Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 10 » Issue 1
Triple therapy with salmeterol/fluticasone propionate 50/250 plus tiotropium bromide improve lung function versus individual treatments in moderate-to-severe Japanese COPD patients: a randomized controlled trial – Evaluation of Airway sGaw after treatment with tripLE
Authors Saito T, Takeda A, Hashimoto K, Kobayashi A, Hayamizu T, Hagan G
Received 5 June 2015
Accepted for publication 6 September 2015
Published 4 November 2015 Volume 2015:10(1) Pages 2393—2404
DOI https://doi.org/10.2147/COPD.S89948
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Richard Russell
Takefumi Saito,1 Akinori Takeda,2 Katsuji Hashimoto,3 Akihiro Kobayashi,4 Tomoyuki Hayamizu,5 Gerald W Hagan6
1Department of Respiratory Medicine, Ibarakihigashi National Hospital, Naka, Ibaraki, 2Center for Pulmonary Medicine, Asahikawa Medical Center, Asahikawa, Hokkaido, 3AMC Nishiumeda Clinic, Osaka, 4Biomedical Science Department, 5Medical Affairs Respiratory Department, GlaxoSmithKline KK, Tokyo, Japan; 6Independent Consultant, Marbella, Spain
Purpose: Triple therapy using salmeterol/fluticasone propionate (FP) and tiotropium bromide is commonly used to treat chronic obstructive pulmonary disease (COPD), but sparse efficacy data exist in COPD patients with fewer symptoms and with a lower dose of inhaled corticosteroid in Japanese patients. The effects of of salmeterol/fluticasone propionate 50/250 µg (SFC250) twice daily plus tiotropium 18 µg (TIO) once daily and individual treatments on lung function were compared.
Patients and methods: Fifty three Japanese COPD patients participated in this randomized, double-blind, double-dummy, Williams square design crossover study. Lung function was assessed by plethysmography and spirometry.
Results: The primary endpoint of postdose specific airway conductance area under the curve (AUC0–4h) on day 28 was significantly higher following SFC250 + TIO (0.854) compared with TIO (0.737, 15.8%) and SFC250 (0.663, 28.8%) alone. SFC250 + TIO significantly improved trough forced expiratory volume in 1 second from baseline versus TIO (0.161 L, P<0.001) and SFC250 (0.103 L, P=0.008). SFC250 + TIO significantly improved residual volume compared with TIO (P<0.001) and SFC250 (P=0.003) on day 28. Nonsignificant improvements were seen in trough inspiratory capacity, total lung capacity, and thoracic gas volume. There was no mean change seen in rescue medication.
Conclusion: Triple therapy using SFC250 + TIO was well tolerated and gave a greater improvement in bronchodilation compared with TIO and SFC250 alone in Japanese patients with COPD. There was improvement in few symptoms, but no mean change was seen in patient-reported outcomes measured by rescue medication use.
Keywords: COPD, fluticasone propionate, plethysmography, salmeterol, spirometry, triple therapy
Corrigendum for this paper has been published.
Introduction
The aims of chronic obstructive pulmonary disease (COPD) pharmacotherapy are to reduce symptoms, reduce the frequency and severity of exacerbations, and improve health status and exercise tolerance.1 Bronchodilators, such as long-acting β2 agonists (LABAs) and long-acting anticholinergics, are often prescribed as monotherapy. However, this strategy is now considered suboptimal, with patients remaining symptomatic.2,3
The combination of inhaled corticosteroid (ICS), fluticasone propionate (FP), and LABA, salmeterol, controls COPD exacerbations better than ICS alone.4–6 Tiotropium bromide 18 μg (TIO) is also proven to provide effective, 24-hour bronchodilation and symptom relief.4,7,8
Salmeterol/fluticasone propionate (SFC) and TIO are frequently coprescribed to treat COPD. Combining long-acting anticholinergics with LABA and ICS, the so-called “triple therapy”, is commonly used in clinical practice in Japan9 and is advocated by current guidelines for the treatment of severe COPD.1,3 The combined use of SFC 50/500 μg plus TIO has demonstrated a significantly greater effect on lung function compared with either of the treatments alone.4,10 However, SFC 50/500 μg is not licensed for treating COPD in Japan. Significantly greater improvements in lung function and changes to airway dimensions were seen with the lower FP dose used in SFC 50/250 μg (SFC250) when combined with TIO compared to TIO, SFC250, and salmeterol alone.11 Other studies have shown the same effects, but did not include a SFC alone arm to fully evaluate the contribution of TIO to the effect of the triple combination.9,12,13 Information about the effects on lung function after treatment with SFC250 plus TIO compared with either of the individual treatments is sparse.11 This is in contrast to the effect with SFC 50/500 μg plus TIO, which was demonstrated by Singh et al using both spirometry and body plethysmography to measure lung function in COPD.4
This study replicates the design of the Singh study with three important differences: 1) we recruited patients with a modified Medical Research Council (mMRC) score of 1 or more, making ours a study in a milder group of COPD patients in terms of symptoms, whereas Singh et al4 recruited patients with a mMRC score greater than 1; 2) we used 250 μg FP not 500 μg; and 3) our treatment period was 28 days not 14 days.
