Back to Journals » Clinical Ophthalmology » Volume 9
Traumatic uveitis in the mid-Atlantic United States
Authors Engelhard S, Patrie J, Prenshaw J, Bajwa A, Monahan R, Reddy A
Received 9 June 2015
Accepted for publication 17 July 2015
Published 5 October 2015 Volume 2015:9 Pages 1869—1874
DOI https://doi.org/10.2147/OPTH.S90153
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Stephanie B Engelhard,1 James Patrie,2 John Prenshaw,1 Asima Bajwa,1 Rose Monahan,1 Ashvini K Reddy1
1Department of Ophthalmology, 2Department of Public Health Sciences, University of Virginia, Charlottesville, VA, USA
Purpose: The purpose of this study was to conduct an in-depth analysis of traumatic uveitis in patients managed in a mid-Atlantic tertiary care center with the goal of better characterizing the clinical features and outcomes of this large and important subset of uveitis patients.
Methods: This was a retrospective, observational study comparing traumatic uveitis patients with nontraumatic uveitis patients seen at the University of Virginia, Charlottesville, VA, USA, from 1984 to 2014.
Results: Fifty-four traumatic uveitis patients (55 eyes) were identified. The patient population was 70.4% male, 57.4% Caucasian, and 37.0% African American. Mean age at diagnosis was 31.2 years; mean duration of follow-up was 5.4 years; and mean number of visits to the clinic was 4. The most common treatment modality was local steroids (77.8%). Glaucoma was medically managed in eight patients (14.8%). Cataract surgery was performed in five patients (9.3%). Mean best-corrected visual acuity at baseline for traumatic uveitis patients was 0.33 logMAR (SD 0.42) at the initial visit and 0.16 logMAR (SD 0.33) at the final visit. Mean baseline intraocular pressure (IOP) in the traumatic uveitis group was 15.5 mmHg (SD 7.4) at the initial visit and 14.6 mmHg (SD 4.0) at the final visit. Patients in the traumatic uveitis cohort tended to have better visual outcomes than those in the nontraumatic uveitis cohort.
Conclusion: In our series, traumatic uveitis patients tended to be young and male and present with unilateral disease, all findings consistent with other reports. Despite relatively good visual outcomes, the traumatic uveitis patients still experienced a high burden of disease, measured both in the number of clinic visits and duration of follow-up. Due to the young mean age of patients, these disease burdens and decreased quality of life are nontrivial, emphasizing the importance of careful management and prompt treatment of this subset of uveitis patients.
Keywords: traumatic uveitis, trauma, iritis, best-corrected visual acuity, intraocular pressure
Introduction
Traumatic uveitis is an important cause of anterior uveitis. In children, it is one of the two leading causes of endogenous uveitis,1 comprising up to 25% of all pediatric uveitis diagnoses;2 however, it remains poorly characterized, perhaps, because of rare diagnostic dilemmas, straightforward management, and relatively good visual outcomes. Most of what is known of traumatic uveitis is gathered from case series and brief reports of ocular trauma and referral patterns, many of which are from the developing world and focus primarily on the mechanism of ocular injury.3–5 The purpose of this study is to conduct an in-depth analysis of all traumatic uveitis cases seen over a 30-year period with the goal of better characterizing the clinical features and outcomes of this large and important subset of uveitis patients. To our knowledge, this is one of only two studies dedicated solely to the analysis and characterization of traumatic uveitis in the literature, and the only such study published in the past 25 years.
