Back to Journals » Clinical Ophthalmology » Volume 8
Ultraviolet damage to the eye revisited: eye-sun protection factor (E-SPF®), a new ultraviolet protection label for eyewear
Authors Behar-Cohen F , Baillet G, de Ayguavives T, Ortega Garcia P, Krutmann J, Peña-García P, Reme C, Wolffsohn J
Received 3 April 2013
Accepted for publication 26 June 2013
Published 19 December 2013 Volume 2014:8 Pages 87—104
DOI https://doi.org/10.2147/OPTH.S46189
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Francine Behar-Cohen,1 Gilles Baillet,2 Tito de Ayguavives,2 Paula Ortega Garcia,3 Jean Krutmann,4 Pablo Peña-García,3,5 Charlotte Reme,6 James S Wolffsohn7
1French Institute of Health and Medical Research, Team 17, Centre de Recherche des Cordeliers, France; 2Research and Development Center, Essilor International, Saint Maur des Fossés, France; 3Department of Research, Fundacion Jorge Alio, Alicante, Spain; 4IUF Leibniz Research Institute for Environmental Medicine, Düsseldorf, Germany; 5Division of Ophthalmology, University Miguel Hernández, Alicante, Spain; 6Laboratory of Retinal Cell Biology, Department of Ophthalmology, University of Zurich, Switzerland; 7Life and Health Sciences, Aston University, Aston Triangle, Birmingham, UK
Abstract: Ultraviolet (UV) radiation potentially damages the skin, the immune system, and structures of the eye. A useful UV sun protection for the skin has been established. Since a remarkable body of evidence shows an association between UV radiation and damage to structures of the eye, eye protection is important, but a reliable and practical tool to assess and compare the UV-protective properties of lenses has been lacking. Among the general lay public, misconceptions on eye-sun protection have been identified. For example, sun protection is mainly ascribed to sunglasses, but less so to clear lenses. Skin malignancies in the periorbital region are frequent, but usual topical skin protection does not include the lids. Recent research utilized exact dosimetry and demonstrated relevant differences in UV burden to the eye and skin at a given ambient irradiation. Chronic UV effects on the cornea and lens are cumulative, so effective UV protection of the eyes is important for all age groups and should be used systematically. Protection of children’s eyes is especially important, because UV transmittance is higher at a very young age, allowing higher levels of UV radiation to reach the crystalline lens and even the retina. Sunglasses as well as clear lenses (plano and prescription) effectively reduce transmittance of UV radiation. However, an important share of the UV burden to the eye is explained by back reflection of radiation from lenses to the eye. UV radiation incident from an angle of 135°–150° behind a lens wearer is reflected from the back side of lenses. The usual antireflective coatings considerably increase reflection of UV radiation. To provide reliable labeling of the protective potential of lenses, an eye-sun protection factor (E-SPF®) has been developed. It integrates UV transmission as well as UV reflectance of lenses. The E-SPF® compares well with established skin-sun protection factors and provides clear messages to eye health care providers and to lay consumers.
Keywords: back reflection, transmission, irradiation, solar irradiance, aging, risk reduction, prevention
Introduction
Awareness of ultraviolet (UV) radiation damage to the skin has risen substantially over recent decades, and effective measures have been implemented to improve UV protection of the skin.1–3 Although evidence has been accumulating that the eye too is vulnerable to UV damage, a comprehensive generally accepted definition of a sun protection factor for the eye (similar to a skin-sun protection factor) is lacking. Protecting the periorbital skin region follows the same rationale as protecting skin in general, but is still widely neglected.4–6 There are unique requirements for protecting the tissues of the eye, since eye function depends on direct exposure to light. The UV burden of the eye differs from the UV burden of the skin.7–10 The importance of the eye protection that clear lenses and contact lenses may offer is still underestimated. Lenses should provide reliable labeling of their protective potential. Several attempts to define “sun protection” or “UV protection” of the eye have been made, but were mainly restricted to UV transmission.11–13 UV reflection, in contrast, is an important and underestimated contributor, as recent research has demonstrated.14
The purpose of this paper is not only to pioneer by introducing an eye-sun protection factor (E-SPF®), to reopen a discussion, and to raise awareness of eye health care professionals and lay persons about UV damage, including novel aspects such as possible harm induced by reflection, but also to stimulate research to evaluate better the potential harm of UV radiation (UVR) and the potential benefit of adequate UV protection.
UV exposure to the eye
UV definition
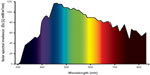
The electromagnetic spectrum ranges from infrared to visible light (Figure 1), and extends down to the shortest gamma rays. Ultraviolet radiation (UVR) is invisible to human perception. The ozone layer absorbs shorter wavelengths more efficiently, therefore the longer range of UV radiation, ie, UV-A (400–320 nm), contributes up to 95% of total UVR. Approximately 5% is contributed by UV-B (320–280 nm, middle UV).15 UV-C (280–220 nm) is absorbed within the atmosphere, mainly in the ozone layer. In recent decades, exposure to UV-A and UV-B radiation has become more relevant in areas with a deficient ozone layer. The composition of UV-A/UV-B (Figure 1) depends on the height of the sun above the horizon, on haze and cloud cover, and on atmospheric pollution.
With decreasing wavelength, spectral energy increases, and higher spectral energy raises the potential for ocular damage16,17 (Figure 2). At 300 nm (UV-B), the biologic damage potential surmounts the damage potential at 325 nm by a factor 600×.18 For ophthalmic standards, the International Organization for Standardization (ISO) 8980/3 norm defines a cutoff at 380 nm for UV-A (380–320 nm, near UV). From the danger potential point of view, experts, ie, ophthalmologists, cell biologists, and photochemists, agreed to include the range between 380 nm and 400 nm, the cutoff of 400 nm for UVR being adopted by the World Health Organization,2 European Council of Optometry and Optics,19 Commission Internationale de L’Eclairage,20 and others. The division of the UV spectrum into the spectral regions was defined in 1932 as UV-A at 400–315 nm, UV-B at 315–280 nm, and UV-C at 280–100 nm, while environmental photobiologists and dermatologists frequently define the regions as UV-A at 400–320 nm, UV-B at 320–290 nm, and UV-C at 290–200 nm.
UV sources
The predominant source of UVR is natural sunlight. Artificial light sources contribute to a lesser extent, but might become more important with the advent of modern and more energy-efficient light sources. In specific settings (eg, welding, microscopes, sun beds without adequate protection21), the UV burden is increased.
Ambient UV: direct radiation, scatter, and reflection
Direct sunlight only partly contributes to ambient UV. Under average conditions, more than 50% of ocular exposure results from scattering and cloud reflection.22 Due to the functional relationship between scattering and wavelength (proportionate to inverse fourth power of wavelength, Rayleigh’s law), UVR is scattered to a considerably higher extent than is visible light, and UV-B even more so than UV-A. Even on a clear sunny day, the main proportion of UV-B incident at the face is contributed by diffuse scatter.
Scattering is increased by clouds and haze. Ground reflection of UV (albedo) depends on the surface, with grass reflecting at low rates (2%–5%), open water 3%–13%, concrete about 10%, and snow 94%. UV intensity to the body is most significantly determined by solar angle. Higher altitude as well as lower latitude increase ambient UVR burden.
In general, adults and children are exposed to about 2%–4% of total available annual UV, while adults working outdoors receive about 10%.23 The average annual UV dose is estimated to be 20,000–30,000 J/m2 for Americans,24 10,000–20,000 J/m2 for Europeans, and 20,000–50,000 J/m2 for Australians, excluding vacation, which can add 30% or more to the UV dose.
