Back to Journals » International Journal of Nanomedicine » Volume 14
Traditional Chinese medicine-combination therapies utilizing nanotechnology-based targeted delivery systems: a new strategy for antitumor treatment
Authors Ma Z, Fan Y, Wu Y, Kebebe D , Zhang B, Lu P, Pi J , Liu Z
Received 12 December 2018
Accepted for publication 12 February 2019
Published 22 March 2019 Volume 2019:14 Pages 2029—2053
DOI https://doi.org/10.2147/IJN.S197889
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Mian Wang
Zhe Ma,1,2 Yuqi Fan,2,3 Yumei Wu,1,2 Dereje Kebebe,1,2,4 Bing Zhang,1,2 Peng Lu,1,2 Jiaxin Pi,1,2 Zhidong Liu1,2
1Engineering Research Center of Modern Chinese Medicine Discovery and Preparation Technique, Ministry of Education, Tianjin University of Traditional Chinese Medicine, Tianjin 300193, China; 2Institute of Traditional Chinese Medicine, Tianjin University of Traditional Chinese Medicine, Tianjin 300193, China; 3School of Integrative Medicine, Tianjin University of Traditional Chinese Medicine, Tianjin 300193, China; 4School of Pharmacy, Institute of Health Sciences, Jimma University, Jimma, Ethiopia
Abstract: Cancer is a major public health problem, and is now the world’s leading cause of death. Traditional Chinese medicine (TCM)-combination therapy is a new treatment approach and a vital therapeutic strategy for cancer, as it exhibits promising antitumor potential. Nanotargeted drug-delivery systems have remarkable advantages and allow the development of TCM-combination therapies by systematically controlling drug release and delivering drugs to solid tumors. In this review, the anticancer activity of TCM compounds is introduced. The combined use of TCM for antitumor treatment is analyzed and summarized. These combination therapies, using a single nanocarrier system, namely codelivery, are analyzed, issues that require attention are determined, and future perspectives are identified. We carried out a systematic review of >280 studies published in PubMed since 1985 (no patents involved), in order to provide a few basic considerations in terms of the design principles and management of targeted nanotechnology-based TCM-combination therapies.
Keywords: cancer, codelivery, combination therapy, nanotargeted drug-delivery system, tumor targeting, TCM
Introduction
Cancer is the leading cause of disease-associated death in China,1 and is now the world’s leading cause of death.2 According to the Global Cancer Report 2018 on the trend of 36 cancers in 185 countries worldwide by the WHO, the global burden of cancer is increasing at an alarming rate (one in eight deaths on average are due to cancer). The report also pointed out that the incidence and mortality rate of cancer continues to rise each year, with developing countries accounting for approximately 60% of the world’s new cases and 70% of annual deaths. In 2018, nearly half the world’s new cases of cancer occurred in Asia, most of which occurred in China.3 Bray et al2 provided a status report on the global burden of cancers using GLOBOCAN 2018. It is estimated that there will be 18 million new cases of cancer and 9.6 million cancer deaths in 2018. Lung cancer and breast cancer are the most frequent cancers in men and women, respectively, and the two leading causes of cancer death. Due to the high incidence and mortality rate of cancer, the global health care burden is also increasing rapidly.
Surgical treatment, chemotherapy, and radiotherapy are the primary treatment methods for cancer.4 If cancer patients are diagnosed early and receive timely surgical treatment, the probability of surviving for 5 years after surgery is greatly improved. However, when cancers are diagnosed late, the vast majority of patients are already in the terminal stages, and thus may have lost the opportunity of surgical treatment. In addition, due to adverse reactions caused by radiotherapy, such as fatigue, gastrointestinal reactions, skin damage, bone-marrow suppression, and cardiotoxicity,5 chemotherapy is still the main method of cancer treatment.
Nevertheless, due to lack of specificity and poor targeting, chemotherapy drugs not only kill tumor cells but also act on normal tissue, causing a reduction in immunity, significant side effects, and low drug efficacy. In addition, cancer patients can develop resistance to a single chemotherapy drug in clinical practice, resulting in a decrease in the subsequent curative effect. Multidrug resistance (MDR) was once considered the leading cause of chemotherapy failure, and may also promote tumor metastasis and recurrence.6 Based on recent statistics from the American Cancer Society, >90% of cancer patients die from different levels of MDR.