Methods
This was a retrospective, observational study of all patients with traumatic uveitis seen in the Department of Ophthalmology at the University of Virginia, Charlottesville, VA, USA. The study was approved by the Institutional Review Board and was conducted in accordance with the principles of the Declaration of Helsinki. A database of 491 patients with a diagnosis of uveitis is maintained in the Department of Ophthalmology at the University of Virginia. The database was reviewed to identify patients diagnosed with traumatic uveitis over a 30-year period from 1984 to 2014. Sixty patients diagnosed with traumatic uveitis (62 eyes) were identified. Six patients (seven eyes) diagnosed with hyphema, penetrating ocular trauma, and ruptured globe injuries were excluded. Fifty-four patients (55 eyes) diagnosed with traumatic uveitis were included in the study. In order to compare traumatic and nontraumatic uveitis patients, a control group of 546 nontraumatic uveitis eyes was established (26 eyes were excluded due to incomplete data). No minimum follow-up period was required for inclusion, and all data in the database were collected from patient medical charts completed at the time of patient visits. The patients’ initial (baseline) and final follow-up visits to the clinic were considered as time points for data collection in the database. Disease classification was recorded in accordance with the Standardization of Uveitis Nomenclature (SUN) Working Group criteria.6
Patient demographic information including date of birth, sex, race/ethnicity, age at presentation, duration of follow-up, and number of clinic visits was reviewed. Clinical information including final diagnosis with etiology, location, laterality, and chronicity of uveitis, best-corrected visual acuity (BCVA), intraocular pressure (IOP), laboratory findings, relevant systemic and associated diagnoses, treatment modalities, and complications was also reviewed. BCVA results were converted to the logMAR units for analysis and are given as logMAR (SD). Visual acuity of counting fingers was recorded as 2.0 logMAR; hand movements, 2.3 logMAR; light perception, 2.6 logMAR; and no light perception, 2.9 logMAR.7,8 Management including the use of local and systemic steroids, antihypertensive drops, intravitreal injections, subtenon steroid injections, antimetabolites, antitumor necrosis factor agents, cataract surgery, pars plana vitrectomy, and glaucoma management (medical and surgical intervention) was recorded.
Categorical data were summarized by frequencies and percentages, and continuous scale data were summarized by the mean and standard deviation distribution as well as by median and interquartile range of the distribution (ie, the range of values between the 25th and 75th percentiles of the distribution). The visual acuity data (logMAR) and the IOP data were analyzed via linear mixed-effects (LME) models. For each model, the initial to final change in outcome (eg, logMAR) and the cohort (nontrauma vs trauma uveitis patients) were treated as the dependent and independent variables, respectively. A subject-specific random effect was utilized as a component of the linear mixed-effects model, to account for intra-subject correlation between the bilateral outcomes of patients with bilateral uveitis. With regard to hypothesis testing, a P≤0.05 decision rule was established a priori as the null hypothesis rejection criterion.
The MIXED procedure of SAS version 9.4 (SAS Institute Inc., Cary, NC, USA) was used to conduct the linear mixed-effects model analyses.
Results
A cohort of 491 patients diagnosed with uveitis at the University of Virginia between 1984 and 2004 was reviewed. Of those 491 patients, 60 patients (12.2%) were diagnosed with traumatic uveitis. After excluding six patients (seven eyes) with hyphema, penetrating ocular trauma, and ruptured globe injuries, 54 traumatic uveitis patients (55 eyes) were included in the study. All 54 patients with traumatic uveitis had anterior uveitis. Three patients (5.6%) had afferent pupillary defects at the initial visit and five patients (9.3%) at the final visit. Patient demographic information is summarized in Table 1.
  | Table 1 Patient demographics and characteristics |
Ophthalmic management and interventions by anatomic location are summarized in Table 2. No patients were prescribed antimetabolites, antitumor necrosis factor agents, or intravitreal injections, and no patients underwent glaucoma surgery during the follow-up period.
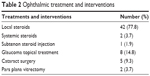  | Table 2 Ophthalmic treatment and interventions |
BCVA was recorded at the patients’ initial and final visits and converted to logMAR. BCVA measurements of the traumatic and nontraumatic uveitis patients were compared for all nontraumatic uveitis patients seen during the study period. Seven eyes of the 54 traumatic uveitis patients were eliminated due to incomplete BCVA data at either the initial or final visit. The remaining 48 eyes in the group were included in the analysis. Similarly, 26 of the 572 eyes of the nontraumatic uveitis patients were eliminated due to incomplete data, leaving 546 eyes for the analysis. Table 3 summarizes the initial and final BCVA for both the traumatic uveitis and nontraumatic uveitis groups for those patients with available BCVA data at initial and final visits. The initial to final change in visual acuity (logMAR) for traumatic and nontraumatic uveitis patients is shown in Figure 1.
