Back to Journals » Clinical Ophthalmology » Volume 14
Treatment-Resistant Bacterial Keratitis: Challenges and Solutions
Authors Egrilmez S ![]() , Yildirim-Theveny Ş
, Yildirim-Theveny Ş ![]()
Received 16 August 2019
Accepted for publication 10 December 2019
Published 29 January 2020 Volume 2020:14 Pages 287—297
DOI https://doi.org/10.2147/OPTH.S181997
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Video abstract presented by Egrilmez S
Views: 1857
Sait Egrilmez,1 Şeyda Yildirim-Theveny2
1Private Office, Izmir, Turkey; 2Adiyaman University, Training and Research Hospital, Adiyaman, Turkey
Correspondence: Sait Egrilmez
Private Office, 1593/1 Sok. No: 4 B Blok D: 41 Mansuroglu Mah, Bayrakli, Izmir 35535, Turkey
Tel +90 505 4504765
Email [email protected]
Abstract: Bacterial keratitis is an important ophthalmic emergency and one of the most common causes of corneal blindness. The main causes of treatment resistance in bacterial keratitis are failure to eliminate predisposing factors, misdiagnosis and mistreatment. At first, exogenous, local and systemic predisposing factors that disturbing ocular surface must be eliminated to improve corneal ulcers and to prevent recurrences. Smears and scrapings for staining and culture are indispensable diagnostic tools for cases of sight-threatening keratitis (centrally located, multifocal, characterized by melting, painful). Main treatment agents in bacterial keratitis treatment are topical antibiotics. Until the results of culture antibiograms reach the ophthalmologist, empirical antibiotic selections based on direct microscopic examination and gram stain findings are the most appropriate initial treatment approach currently. S. aureus and coagulase-negative staphylococci (CoNS), the most common gram-positive agents, have resistance rates of more than 30% for fluoroquinolone and methicillin. Multidrug resistance rates are similarly high in these microorganisms. P. aeruginosa is the most common gram-negative micro-organism, in case of multidrug-resistant isolates, both functional and anatomical prognosis of the eyes are very poor. In cases of sight-threatening and resistant keratitis, antibiotic susceptibility testing containing imipenem, colistin, and linezolid is seeming to be an important requirement. Despite its efficiency limited to superficial cases, a nonpharmaceutical anti-infective treatment option such as corneal crosslinking for bacterial keratitis is an emerging hope, while antibiotic resistance increases.
Keywords: bacterial keratitis, antibiotic resistance, multidrug resistant, corneal crosslinking
Introduction
According to data from the Vision Loss Expert Group of the Global Burden of Disease study,1 36 million people are blind and 216.6 million people have moderate or severe visual impairment worldwide. Non-trachomatous corneal opacities are the fifth leading cause of vision loss, at a rate of 3.21%. However, when reversible vision losses such as cataract (35.15%) and uncorrected refraction defects (20.28%) are subtracted, the contribution of corneal opacities to vision loss is greater than twice the reported figure.1 Corneal infections are among the most common causes of corneal haze, and viral, bacterial, and fungal infections are the leading causes of microbial keratitis.2
Prevalence
Both the prevalence and etiologies of microbial keratitis differ substantially between developed and developing countries. Erie et al3 reported that the incidence of microbial keratitis in Minnesota rose from 2.5 per 100,000 in the 1950s to 11 per 100,000 in the 1980s. Jeng et al4 reported this rate to be 27.6 per 100,000 in Northern California in the years 1998–1999, with subgroup analysis yielding rates of 130.4 per 100,000 among contact lens users and 238.1 per 100,000 among HIV-positive individuals. Seal et al5 reported an incidence rate of 3.6 per 100,000 in Scotland in 1995, while Ibrahim et al6 reported a rate of 40.3 per 100,000 in Portsmouth, England in 2006. The increase in microbial keratitis in developed countries has been linked to the widespread use of contact lenses.7
The rate of microbial keratitis is much higher in developing countries, where access to health services is limited and risky occupations such as farming, and agriculture are more prevalent. Microbial keratitis has been reported at rates of 113 per 100,000 in Madurai8 (Tamil Nadu, India) and 799 per 100,000 in Nepal.9
A review by Ung et al7 who conducted an etiological study including the three most extensive studies of each continent, points to a very important conclusion about the etiology of microbial keratitis. In all of the 15 studies representing North America, South America, Europe, the Middle East and Africa, Europe, and Oceania, most cases of microbial keratitis were bacterial in origin, whereas fungal cases outnumbered bacterial cases in four of the six studies representing South Asia and East Asia.
This article will examine bacterial keratitis, one of the major ophthalmic emergencies, with emphasis on treatment resistance.
The main causes of treatment resistance in bacterial keratitis are:
- Inability or failure to eliminate predisposing risk factors
- Misdiagnosis
- Drug resistance and drug toxicity (mistreatment)
Risk Factors
The most common pathogens associated with bacterial keratitis are Staphylococcus aureus, Staphylococcus epidermidis, Streptococcus pneumoniae, and Pseudomonas aeruginosa.2,7 However, these agents are not capable of causing infection in a healthy cornea. A healthy corneal epithelium is a protective barrier against bacterial infections, with the exception of a few bacterial species such as Neisseria, Corynebacterium, Haemophilus, and Listeria species.2 Infections usually occur as a result of conditions that disrupt this important barrier, which are outlined below:2,10
1. Exogenous local factors
- Contact lens usage (overnight wear, inadequate disinfection, use of contact lenses without a doctor’s prescription and follow-up, or swimming, using a hot tub, or showering while wearing contact lenses)
- Topical corticosteroids, topical nonsteroidal anti-inflammatory drugs
- Topical antibiotics and glaucoma medications (chronic and/or multiple ophthalmic drug usage containing preservatives, especially benzalkonium chloride)
- Past ocular trauma (corneal abrasions or epithelial defects)
- Past ocular surgery (filtering bleb, glaucoma tube exposure, loose or broken corneal sutures, etc.)