Data regarding the use of SFC250 plus TIO in Japan are limited. Therefore, our aim was to compare the effects of SFC250 twice daily plus TIO once daily with the individual treatments on lung function and volume in Japanese COPD patients to provide further evidence to support the effect of “triple therapy” for appropriate patients.
Patients and methods
Patient population
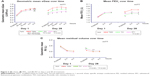
Figure 1 shows the study design with dosing regimen and period. Patients with COPD diagnosed according to current guidelines were recruited from February to June 2013 (last patient’s last visit: November 2013) by three centers across Japan. Patients were aged 40–80 years (inclusive) and had a smoking history of >10 pack years, postbronchodilator forced expiratory volume in 1 second (FEV1) ≥30% to ≤75% predicted normal, postbronchodilator FEV1/forced vital capacity (FVC) ratio <70%, and mMRC Dyspnoea Scale score ≥1.
  | Figure 1 Study design flow diagram. |
Exclusion criteria included a diagnosis of asthma; any respiratory disorder other than COPD; previous lung surgery; current pulmonary rehabilitation; regular long-term oxygen use ≥12 hours/day; exacerbation; use of parenteral, oral, or depot corticosteroids; change in COPD medication; use of antibiotics in the 4 weeks before screening; or hospitalization within the past year for an exacerbation.
All patients gave written informed consent. The study protocol was approved by the appropriate institutional review boards (Asahikawa Medical Center Institutional Review Board; ID# 12-b-1, Ibarakihigashi National Hospital Institutional Review Board, and AMC Nishi-umeda Clinic Institutional Review Board), and the study was conducted in accordance with good clinical practice guidelines and the Declaration of Helsinki 2008.
Study design
This was a randomized, double-blind, double-dummy, Williams square design crossover study.14 Patients entered a 2-week washout period having discontinued all existing COPD medications. Eligible patients were then randomized to one of six treatment sequences (Figure 1). Each patient received all three treatments of SFC250 twice daily plus TIO once daily, SFC250 twice daily, or TIO once daily, for 4 weeks. SFC250 was administered via Diskus™ inhaler (GlaxoSmithKline, Middlesex, UK), TIO via HandiHaler® (Boehringer Ingelheim, Ingelheim, Germany). Two-week washout periods separated each treatment period. After randomization, only salbutamol was allowed in addition to the study medication.
Lung function measurements were performed on days 1 and 28 of each treatment period, and salbutamol was withheld for 6 hours premeasurement. Follow-up was 2 weeks after the end of treatment or study withdrawal. Total study duration was 20 weeks.
Patients were randomly assigned to a treatment sequence by site staff using the randomization schedule from the validated GlaxoSmithKline Registration and Medication Ordering System. Treatment compliance was assessed at each posttreatment visit by recording the number of doses remaining in each Diskus™ and the number of capsules remaining for each HandiHaler®.
Assessments
Pulmonary function
Specific airway conductance (sGaw) and specific airway resistance (sRaw) were recorded pre- and postdose (30, 75, 120, and 240 minutes) on days 1 and 28 of each treatment period. FEV1, FVC, inspiratory capacity (IC), residual volume (RV), total lung capacity (TLC), and thoracic gas volume (TGV) were assessed pre- and postdose (120 and 240 minutes) on days 1 and 28 of each treatment period after sGaw was recorded. sGaw, FEV1, FVC, IC, and TGV were assessed using a constant volume plethysmograph (MasterScreen® Body, eResearch Technology GmbH, Philadelphia, PA, USA); sRaw, TLC, and RV were calculated from these parameters. Triplicate measurements were recorded and the mean was used for analysis.
Diary cards and symptoms
Patients completed daily record cards to record the number of occasions they used rescue medication over a 24-hour period between the first dose on that day and the first dose on the next day.