  | Table 3 Summary statistics for initial and final logMAR and initial to final change in logMAR for patients with both initial and final logMAR measurements |
  | Figure 1 Initial to final change in visual acuity (logMAR) for traumatic and nontraumatic uveitis patients. |
IOP was recorded for all traumatic and nontraumatic uveitis patients at the initial and final visits. IOP measurements of the traumatic and nontraumatic patients were compared for all patients seen during the study period. Fourteen eyes of the 54 traumatic uveitis patients were excluded due to incomplete IOP data. The remaining 41 eyes in the traumatic uveitis group were included in the analysis. Similarly, 41 of the 572 eyes of the nontraumatic uveitis patients were eliminated due to incomplete data, leaving 531 eyes for the analysis. Table 4 summarizes the initial and final IOP measurements for both the traumatic uveitis and nontraumatic uveitis groups for those patients with available IOP data at the initial and final visits. The initial to final change in IOP (mmHg) for traumatic and nontraumatic uveitis patients is shown in Figure 2. A summary of clinical outcomes of patients prescribed steroids and those not prescribed steroids is found in Table 5.
  | Table 4 Summary statistics for initial and final logMAR and initial to final change in logMAR for patients with both initial and final logMAR measurements |
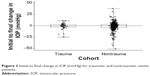  | Figure 2 Initial to final change in IOP (mmHg) for traumatic and nontraumatic uveitis patients. |
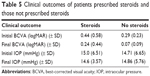  | Table 5 Clinical outcomes of patients prescribed steroids and those not prescribed steroids |
Discussion
This retrospective study reports and compares the demographics, treatments, and outcomes of a population of 54 traumatic uveitis patients at a tertiary referral center in Charlottesville, VA, USA, over a 30-year period. Traumatic uveitis is poorly characterized in the literature, with most coverage existing in the form of case reports or brief mentions in studies focusing on ocular trauma; however, a number of traumatic uveitis are quite high in certain populations, which highlights the importance of studying this subset of patients. Although traumatic uveitis is generally considered less complicated than endogenous forms of uveitis, the mean duration of dynamic follow-up in our cohort was 5.40 years (SD 9.09), suggesting that traumatic uveitis can be associated with a significant disease burden and challenging management, especially because it disproportionately affects younger patients.
The pathophysiology of traumatic uveitis is incompletely understood. Rosenbaum et al theorized that a possible mechanism for the development of intraocular inflammation secondary to nonpenetrating trauma could be akin to similar processes in other organ systems.9 They posit that traumatic uveitis may be similar to the Koebner phenomenon, whereby minor skin trauma precipitates psoriasis flares in approximately 25% of patients.9 This study – which is to our knowledge, the only other study focused solely on traumatic uveitis except for the current series – reported that 4.8% of uveitis patients suspected that the cause of their uveitis was related to previous nonpenetrating trauma.9 At our center, traumatic uveitis following nonpenetrating trauma was more than double that found in the earlier study.
One ocular trauma study conducted in Tanzania reported that traumatic uveitis was the most common traumatic diagnosis at the time of presentation, representing 23% of traumatic diagnoses, 18% of eclipsing ruptured globe, and 17% of corneal ulcer.4 Furthermore, in the group with patients aged 20 years or less, traumatic uveitis accounted for 27% of traumatic diagnoses, and in patients aged over 60 years, it accounted for 29% of such diagnoses.4 In that study, 72% of traumatic uveitis patients presented with severe visual acuity deficits, which was defined as visual acuity <3/60 (1.3 logMAR). However, the report notes that there were too few final visual acuities documented to draw any conclusion about final visual outcomes in these patients.