2. Endogenous local factors and conditions
Eyelid disorders (lagoftalmus, ectropion, entropion, blepharitis, trichiasis, distichiasis)
Lacrimal disorders (dry eye, dacryocystitis)
- Conjunctival disorders (vernal keratoconjunctivitis, chemical burn, multiple conjunctival surgery, xerophthalmia, ocular pemphigoid)
- Corneal disorders (bullous keratopathy, neurotrophic keratopathy, herpetic eye diseases, and past corneal surgeries including keratoplasty, corneal-crosslinking, and keratorefractive surgery)
3. Systemic risk factors
- Diabetes mellitus
- Debilitating illness, immunocompromised state, alcoholism, coma
- Connective tissue diseases
- Dermatological/mucous membrane disorders (Atopy, Stevens-Jonhson syndrome, pemphigus)
- Acoustic neuroma or neurological surgery causing damage to the fifth and/or seventh cranial nerves
- Graft-versus-host disease
Identifying the above risk factors through medical history, examination, testing, and appropriate consultations and managing them correctly will not only make treatment possible but also help prevent recurrence.
The Challenge of Diagnosis
The main signs and symptoms of bacterial keratitis are pain, redness, blurred vision, discharge, corneal infiltrates, ulcerations, photophobia, and anterior chamber inflammation. These symptoms and findings should be supported by medical history, and all of the above-mentioned risk factors should be evaluated by physical examination and slit-lamp examination.
Suppurative stromal infiltrates greater than 1 mm in diameter and located in the central cornea are suggestive of bacterial keratitis.2 These infiltrates have indistinct edges and are usually accompanied by edema and leukocyte infiltration. There is typically an epithelial defect, and anterior chamber reaction is also common.2,10
Pain, rapid stromal thinning, and descemetocele should immediately suggest a Pseudomonas infection. Yellow-green purulent discharge and ground-glass appearance and loss of transparency in the adjacent corneal stroma are other important clinical features suggesting Pseudomonas.2 In cases of staphylococcal keratitis, the corneal ulcer is usually round and circumscribed by an epithelial defect, with minimal edema in the adjacent cornea.2 Streptococcus pneumoniae can cause serpiginous ulcers that infiltrate and span the cornea.2
Cornea specialists can distinguish bacterial keratitis from fungal keratitis based on biomicroscopic appearance. In a study based on 80 photographs of keratitis cases (40 bacterial, 40 fungal) in which the etiological agent was confirmed by direct culture, Dalmon et al11 found that 15 cornea specialists distinguished bacterial keratitis from fungal keratitis at a rate of 66%, correctly predicted Gram staining results at a rate of 46%, and predicted genus and species at rates of 25% and 10%, respectively. Although bacterial/fungal differentiation was better than by chance, predictions of Gram staining, genus, and species were very low. In routine practice, the predominant approach to community-acquired bacterial keratitis is empirical antibiotic therapy without obtaining smears or culture. However, clinical findings are not sufficient for the diagnosis of microbial keratitis and staining and culture remain the gold standard.
When are Smears and Culture Necessary?
In the Preferred Practice Pattern report for bacterial keratitis issued by the American Academy of Ophthalmology (AAO) in 2018, smears and culture are specifically recommended in the following cases:10
- If the infiltrate is centrally located, with large and/or significant stromal involvement or melting
- If infection is unresponsive to chronic or broad-spectrum antibiotic therapy
- If there is a previous history of corneal surgery
- If atypical clinical findings suggestive of fungal, amoebic, or mycobacterial infection are present (eg, eye pain severe enough to disrupt or prevent sleep)
- If infiltrates are multifocal
In addition, smears and culture were also found to be helpful in cases with history of injury by plant-based materials or swimming/using a hot tub while wearing contact lenses.10
Sample Collection for Smears and Culture
While conjunctival smears can be collected without anesthetic drops, corneal smears usually require topical anesthetic. Local anesthetic options that do not contain preservatives should be preferred in order to increase the likelihood of growth in culture, and tetracaine should be avoided due to its antimicrobial effect.10 The sample can be obtained under a biomicroscope or surgical microscope with a flame-sterilized spatula (Kimura), 25-gauge needle, or sterile blades. Scraping should begin slightly outside the border of the infectious focus and deepen in the infiltrate zone so as to reach the ulcer surface.2 Because the middle of the purulent material consists predominantly of inflammatory cells, samples collected from the region near the infiltrate edge will be more helpful in capturing the causative agent.2,10
Staining for Bacterial Keratitis
Staining a slide prepared from the swab material with appropriate dyes, examining it under a microscope is important in terms of rapidly detecting the agent and demonstrating its presence even in a patient receiving antibacterial therapy. The following three stains, found in almost every hospital, will suffice for bacterial keratitis:
- Gram staining: It distinguishes bacterial keratitis from fungal keratitis with higher accuracy, allows the recognition of amoebae, and distinguishes between gram-negative and gram-positive bacteria.12
- Giemsa staining: Demonstrates bacteria, fungi, Chlamydia, and Acanthamoeba.
- Acid-fast staining: Enables visualization of Mycobacterium and Nocardia.
All three dyes are readily available in hospitals and rapid; Gram staining is completed in 5 mins, Giemsa staining in 2 mins, and acid-fast staining in 1 hr.10
Diagnostic Methods Other Than Staining
Although not as common as the above-mentioned methods, options such as in vivo confocal microscopy and molecular diagnostic techniques such as polymerase chain reaction (PCR) can also be used for direct inspection in centers with the necessary facilities. In addition to bacteria and fungi, in vivo confocal microscopy allows the direct observation of parasites like Acanthamoeba that are not routinely cultured.10 In vivo confocal microscopy has grown in popularity in recent years because of its speed and high sensitivity in detecting larger organisms such as filamentous fungus, Acanthamoeba, and Nocardia bacteria.
Why Is Culture Necessary?
Culture is the only way to determine which antibiotics the agent is susceptible to. Therefore, culture is extremely valuable both to avoid unnecessary drug use and to shorten the duration of treatment through the selection of an effective antibiotic. Culture is an indispensable diagnostic tool for cases of sight-threatening keratitis (centrally located, multifocal, characterized by melting, painful).
Inoculation Onto Culture Medium
Inoculating corneal scrapings directly into culture material or obtaining the scrape sample where inoculation will be done and inoculating immediately will increase the success of cultures. However, if these options are not feasible, samples can also be placed in transport media.10
What additional samples other than smear and scrapings can be sent for culture?