Therefore, the treatment of cancer should be changed from an initial single medication to combination therapy. The combination of two or more active antitumor ingredients plays a crucial role in complementarity and synergy, and has become the preferred scheme in cancer treatment. Notably, the combination of traditional Chinese medicines (TCMs) with chemotherapeutic drugs and the combination of various TCMs, which involves multiple targets and multiple signaling pathways, have improved efficacy compared with drugs with a single molecular target and become a new strategy for tumor therapy in recent years.7 Due to a great deal of investment and rapid development, nanotechnology is already used in various fields of biomedical science.8 Novel nanoformulation-based drug-delivery systems, such as liposomes, nanoparticles (NPs), vesicles, mesoporous silica NPs (MSNs), and micelles, provide promise in overcoming current limitations, including poor targeting, insufficient absorption, poor pharmacokinetics and bioavailability, and limited biodistribution.9–11
In this review, the anticancer activity of TCM compounds is introduced. The combined use of TCMs for antitumor therapy is analyzed and summarized. These combination therapies using a single nanocarrier system, namely codelivery, are analyzed to determine their potential in prolonging drug duration in vivo, targeting drug delivery, and reducing toxicity (Figure 1). Matters requiring attention and future perspectives in this field are also reviewed, in order to accelerate the clinical application of combination antitumor therapy using targeted nanotechnology.
Antitumor effects of TCMs
Herbs, animals, and minerals are used widely as health foods and medicines to remedy various diseases in Asia, and have been collected and recorded as effective and traditional therapies in the TCM literature. For example, artemisinin was isolated by Youyou Tu at the China Academy of Traditional Chinese Medicine in Beijing, and is now an effective medicine in the treatment of malaria. As a result, Tu won the Nobel Prize in Physiology or Medicine in 2015. In most developing countries, 80% of the population continue to use traditional medicines for primary health care.12 From 2016 to 2017, the total amount of TCM herbal medicines and other related products exported to the Belt and Road Initiative countries reached US$295 million. In addition, the WHO also recognized traditional medicine in its influential global medical compendium.13
Worldwide, including in Western countries, TCM has been increasingly used in the past few decades, and is well known for its vital role in cancer prevention and treatment. A number of studies have confirmed that the active ingredients in TCM (curcumin [Cur], gambogic acid, and baicalein [BA], among others) are able to effectively induce apoptosis, interfere with tumor progression, inhibit tumor development, inhibit angiogenesis, cause cell-cycle arrest, and block metastasis. A summary of the antitumor effects of drugs isolated from TCMs is shown in Table 1. Structures of TCM compounds are shown in Figure 2.
  | Table 1 Antitumor effects of traditional Chinese medicine |
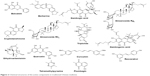  | Figure 2 Chemical structures of the active components in traditional Chinese medicine. |
Antitumor effects of TCM-combination therapy
Initially, cancer therapy consisted of a single drug, which could involve a single target. However, malignant disease is caused by many complicated factors, and treatment with a single drug is not adequate. Patients are usually susceptible to drug resistance after sequential cycles of therapy with these chemotherapy drugs,100 and a single medication frequently causes serious side effects. For instance, although cisplatin is clinically effective, it lacks selectivity for tumor tissue, resulting in serious side effects, such as kidney-function damage,101 neurotoxicity,102 ototoxicity,103 and the emergence of MDR, resulting in the failure of chemotherapy.104 In addition, long-term or high-dose cisplatin treatment can also cause severe anemia.105 Therefore, the clinical application of single drugs, such as cisplatin, has been greatly restricted.