A study of ocular emergencies in Kinshasa (Democratic Republic of the Congo) reported traumatic iritis in 17.6% of patients with ocular trauma.3 A Turkish study reported traumatic iritis in 48 of 98 patients hospitalized for ocular trauma.5 Similarly, a Glasgow study reported traumatic uveitis in 24 patients (0.4% of patients with ocular trauma).10 In a UCLA study, traumatic uveitis was the third most common cause of unilateral anterior uveitis, representing 5.7% of all anterior uveitis cases.11 In all of these studies, the authors did not provide any additional information about the clinical course or management in this subset of patients. A British study reported that patients with severe traumatic uveitis rapidly developed mature cataracts, elevated IOP, and vitreous hemorrhage.12
An Egyptian ocular trauma study found that 15.7% of ocular injuries in that cohort were diagnoses of traumatic iritis.13 No additional information about those patients is available. A study from the US military found that traumatic iritis was one of the most common ocular complaints of military personnel injured during Operations Desert Shield and Desert Storm.14 A number of other reports note a small number of traumatic uveitis cases, but do not comment on the clinical course, management, or outcomes for these patients as a group.15–18
In our traumatic uveitis population, the male to female ratio was 2.4:1. This ratio is similar to that reported in reports of all ocular trauma, suggesting a difference in rates of risk-taking behavior between sexes.3–5 The mean age of traumatic uveitis diagnosis in our population was 31.2 years (SD 17.03), which is considerably lower than the mean age of diagnosis of all types of uveitis (n=491) at the University of Virginia, which was 46.0 years (SD 21.4). From the reports on ocular trauma, it was also found that young patients are at greater risk for ocular injury.4,5 At our site, 25.6% of uveitis patients aged 0 to 18 years were diagnosed with traumatic uveitis, reflecting the importance of traumatic uveitis in the pediatric uveitis clinic.2 BenEzra et al reported traumatic uveitis in ten of the 110 pediatric uveitis population studied.19 Cunningham reported that traumatic uveitis is one of the leading causes of uveitis in children, along with juvenile idiopathic arthritis.1
In our cohort, most patients (77.8%) were prescribed local steroids, while only 3.7% were prescribed systemic steroids. Although eight patients (14.8%) developed glaucoma during the follow-up period, all were medically managed and none required glaucoma surgery. Five patients (9.3%) in our cohort underwent cataract surgery, and only two (3.7%) underwent pars plana vitrectomy. Rates of complications in our cohort were significantly lower than those in our entire uveitis population of 491 patients. In the overall uveitis population, 23.6% of patients developed glaucoma, with 8.8% of all uveitis patients requiring glaucoma surgery. Higher cataract surgery numbers were found in 26.4% of the overall uveitis population, as were rates of pars plana vitrectomy, which were performed in 9.4% of all uveitis patients. This difference is due in part to the fact that all of our traumatic uveitis patients were diagnosed with anterior uveitis, which in general has better outcomes than intermediate, posterior, and panuveitis, and in part due to the comparatively straightforward management of traumatic uveitis.
The difference in the mean change in logMAR between the traumatic and nontraumatic uveitis groups was 0.18 logMAR (95% CI: -0.03 to 0.39 logMAR), which approached, but did not achieve, statistical significance (P=0.099). The lack of statistical significance indicates that although traumatic uveitis may have overall better results than other uveitis etiologies, positive outcomes are not guaranteed in traumatic uveitis, and it should be approached as aggressively as other etiologies. Similarly, the difference in the mean change in IOP between the traumatic and nontraumatic uveitis groups was 0.19 mmHg (95% CI: -2.40 to 2.78 mmHg), which was not significant (P=0.885), indicating that traumatic uveitis patients did not have significantly better IOP outcomes, again highlighting the fact that traumatic uveitis requires careful management.
Visual acuity improvement was approximately equal between patients prescribed steroids and those who did not receive steroids; however, the group that did not receive steroids had better final BCVA than did the steroid group. This likely reflects the fact that patients with milder, self-limited disease did not require steroid treatment. Consequently, vision in these patients improved without steroid intervention. The steroid group, however, may have had more severe disease requiring intervention. IOP values did not vary significantly between the steroid and nonsteroid groups.
Our results echo those found in the Rosenbaum et al study, as well as other series, which also found that most traumatic uveitis patients were young and male.9 However, the study also reported that one-third of the patients had bilateral inflammation, while only one patient in our series had bilateral disease. The authors suggest that the high rate of bilateral disease in the study may be explained by patients misattributing the inflammation as secondary to the trauma, when in reality it might have been a coincidence that some trauma occurred at or near the time of the development of uveitis. Furthermore, many patients in the series had known underlying systemic conditions that predispose to the development of uveitis including sarcoidosis, ankylosing spondylitis, Reiter’s syndrome, or were HLA-B27 positive.9
The major limitation of this study is its retrospective nature, which increased susceptibility to referral, treatment, and data collection biases. The ophthalmology clinic at the University of Virginia treats patients from the entire state of Virginia as well as referrals from other areas. Because there was not a standardized follow-up period required for inclusion in the study, it is possible that the visual and IOP outcomes represent only patients with more severe disease or those with longer follow-up periods.