As contact lens users who have already received antibiotherapy may have low culture positivity, it is helpful to also culture the contact lens itself, the lens case, and the lens solution.10,13
If there is a corneal suture at the infiltration site, the suture should be removed and sent for culture. In the case of deep corneal abscesses, a 7-0 or 8-0 vicryl or silk suture can be passed through the abscess focus, allowing the microbial agent to be smeared on the fibrils of the suture, which is then cultured.10
Corneal biopsy is rarely performed because it will impact refraction and reduce tissue integrity. It may be necessary for treatment-resistant cases in which clinical profile suggests an infection, but no growth is detected in direct inspection or culture. To avoid affecting refraction, lamellar tissue containing the boundaries of the lesion can be obtained from the peripheral cornea with 2–3 mm skin trephines or knives under a microscope. In addition, corneal tissue may be removed during full-thickness patch grafting or penetrating keratoplasty in cases with loss of tissue integrity. The tissue samples obtained must be divided so that half can be sent to microbiology and the other half to histopathological examination.14
Histopathological examination provides the opportunity to detect amoeba and yeast or mold fungus without waiting for growth in culture.
Culture media for bacterial keratitis:
Blood agar: Suitable for aerobic and facultative anaerobic bacteria such as P. aeruginosa, S. aureus, S. epidermidis, and S. pneumoniae.2,10,12
Chocolate agar: Suitable for aerobic and facultative anaerobic bacteria such as H. influenzae, N. gonorrhea, and Bartonella species.2,10,12
Löwenstein-Jensen medium: Suitable for Mycobacterium and Nocardia species2,10,12
BHI (brain heart infusion [Oxid]) medium: transport medium for aerobic and facultative anaerobic bacteria.10
Differential Diagnosis
Differential diagnosis is required for both infectious and noninfectious corneal ulcers. Fungi, amoebae (Acanthamoebae), and nematodes from infectious ulcers can also cause infiltrative keratitis. On the other hand, the possibility of superinfection by these agents with bacterial keratitis should also be kept in mind. Some members of the herpes virus family (herpes simplex, herpes zoster, Epstein-Barr) may produce immune-mediated infiltrates that mimic bacterial, fungal, and acanthamoeba keratitis.
Although clinical presentation, history, and symptoms provide some guidance, culture of corneal swab and scrape samples is the main differential diagnostic method.
If there is clinical uncertainty regarding the etiology until microbiological analysis results are available, then empirical treatment should also include antibacterial therapy.
Connective tissue diseases and vasculitic disorders also produce noninfectious corneal infiltrates. These types of immune ulcers are generally parallel to the limbus, arc-shaped, and peripheral.2
Management
Prevention
Avoiding and mitigating predisposing risk factors is the top priority. This approach will make treatment more effective and prevent recurrences. Restoring the integrity of the corneal epithelium, an impenetrable barrier for most bacteria, will provide resistance to infection and improve patient comfort. The following steps are key to protecting the corneal epithelium:
- Complying with the principles of proper contact lens use
- Discontinuing damaging topical drugs and/or replacing them with gentler alternatives
- Eliminating irritation caused by explants or sutures on the ocular surface
- Correcting eyelid and eyelash disorders
- Treating persistent epithelial defects with artificial tears, autologous serum or platelet-rich plasma15
- Treating adjacent tissue infections such as dacryocystitis, meibomian gland disease, and canaliculitis
- Repairing thinned or perforated corneas with amniotic membrane and lamellar or full-thickness corneal grafts
- Managing systemic diseases through appropriate consultations
Initial Treatment
Unlike infections in other tissues, bacterial keratitis is treated primarily with topical antibiotics. Topical treatment is more effective due to the avascular nature of the tissue and the presence of the blood-aqueous humor barrier. Systemic antibiotherapy is added in extreme cases with scleral involvement and intraocular infectious spread, as well as cases of gonococcal keratitis.2,10,12
In patients with central keratitis foci and in severe cases, a loading dose can be administered at 5- to 15 min intervals and the drug can be given hourly thereafter.2,10 In the presence of anterior chamber reaction, cycloplegic agents are useful for providing comfort and preventing the formation of synechiae.
In cases of bacterial keratitis where direct microscopic inspection and staining have been done but culture antibiogram results have not been returned yet, empirical treatment is guided by Gram staining results. The relevant table in the AAO 2018 Preferred Practice Pattern report for bacterial keratitis is summarized in Table 1 as follows:10
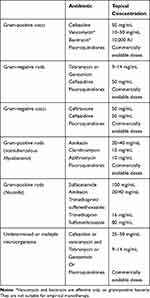 |
Table 1 Recommended Antibiotic Therapy for Bacterial Keratitis by AAO |
The AAO 2018 report on bacterial keratitis cites strong and high-quality evidence that fluoroquinolone monotherapy is at least as effective as combination therapy with fortified drops.10 However, there is no randomized controlled trial comparing fluoroquinolones and fortified topical drug combinations with respect to the treatment of severe keratitis. The use of fortified topical antibiotics is recommended for large and/or visually significant corneal infiltrates, especially if hypopyon is present.10
Ciprofloxacin 0.3%, ofloxacin 0.3%, and levofloxacin 1.5% have been approved by the US Food and Drug Administration (FDA) for the treatment of bacterial keratitis.10 In addition, although it does not have FDA approval for this indication, moxifloxacin has long been used in the treatment of bacterial keratitis. The results of a single-center, prospective, randomized clinical trial with moxifloxacin published in 2007 indicated that there was no difference in treatment efficacy between the fortified cefazolin/tobramycin, moxifloxacin, and ofloxacin groups.16 In a multicenter, randomized, placebo-controlled, double-blind clinical trial named Steroids for Corneal Ulcers Trial (SCUT), topical moxifloxacin therapy was administered to all of the 500 study subjects and the outcomes were published in 2012.17 When this study was analyzed in terms of drug susceptibility, the moxifloxacin susceptibility of the microorganism was reported to mediate visual acuity achieved at week 3 of treatment.18
In early 2019, the Antibiotic Resistance Monitoring in Ocular Microorganisms (ARMOR) surveillance program published its antibiotic resistance reports on 4829 isolates from the years 2009–2016.19 Moxifloxacin resistance was observed in 33.6% of all S. Aureus strains (1695 isolates) and in 72.8% of methicillin-resistant S. aureus (MRSA) (621 isolates). Among the coagulase-negative staphylococci (CoNS) (1475 isolates), moxifloxacin resistance was observed in 31.1% overall and in 51.5% of methicillin-resistant CoNS. Although the resistance rate had not increased during the 8-year surveillance, it was emphasized that the high in vitro resistance rate should be considered when treating patients with ocular infections.19 In a 20-year follow-up study (1993–2012) conducted in the USA, Chang et al20 reported an MRSA rate of 30.7% and increasing resistance to fourth-generation fluoroquinolones. In another such study (1996–2015), Peng et al21 reported an increase in MRSA rate. In a 20-year follow-up study conducted in Taiwan, Liu et al22 compared susceptibility data from the years 1992–2001 with those from 2007 to 2016, and reported rising rates of antibiotic resistance among gram-positive bacteria as well as a significant increase in oxacillin resistance. Fortunately, these same studies that report fluoroquinolone resistance, methicillin resistance, and even multidrug resistance have also reported complete susceptibility to vancomycin.19,20 In addition, besifloxacin 0.6%, the newest commercial ophthalmic fluoroquinolone, was found to be superior to all other ophthalmic fluoroquinolones against methicillin- and ciprofloxacin-resistant S. aureus and S. epidermidis in in vitro studies.23 Data regarding besifloxacin are limited to the results of one multicenter, retrospective clinical trial on the treatment of bacterial keratitis, in which its level of efficacy was similar to that of moxifloxacin.24 In addition, besifloxacin is only available in ophthalmic form, suggesting less risk of resistance compared to other fluoroquinolones.25