Ocular factors: exposure geometry and anatomic conditions
Recent studies underscore the need for a clear distinction between general UV irradiance and UV incident at the eye. The burden for the skin and that for structures of the eye differ significantly. For geometric and anatomic reasons, exposure of the eye is much more influenced by ground reflection than is the skin.25 While ambient UVR peaks around noon, UVR reaching the eye depends largely on the solar angle. Depending on the time of year and latitude, ocular UVR can peak in the early morning and afternoon, at a time when the potential for UV damage to the skin or eye is usually considered to be reduced compared with the peak of the day. For summer in Japan, Sasaki et al demonstrated the UV-B burden (at the level of the cornea) between 8 am and 10 am and between 2 pm and 4 pm to be nearly twice as high as that at noon,8 even though the total amount of ambient UVR is low in the morning (Figure 3).
  | Figure 3 Hourly average of UV-B intensity in the central eye when facing towards and away from the sun (in volts). |
UV exposure to the eye is also determined by geometric factors related to the orbital anatomy and by natural protective mechanisms, including squinting and pupil constriction.22 The geometric features of the face explain the increasing exposure of UV in the morning related to solar altitude. At higher solar angles, the upper orbital rim and the brow area cast a shadow on the eye. Medially, the eye is protected by the nose; temporally, direct and reflected radiation has wide access. Even when facing away from the sun, the eye is exposed to reflected UVR.
By squinting, ocular exposure is reduced. The “aperture” of the eye is further determined by facial characteristics (eyelids, brow ridge, cheeks). Eyelids and pupil size react to the overall luminance. Pupil size affects lenticular and retinal UV exposure, which depends on multiple factors, such as age and ambient illumination. At the age of 20–29 years, mean dark-adapted pupil diameter has been shown to be 7.33 ± 0.81 mm (2 minutes at 1 lux with preadaptation), with progressive reduction over the decades to 4.85 ± 0.54 mm at the age of 80–89 years.26
Interestingly, behind sunglasses, the pupil does not dilate more, as a comprehensive analysis of geometric and radiometric aspects of ocular irradiation showed in an analysis of 400 retail sunglasses.27
Another eye-specific effect (as opposed to the skin burden) is the focusing effect of the cornea, especially a peripheral light-focusing effect as described by Coroneo.28,29 UVR incident from the periphery is refracted into the eye, and due to the focusing effect of the cornea, UV radiation is on average 22-fold stronger at the nasal limbus, which is the typical site for pterygium and pinguecula. Moreover, UV radiation is on average eight times stronger at the nasal lens cortex, the typical site for cortical cataract.30 These well known “sun terraces” according to anatomic features are also described by dermatologists. They translate into a higher incidence of malignancies and accelerated aging in areas that are exposed to UVR.
Back reflection of antireflective coatings
The aforementioned aspects are relevant for the unprotected eye as well as for eyes protected by lenses. In spectacle lens wearers, a further important source of UVR has been identified recently, ie, back reflectance by antireflective coatings in glasses.
Mannequin experiments with dosimetry (International Light ACTS270 detector for UV-B, and International Light SCS280 detector for UV-B and UV-A) demonstrated reduced transmission of UV-B (mostly <1%) by bargain sunglasses. Nevertheless, the ocular exposure was markedly greater than the transmission, indicating that indirect pathways contribute to ocular exposure.31
In prescription glasses, 6.6% of incident UVR reached the eye dosimeter placed in the eye socket of a mannequin, even in lenses covered with black opaque tape.32 In this experiment, 43 spectacle prescription lenses (glass and plastic) and 39 contact lenses left for disposal at an optician’s office were tested. Data on lens power were not provided.
Irradiation increases with increasing distance of the spectacle lenses to the forehead. A comparison between spectacles and sunglasses contacting the forehead versus displaced 6 mm showed a 5–10-fold increase in ocular exposure, depending on detector aperture (4 mm or 10 mm), so the wearing position is of relevance but does not sufficiently explain the dose measured at the eye socket.31,32
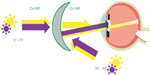
In spectacle wearers (sunglasses and clear lenses), UVR is reflected from the back surface of the lens and thus enters the eye, so even UVR incident from behind the wearer can reach the ocular surface (Figure 4).33,34
Antireflective coatings play an important role here, since most antireflective coatings reduce reflection only in the visible range of wavelengths. Antireflective coating of lenses (clear lenses and sunglasses) increases visual comfort for wearers, because it improves contrast, transparency, and transmission of visible light (typically by over 98%, depending on the angle of incidence). Antireflective coating suppresses glare and ghost images and increases image quality at the retina level, while secondarily improving cosmetic appearance. To fulfill these functions, an antireflective coating is applied to both sides of the lens, reducing direct and internal reflections at each surface, and, thereby reducing glare from light sources in front of and behind the wearer. There are single, double, and broadband antireflective coatings available. The multilayer coating reduces residual reflection over a broad band of visible wavelengths down to almost 0% between 430 nm and 660 nm.
Citek demonstrated that antireflective coatings reflect UVR at unacceptably high levels.14 Depending on the coating, some lenses exhibited up to 76.9% reflection of erythemal UV (200–315 nm). The majority of lenses tested showed negligible UV transmittance (0.0%–0.012%), but at the same time, lenses showed a reflectance between 4.21% and 62.11% in UV-A (near UV, in his measurement defined as 315–380 nm), and between 2.18% and 76.89% in high-intensity UV-B (middle or erythemal UV, 290–315 nm). In all lenses tested, reflectance in the UV range averaged at 25% of any UV band. In 35.7% of lenses, more than 10% of UV-A and UV-B was reflected (on average 38.68%). UV reflectance was less pronounced for tinted lenses, but was still between 4.05% and 5.99% of near UV-A and UV-B.
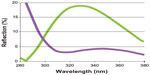
One factor influencing the efficiency of an antireflective coating is spectral bandwidth.35 By extending efficiency (destructive interferences) to a larger bandwidth from visible to UV, reflection of UVR can be reduced markedly (Figure 5).
  | Figure 5 (A) Back side reflection measurement at 30° of a standard clear lens with multilayer antireflective coating. At 300 nm, more than 15% of UVR is reflected (green line). A lens with enhanced efficiency (pink line) reduces the back reflection in the UV range markedly (Crizal UV™, Essilor International SA, F-Charenton-le-Pont, France). (B) Reflectance characteristics for various clear high-index materials with antireflective coating and uncoated crown glass and acrylic. |
Dosimetry and quantification
Assessing potential UVR hazard quantitatively is a complex task, and even the physical assessment of UV burden incident at the eye remains challenging.
UV burden without lenses
The solar ultraviolet index was developed by the World Health Organization and other organizations to offer an international standard index for UV burden.36 It is calculated from direct measurement of UV spectral power under standardized conditions to assess the risk of UV damage to the skin.
Studies by Sasaki et al showed that this index is an invalid indicator to determine the need for eye protection and can even be seriously misleading.8 They performed objective measurements with mannequin heads at defined conditions with regard to a variety of influencing factors, such as altitude, latitude, solar angle, reflectance of airborne particles, reflectance of ground and surroundings, anatomic features, posture, and facing to or away from the sun.22,25,37 UV burden was assessed by UV-B sensors mounted onto the eye sockets of the mannequin heads as well as at the top of the head to compare eye exposure with overall exposure.
UV incident at the ocular surface has been measured by contact lens UV dosimetry to determine the ratio of ocular-to-ambient UV exposure, and was reported to range from 4% to 23% at solar noon.38,39
Walsh et al employed a photodiode sensor array detector located on the surface of the cornea to measure the spectrum of radiation refracted across the cornea.40 Thus, the Coroneo effect could be demonstrated in vivo, and Kwok et al contributed further evidence by using UV sensors to determine the intensity of peripheral light-focusing that is focused on the nasal limbus versus the angle of incidence for incoming temporal light.41,42 The maximum intensity occurred at around an angle of 120° from the sagittal plane.