Safety
Adverse events (AEs) were documented at each visit; vital signs and 12-lead electrocardiograms were taken at screening only.
Statistical methods
The treatment comparisons were SFC250 + TIO versus TIO and SFC250 + TIO versus SFC250 and were considered of equal importance. Based on previous data,15 a 15% decrease in sGaw for the individual treatments compared with SFC250 + TIO was considered clinically relevant. To detect a 15% decrease in AUC0–4h sGaw between one individual treatment and SFC250 + TIO with 90% power, a two-sided 0.025 significance level and a within-patient standard deviation of 0.1652 (log transformed sGaw) and to ensure ≥90% power to detect 15% difference in sRaw using a standard deviation of 0.1725, 48 randomized patients were needed; so, 36 patients completed ≥2 treatment periods.
The modified intent-to-treat (mITT) population was the primary population for the efficacy analysis (Figure 2).
The primary efficacy endpoint was postmorning dose, AUC0–4h sGaw on day 28. sGaw AUC0–4h was log transformed and analyzed using a mixed model with treatment, period, baseline, and mean baseline included as fixed effects, and patient fit as a random effect. Analyses were adjusted for baseline (log-transformed predose value on day 1 of each treatment period) and mean baseline (mean of the three baselines for each patient) to account for the patient’s status at the beginning of each treatment. Denominator degrees of freedom were approximated and bias was corrected in the estimated variance–covariance matrix of the fixed effects.16
Treatment ratios for the comparison of SFC250 + TIO with TIO and SFC250 were calculated from the antilog of the difference between least square means. The 97.5% and 95% confidence intervals were calculated using pooled estimates of variance for the difference, and the antilog values were reported. Adjusted means and differences in logged data were converted to geometric mean ratios. An adjusted geometric mean ratio =1 indicated no difference between the treatments or time points compared; >1 indicated a positive difference, and <1 indicated a negative difference. The 97.5% confidence interval was used to draw conclusions regarding the relative efficacy of the treatments to account for the two equally important treatment comparisons, with a P-value of <0.025 being significant.
The 2-week washout period between treatment periods was considered sufficient to minimize carryover effects, so treatment by period interaction and carryover effects were not formally investigated.
Secondary end points, FEV1, FVC, IC, RV, TLC, TGV, and postdose sRaw and sGaw, were analyzed in a similar way to the primary efficacy parameter, although FEV1 and FVC were not log transformed prior to analysis. Analyses for days 1 and 28 were performed separately. The number of occasions salbutamol was used as rescue medication in a 24-hour period was recorded. All on-treatment AEs were summarized. Events with onset during the washout and follow-up periods were considered to have onset on the previously received treatment.
Results
Patient demographics and disposition
Patient demographic and baseline characteristics are summarized in Table 1. Of the 59 patients screened, 53 were randomized, of which 50 comprised the mITT population (Figure 2). During each treatment period, treatment compliance was >80% or <120%.
Efficacy
Pulmonary function
sGaw
AUC0–4h sGaw on day 28, following the postmorning dose of SFC250 + TIO, showed significant and clinically meaningful improvements over TIO (15.8%) and SFC250 (28.8%) alone. Ratios of SFC250 + TIO over TIO and SFC250 were statistically significant on days 1 and 28 (Table 2).
SFC250 + TIO also produced improvements in trough and postdose mean sGaw on days 1 and 28 at all timepoints (Figure 3A; Tables S1 and S2) with significant differences versus components on day 28. Changes in sRaw reflected those of sGaw (Tables 3, S1, S2).
Spirometry
SFC250 + TIO and its individual components improved mean FEV1 over time on days 1 and 28 (Figure 3B). On day 28, SFC250 + TIO provided significantly greater improvements in trough FEV1 compared with TIO and SFC250 (Table 3) and in postdose FEV1 (Tables S3 and S4). No significant treatment differences were seen on day 1 (Tables S3 and S4).
Lung volumes
On day 28, SFC250 + TIO provided a significantly greater improvement in trough RV but not in trough IC, TLC, or TGV compared with TIO and SFC250 (Table 3). On day 28, SFC250 + TIO produced improvements in postdose RV compared to both components (Figure 3C). Statistical significance was observed for postdose RV immediately after dosing (time =0) on day 28 for SFC250 + TIO compared to TIO and SFC250 (Tables S3, S4). Improvements on day 28 were also seen in postdose TGV, IC, and TLC following treatment with SFC250 + TIO compared to each component, but none were significant (Table S4). Improvements were seen for all parameters for their adjusted mean values on day 1, but none were significant (Table S3).