Nontuberculous mycobacteria were previously called atypical mycobacteria. In cases of keratitis following refractive surgery, normally rare agents like Mycobacterium may be more observed at higher frequency in addition to more commonly detected agents.26–30 They cause slow-progressing keratitis and treatment may require multiple antibiotics, as shown in Table 1.10
Nocardia, a Gram-positive rod, causes slow progressive infections after ulceration due to minor trauma, especially when exposed to contaminated soil.31 In nocardia keratitis; superficial, needle-shaped multifocal leaks are observed in a wreath-like configuration.31
Early presentation determines the characteristic clinical picture of Nocardia.32 Microbiological verification is important as it can be mixed with fungal keratitis. The preferred drug for Nocardia keratitis is amikacin,32 trimethoprim-sulfamethoxazole combination, and sulfacetamide eye drops.33
Antibiotics in Multidrug Resistance
Multidrug-resistant (MDR) refers to “acquired resistance (to be non-susceptibility) to at least one agent in three or more antimicrobial categories” specifically, not “more than one”, in accordance with the dictionary meaning of the word.34 The other term that “extensively drug-resistant (XDR)” has a specific mean also. XDR was defined by same international panel of experts as “non-susceptibility to at least one agent in all but two or fewer antimicrobial categories”34 It simply means that bacterial isolates remain susceptible to only one or two categories. Pandrug-resistant (PDR) was defined as “non-susceptibility to all agents in all antimicrobial categories”, which is the most understandable definition.34
Since MRSA and methicillin-resistant CoNS are also highly resistant to cephalosporins and fluoroquinolones, topical vancomycin is the most commonly used medication in these cases.35 Vancomycin-resistant microorganisms have also been reported.36 Multidrug resistance was observed in 32.0% of all S. aureus and 76.2% of MRSA.19 Multidrug resistance is similarly high in CoNS also. It was observed in 40.7% of all CoNS and 73.5% of methicillin-resistant CoNS.19 Linezolid is effective against many resistant gram-positive bacteria, including vancomycin-resistant Enterococci (VRE) and MRSA.35 Topical linezolid drops are reported to be well tolerated by patients, even when used on previously damaged ocular surfaces. Budak et al37 reported that linezolid/ceftazidime topical combination therapy provided the desired treatment comfort for seven patients with bacterial keratitis who could not continue vancomycin/ceftazidime topical combination therapy due to vancomycin intolerance. Optimum concentration and frequency of use have not been definitively determined. However, it was used at a concentration of 0.2% in a case series of three patients.38 It has good corneal penetration according to pharmacokinetic studies on animal models, and toxicity has not been reported.39 Sueke et al40 reported that gram-positive bacteria, including MRSA, were not resistant to linezolid.
For multidrug-resistant gram-negative bacteria, it becomes more necessary to go beyond the drugs ophthalmologists are familiar with. Dave et al41 investigated antibacterial susceptibility in cases of endophthalmitis leading to evisceration and reported that the antibiotic with the highest susceptibility rate for gram-positive bacteria was vancomycin (136/147, 92.51%). Among gram-negative bacteria, susceptibility to imipenem was highest (24/29, 82.75%) while susceptibility to ceftazidime was just 50.81% (31/61). Vazirani et al42 reported less than 15% susceptibility to aminoglycosides, cephalosporins, and fluoroquinolones in their series of 23 patients with multi-drug resistant P. aeruginosa (MDR-PA) keratitis. Antibiotic susceptibility was detected in only 10 of the 23 eyes with MDR-PA keratitis (43.38%), all of which were susceptible to colistin and imipenem.42 In another 15 eyes with MDR-PA keratitis, Fernandes et al43 detected susceptibility to imipenem in eight eyes, colistin in four eyes, neomycin in two eyes, and no susceptibility to any drug in one eye.
Colistin, a member of the polymyxin group also referred to as polymixin E, is also used intravenously.44 Originally discovered in 1949, this antibiotic has a very narrow antimicrobial spectrum.44 It is effective against Acinetobacter species, Klebsiella species, Enterobacter species, E. coli, and especially P. aeruginosa.44 It was abandoned 50 years ago due to the serious side effects of systemic use (nephrotoxicity, neurotoxicity).44 Because topical use is more effective and frequently used for keratitis, ophthalmologists are able to utilize colistin for MDR-PA without facing the side effects associated with systemic use.
Imipenem is a broad-spectrum antibiotic from the carbapenem group. It is effective against both gram-positive and gram-negative bacteria, including extended-spectrum beta-lactamase-producing (ESBL) bacteria and enterobacteria.45 In addition to being the other antibiotic (besides colistin) for which high susceptibility has been reported in MDR-PA,41–43,46–48 imipenem is also effective in atypical keratitis (nocardia32,49 nontuberculosis mycobacteria29,50,51) and it has also been a preferred antibiotic in resistant and polymicrobial keratitis due to its broad spectrum of suitability for empirical monotherapy.52
Jain et al53 treated eight patients with MDR-PA with ciprofloxacin and 5% cefazolin in the first 48 hrs, before antibiotic susceptibility test results were reported, then with topical colistin 0.19%. They reported recovery with scarring in four cases, medical recovery requiring cyanoacrylate supplementation in three cases, and recourse to sclerocorneal patch surgery in one case. Of 12 cases of culture-confirmed MDR-PA keratitis, Chatterjee and Agrawal46 achieved no visual improvement in any of the eight eyes treated with fluoroquinolones alone, and reported four eviscerations, one case of phthisis bulbi, and one eye that required corneal graft. After imipenem and colistin were introduced to culture antibiograms in 2011 and 2012, four of four eyes tested were susceptible to imipenem and three of three eyes tested were also susceptible to colistin. After initial treatment with fluoroquinolone, the three eyes switched to susceptible drug therapy (colistin) within 2 days resulted in recovery with scarring, while the one eye switched in 4 days resulted in therapeutic keratoplasty. In patients with MDR-PA, the likelihood of clinical success is reduced and loss of anatomic integrity increases when imipenem or colistin therapy is not initiated, or even initiated after more than a 48 hrs delay.42,43,46,53 Antibiotic selection in multidrug resistance can be summarized as shown in Table 2.