Cancer therapy urgently requires a new therapeutic approach to overcome these shortcomings. Combination-drug therapy is a new mode of treatment, and has gradually gained the attention of researchers.106 Combination therapy involves the simultaneous or sequential use of two or more medicines for therapeutic purposes, and gradually plays a meaningful role in a complementary way, has synergistic action, and alleviates adverse reactions. It can not only produce a better therapeutic effect by regulating multiple signaling pathways in abnormal cells and act on multiple targets simultaneously but also reduce the occurrence of MDR and reduce both the dosage and side effects. The combination of two or more active antitumor ingredients is now a vital treatment method for tumors, and has received US Food and Drug Administration (FDA) approval.107
Based on classic TCM theory, the combination of antitumor TCMs exhibits promising potential in cancer treatment such as: 1) enhancing the therapeutic efficacy of chemotherapeutic drugs – due to the combined effects of Cur and cisplatin determined in vitro and in vivo, experimental results demonstrate that Cur can enhance the antitumor effect of cisplatin in A549 cells in vitro, the combination markedly inhibiting tumor growth and promoting apoptosis in the A549-xenograft mouse model;108 2) achieving synergistic therapeutic effects – resveratrol and Cur synergistically cause apoptosis in breast cancer cells by p2 (Waf/Cip1)-mediated inhibition of the Hedgehog–Gli cascade;109 3) reversing drug resistance – the combination of cryptotanshinone and cisplatin leads to cell death and apoptosis, and cryptotanshinone reverses cisplatin resistance in human lung carcinoma A549 cells by downregulating the Nrf2 pathway;110 4) reducing the dose of drugs – combination therapy with triptolide and cisplatin completely suppresses tumor growth, suggesting that lower concentrations of cisplatin and triptolide may produce a synergistic anticancer effect;111 and 5) prolonging survival – As2O3 combined with ginsenoside Rg3 can significantly inhibit the proliferation of NCIH1299 cells and prolong survival of tumor-bearing nude mice, with a significant effect on lung cancer treatment.112 In addition, TCM-combination therapy results in good prognosis, has fewer adverse reactions, has long-lasting curative effects, regulates the expression of intracellular marker proteins, and reduces the side effects of drugs.112–115 Further superior effects are shown in Table 2. The anti–lung cancer and anti–breast cancer mechanisms of TCM-combination therapy are shown in Figures 3 and 4. TCM-combination therapy achieves the effects that single chemotherapeutic drugs fail to achieve, and has become the main direction in clinical and experimental research on antitumor therapy.116
Nevertheless, there are three possible interactions in drug combinations: antagonistic, additive, and synergistic effects. Therefore, if we do not understand interactions among drugs, blindly combining drugs will not only fail to achieve the desired response but also lead to reduced efficacy and increased toxicity, and even produce drug-borne diseases. For instance, the combination of paclitaxel (Ptx) and BA shows antagonism in breast cancer MCF7 cells,117 and Liu et al118 suggested that the combination of gambogic acid and bortezomib should be avoided in patients. In addition, attention should be paid to the proportion and sequence of the two drugs in combination.
Applications of targeted nanotechnology in TCM-combination therapy
During the early 20th century, Paul Ehrlich proposed the concept of targeted drugs, which consisted of three components: the drug, targeting moiety, and drug carrier. The main aim was to deliver the drug to the specific target organ under the specific guiding mechanism.168 Targeted preparations are characterized by increasing the intensity of pharmacological action in target tissue, controlling drug release, and decreasing systemic adverse reactions. Targeted drug-delivery systems have become one of the important high-profile topics in modern pharmacy. Nanotargeted drug-delivery systems have remarkable advantages in improving the bioavailability of drugs, enhancing the targeting ability of drugs, improving the distribution and pharmacokinetic properties of antitumor drugs in vivo and in vitro, increasing the stability of drugs, solubilizing poorly soluble drugs, protecting drugs from degradation in vivo, intelligently regulating the release of components, enhancing efficacy, and reducing toxicity.169–172 Moreover, metastasis of neoplastic cells is the major cause of death in cancer patients,173,174 and nanosize drug-delivery systems also provide an encouraging strategy for lymphatic metastases.175 In 2004, the National Cancer Institute (NCI) launched the NCI Cancer Nanotechnology Alliance, which aims to use nanotechnology to combat cancer.176
In recent years, many types of nanopreparations of TCMs, which involve the combination of nanotargeted drug-delivery systems and the advantages of TCM components in the treatment of tumors, have been reported.177–179 Simultaneously, nanotargeted drug-delivery systems are also promising multidrug carriers and allow the development of drug combinations by systematically controlling drug release and delivering drug to solid tumors.180 Codelivery of multiple antitumor agents in a single well-designed nanocarrier has significant advantages over monotherapy.181,182 Generally, drug targeting can be classified into three categories: passive targeted preparations, active targeted preparations, and other physicochemical targeted drug-delivery systems.