Diary cards and symptoms
Rescue medication usage was similar across all three groups (number of occasions per 24-hour period) for the 70% of patients in the mITT population who were mMRC =1; similar rescue medication usage was also seen in approximately 30% of patients with mMRC ≥2, although usage was numerically higher than that in the milder group (Table 4).
Safety
All treatments were well tolerated, with AEs reported by 33% (SFC + TIO), 16% (TIO), and 22% (SFC) of patients (Table 5). Two serious AEs were reported, both following SFC250 + TIO treatment, and were considered unrelated to the study treatment: prinzmetal angina (one patient) was of moderate intensity and lumbar spinal stenosis (one patient) was of mild intensity.
  | Table 5 Adverse events occurring in greater than 3% patients (safety population) |
Discussion
Repeat-dose SFC250 + TIO was more effective in improving sGaw compared to TIO or SFC250, with significant improvements observed on days 1 and 28 up to 4 hours postdose. These improvements were considered clinically relevant and met the primary aim of the study to increase sGaw AUC0–4h by 15%.
This was the first statistically powered study to be performed in Japanese patients with mild COPD investigating the effects of triple therapy on lung function using SFC250. The improvements we saw in sGaw were comparable with those observed in the Western population (22% improvement over TIO and 27% over SFC 50/500 μg alone) studied by Singh et al4 over 14 days.
Although previous studies have investigated the benefits of triple therapy in a Japanese COPD population,9 this study was the first to use whole-body plethysmography and to include an SFC250 arm to fully evaluate the contribution of TIO under blinded conditions. sGaw is a more sensitive measurement for detecting lung function changes in COPD patients than the more commonly used FEV1, therefore, a smaller sample size can be used in clinical trials.4,15,17,18 Plethysmography is not routinely used in clinical practice or studies due to its complexity, however, as it was used in the study conducted by Singh et al4 it was important to include it in our study to provide a comparison. It has been postulated that sGaw provides data on the large central airways.19–21 Therefore, to give a comprehensive assessment of lung function and lung volumes, we also included the more traditional measurements of FEV1 and FVC; these describe the more clinically relevant small airways. Improvements in these parameters reflected those of sGaw on day 28 and concurred with previous data using SFC25011 and SFC 50/500.4 This is consistent with our finding of a nonsignificant trend in the difference in sGaw between TIO and SFC250 by day 28 (but not day 1), although FEV1 changes were similar.
Day 28 trough RV, TGV IC, and TLC confirmed that triple therapy was more effective in enhancing lung volume and reducing lung hyperinflation than each individual component, and this was in agreement with previous data.4 SFC250 + TIO significantly improved trough RV compared to each component, which concurred with other data.4 TGV represented a novel aspect of our study not measured previously.4
Improvements were seen in all parameters on day 1 postdose, but significant differences between SFC250 + TIO and SFC250 or TIO alone were only observed for sGaw and its derivative, sRaw.
The majority of patients in this study were mMRC =1 (70%) and did not use rescue medication during the study while on active treatment. Although SFC250 + TIO is widely used, there is limited evidence to support this approach, particularly using FP 250 μg in milder COPD patients of Japanese origin. Published data largely advocate the use of triple therapy in Western COPD populations and using SFC 50/500.4,10,22–24 A parallel group, 16-week study compared the effects of SFC250 + TIO on lung function (FEV1, FVC, RV, TLC, IC, and functional residual capacity) with SFC250, TIO, and salmeterol. Triple therapy produced significantly greater improvements in FEV1, FVC, RV, and TLC versus TIO, salmeterol, and SFC250 (except FVC).11 However, the study was open label, which may have introduced bias, with a small sample size per arm, (range: 14–16) and body plethysmography was not utilized to measure lung function.11
Our study had limitations. It was powered to explore the effect of triple therapy on lung function rather than symptoms and was not designed to look at the effect on exacerbations. Adequately powered studies are now needed to explore the longer term effects of triple therapy, including the impact on symptoms and exacerbation rates. Our observations are relevant to a Japanese population with milder COPD rather than a particular subgroup or phenotype. ICS + LABA is recommended for patients who have risk of exacerbation (GOLD Group C/D); triple therapy is recommended for GOLD group D.1 However, our study was conducted in patients with a low risk of exacerbation to assess pulmonary function parameters as efficiently as possible: exacerbations can cause daily variation in COPD symptoms, including lung function. Furthermore, exacerbations may increase the risk of patient withdrawal from the study, which would impact outcome validity in a crossover study. The crossover design may also make the safety analysis of each treatment more difficult due to the possibility of carryover effects. Carryover effects on efficacy endpoints were considered unlikely and negligible, so a crossover design had the advantage that each patient was their own control. The study design could not establish when and how symptoms and exacerbations should be used to optimize treatment. A study to answer this question is ongoing.2
Conclusion
SFC250 + TIO treatment for 28 days led to greater improvements in bronchodilation compared with TIO or SFC250 and was well tolerated in Japanese COPD patients. The advantages of triple therapy were observed across a range of physiologically important parameters. Therefore, our study substantiates current knowledge that SFC250 + TIO is more effective than either TIO or SFC250 in improving lung function.