 |
Table 2 Useful Antibiotics in Multidrug Resistance for Bacterial Keratitis |
Modification of Therapy
The effectiveness of the treatment plan is evaluated completely based on clinical response in the first few days, before antibiotic susceptibility is known. If the response is poor, treatment should be modified according to culture and antibiotic susceptibility testing. However, if clinical response is good, treatment should not be altered just because of laboratory findings.2,10,54
If stabilization and improvement are not observed with initial treatment within the first 48 hrs, the treatment should be modified.10 The AAO summarized the qualities of favorable treatment response as follows:10
- Reduced pain
- Less purulent discharge
- Regression of valvular edema and conjunctival hyperemia
- Decrease in the size and depth of infiltrates, with more defined borders
- Regression of stromal edema, cessation of melting
- Reduced anterior chamber reaction
- Start of epithelialization
Patients should be monitored carefully for these signs, and the number of drugs and frequency of application must be reduced when clinical response is achieved. Discontinuing unnecessary drugs is easier when the microorganism and its drug susceptibility are determined. In order to accelerate the healing of persistent epithelial defects, treatment can be reinforced with lubrication, platelet-rich plasma,15 amniotic membrane transplantation, and bandage contact lenses.10 Once antibiotics have been reduced to 3–4 drops per day, they should be discontinued directly without further tapering in order to avoid promoting the development of resistance by the use of subtherapeutic doses.10
If cultures are negative and the desired therapeutic response cannot be obtained, the patient may be referred to centers equipped with in vivo confocal microscopy and PCR. If cultures will be repeated, antibiotherapy should be discontinued for 12–24 hrs and additional culture media (such as Löwenstein-Jensen medium) should be inoculated so as to include atypical species of bacteria that were not previously considered.2,10,54
Preventing the Spread of Resistance in Ophthalmology
Choosing the Appropriate Dose
Using a very low dose (subtherapeutic dosage) or too short duration of antimicrobial usage increase antimicrobial resistance.55 Most antibiotics, particularly those used in ophthalmology, are topical or intracameral, and therefore most studies regarding systemic administration pharmacokinetics (PK) and pharmacodynamic (PD) parameters may not be feasible and may even be misleading.55
Using the Right Antibiotic
In an ideal world, an antibiotic is prescribed following the resistance tests after identification of the pathogen. However, in real-world practice, this is not always possible, so antibiotics are often prescribed empirically.
One of the important issues related to conventional culture and disc diffusion resistance testing is a relatively long time. Identification of the pathogen takes 24–48 hrs and the sensitivity takes another 24 hrs to complete.56
To address this problem, a number of rapid diagnostic tests are being developed and employed.56,57 These include many polymerase chain reaction (PCR) and peptide nucleic acid fluorescence in situ hybridization (PNA-FISH) tests from various manufacturers.56 Both tests work by identifying known resistance or species-specific coding sequences. Results from these tests are available within 45 min to 6 h.56,57 In the future, these innovations may help ophthalmologists in choosing the right antimicrobial and reserving antibiotics of last resort, while remaining confident in the therapeutic effect of the antibiotic prescribed.
Use of Antibiotics When Proved to Be Tangible Benefit
All antibiotic stewardship programs give the same message: use antibiotics only when necessary.58 Despite the widespread practice, the evidence supporting preoperative topical antibiotics is not compelling.59 Antibiotic prophylaxis in intravitreal injection (IVI) and cataract surgery might be questioned by this aspect.60 One such alternative already employed in ophthalmology is the use of effective antiseptics such as povidone iodine and chlorhexidine instead of antibiotics.61 An additional benefit of not using antibiotic prophylaxis in intravitreal injection is to estimate an annual saving of $ 300 million compared to the use of antibiotic prophylaxis for each IVI in the United States.60 In addition, antibiotics are often misused in the treatment of viral and allergic conjunctivitis.60 Research shows that up to 80% of cases of conjunctivitis are caused by virus that does not require antibiotic treatment and is usually self-limiting.62
A Nonpharmaceutical Anti-Infective Treatment Option: Corneal Crosslinking
Non-pharmaceutical anti-infective treatment options, such as corneal cross-linking, are also a relatively new option for anti-infective treatment, especially in cases of superficial bacterial keratitis.63–65 Riboflavin is activated by ultraviolet (UV) light and has long been used for water, surface, and air disinfection. Riboflavin photoactivation has also been used for many years to neutralize pathogens during the preparation of blood products, a procedure known as photochemical pathogen inactivation. This technique inactivates various pathogens such as bacteria and viruses in the donor blood, thereby greatly reducing the risk of blood-borne infections in the recipients.66 The interaction of UV light and riboflavin damages the DNA and RNA of bacterial and viral pathogens and prevents their protein synthesis and replication, leading to the death of the microorganism.67 In addition, corneal crosslinking (CXL) renders the cornea resistant to proteolytic enzymes produced by bacteria.68
Based on the results of similar studies, Makdoumi et al63 published their outcomes of CXL as primary treatment for bacterial keratitis in 2011. They reported that only one patient needed additional antibiotic therapy due to suspected infection progression and that complete corneal epithelial recovery was achieved in six patients. This study was the first clinical series in which UVA-photosensitized riboflavin was administered without antibiotics for the treatment of bacterial keratitis.63 It demonstrated that CXL therapy can be used not only as adjuvant treatment but also as primary treatment in the early stages of infectious ulcerative keratitis. However, Makdoumi et al emphasized that none of the patients in their study had severe or advanced keratitis. The use of CXL besides the Dresden protocol for the treatment of infectious keratitis has been named PACK-CXL (photoactivated chromophore for keratitis).64
The most recent review on CXL therapy for infectious keratitis reports that the CXL procedure is promising in cases in which there is corneal thinning and the causative organism has not spread to the posterior stroma.69 The sterilizing and antiangiogenic effect of PACK-CXL may increase the lifespan of the corneal graft on inflamed and vascularized recipient beds.69
Conclusion
The main causes of treatment resistance in bacterial keratitis are failure to eliminate predisposing factors, misdiagnosis and mistreatment.