Passive targeted drug-delivery systems
In a passive targeted drug-delivery system, lipids, adipoids, proteins, and biodegradable high-molecular-weight polymers are mainly used as carriers, and the drug is encapsulated or embedded into various colloidal systems, forming stable structures, such as polymeric NPs (PNPs), micelles, nanovesicles, and liposomes, to increase drug concentration in tumor cells, decrease drug distribution in blood and other organs, and prevent toxicity and adverse reactions. This spontaneous accumulation, or “passive” targeting, is particularly effective against tumors, due to leaky angiogenic vessels and poor lymphatic drainage of the tumor, which is currently referred to as the enhanced permeability and retention (EPR) effect. That is to say, the high permeability of tumor blood vessels allows nanosystems to enter the interstitial spaces of the tumor, while impaired lymphatic filtration allows these nanosystems to remain there. This phenomenon does not exist in normal tissue. Currently, EPR-mediated drug delivery is considered effective in delivering drugs into tumors, especially nanocarriers (Figure 5). The size of the particles is also closely related to their distribution.183 The different sizes of these nanosystems decide the in vivo distribution behavior. Nanopreparations of <100 nm can be slowly accumulated in bone marrow; nanocarriers of 100–200 nm are apt to become enriched in the solid-tumor site; nanosystems of 0.2–3 μm are taken by macrophagocytes in the liver and spleen and particles of >7 μm are often intercepted by pulmonary capillary beds and enter the pulmonary tissues or alveoli.184
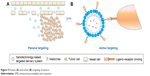  | Figure 5 Passive (A) and active (B) targeting of tumors. |
Liposomes
Liposomes are microvesicles with one or more aqueous cavities formed by the encapsulation of one or two amphiphilic molecular double-layer membranes, and the drug is encapsulated or embedded into the liposomes to form the liposome drug. Due to the similarity between the structure and the biological membrane, the encapsulation of water-soluble and fat-soluble drugs can reduce the drug dose, lower drug toxicity, delay release, lower in vivo elimination speed, change in vivo distribution of the drug, and achieve targeted release. Due to these advantages, liposome drugs have attracted considerable attention, and many studies have been carried out on them.185
Plumbagin is a quinonoid isolated from the roots of Plumbago zeylanica (bai hua dan in Chinese).186 It has high antiproliferative activity against a variety of tumor cell lines,187,188 and its anticancer properties have also been demonstrated in vivo.186 Celecoxib and plumbagin are two antitumor drugs that synergistically kill melanoma cells instead of normal cells. The combined use of these two agents in traditional approaches was not possible, due to their poor bioavailability and toxicity concerns. In order to circumvent these challenges, Raghavendra et al182 developed a nanoliposome containing celecoxib and plumbagin, named CelePlum 777, which has good stability and can release these two drugs at optimal proportion to achieve the maximum synergistic killing effect. Compared to nanoliposomes containing individual drugs, CelePlum 777 can enhance the inhibition of melanoma-cell proliferation in vitro and reduce the growth of melanoma tumors, with negligible systemic toxicity in nude mice. The goal of loading different individual drugs into a nanoliposome that releases the drugs at synergizing ratios was realized.
Previous studies have shown that Cur can reduce side effects caused by cisplatin (cis-diamminedichloroplatinum [CDDP]), including ototoxicity,189 nephrotoxicity,190 and neurotoxicity.191 In addition, Cur can also overcome resistance to CDDP and improve the sensitivity of hepatocellular carcinoma cells to CDDP.192 However, due to poor water-solubility and the different pharmacokinetics of CDDP and Cur,193 the cocktail of both drugs controlling the drug proportions and dose regimen at target cancer cells would be challenging. Based on a previous approach and advantages of the liposome, Cheng et al194 developed a liposomal delivery system using a reverse-microemulsion and film-dispersion method, which coencapsulated CDDP and Cur and transplanted them into hepatocellular carcinoma cells. The antitumor activity of CDDP-Cur liposomes against HepG2 cells was higher than that of free drug or encapsulated-monodrug therapy, and retention was prolonged (t½=2.38 hours). Therefore, coloaded liposomes can be used as an effective treatment for tumors, with great clinical application potential.