Measurements have been predominantly performed for UV-B radiation32,43–45 due to its higher spectral function efficiency.46 Since the damage potential of shorter wavelengths is higher, these gain more importance even if the total ratio within the incident UV spectrum is small. Chaney et al46 provided a re-evaluation of the UV hazard function, ie, S(λ). This establishes a photobiologic action spectrum that describes the relative effectiveness of different wavelengths in causing a photobiologic effect (relative effectiveness of different wavelengths to elicit a biologic response).33 This function has been used for 30 years in the ophthalmic industry and in standards and norms for transmission. Dosimetric concepts are explained in more detail in a paper by Sliney.25
UV burden with lenses
UV transmission is blocked by most spectacle lenses. Present standards for ophthalmic and solar lenses, such as the Short Wavelength Technical Report International Standard ISO 8980-3,47 are defined for UV transmission (TUV) only. In this standard, the UV performance calculation is carried out using two functions:
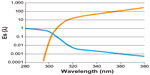
- direct sun radiation spectrum Es(λ) received at the earth’s surface, with a small amount of UV-B compared with UV-A (Figure 6, orange line)
- relative efficiency spectral function S(λ)46 or “function of UV risk”, which shows that UV-B is more dangerous than UV-A. This S(λ) function expresses the biologic risk linked to photochemical deterioration of the cornea when exposed to UV (Figure 6, blue line, arbitrary units).
Therefore, we have applied these functions to evaluate transmission TUV using the formula, hereafter, T(l) being the transmission of a lens and dl being the spectral interval (“l” reads lambda):
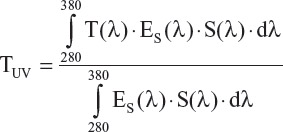
To assess UV reflection (RUV) (Figure 7) based on the current ISO standard, an eye UV exposure function has to be taken into account (Figure 7), R(l) being the back reflectance of a lens (“l” reads lambda):
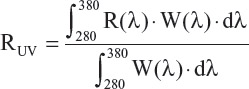
with W(λ) = Es(λ) × S(λ), Eye UV exposure function.
Absorption and transmission within the eye
The damage potential, absorption, and transmission of UVR within structures of the eye are crucial factors to assess. Hoover48 computed a mathematical model to determine solar UVR and particularly spectral transmittance of ocular media.
UVR incident at the cornea level is 100% absorbed by the cornea at wavelengths below 280 mm.25,49,50 In UV spectra shorter than 310 nm, the epithelium and Bowman’s layer have significantly higher absorption coefficients than does the stroma, and absorption is greater in the peripheral cornea than in the center.48,49 The crystalline lens of the adult absorbs almost all incident energy to wavelengths of nearly 400 nm. The absorptive peak of the lens changes with age, ie, at age 65 years, peak absorption is at 450 nm, and at age 8 years, peak absorption is at 365 nm.48 In newborns and children, a small transmission window occurs at around 320 nm, remaining until the age of 10 years (Table 1). Remarkable interindividual differences have been shown in post mortem eyes, for example, with the eye of a 24-year-old showing a pronounced window at 330 nm.50 Weale showed that a significant rise in absorbance occurs throughout the UV-A spectrum.51
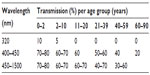  | Table 1 Ultraviolet transmission (cornea, aqueous, and lens) according to age group |
UV hazard to structures of the eye
Animal models cannot be translated directly into a similar risk in humans, since characteristics of animal and human eyes differ, for example, in lens characteristics or in UV absorption potential of the cornea, with the rodent cornea absorbing UV only below 300 nm. Despite their limitations, animal studies and human epidemiologic studies pointing in the same direction build a strong basis for sufficiently reliable correlations. Table 2 shows correlations that have been agreed upon in numerous studies, even for UVR alone. Chromophores are abundantly present in DNA, thus rendering it sensitive to mutagenesis.49 One role of UVR is attributed to damaging germinal epithelial cells such as corneal or lens cells by interference with DNA.
  | Table 2 Eye diseases having a correlation with UVR |
Acute UV-induced damage
Acute UVR at sufficiently high doses induces acute photokeratitis, welder’s flash, or arc eye.49 Photokeratitis following short wave UVR is easy to demonstrate, for example, by comparison with eyes protected by UV blocking contact lenses. Photokeratitis induced by irradiation with 180–400 nm UVR is a temporary photochemical injury to corneal cells. It presents with ocular pain several hours after acute exposure, with symptoms lasting for 24–48 hours. Laboratory studies have shown damage to keratinocytes and epithelial cells of the cornea, with strong healing properties. Acute responses to above-threshold doses are epithelial cell death and related conjunctival trauma.53 In rare cases, higher energy exposure can lead to permanent endothelial cell damage. The subclinical photokeratitis level is approximately 30–40 J/m2 normalized to the UV hazard (photokeratitis) action spectrum peak of 270 nm (as defined by American Conference of Governmental Industrial Hygienists and International Commission on Non-Ionizing Radiation Protection and stated in EU Directive 2006/25/EC). The radiant exposure at 300 nm that would be equivalent to the corneal exposure of 30–40 J/m2 at 270 nm is 100 J/m2. UVR exposure can also induce photoconjunctivitis (irritation, swelling, redness, pruritus), and eyelid sunburn.
Chronic UV-induced damage
On an individual and epidemiologic basis, chronic UV exposure is of much higher relevance than is acute UV-induced damage. Several eye diseases have been attributed to UVR, mainly based on epidemiologic and animal studies.56-58
Cornea: pterygium, pinguecula, climatic droplet keratopathy
The cornea is most exposed to UVR, absorbing the greatest part, not only from direct irradiation but also from oblique rays arriving at an angle of up to 110°, which are then reflected across the cornea and anterior chamber into the limbal area. Absorbing UVR puts these structures at risk. In climatic droplet keratopathy, translucent material characteristically accumulates in the corneal stroma, most prominently in the band between the lids. The incidence is greater in regions high in UVR and in persons who spend considerable time outdoors; this is attributed to UV exposure inducing denaturation of plasma proteins.58
Outdoor work is a recognized factor for development of pterygium.59–62 Pterygium was found to be almost twice as frequent among persons who work outdoors, but only one fifth as likely among those who always use sunglasses outdoors.61 In areas with higher ambient UV burden (close to the equator,63,64 higher altitude84), and in regions with higher ground reflectivity, prevalence is higher.65–67
The more common site for pterygium is the nasal conjunctiva. Coroneo explained this (as well as the advent of typical cataract “spokes” in the nasal quadrant of the lens) by incident oblique rays arriving at the peripheral cornea that are focused at this very predilective site.28,30
UV-B radiation induces oxidative stress in corneal epithelial cells, and upregulation of proinflammatory cytokines has been shown.68 In a first step, pinguecula develop, followed later by pterygia which can reduce vision and often necessitate surgery.
Dry eye disease
Tears absorb incident UV and contain antioxidants. With decreasing tear film production in older individuals, a reduced antioxidant supply as well as UV-absorbing properties could predispose to more severe UV corneal damage.69
Cortical cataract
The human lens absorbs near UV and far infrared light (<400 nm and >800 nm, respectively). It is known that UV light induces cataract,70–82 with a damage threshold at 350 nm of 60 mJ/cm2. The corresponding value for 310 nm is 0.75 mJ/cm2. An association between sunlight exposure and cataract formation has been suggested for over a century. Of interest in this article is the contribution of UV, especially UV-B as being biologically more harmful.