- Exogenous, local and systemic predisposing factors that disturbing ocular surface must be eliminated.
- Smears and scrapings for staining and culture are indispensable diagnostic tools
- Empirical antibiotic selections based on direct microscopic examination and gram stain findings are the most appropriate initial treatment approach currently.
- S. aureus and coagulase-negative staphylococci (CoNS), the most common gram-positive agents, have resistance rates higher than 30% for fluoroquinolone and methicillin.
- P. aeruginosa is the most common gram-negative micro-organism, in case of multidrug-resistant isolates, both functional and anatomical prognosis of the eyes are very poor.
- In cases of sight-threatening and resistant keratitis, antibiotic susceptibility testing containing imipenem, colistin, and linezolid is seeming to be an important requirement.
- Despite its efficiency limited to superficial cases, a nonpharmaceutical anti-infective treatment option such as corneal crosslinking for bacterial keratitis is an emerging hope, while antibiotic resistance increases.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Flaxman SR, Bourne RRA, Resnikoff S, et al. Global causes of blindness and distance vision impairment 1990-2020: a systematic review and meta-analysis. Lancet Glob Health. 2017;5:e1221–e1234.
2. Tabbara KF, Bou Chacra CT. Infectious keratitis. In: Tabbara KF, Abu El-Asrar AM, Khairallah M, editors. Ocular Infections. New York, NY: Springer Berlin Heidelberg; 2014:73–94.
3. Erie JC, Nevitt MP, Hodge DO, Ballard DJ. Incidence of ulcerative keratitis in a defined population from 1950 through 1988. Arch Ophthalmol. 1993;111:1665–1671. doi:10.1001/archopht.1993.01090120087027
4. Jeng BH, Gritz DC, Kumar AB, et al. Epidemiology of ulcerative keratitis in Northern California. Arch Ophthalmol. 2010;128:1022–1028. doi:10.1001/archophthalmol.2010.144
5. Seal DV, Kirkness CM, Bennett HG, Peterson M; Keratitis Study G. Population-based cohort study of microbial keratitis in Scotland: incidence and features. Cont Lens Anterior Eye. 1999;22:49–57. doi:10.1016/S1367-0484(99)80003-4
6. Ibrahim YW, Boase DL, Cree IA. Epidemiological characteristics, predisposing factors and microbiological profiles of infectious corneal ulcers: the Portsmouth corneal ulcer study. Br J Ophthalmol. 2009;93:1319–1324. doi:10.1136/bjo.2008.151167
7. Ung L, Bispo PJM, Shanbhag SS, Gilmore MS, Chodosh J. The persistent dilemma of microbial keratitis: global burden, diagnosis, and antimicrobial resistance. Surv Ophthalmol. 2019;64:255–271. doi:10.1016/j.survophthal.2018.12.003
8. Gonzales CA, Srinivasan M, Whitcher JP, Smolin G. Incidence of corneal ulceration in Madurai district, South India. Ophthalmic Epidemiol. 1996;3:159–166. doi:10.3109/09286589609080122
9. Upadhyay MP, Karmacharya PC, Koirala S, et al. The Bhaktapur eye study: ocular trauma and antibiotic prophylaxis for the prevention of corneal ulceration in Nepal. Br J Ophthalmol. 2001;85:388–392. doi:10.1136/bjo.85.4.388
10. Lin A, Rhee MK, Akpek EK, et al. Bacterial keratitis preferred practice pattern(R). Ophthalmology. 2019;126:P1–P55. doi:10.1016/j.ophtha.2018.10.018
11. Dalmon C, Porco TC, Lietman TM, et al. The clinical differentiation of bacterial and fungal keratitis: a photographic survey. Invest Ophthalmol Vis Sci. 2012;53:1787–1791. doi:10.1167/iovs.11-8478
12. Austin A, Lietman T, Rose-Nussbaumer J. Update on the management of infectious keratitis. Ophthalmology. 2017;124:1678–1689. doi:10.1016/j.ophtha.2017.05.012
13. Karaca I, Barut Selver O, Palamar M, Egrilmez S, Aydemir S, Yagci A. Contact lens-associated microbial keratitis in a tertiary eye care center in Turkey. Eye Contact Lens. 2019;1. doi:10.1097/ICL.0000000000000617
14. Hwang DG. Lamellar flap corneal biopsy. Ophthalmic Surg. 1993;24:512–515.
15. Kim KM, Shin YT, Kim HK. Effect of autologous platelet-rich plasma on persistent corneal epithelial defect after infectious keratitis. Jpn J Ophthalmol. 2012;56:544–550. doi:10.1007/s10384-012-0175-y
16. Constantinou M, Daniell M, Snibson GR, Vu HT, Taylor HR. Clinical efficacy of moxifloxacin in the treatment of bacterial keratitis: a randomized clinical trial. Ophthalmology. 2007;114:1622–1629. doi:10.1016/j.ophtha.2006.12.011
17. Srinivasan M, Mascarenhas J, Rajaraman R, et al. Corticosteroids for bacterial keratitis: the steroids for corneal ulcers trial (SCUT). Arch Ophthalmol. 2012;130:143–150. doi:10.1001/archophthalmol.2011.315
18. Oldenburg CE, Lalitha P, Srinivasan M, et al. Moxifloxacin susceptibility mediates the relationship between causative organism and clinical outcome in bacterial keratitis. Invest Ophthalmol Vis Sci. 2013;54:1522–1526. doi:10.1167/iovs.12-11246
19. Thomas RK, Melton R, Asbell PA. Antibiotic resistance among ocular pathogens: current trends from the ARMOR surveillance study (2009–2016). Clin Optom (Auckl). 2019;11:15–26. doi:10.2147/OPTO.S189115
20. Chang VS, Dhaliwal DK, Raju L, Kowalski RP. Antibiotic resistance in the treatment of staphylococcus aureus keratitis: a 20-year review. Cornea. 2015;34:698–703. doi:10.1097/ICO.0000000000000431
21. Peng MY, Cevallos V, McLeod SD, Lietman TM, Rose-Nussbaumer J. Bacterial keratitis: isolated organisms and antibiotic resistance patterns in San Francisco. Cornea. 2018;37:84–87. doi:10.1097/ICO.0000000000001417