Acknowledgments
Funding for this study was provided by GSK (NCT01751113). All listed authors meet the criteria for authorship set forth by the International Committee for Medical Journal Editors. The authors wish to acknowledge the following individuals for their contributions to the clinical study and development of this paper: members of GSK team, Kazuyoshi Tenjinbaru, Yuko Amagasa, Yu Jincho, Miho Hashio, Ken Tanaka, Masato Tsukada, Hideki Hitosugi, members of the GSK monitoring team, eResearch Technology GmbH for their contribution to this study, including the system development of the body box, as well as data management of and associated staff training for all lung function tests. Editorial support in the form of development of draft outline, development of manuscript first draft, editorial suggestions to draft versions of this paper, assembling tables and figures, collating author comments, and referencing was provided by Kathryn White at Cathean Ltd and was funded by GSK.
Author contributions
Dr Takefumi Saito, Akinori Takeda, and Katsuji Hashimoto were each a principal investigator. Akihiro Kobayashi was a statistician for this study. Gerald W Hagan and Tomoyuki Hayamizu contributed to the study protocol development. All authors were involved in the acquisition, analysis of data, drafting, critical revision of the manuscript, and the final approval of the paper to be published.
Disclosure
GlaxoSmithKline KK (GSK) was the funding source and was involved in all stages of the study protocol development. GSK also funded all costs associated with the development, data analysis, and publishing of this paper. All authors took final responsibility for submitting this paper for publication.
Akihiro Kobayashi and Tomoyuki Hayamizu were employees of GSK at the time of the study conduct with no other relationships or activities that could appear to have influenced the submitted work. Takefumi Saito discloses having honoraria for speaking, and his hospital has received research grants from the commercial entity that sponsored the study. He has received speaking fee from Boehringer Ingelheim, Pfizer, MSD, Actelion, Asahi Kasei, Astellas, Abbott, Otsuka, Kyorin, Shionogi, Daiichi Sankyo, Taisho Toyama, Taiho, Takeda, Chugai, Eli Lilly, and Bayer. Akinori Takeda discloses having received honoraria for speaking, travel assistance, and consultancy fees from GSK for participating on the advisory board meeting, and his hospital has received research grants from the commercial entity that sponsored the study. He has received speaking fees from Boehringer Ingelheim, Astellas, AstraZeneca, Novartis, and Kyorin. Katsuji Hashimoto discloses that his clinic has received research grants from the commercial entity that sponsored the study. Gerald W Hagan discloses having received consultant fee and travel assistance from GSK and reports ownership of GSK shares. He has received consultancy fee from Almirall, Bayer, Merck, Mundipharma, Novartis, and Nycomed. He has previously worked for GSK. The authors report no other conflicts of interest in this work.