UV cataract can be generated in animals and in vitro, with a solid body of evidence70 to show an association between UV exposure and cataract formation. Underlying photochemical processes that change the molecular composition of the lens have been described.71,72
Human studies followed three types of study designs, ie, geographic correlation studies, cross-sectional prevalence studies, and case-control studies.74–79 Geographic studies assess the UV burden of the region of residency, but cannot rule out confounding factors that are explained by the same geographic localization, such as visible light intensity and diet.80–82 In cross-sectional studies, participants with cataract are assessed at a single time point and questioned as to their recalled exposure to UV-B. In a case-control study, patients with cataract are followed over time with some form of UV-reducing strategy compared with matched controls. Results of cross-sectional studies are shown in Table 3. The Chesapeake Bay study,78 which examined the dose relationship between UV-B and cortical cataract, demonstrated a 3.3-fold higher risk for men in the highest quartile of UV exposure. In the Beaver Dam study,79 which employed a similar questionnaire, a relationship was shown for men, but not for women, who in general had lower exposures to UV-B. Altogether, the magnitude of risk shown in the Chesapeake Bay study indicates a weak to moderate correlation. The weakness of correlation might be attributable to confounding factors.
  | Table 3 Results of cortical cataract studies assessing individual exposure |
In examining the prevalence of nuclear cataract, the same studies failed to show an association. There is evidence available to suggest that exposure to UV at younger ages predisposes to nuclear cataracts later in life.83
The Salisbury Evaluation Project assessed UV-B exposure in a population of 2,500 adults in Maryland by utilizing a short questionnaire. Ocular exposure to UV-B was relatively moderate, but an association between exposure and cortical opacity could be demonstrated.83 Contribution of lens region to cortical cataract severity was examined in a sample of lenses from this study. By digital recording, Abraham et al demonstrated a differential effect of UV light exposure on cataract rate across regions of the lens, with a most prominent effect in the lower regions of the lens in those with higher lifetime exposure.72
The amount of cataract attributable to UVR has been estimated in the Global Burden of Disease due to Ultraviolet Radiation Study.84 The authors concluded the cataract burden could be reduced by roughly 5% with appropriate UV protection (assuming 25% of cataracts are UV-related cortical cataracts), or even by higher rates if it is assumed that UV also contributes to the development of other forms of cataract. In Australia, cataract rates are 11.3% for the population over 40 years,82 and in the US, cataract rates of 17.2% for this population are reported.85 Cataract not only has disabling consequences, but is also linked with increased mortality. Mixed opacities with nuclear opacity were significantly associated with a two-fold increased 2-year mortality, independent of confounding factors.108
Sasaki et al showed the highest prevalence of early cortical cataract to be in the lower nasal quadrant in patients of widespread geographic residency (Singapore, Melbourne, Reykjavik).81 The risk for early cataract in the lower nasal quadrant compared with the upper temporal hemisphere was highest for patients in Singapore. Participants of different races in different regions showed marked differences, for example in age of onset, but at the same time, predilection sites were similar.
A recent study comparing mountain guides with age-matched controls showed significant differences in incidence of cortical cataract (31% versus 10%), with 5% having undergone cataract surgery at an average age of 59 years (versus 0% in controls). Pterygia and pinguecula were significantly more frequent than in the controls (9% versus 0% and 58% versus 22%, respectively).86
Taken together, these results and additional newer studies provide evidence for a relationship between UVR and cortical cataract formation.
Premature presbyopia
Little is known about the effects of UVR effects on presbyopia. Heat-induced denaturation of proteins in the crystalline lens is linked to reduced ability to focus and to cataract formation.109 A high incidence of presbyopia occurs at younger ages in countries with high levels of UV.87,88 Studies are underway to compare contact lens wear in a group with UV-blocking contact lenses and in a control group with only minimally UV-blocking contact lenses for a follow-up period of 5 years.89 It is hypothesized that UV damage can accelerate presbyopia.
Uveal melanoma
Cutaneous malignant melanoma is strongly associated with UV exposure (mostly UV-A), and the molecular signature of UV-induced DNA damage can be found in cutaneous melanoma.90,91 Such interaction could not be found for uveal melanoma, the molecular signature of which is different to that of cutaneous malignant melanoma.92 However, some studies indicate that UV radiation could be associated with a higher risk of iris melanoma in individuals having a light iris color.93
Age-related macular degeneration
The association between UVR and age-related macular degeneration remains controversial. By far the most important contributor to development of age-related macular degeneration is blue light. The amount of UV reaching the retina is very low in human adults. In general, different geometric factors (or not having them adequately incorporated into the hypotheses)37 and the strong influence of genetic factors contribute to the fact that epidemiologic studies looking for an association between UV burden and eye disease have produced mixed and inconsistent findings.
UV-related skin aging and periorbital skin diseases
UVR exerts acute and chronic effects on human skin. Acute responses of normal human skin represent a form of inflammation.85 Clinical manifestations of acute exposure include erythema, swelling, pain, and pruritus, while delayed reactions are increaed pigmentation and thickness of the epithelium. UV exposure of the skin also modulate immunologic recations and intervenes in Vitamin D synthesis.94
Chronic effects include photoaging and photocarcinogenesis. Photoaging is the term used to describe the clinical, histologic, and functional changes in chronically sun-exposed skin areas, including the periorbital skin. Clinical manifestations of photoaged skin include dryness, actinic keratosis, irregular pigmentation, lentigines, wrinkling, stellate pseudoscars, telangiectasia, and inelasticity. Since the periorbital region is not only highly exposed to the sun but is also visually apparent, the perception of an individual’s age is primarily influenced by the extent of his/her cutaneous photodamage.94–96
Mitochondrial DNA is a chromophore for UV-B and UV-A radiation, so the mitochondria of epidermal keratinocytes and dermal fibroblasts are subject to damage by UVR, leading to point mutations and large-scale deletions which are pathophysiologically causal for photoaging. Certain DNA deletions are increased by up to 10-fold in photoaged skin compared with sun-protected skin in the same individual.97 Studies support the hypothesis that mitochondrial DNA deletions are an important basis for premature aging at the organ level (photoaging).98
Another gene-independent mechanism of damage, ie, activation of the arylhydrocarbon receptor, has been shown to contribute in a relevant way to the UV-B stress response in human skin cells, activating pathways such as increased transcription of genes including cyclooxygenase-2. Photocarcinogenesis includes the development of actinic keratosis, squamous cell carcinoma, basal cell carcinoma, and malignant melanoma. DNA damage to keratinocytes is well demonstrated and an established risk factor for malignancies in the periorbital region. DNA is one of the most prominent chromophores that absorb UV-B.
Among malignant tumors, skin malignancies have the highest incidence. UVR is a risk factor for actinic keratoses (a premalignant condition), squamous cell carcinoma, basal cell carcinoma, and malignant melanoma. The periorbital region is small in absolute size, but 5%–10% of all nonmelanoma skin cancers occur on the eyelids (Figure 8).99 According to the Australian database for squamous cell carcinoma and basal cell carcinoma, lid tumors are mostly located at the lower lid in approximately 50% of cases (squamous cell carcinoma, 68%; basal cell carcinoma, 54%), and in the medial canthus (squamous cell carcinoma, 24%; basal cell carcinoma, 41%). Since nonmelanoma skin cancer is frequent (one in five Americans will develop a skin cancer during their lifetime), protection is crucial. Application of sunscreen to the lid area is poorly tolerated and impractical.100
  | Figure 8 Location of eyelid malignancies: percentages for 174 tumors. |
About 90% of nonmelanoma skin cancers are associated with exposure to UVR.50 In malignant melanoma, about 86% of cases are attributed to exposure to UVR.5
Current attempts at eye protection
Populations at risk
Sasaki et al suggest that UV protection of the eye should be provided full time throughout the day and year, given that the UV ocular burden might be higher than supposed by solar irradiation.8
Since there is a cumulative burden throughout life, UVR generally poses risks, but there are populations of special concern. In children, increased UV transmission to the retina has been shown since the lens is still clear and the pupil is wider than that in adults.16 This UV exposure can contribute to retinal stress with delayed consequences, ie, formation of lipofuscin, which might be a contributory factor in age-related macular degeneration.101
High ocular UV exposure is seen at higher altitudes, frequent outdoor work, and ample leisure time exposure. Individual factors, such as a thin cornea, larger pupil size, lid and orbital anatomy, hyperopia, aphakia (in the absence of a UV-filtering intraocular lens), and genetic predisposition play a role. Photosensitizing drugs, such as psoralens, phenothiazines, hydrochlorothiazide, porphyrins, nonsteroidal anti-inflammatory drugs, antiarrhythmics (cordarone/amiodarone), tetracyclines, chloroquine, and St John’s wort, increase the susceptibility to UVR damage.