22. Liu HY, Chu HS, Wang IJ, Chen WL, Hu FR. Microbial keratitis in Taiwan: a 20-year update. Am J Ophthalmol. 2019.
23. Miller D, Chang JS, Flynn HW, Alfonso EC. Comparative in vitro susceptibility of besifloxacin and seven comparators against ciprofloxacin- and methicillin-susceptible/nonsusceptible staphylococci. J Ocul Pharmacol Ther. 2013;29:339–344. doi:10.1089/jop.2012.0081
24. Schechter BA, Parekh JG, Trattler W. Besifloxacin ophthalmic suspension 0.6% in the treatment of bacterial keratitis: a retrospective safety surveillance study. J Ocul Pharmacol Ther. 2015;31:114–121. doi:10.1089/jop.2014.0039
25. Deschenes J, Blondeau J. Besifloxacin in the management of bacterial infections of the ocular surface. Can J Ophthalmol. 2015;50:184–191. doi:10.1016/j.jcjo.2014.12.013
26. Bostan C, Slim E, Choremis J, et al. Successful management of severe post-LASIK Mycobacterium abscessus keratitis with topical amikacin and linezolid, flap ablation, and topical corticosteroids. J Cataract Refract Surg. 2019;45:1032–1035. doi:10.1016/j.jcrs.2019.03.001
27. John T, Velotta E. Nontuberculous (atypical) mycobacterial keratitis after LASIK: current status and clinical implications. Cornea. 2005;24:245–255. doi:10.1097/01.ico.0000151565.63107.64
28. Ko J, Kim SK, Yong DE, Kim TI, Kim EK. Delayed onset mycobacterium intracellulare keratitis after laser in situ keratomileusis: a case report and literature review. Medicine (Baltimore). 2017;96:e9356. doi:10.1097/MD.0000000000009356
29. Liu HY, Chu HS, Chen WL, Hu FR, Wang IJ. Bilateral non-tuberculous mycobacterial keratitis after small incision lenticule extraction. J Refract Surg. 2018;34:633–636. doi:10.3928/1081597X-20180827-01
30. Solomon A, Karp CL, Miller D, Dubovy SR, Huang AJ, Culbertson WW. Mycobacterium interface keratitis after laser in situ keratomileusis. Ophthalmology. 2001;108:2201–2208. doi:10.1016/S0161-6420(01)00851-X
31. Sridhar MS, Gopinathan U, Garg P, Sharma S, Rao GN. Ocular nocardia infections with special emphasis on the cornea. Surv Ophthalmol. 2001;45:361–378. doi:10.1016/S0039-6257(00)00207-1
32. Lalitha P, Tiwari M, Prajna NV, Gilpin C, Prakash K, Srinivasan M. Nocardia keratitis: species, drug sensitivities, and clinical correlation. Cornea. 2007;26:255–259. doi:10.1097/ICO.0b013e318033d853
33. Johansson B, Fagerholm P, Petranyi G, Claesson Armitage M, Lagali N. Diagnostic and therapeutic challenges in a case of amikacin-resistant Nocardia keratitis. Acta Ophthalmol. 2017;95:103–105. doi:10.1111/aos.2017.95.issue-1
34. Magiorakos AP, Srinivasan A, Carey RB, et al. Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: an international expert proposal for interim standard definitions for acquired resistance. Clin Microbiol Infect. 2012;18:268–281. doi:10.1111/j.1469-0691.2011.03570.x
35. Elsahn AF, Yildiz EH, Jungkind DL, et al. In vitro susceptibility patterns of methicillin-resistant Staphylococcus aureus and coagulase-negative Staphylococcus corneal isolates to antibiotics. Cornea. 2010;29:1131–1135. doi:10.1097/ICO.0b013e3181d2ce25
36. Hernandez-Camarena JC, Bautista-de Lucio VM, Navas A, Ramirez-Miranda A, Graue-Hernandez EO. Delayed-onset post-keratoplasty endophthalmitis caused by vancomycin-resistant enterococcus faecium. Case Rep Ophthalmol. 2012;3:370–374. doi:10.1159/000344006
37. Akova Budak B, Baykara M, Kivanc SA, Yilmaz H, Cicek S. Comparing the ocular surface effects of topical vancomycin and linezolid for treating bacterial keratitis. Cutan Ocul Toxicol. 2016;35:126–130. doi:10.3109/15569527.2015.1058273
38. Tu EY, Jain S. Topical linezolid 0.2% for the treatment of vancomycin-resistant or vancomycin-intolerant gram-positive bacterial keratitis. Am J Ophthalmol. 2013;155:1095–1098 e1. doi:10.1016/j.ajo.2013.01.010
39. Ekdawi NS, Fiscella R, Schreckenberger P, Tu E. Topical linezolid in streptococcus pneumoniae corneal ulcer model in rabbits. Invest Ophthalmol Vis Sci. 2005;46:4910.
