberg M, Barenbrock M, Kosch M. Elevated sympathetic nerve activity: the link between low birth size and adult-onset metabolic syndrome? J Hypertens. 2004; 22(6):1087–1089. | |
Jansson T, Lambert GW. Effect of intrauterine growth restriction on blood pressure, glucose tolerance and sympathetic nervous system activity in the rat at 3–4 months of age. J Hypertens. 1999;17(9):1239–1248. | |
Watanabe K, Ichinose M, Fujii N, Matsumoto M, Nishiyasu T. Individual differences in the heart rate response to activation of the muscle metaboreflex in humans. Am J Physiol Heart Circ Physiol. 2010; 299(5):H1708–H1714. | |
Murphy MN, Mizuno M, Mitchell JH, Smith SA. Cardiovascular regulation by skeletal muscle reflexes in health and disease. Am J Physiol Heart Circ Physiol. 2011;301(4):H1191–H1204. | |
Grote L, Zou D, Kraiczi H, Hedner J. Finger plethysmography – a method for monitoring finger blood flow during sleep disordered breathing. Respir Physiol Neurobiol. 2003;136(2–3):141–152. | |
Jarvis SS, VanGundy TB, Galbreath MM, et al. Sex differences in the modulation of vasomotor sympathetic outflow during static handgrip exercise in healthy young humans. Am J Physiol Regul Integr Comp Physiol. 2011;301(1):R193–R200. | |
Taylor JA, Carr DL, Myers CW, Eckberg DL. Mechanisms underlying very-low-frequency RR-interval oscillations in humans. Circulation. 1998;98(6):547–555. | |
Smith SA, Williams MA, Leal AK, Mitchell JH, Garry MG. Exercise pressor reflex function is altered in spontaneously hypertensive rats. J Physiol. 2006;577(Pt 3):1009–1020. | |
Schäffer L, Burkhardt T, Müller-Vizentini D, et al. Cardiac autonomic balance in small-for-gestational-age neonates. Am J Physiol Heart Circ Physiol. 2008;294(2):H884–H890. | |
Chifamba J, Chakanyuka KE, Longo-Mbenza B, Mahachi CB, Mufunda J. Comparison of exercise-induced hypertension in low birth weight and normal birth weight young black adults in Zimbabwe. High Blood Press Cardiovasc Prev. 2012;19(3):123–127. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.