Some contact lenses have been shown to reduce UVR markedly, although this differs between brands.40,102–104 Soft contact lenses have sufficient diameter (typically around 14 mm) to protect the limbal cells, but do not offer protection for the periorbital skin and lids.
Sunglasses reduce transmission, but may increase back reflection. Clear lenses also reduce transmission, but a universal scale to address their protective potential has been lacking. The European Council of Optometry and Optics states:
“The ideal UVR-blocker should transmit only visible radiation and block solar UVR incident on the eye from all directions.”19
Effectiveness of protective measures
Sun protection clearly reduces the incidence of skin cancer, but clear evidence for the benefits of sun protection for the eye is still lacking. One study showed a 3.6–4.6-fold increased risk of pterygium in persons rarely using sunglasses or hats.60Another study provided evidence for the usefulness of almost always wearing sunglasses; compared with rarely or never wearing hats or sunglasses, the risk for nuclear cataract was reduced. The failure to show more clearcut evidence might be due to not taking into account confounding factors such as back reflection or frame characteristics.77 In vitro methods for evaluating UV damage to ocular epithelial cells and the UV-filtering properties of ophthalmic materials on a biologic level have been tested.55 Human corneal epithelial cells, lens epithelial cells, and retinal pigment epithelial cells showed reduced viability after UVR in a dose-dependent manner. At 400 nm filter protection, cell viability was unchanged compared with nonirradiated cells; at 320 nm filter protection, cell viability was reduced, but less so than without any filtering. Confocal microscopy has been used to show dose-dependent UV exposure degradation of the nuclei and mitochondria in ocular cells, as well as Annexin V staining to document the number of apoptotic cells (decreasing after UVR).55
To assess the biologically beneficial effects of class 1 UV-blocking contact lenses, studies have been performed in post mortem human lenses and in a human lens epithelial cell line (HLE B-3). These models demonstrated protective effects regarding protein alterations, including enzymes, structural proteins, and the cytoskeleton, that are typically induced by UV-B.13
Eye-sun protection factor
For all the reasons discussed, we propose a comprehensive eye-sun protection factor (E-SPF®). It should be relevant for transmission, back reflectance, and protection of structures of the eye and periorbital skin. With a generally accepted E-SPF®, that would be used ideally by all lens producers, eye health care professionals and consumers will be able to compare the UVR-protective properties of lenses, be they clear prescription lenses, contact lenses, or sunglasses (prescription or nonprescription).
The clothing industry uses the ultraviolet protection factor, based mainly on transmission measurements for three classes of clothes (European Standard EN 13758).12,105
In the skin care industry, the sun protection factor (SPF) for sunscreen is now well established (ie, the COLIPA [European Cosmetics Trade Association] method,106 European Commission Recommendation [2006/647/EC]) and is determined either by in vitro or in vivo measurements. It is important to note that SPF labeling addresses UV-B protection only; if UV-A protection is claimed, the UV-A protection factor should be at least one third of the SPF value.107
Interestingly, the erythema action spectrum, ie, E(λ), used for sunscreen SPF calculation (CIE 1987, ECR [2006/647/EC]) is similar to the eye UV hazard function, ie, Sλ, for photokeratitis and acute cataract used in ISO Norm 8980-3 (transmittance specifications and test methods, spectacle lenses).47 Therefore, there is a strong rationale to address eye and skin protection with the same function.
For the eyewear industry, different approaches have been proposed to raise public awareness of a protection factor, mainly for sunwear plano lenses. However, these factors have remained either localized in use or have not gained widespread use, and sometimes have been difficult to understand. The Australian eye protection factor is based on the UV absorption efficiency of solar lenses (10 classes).12 The FUBI (Frame coverage, Ultraviolet, Blue and Infrared) system also considers frame parameters (coverage) in addition to transmission on solar lenses.11
For contact lenses, no protection factor has been established for lay public information. Recently, some contact lenses have been developed to provide increased UV protection by adding UV absorbers (class 1 and 2 contact lenses).13
To address the multifaceted field of UV hazard comprising UV absorption of clear and solar lenses as well as UV reflection by antireflective lenses,14 an E-SPF® has been developed. This E-SPF® is an intrinsic property of a lens.120 It is characterized by UV transmittance and the back reflectance properties of the lens in question.
As depicted in Figure 9, the transmission properties of a lens are more relevant under conditions in which the wearer is facing the sun, whereas UV reflectance becomes more important in a situation of back exposure in a solid angle between 135° and 150° from the sagittal plane.
To define eye sun protection, we propose an E-SPF® formula as follows:
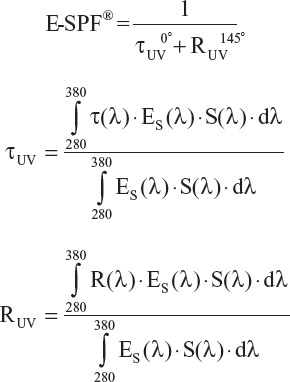
where
τ (λ) = transmission of the lens;
Es(λ) = spectral distribution for solar irradiation;
S(λ) = relative spectral distribution efficiency;
R(λ) = back reflectance of the lens;
τUV = weighted UV Transmission of the lens; and
RUV = weighted back reflectance of the lens.
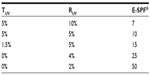
The E-SPF® is an intrinsic value of a lens based on physical measurements of the material itself, and allows direct comparison of the UV protection offered by different lenses on the market.
Table 4 shows that the E-SPF® obtained matches the sunscreen SPF labeling well, and could help the lay consumer to understand the level of protection provided by spectacles as with SPF sunscreen-labeling commonly used for skin protection.
The UV-A protection factor for the skin is also based on the relevant action spectrum (assessed by pigment darkening, PPD), the spectral range, ie, the spectrum of an UV-A lamp (SUV-A) and transmission T(λ).106 This skin-sun protection factor is validated and based on an in vivo measurement (pigment darkening), thus ascertaining biologic relevance.

As E-SPF® is based on transmission and back reflectance of the lens, it gives a clear indication of its intrinsic properties for the eye protection itself and subsequently for the periorbital area as well. Consumers and eye-care professionals have to be aware that additional factors, such as the spectacle frame, anatomic features of the individual, solar angle, reflection, and diffuse UVR which might enter the space between the frame and the eye, all play a role.
The E-SPF® does not intend to match the skincare sun protection factor because the UV shielding mechanisms are different. That is why a new label dedicated to eye protection is proposed to drive awareness, taking advantage of the already well known SPF.
The methods of determination are different. For the skin-sun protection factor, in vivo/in vitro UV-B or UV-A transmission of sun block creams applied directly onto the skin are considered (COLIPA method). For lenses, in vitro UV-A/B transmission and back reflectance of an antireflective lens which is not applied right onto the eye are taken into account for determination of E-SPF®. However, there is an analogy between the two methods, leading to the same order of magnitude in terms of value.
Future tasks and research agenda
Research should not only quantify the benefit of UV eye protection but also address the question of whether UV incident at the eye might initiate positive effects that should be maintained. For the skin, UVR plays an important role for vitamin D synthesis and in regulating important aspects of the immune system.
Vitamin D synthesis is not likely to be affected by protecting the eye against UVR, but the interaction between UVR and the immune system of the eye is unclear. The immune system of the eye has specific properties, ie, the eye is “immune-privileged”. Whether UVR plays a detrimental or protective role on the immune system of the eye is still unknown. A recent study showed upregulation expression of inflammatory and anti-oxydant mediators in corneal epithelial cells.68 However, at present, there is no evidence for beneficial effects of UVR on eye tissues comparable with the beneficial effects of UV irradiating skin.