40. Sueke H, Kaye S, Neal T, et al. Minimum inhibitory concentrations of standard and novel antimicrobials for isolates from bacterial keratitis. Invest Ophthalmol Vis Sci. 2010;51:2519–2524. doi:10.1167/iovs.09-4638
41. Dave TV, Dave VP, Sharma S, et al. Infectious endophthalmitis leading to evisceration: spectrum of bacterial and fungal pathogens and antibacterial susceptibility profile. J Ophthalmic Inflamm Infect. 2019;9:9. doi:10.1186/s12348-019-0174-y
42. Vazirani J, Wurity S, Ali MH. Multidrug-resistant pseudomonas aeruginosa keratitis: risk factors, clinical characteristics, and outcomes. Ophthalmology. 2015;122:2110–2114. doi:10.1016/j.ophtha.2015.06.007
43. Fernandes M, Vira D, Medikonda R, Kumar N. Extensively and pan-drug resistant Pseudomonas aeruginosa keratitis: clinical features, risk factors, and outcome. Graefes Arch Clin Exp Ophthalmol. 2016;254:315–322. doi:10.1007/s00417-015-3208-7
44. Martis N, Leroy S, Blanc V. Colistin in multi-drug resistant Pseudomonas aeruginosa blood-stream infections: a narrative review for the clinician. J Infect. 2014;69:1–12. doi:10.1016/j.jinf.2014.03.001
45. Sueke H, Shanker J, Neal TJ, Horsburgh M, Gilbert R, Kaye SB. New Developments in Antibacterial Chemotherapy for Bacterial Keratitis. In: Reinhard T, Larkin F, editors. Corneal Disease: Recent Developments in Diagnosis and Therapy. Berlin, Heidelberg: Springer Berlin Heidelberg. 2013.19–35
46. Chatterjee S, Agrawal D. Multi-drug resistant Pseudomonas aeruginosa keratitis and its effective treatment with topical colistimethate. Indian J Ophthalmol. 2016;64:153–157. doi:10.4103/0301-4738.179721
47. Fernandez-Ferreiro A, Gonzalez-Barcia M, Gil-Martinez M, et al. Evaluation of the in vitro ocular toxicity of the fortified antibiotic eye drops prepared at the hospital pharmacy departments. Farm Hosp. 2016;40:352–370. doi:10.7399/fh.2016.40.5.10416
48. Jain R, Murthy SI, Motukupally SR. Clinical outcomes of corneal graft infections caused by multi-drug resistant Pseudomonas aeruginosa. Cornea. 2014;33:22–26. doi:10.1097/ICO.0000000000000011
49. Patel NR, Reidy JJ, Gonzalez-Fernandez F. Nocardia keratitis after laser in situ keratomileusis: clinicopathologic correlation. J Cataract Refract Surg. 2005;31:2012–2015. doi:10.1016/j.jcrs.2005.02.049
50. Broadway DC, Kerr-Muir MG, Eykyn SJ, Pambakian H. Mycobacterium chelonei keratitis: a case report and review of previously reported cases. Eye (Lond). 1994;8(Pt 1):134–142. doi:10.1038/eye.1994.27
51. Hu FR, Wang IJ. Comparison of topical antibiotics for treating mycobacterium chelonae keratitis in a rabbit model. Curr Eye Res. 1998;17:478–482. doi:10.1076/ceyr.17.5.478.5195
52. Turkyilmaz K, Kurt A, Dilek AR, Sekeryapan B, Erturk A. A case of suture-related bacterial keratitis and its treatment with topical imipenem. J Ocul Biol Dis Infor. 2011;4:141–144. doi:10.1007/s12177-012-9081-2
53. Jain R, Murthy SI, Motukupally SR, Jain M. Use of topical colistin in multiple drug-resistant Pseudomonas aeruginosa bacterial keratitis. Cornea. 2014;33:923–927. doi:10.1097/ICO.0000000000000184
54. Hong AR, Shute TS, Huang AJW. Bacterial Keratitis. In: Mannis MJ, Holland EJ, editors. Cornea.
55. Grzybowski A, Brona P, Kim SJ. Microbial flora and resistance in ophthalmology: a review. Graefes Arch Clin Exp Ophthalmol. 2017;255:851–862. doi:10.1007/s00417-017-3608-y
56. Goff DA, Jankowski C, Tenover FC. Using rapid diagnostic tests to optimize antimicrobial selection in antimicrobial stewardship programs. Pharmacotherapy. 2012;32:677–687. doi:10.1002/phar.2012.32.issue-8
57. Wenzler E, Wong JR, Goff DA, Jankowski CA, Bauer KA. Controversies in antimicrobial stewardship: focus on new rapid diagnostic technologies and antimicrobials. Antibiotics (Basel). 2016;5:6.
58. Pollack LA, Srinivasan A. Core elements of hospital antibiotic stewardship programs from the centers for disease control and prevention. Clin Infect Dis. 2014;59 Suppl 3:S97–S100. doi:10.1093/cid/ciu542
59. Grzybowski A, Kuklo P, Pieczynski J, Beiko G. A review of preoperative manoeuvres for prophylaxis of endophthalmitis in intraocular surgery: topical application of antibiotics, disinfectants, or both? Curr Opin Ophthalmol. 2016;27:9–23. doi:10.1097/ICU.0000000000000216
60. Parke DW
61. Dossarps D, Bron AM, Koehrer P, Aho-Glele LS, Creuzot-Garcher C, net F. Endophthalmitis after intravitreal injections: incidence, presentation, management, and visual outcome. Am J Ophthalmol. 2015;160:17–25 e1. doi:10.1016/j.ajo.2015.04.013
62. Azari AA, Barney NP. Conjunctivitis: a systematic review of diagnosis and treatment. JAMA. 2013;310:1721–1729. doi:10.1001/jama.2013.280318
63. Makdoumi K, Mortensen J, Crafoord S. Infectious keratitis treated with corneal crosslinking. Cornea. 2010;29:1353–1358. doi:10.1097/ICO.0b013e3181d2de91
64. Said DG, Elalfy MS, Gatzioufas Z, et al. Collagen cross-linking with photoactivated riboflavin (PACK-CXL) for the treatment of advanced infectious keratitis with corneal melting. Ophthalmology. 2014;121:1377–1382. doi:10.1016/j.ophtha.2014.01.011
65. Ting DSJ, Henein C, Said DG, Dua HS. Effectiveness of adjuvant photoactivated chromophore corneal collagen cross-linking versus standard antimicrobial treatment for infectious keratitis: a systematic review protocol. JBI Database System Rev Implement Rep. 2019.
66. Ruane PH, Edrich R, Gampp D, Keil SD, Leonard RL, Goodrich RP. Photochemical inactivation of selected viruses and bacteria in platelet concentrates using riboflavin and light. Transfusion. 2004;44:877–885. doi:10.1111/trf.2004.44.issue-6
67. Martins SA, Combs JC, Noguera G, et al. Antimicrobial efficacy of riboflavin/UVA combination (365 nm) in vitro for bacterial and fungal isolates: a potential new treatment for infectious keratitis. Invest Ophthalmol Vis Sci. 2008;49:3402–3408. doi:10.1167/iovs.07-1592
68. Spoerl E, Wollensak G, Seiler T. Increased resistance of crosslinked cornea against enzymatic digestion. Curr Eye Res. 2004;29:35–40. doi:10.1080/02713680490513182
69. Bonzano C, Di Zazzo A, Barabino S, Coco G, Traverso CE. Collagen cross-linking in the management of microbial keratitis. Ocul Immunol Inflamm. 2019;27:507–512. doi:10.1080/09273948.2017.1414856
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