Conclusion
Traumatic uveitis patients tend to be young and male and present with unilateral disease, and tend to have better visual outcomes than patients in the nontraumatic uveitis cohort. In spite of relatively good visual outcomes, the traumatic uveitis patients still experienced a high burden of disease, measured both in number of clinic visits and duration of follow-up. Because they tend to be younger than other patients with uveitis, the impact of disease burdens and decreased quality of life may be amplified, highlighting the importance of careful management and prompt treatment of this subset of uveitis patients.
Disclosure
The authors report no conflicts of interest in this work.
References
Cunningham ET. Uveitis in children. Ocul Immunol Inflamm. 2000;8(4):251–261. | ||
Engelhard SB, Bajwa A, Reddy AK. Causes of uveitis in children without juvenile idiopathic arthritis. Clin Ophthalmol. 2015;9:1121–1128. | ||
Kaimbo WK, Spileers W, Missotten L. Ocular emergencies in Kinshasa (Democratic Republic of Congo). Bull Soc Belge Ophtalmol. 2002;284:49–53. | ||
Abraham D, Vitale S, West S, Isseme I. Epidemiology of eye injuries in rural Tanzania. Neuroophthalmology. 1999;6(2):85–94. | ||
Oner A, Kekec Z, Krakucuk S, Ikizceli I, Sözüer EM. Ocular trauma in Turkey: a 2-year prospective study. Adv Ther. 2006;23(2):274–283. | ||
Standardization of Uveitis Nomenclature (SUN) Working Group. Standardization of uveitis nomenclature for reporting clinical data. Results of the First International Workshop. Am J Ophthalmol. 2005;140(3):509–516. | ||
Lange C, Feltgen N, Junker B, Schulze-Bonsel K, Bach M. Resolving the clinical acuity categories “hand motion” and “counting fingers” using the Freiburg Visual Acuity Test (FrACT). Graef Arch Clin Exp. 2009;247(1):137–142. | ||
Kwon YH, Kim CS, Zimmerman MB, Alward WL, Hayreh SS. Rate of visual field loss and long-term visual outcome in primary open-angle glaucoma. Am J Ophthalmol. 2001;132(1):47–56. | ||
Rosenbaum JT, Tammaro J, Robertson JE. Uveitis precipitated by nonpenetrating ocular trauma. Am J Ophthalmol. 1991;112(4):392–395. | ||
MacEwen CJ. Eye injuries: a prospective survey of 5671 cases. Br J Ophthalmol. 1989;73(11):888–894. | ||
McCannel CA, Holland GN, Helm CJ, Cornell PJ, Winston JV, Rimmer TC; UCLA Community-Based Uveitis Study Group. Causes of uveitis in the general practice of ophthalmology. Am J Ophthalmol. 1996;121(1):35–46. | ||
Eagling EM. Ocular damage after blunt trauma to the eye. Its relationship to the nature of the injury. Br J Ophthalmol. 1974;58(2):126–140. | ||
Soliman MM, Macky TA. Pattern of ocular trauma in Egypt. Graef Arch Clin Exp. 2008;246(2):205–212. | ||
Heier JS, Enzenauer RW, Wintermeyer SF, Delaney M, LaPiana FP. Ocular injuries and diseases at a combat support hospital in support of Operations Desert Shield and Desert Storm. Arch Ophthalmol. 1993;111(6):795–798. | ||
Oruc S, Kaplan AD, Galen M, Kaplan HJ. Uveitis referral pattern in a Midwest University Eye Center. Ocul Immunol Inflamm. 2003;11(4): 287–298. | ||
Rathinam SR, Krishnadas R, Ramakrishnan R, et al. Population-based prevalence of uveitis in Southern India. Br J Ophthalmol. 2011;95(4):463–467. | ||
Ramstead C, Ng M, Rudnisky CJ. Ocular injuries associated with Airsoft guns: a case series. Can J Ophthalmol. 2008;43(5):584–587. | ||
Khan M, Reichstein D, Recchia FM. Ocular consequences of bottle rocket injuries in children and adolescents. Arch Ophthalmol. 2011;129(5):639–642. | ||
BenEzra D, Cohen E, Maftzir G. Uveitis in children and adolescents. Br J Ophthalmol. 2005;89(4):444–448. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