The role of lens diameter or of the prescription itself (magnifying or dispersing effect linked to diopters), frame parameters and shape, different environmental reflections, including skin types, manner of wearing spectacles, morphotypes (eg, Caucasian, Asian, African), and further relevant questions should be addressed. Ray-trace modeling with associated radiometry and in vivo measurements are needed.
Models to assess the protective benefit of E-SPF® should be developed. For an educated guess, the minimal erythemal dose can be determined by positioning the lens over the skin. Skin protected by sunscreen will serve as a positive control. Further studies are possible using excised cornea and lenses.
Public education should be given more attention. Eye care professionals, health care professionals, and the industry should inform the lay public about the hazards of UVR and of the new possibility of quantifying a level of protection. A common belief is that all sunglasses protect the eyes from UV. There is also a need to better convey information on the UV-protecting properties of clear lenses. The public should understand better that protection is not only needed from direct sunlight but also from reflected radiation, even when wearing (sun) glasses and at times when sun glasses seem unnecessary. UV protection is particularly important in children of young age because of higher UV transmittance by the child eye structures.UV protection is particularly important in children of young age because of UV higher transmittance by the child eye structures. Similarities and differences between eye and skin protection should be explained (eg, time of the day, reflectance). Eye care professionals and dermatologists should increase cooperation to harmonize messaging and treatment strategies.
Conclusion
With increasing life expectancy and changing life habits (such as travelling, outdoor sports, and new artificial light sources), the cumulative effects of UVR in the periorbital region (ie, malignancies), at the cornea and conjunctiva (ie, pterygia) and lens are of increasing public health relevance. Protective measures should be put into place, given that the level of eye protection is generally too low, and sun protection considerations for the skin are not the same as those required for the eyes. For example, the UV burden of the eye might peak at hours of the day when the skin hazard is far lower. In order to have an impact on a population scale, standardization of an E-SPF® is desirable, not only for lay consumers, but also for eye health care professionals and for health care professionals in general.
The notion “wearing sun glasses protects against UVR” is incomplete or might even be misleading and should be replaced by a message that “clear lenses and/or sunglasses with high E-SPF® worn on a regular basis from childhood on provide useful UVR protection”. Despite considerable advantages and benefits which antireflective coatings offer, comprehensive measurements demonstrated that back reflection caused by some antireflective coatings of lenses might contribute to UV burden. The importance of improving assessments of UVR burden to the eye has been unequivocal. Additionally, surrogate parameters should be developed to better assess UV-protective properties and their benefit on an individual as well as on a population basis. This paper reports and encourages the universal use of an E-SPF® to deliver a unified, simple-to-understand message, and we encourage lens manufacturers to adhere to a shared standard.
Disclosure
Authors are either employees of Essilor, or have been consultants for Essilor in a group dedicated for writing this paper. The authors report no other conflicts of interest in this work.
References
Koh HK, Geller AC, Miller DR, Grossbart TA, Lew RA. Prevention and early detection strategies for melanoma and skin cancer. Current status. Arch Dermatol. 1996;132(4):436–443. | |
World Health Organization. [webpage on the Internet]. http://www.who.int/uv/uv_and_health/en/. Accessed August 28, 2013. | |
Diffey BL. Sunscreens as a preventative measure in melanoma: an evidence-based approach or the precautionary principle? Brit J Dermatol. 2009;161(3):25–27. | |
Parkin DM, Mesher D, Sasieni P. 13. Cancers attributable to solar (ultraviolet) radiation exposure in the UK in 2010. Brit J Cancer. 2011; 105(2):S66–S69. | |
Cook BE, Jr., Bartley GB. Treatment options and future prospects for the management of eyelid malignancies: an evidence-based update. Ophthalmology. 2001;108(11):2088–2098. | |
Bergmanson JPG, Ostrin LG, Walsh JE, Tschen J. Correlation between ultraviolet radiation exposure of the eyelids and location of skin cancer. Assoc Research Vision Ophthalmology. 2001;42(4):s335. | |
Pakrou N, Casson R, Fung S, Ferdowsi N, Lee G, Selva D. South Australian adolescent ophthalmic sun protective behaviours. Eye. 2008;22(6):808–814. | |
Sasaki H, Sakamoto Y, Schnider C, et al. UV-B exposure to the eye depending on solar altitude. Eye Contact Lens. 2011;37(4):191–195. | |
Bergmanson JP, Sheldon TM. Ultraviolet radiation revisited. The CLAO journal: official publication of the Contact Lens Association of Ophthalmologists, Inc. 1997;23(3):196–204. | |
Diffey BL. Human exposure to ultraviolet radiation. Seminars in Dermatology. 1990;9(1):2–10. | |
Hall GW, Schultmeyer M. The FUBI system for solar rating nonprescription eyewear. Optometry. 2002;73(7):407–417. | |
Gies PH, Roy CR, Toomey S, McLennan A. Protection against solar ultraviolet radiation. Mutation Res. 1989;422(1):15–22. | |
Andley UP, Malone JP, Townsend RR. Inhibition of lens photodamage by UV-absorbing contact lenses. Invest Ophthalmol Vis Sci. 2011;52(11):8330–8341. | |
Citek K. Anti-reflective coatings reflect ultraviolet radiation. Optometry. 2008;79(3):143–148. | |
Klein R, Klein BE, Jensen SC, Meuer SM. The five-year incidence and progression of age-related maculopathy: the Beaver Dam Eye Study. Ophthalmology. 199;104(1):7–21. | |
Reme C, Reinboth J, Clausen M, Hafezi F. Light damage revisited: converging evidence, diverging views? Graefe’s Arch Clin Experiment Ophthalmol. 1996;234(1):2–11. | |
van Norren D, Gorgels TG. The action spectrum of photochemical damage to the retina: a review of monochromatic threshold data. Photochem Photobiol. 2011;87(4):747–753. | |
Kolozsvari L, Nogradi A, Hopp B, Bor Z. UV absorbance of the human cornea in the 240- to 400-nm range. Invest Ophthalmol Vis Sci. 2002;43(7):2165–2168. | |
Bergmanson J, Walsh J, Söderberg P. European Council of Optometry and Optimcs. [webpage on the Internet]. Position Paper on Ocular Ultraviolet Radiation. Position Paper. 09/2009. http://www.ecoo.info/category/position-paper/. Accessed August 28, 2013. | |
International Commission on Illumination. International lighting vocabulary [French]. 4th edition Geneva, Switzerland: Bureau Central de la Commission Electrotechnique Internationale, 1987. CIE publication no. 17.4. | |
World Health Organization. [webpage on the Internet]. Sunbeds, tanning and UV exposure. Fact Sheet No 287, Interim revision April 2010. http://www.who.int/mediacentre/factsheets/fs287/en/. Accessed August 28, 2013. | |