References
Global Initiative for Chronic Obstructive Lung Disease (GOLD). Revised ed 2011; 2014. Available from: http://www.goldcopd.org/uploads/users/files/GOLD_Report_2014_Jan23.pdf | ||
Betsuyaku T, Kato M, Fujimoto K, et al. A study to assess COPD symptom-based management and to optimise treatment strategy in Japan (COSMOS-J) based on GOLD 2011. Int J Chron Obstruct Pulmon Dis. 2013;8:453–459. | ||
The Japanese Respiratory Society. Guidelines for the Diagnosis and Treatment of COPD. 4th ed. Tokyo, Japan: Japanese Respiratory Society; 2013. | ||
Singh D, Brooks J, Hagan G, Cahn A, O’Connor BJ. Superiority of “triple” therapy with salmeterol/fluticasone propionate and tiotropium bromide versus individual components in moderate to severe COPD. Thorax. 2008;63(7):592–598. | ||
Bourbeau J, Christodoulopoulos P, Maltais F, Yamauchi Y, Olivenstein R, Hamid Q. Effect of salmeterol/fluticasone propionate on airway inflammation in COPD: a randomized controlled trial. Thorax. 2007;62(11):938–943. | ||
Johnson M. Corticosteroids: potential beta2-agonist and anticholinergic interactions in chronic obstructive pulmonary disease. Proc Am Thorac Soc. 2005;2(4):320–325. | ||
Littner MR, Ilowite JS, Tashkin DP, et al. Long-acting bronchodilation with once daily dosing of tiotropium (Spiriva) in stable chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2000;161(4 Pt 1):1136–1142. | ||
Casaburi R, Mahler DA, Jones PW, et al. A long-term evaluation of once-daily inhaled tiotropium in chronic obstructive pulmonary disease. Eur Respir J. 2002;19:217–224. | ||
Hoshino M, Ohtawa J. Effects of adding salmeterol/fluticasone propionate to tiotropium on airway dimensions in patients with chronic obstructive pulmonary disease. Respirology. 2011;16(1):95–101. | ||
Cazzola M, Andò F, Santus P, et al. A pilot study to assess the effects of combining fluticasone propionate/salmeterol and tiotropium on the airflow obstruction of patients with severe-to-very severe COPD. Pulm Pharmacol Ther. 2007;20(5):556–561. | ||
Hoshino M, Ohtawa J. Effects of tiotropium and salmeterol/fluticasone propionate on airway wall thickness in chronic obstructive pulmonary disease. Respiration. 2013;86(4):280–287. | ||
Jung KS, Park HY, Park SY, et al; for Korean Academy of Tuberculosis and Respiratory Diseases study group; Korea Chronic Obstructive Pulmonary Disease study group. Comparison of tiotropium plus fluticasone propionate/salmeterol with tiotropium in COPD: a randomized controlled study. Respir Med. 2012;106(3):382–389. | ||
Hanania NA, Crater GD, Morris AN, Emmett AH, O’Dell DM, Niewoehner DE. Benefits of adding fluticasone propionate/salmeterol to tiotropium in moderate to severe COPD. Respir Med. 2012;106(1):91–101. | ||
Williams EJ. Experimental designs balanced for the estimation of residual effects of treatments. Aust J Sci Res Ser A. 1949;2:149–168. | ||
Borrill ZL, Houghton CM, Tal-Singer R, et al. The use of plethysmography and oscillometry to compare long acting bronchodilators in patients with COPD. Br J Clin Pharmacol. 2008;65(2):244–252. | ||
Kenward MG, Rogers JH. Small sample inference for fixed effects from restricted maximum likelihood. Biometrics. 1997;53(3):983–997. | ||
Borrill Z, Houghton C, Woodcock AA, Vestbo J, Singh D. Measuring bronchodilation in COPD clinical trials. Br J Clin Pharmacol. 2005;59(4):379–384. | ||
van Noord JA, Smeets J, Clement J, Van de Woestijne KP, Demedts M. Assessment of reversibility of airflow obstruction. Am J Respir Crit Care Med. 1994;150(2):551–554. | ||
Larbonis A, Delwiche J-P, Jamart J, Vandenplas O. Comparison of FEV1 and specific airway conductance in assessing airway response to occupational agents. Allergy. 2003;58:1256–1260. | ||
Macklem PT, Mead J. Resistance of central and peripheral airways measured by a retrograde catheter. J Appl Physiol. 1967;22(3):395–401. | ||
Bouhuys A, Van De Woestijne KP. Respiratory mechanics and dust exposure in byssinosis. J Clin Invest. 1970;49(1):106–118. | ||
Villar B, Pombo V. Bronchodilator efficacy of combined salmeterol and tiotropium in patients with chronic obstructive pulmonary disease. Arch Bronconeumol. 2005;41(3):130–134. | ||
Aaron SD, Vandemheen KL, Fergusson D, et al; Canadian Thoracic Society/Canadian Respiratory Clinical Research Consortium. Tiotropium in combination with placebo, salmeterol, or fluticasone–salmeterol for treatment of chronic obstructive pulmonary disease: a randomized trial. Ann Intern Med. 2007;146(8):545–555. | ||
Short PM, Williamson PA, Elder DHJ, Lipworth SIW, Schembri S, Lipworth BJ. The impact of tiotropium on mortality and exacerbations when added to inhaled corticosteroids and long-acting β-agonist therapy in COPD. Chest. 2012;141(1):81–86. |
Supplementary materials
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.