Sliney DH. Ocular exposure to environmental light and ultraviolet—the impact of lid opening and sky conditions. Developments Ophthalmol. 1997;27:63–75. | |
Godar DE, Pope SJ, Grant WB, Holick MF. Part 2. Outdoor UV doses of adult Americans and vitamin D3 production. Dermatoendocrinol. 2011;3(4):243–250. | |
Godar DE, Pope SJ, Grant WB, Holick MF. Solar UV doses of young Americans and vitamin D3 production. Environ Health Perspect. 2012;120(1):139–143. | |
Sliney DH. Geometrical assessment of ocular exposure to environmental UV radiation—implications for ophthalmic epidemiology. J Epidemiol. Japan Epidemiological Association. 1999;9(6):S22–S32. | |
Bradley JC, Bentley KC, Mughal AI, Bodhireddy H, Brown SM. Dark-adapted pupil diameter as a function of age measured with the NeurOptics pupillometer. J Refract Surg. 2011;27(3):202–207. | |
Hoover HL. Sunglasses, pupil dilation, and solar ultraviolet irradiation of the human lens and retina. Applied Optics. 1987;26(4):689–695. | |
Coroneo MT. Pterygium as an early indicator of ultraviolet insolation: a hypothesis. Brit J Ophthalmol. 1993;77(11):734–739. | |
Coroneo M. Ultraviolet radiation and the anterior eye. Eye Contact Lens. 2011;37(4):214–224. | |
Coroneo MT, Müller-Stolzenburg NW, Ho A. Peripheral light focusing by the anterior eye and the ophthalmohelioses. Ophthalmic Surg. 1991;22(12):705–711. | |
Rosenthal FS, Bakalian AE, Lou CQ, Taylor HR. The effect of sunglasses on ocular exposure to ultraviolet radiation. Amer J Public Health. 1988;78(1):72–74. | |
Rosenthal FS, Bakalian AE, Taylor HR. The effect of prescription eyewear on ocular exposure to ultraviolet radiation. Amer J Public Health. 1986;76(10):1216–1220. | |
Sliney DH. Photoprotection of the eye—UV radiation and sunglasses. J Photochemistry Photobiology B. 2001;64(2–3):166–175. | |
Sakamoto Y, Kojima M, Sasaki K. [Effectiveness of eyeglasses for protection against ultraviolet rays]. Nippon Ganka Gakkai Zasshi. 1999;103(5):379–385. Japanese. | |
Willey RR. Practical Design and Production of Optical Thin Films. 2nd ed. New York: Marcel Dekker. 2002. 319–335 p. | |
World Health Organization. Global solar UV index–a practical guide. Geneva, Switzerland: WHO publications, 2002. Available from: URL http://www.who.int/uv/publications/fr/GlobalUVI.pdf. Accessed October 16, 2013. | |
Sliney DH. Risks of occupational exposure to optical radiation. La Medicina del lavoro. Mar–Apr 2006;97(2):215–220. | |
Sydenham MM, Collins MJ, Hirst LWS. Measurement of ultraviolet radiation at the surface of the eye. Invest Ophthalmol Vis Sci. 1997;38(8):1485–1492. | |
McLaren K, Watson W, Sanfilippo P, Collins MJ, Sydenham M, Hirst L. Contact lens dosimetry of solar ultraviolet radiation. Clin Experimental Optometry. 1997;80(6):204–210. | |
Walsh JE, Bergmanson JP, Wallace D, et al. Quantification of the ultraviolet radiation (UVR) field in the human eye in vivo using novel instrumentation and the potential benefits of UVR blocking hydrogel contact lens. Brit J Ophthalmol. 2001;85(9):1080–1085. | |
Kwok LS, Daszynski DC, Kuznetsov VA, Pham T, Ho A, Coroneo MT. Peripheral light focusing as a potential mechanism for phakic dysphotopsia and lens phototoxicity. Ophthalmic Physiol Opt. 2004;24(2):119–129. | |
Kwok LS, Kuznetsov VA, Ho A, Coroneo MT. Prevention of the adverse photic effects of peripheral light-focusing using UV-blocking contact lenses. Invest Ophthalmol Vis Sci. 2003;44(4):1501–1507. | |
Rosenthal FS, Safran M, Taylor HR. The ocular dose of ultraviolet radiation from sunlight exposure. Photochem Photobiol. 1985;42(2):163–171. | |
Rosenthal FS, Phoon C, Bakalian AE, Taylor HR. The ocular dose of ultraviolet radiation to outdoor workers. Invest Ophthalmol Vis Sci. 1988;29(4):649–656. | |
Sliney DH. Ultraviolet radiation effects upon the eye. Problems of dosimetry. Radiation Protection Dosimetry. 1997;72(3–4):197–206. | |
Chaney EK, Sliney DH. Re-evaluation of the ultraviolet hazard action spectrum—the impact of spectral bandwidth. Health Physics. 2005;89(4):322–332. | |
ISO standard 8980-3:2013. Ophthalmic optics, uncut finished spectacle lenses. Part3: Transmittance specifications and test methods. http://www.iso.org/iso/home/store/catalogue_tc/catalogue_detail.htm?csnumber=51052. Accessed October 18, 2013. | |
Hoover HL. Solar ultraviolet irradiation of human cornea, lens, and retina: equations of ocular irradiation. Applied Optics. 1986;25(3):359–368. | |
Behar-Cohen FMC, Viénot F, Zissis G, et al. Light-emitting diodes (LED) for domestic lighting: Any risks for the eye? Progress Retinal Eye Research. 2011;30:239–257. | |
Sliney DH. How light reaches the eye and its components. Int J Toxicol. 2002;21(6):501–509. | |
Weale RA. Age and the transmittance of the human crystalline lens. J Physiology. 1988;395:577–587. | |
Bachem A. Ophthalmic ultraviolet action spectra. Am J Ophthalmol. 1956;41(6):969–975. | |
Yam JC, Kwok AK. Ultraviolet light and ocular diseases. Int Ophthalmol. May 31, 2013. | |
Cullen AP. Ozone depletion and solar ultraviolet radiation: ocular effects, a United Nations environment programme perspective. Eye Contact Lens. 2011;37(4):185–190. | |
Youn HY, McCanna DJ, Sivak JG, Jones LW. In vitro ultraviolet-induced damage in human corneal, lens, and retinal pigment epithelial cells. Molecular Vision. 2011;17:237–246. | |
Young RW. The family of sunlight-related eye diseases. Optometry Vis Sci. 1994;71(2):125–132. | |
Lindgren G, Diffey BL, Larko O. Basal cell carcinoma of the eyelids and solar ultraviolet radiation exposure. Brit J Ophthalmol. 1998;82(12):1412–1415. | |
Gray RH, Johnson GJ, Freedman A. Climatic droplet keratopathy. Surv Ophthalmol. 1992;36(4):241–253. | |
Coroneo MT, Di Girolamo N, Wakefield D. The pathogenesis of pterygia. Curr Opin Ophthalmol. 1999;10(4):282–288. | |
Lu P, Chen X, Kang Y, Ke L, Wei X, Zhang W. Pterygium in Tibetans: a population-based study in China. Clin Experiment Ophthalmol. 2007;35(9):828–833. | |
Luthra R, Nemesure BB, Wu SY, Xie SH, Leske MC, Barbados Eye Studies Group. Frequency and risk factors for pterygium in the Barbados Eye Study. Arch Ophthalmol. 2001;119(12):1827–1832. | |
Shiroma H, Higa A, Sawaguchi S, et al. Prevalence and risk factors of pterygium in a South western island of Japan: the Kumejima Study. Am J Ophthalmol. 2009;148(5):766–771. | |
Moran DJ, Hollows FC. Pterygium and ultraviolet radiation: a positive correlation. Brit J Ophthalmol. 1984;68(5):343–346. | |
Heriot WJ, Crock GW, Taylor R, Zimmet P. Ophthalmic findings among one thousand inhabitants of Rarotonga, Cook Islands. Austral J Ophthalmol. 1983;11(2):81–94. | |
Taylor HR. A historic perspective of pterygium. In: Taylor HR, editor. Pterygium. The Hague: Kugler Publications; 2000. | |
Norn M. Spheroid degeneration, keratopathy, pinguecula, and pterygium in Japan (Kyoto). Acta Ophthalmol. 1984;62(1):54–60. | |
Norn MS. Prevalence of pinguecula in Greenland and in Copenhagen, and its relation to pterygium and spheroid degeneration. Acta Ophthalmol. 1979;57(1):96–105. | |
Black AT, Gordon MK, Heck DE, Gallo MA, Laskin DL, Laskin JD. UVB light regulates expression of antioxidants and inflammatory mediators in human corneal epithelial cells. Biochem Pharmacol. 2011;81(7):873–880. | |
Choy CK, Cho P, Benzie IF. Antioxidant content and ultraviolet absorption characteristics of human tears. Optometry Vision Sci. 2011;88(4):507–511. | |
Hockwin O, Kojima M, Sakamoto Y, et al. UV damage to the eye lens: further results from animal model studies: a review. J Epidemiol Japan Epidemiol Assoc. 1999;9(6):S39–S47. | |
Varma SD, Kovtun S, Hegde KR. Role of ultraviolet irradiation and oxidative stress in cataract formation—medical prevention by nutritional antioxidants and metabolic agonists. Eye Contact Lens. 2011;37(4):233–245. | |
Abraham AG, Cox C, West S. The differential effect of ultraviolet light exposure on cataract rate across regions of the lens. Invest Ophthalmol Vis Sci. 2010;51(8):3919–3923. | |
McCarty CA, Taylor HR. A review of the epidemiologic evidence linking ultraviolet radiation and cataracts. Dev Ophthalmol. 2002;35:21–31. | |
Dolin PJ. Ultraviolet radiation and cataract: a review of the epidemiological evidence. Brit J Ophthalmol. 1994;78(6):478–482. | |
Sasaki K, Sasaki H, Kojima M, et al. Epidemiological studies on UV-related cataract in climatically different countries. J Epidemiol. / Japan Epidemiological Association. 1999;9(6 Suppl):S33–S38. | |
The Italian-American Cataract Study Group. Risk factors for age-related cortical, nuclear, and posterior subcapsular cataracts. Am J Epidemiol. 1991;133(6):541–553. | |
West SK, Duncan DD, Munoz B, et al. Sunlight exposure and risk of lens opacities in a population-based study: the Salisbury Eye Evaluation project. J Am Med Assoc. 1998;280(8):714–718. | |
Taylor HR, West SK, Rosenthal FS, et al. Effect of ultraviolet radiation on cataract formation. New Engl J Med. 1988;319(22):1429–1433. | |
Cruickshanks KJ, Klein BE, Klein R. Ultraviolet light exposure and lens opacities: the Beaver Dam Eye Study. Am J Public Health. 1992;82(12):1658–1662. | |
Mitchell P, Cumming RG, Attebo K, Panchapakesan J. Prevalence of cataract in Australia: the Blue Mountains eye study. Ophthalmology. 1997;104(4):581–588. | |
Sasaki H, Kawakami Y, Ono M, et al. Localization of cortical cataract in subjects of diverse races and latitude. Invest Ophthalmol Vis Sci. 2003;44(10):4210–4214. | |
McCarty CA, Mukesh BN, Fu CL, Taylor HR. The epidemiology of cataract in Australia. Am J Ophthalmol. 1999;128(4):446–465. | |
Neale RE, Purdie JL, Hirst LW, Green AC. Sun exposure as a risk factor for nuclear cataract. Epidemiology. 2003;14(6):707–712. | |
Lucas RM. An epidemiological perspective of ultraviolet exposure – public health concerns. Eye Contact Lens. 2011;37(4):168–175. | |
Congdon N, Vingerling JR, Klein BE, et al. Prevalence of cataract and pseudophakia/aphakia among adults in the United States. Arch Ophthalmol. 2004;122(4):487–494. | |
El Chehab H BJ, Herry JP, Chave N, et al. [Ocular phototoxicity and altitude among mountain guides.]. J Franc Ophtalmol. 2012. Epub 2012 November 12, 2012. | |
Olurin O. Refractive errors in Nigerians: a hospital clinic study. Ann Ophthalmol. 1973;5(9):971–976. | |
Burke AG, Patel I, Munoz B, et al. Population-based study of presbyopia in rural Tanzania. Ophthalmology. 2006;113(5):723–727. | |
Wolffsohn JS EF, Bartlett H, Sheppard A, et al. Does Blocking Ultra-Violet Light with Contact Lenses Benefit Eye Health? British Contact Lens Association Conference; 2012. May 2012; Birmingham. | |
Moan J, Porojnicu AC, Dahlback A.Ultraviolet radiation and malignant melanoma. Adv Exp Med Biol. 2008;624:104–16. doi: 10.1007/978–0-387–77574–6_9. | |
Mouret S, Forestier A, Douki T.The specificity of UVA-induced DNA damage in human melanocytes. Photochem Photobiol Sci. Jan 2012;11(1):155–62. doi: 10.1039/c1pp05185 g. Epub October 10, 2011. | |
Griewank KG, Murali R. Pathology and genetics of uveal melanoma. Pathology. 2013;45(1):18–27. | |
Schmidt-Pokrzywniak A, Jöckel KH, Bornfeld N, Sauerwein W, Stang A. Positive interaction between light iris color and ultraviolet radiation in relation to the risk of uveal melanoma: a case-control study. Ophthalmology. 2009;116(2):340–348. | |
Lim HW, Soter NA. Clinical photomedicine. New York: M. Dekker;1993. xv, 415. | |
Gilchrest BA, Krutmann J. Skin aging. Berlin, New York: Springer; 2006. xvii, 198. | |
Yaar M. Clinical and histological features of intrinsic versus extrinsic skin aging. In: Gilchrest BA, Krutmann J, eds. Skin Aging. Heidelberg: Springer; 2006. 9–22. | |
Krutmann J, Schroeder P.Role of mitochondria in photoaging of human skin: the defective powerhouse model. J Investig Dermatol Symp Proc. 2009 Aug;14(1):44–49. doi: 10.1038/jidsymp.2009.1. | |
Schroeder P, Gremmel T, Berneburg M, Krutmann J. Partial depletion of mitochondrial DNA from human skin fibroblasts induces a gene expression profile reminiscent of photoaged skin. J Invest Dermatol. 2008;128(9):2297–2303. | |
Cook BE, Jr., Bartley GB. Epidemiologic characteristics and clinical course of patients with malignant eyelid tumors in an incidence cohort in Olmsted County, Minnesota. Ophthalmology. 1999;106(4):746–750. | |
Malhotra R, James CL, Selva D, Huynh N, Huilgol SC. The Australian Mohs database: periocular squamous intraepidermal carcinoma. Ophthalmology. 2004;111(10):1925–1929. | |
Gaillard ER, Merriam J, Zheng L, Dillon J. Transmission of light to the young primate retina: possible implications for the formation of lipofuscin. Photochem Photobiol. 2011;87(1):18–21. doi: 10.1111/j.1751–1097.2010.00837.x. Epub November 19, 2010. | |
Walsh JE, Bergmanson JP. Does the eye benefit from wearing ultraviolet-blocking contact lenses? Eye Contact Lens. 2011;37(4):267–272. | |
Walsh JE, Bergmanson JP, Saldana G, Jr., Gaume A. Can UV radiation-blocking soft contact lenses attenuate UV radiation to safe levels during summer months in the southern United States? Eye Contact Lens. 2003;29(1):S174–S179; discussion S90–S91, S92–S94. | |
Chandler H. Ultraviolet absorption by contact lenses and the significance on the ocular anterior segment. Eye Contact Lens. 2011;37(4):259–266. | |
Gies P, Roy C, McLennan A, et al. Ultraviolet protection factors for clothing: an inter-comparison of measurement systems. Photochem Photobiol. 2003;77(1):58–67. | |
Matts PJ, Alard V, Brown MW, Ferrero L, Gers-Barlag H, Issachar N, Moyal D, Wolber R. The COLIPA in vitro UVA method: a standard and reproducible measure of sunscreen UVA protection. Int J Cosmet Sci. 2010;32(1):35–46. | |
Commission Recommendation of 22 September 2006 on the efficacy of sunscreen products and the claims made relating thereto. Notified under document number C(2006) 4089. EUR-Lex, Official Journal of the European Union. 2009;49: Legislation 265/39. http://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2006:265:0039:0043:EN:PDF. Accessed October 18, 2013. | |
West SK, Munoz B, Istre J, et al. Mixed lens opacities and subsequent mortality. Arch Ophthalmol. 2000;118(3):393–397. | |
Truscott RJ. Presbyopia. Emerging from a blur towards an understanding of the molecular basis for this most common eye condition. Exp Eye Res. 2009 Feb;88(2):241–247.Epub 2008 Jul 15. |
 © 2013 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2013 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.