Polymeric nanoparticles
NPs are solid colloid particles 10–100 nm in size formed by drug dissolution, encapsulation, or adsorption on macromolecular materials. NPs within the particle-size range of 10–100 nm can hide the physicochemical characteristics of the drug, and the in vivo process of the drugs depends on the physicochemical characteristics of the carriers.195 NPs are characterized by a relatively simple preparation process, a significant solubilization effect on active components, significantly improved drug targeting to tumors, and proneness to surface modification. They can improve drug stability, reduce digestive tract stimulation, achieve sustained release or controlled release,196 effectively overcome multiple physiological barriers encountered in vivo, and achieve accurate, safe, efficient, and targeted therapeutic effects. Coating the surface of NPs with polyethylene glycol (PEG), or “PEGylation”, is a commonly used approach to improve the efficiency of drug delivery to target cells and tissue. PEGylation is capable of achieving prolonged blood circulation of nanocarriers, and can improve colloidal stability by providing steric surface hindrance. In addition, it has the ability to improve particle dispersion and decrease hemolysis.197,198 Polylactic-co-glycolic acid (PLGA), a biodegradable polymer, is atoxic to final degradation products and has been approved by the FDA.181,199 The antitumor effects of codelivered PNPs in TCM combinations are shown in Table 3.
  | Table 3 Polymeric nanoparticles (NPs) for codelivery of antitumor agents |
Lipid–polymer hybrid nanoparticles
As indicated, it has been demonstrated by the increasing numbers of research reports that biodegradable PNPs and liposomes have become the two main types of active TCM nanocarriers. Lipid–polymer hybrid NPs (LPNs) are nuclear-shell NPs formed by polymer core–lipid/lipid–PEG shells206,207 that have the advantages of biodegradable PNPs and bionic liposomes.
Li et al208 prepared LPNs and PNPs loaded with cisplatin and Cur. Results indicated that LPNs had higher anticancer efficacy than PNPs and free drugs. Cytotoxicity was highest in vitro and antitumor effect best in vivo. Therefore, LPNs can be used as a targeted and synergistic nanodrug codelivery system for tumor chemotherapy. In a similar study, Zhu et al209 developed vincristine–quercetin (Qct) dual-loaded LPNs. The experimental results proved that the LPNs loaded with both drugs exhibited better antitumor efficacy in vitro and in vivo.
Ruttala et al210 developed nanocarriers loaded with Ptx and Cur using a method different from the previously mentioned studies. Ptx-loaded albumin NPs were prepared and encapsulated in PEGylated hybrid liposomes containing Cur by a thin-film hydration method. This combination guaranteed the release of Ptx and Cur in a sustained and sequential manner. Compared with a cocktail combination, the dual-drug-loaded nanocarrier had a better cytotoxic effect at a lower dose. Therefore, such coloading drug-delivery systems can be used as a promising treatment method to improve clinical efficacy in various malignant tumors. NPs containing genistein and Ptx have also showed similar experimental results.211
Nanostructured lipid carriers
Nanostructured lipid carriers (NLCs) are novel lipid NPs and mixtures of solid and liquid lipids, which have the advantages of excellent drug-loading capacity and sustained-release properties. Jiang et al212 prepared etoposide (Etp)- and Cur-loaded NLCs by the solvent-injection technique. Etp-Cur NLCs had the highest cytotoxicity in vitro and higher accumulation in tumor tissues in vivo compared with other preparations, including Etp NLCs, Etp + Cur solution, Etp solution, and NLCs. In addition, Etp-Cur NLCs displayed low cytotoxicity in normal tissue in vivo, suggesting that NLCs could serve as a promising therapeutic strategy in the treatment of tumors.
Mesoporous silica nanoparticles
MSNs have attracted much attention due to their potential biomedical applications. MSNs possess many attractive features for application in the delivery of TCMs, such as the size of tuning particles/pores, large surface, large pore volume, high loading capacity, mass producibility, biocompatibility, and chemical inertia.213,214 TCMs can be dissolved in surfactant micelles, simultaneously hydrolyzed, and concentrated with silica to form NPs.
Jia et al215 prepared NPs using the self-assembly in situ drug-loading approach, in order to deliver the anticancer drug Ptx and the MDR-reversal agent tetrandrine (Tet) to increase the intracellular concentration of Ptx, enhance its antitumor effect, and minimize the exposure of normal cells to Ptx and Tet. The study demonstrated that Tet significantly increased the accumulation of NPs in cells. Furthermore, Ptx-Tet–cetyltrimethylammonium bromide MSNs suppressed tumor-cell growth more efficiently than the delivery of Ptx (Ptx–cetyltrimethylammonium bromide MSNs) or free Ptx alone. The prepared NPs released the drugs easily in the acidic environment of tumors, and thus, side effects and toxicity in normal tissue and organs were reduced. This nanocarrier may have important potential in clinical applications to avoid MDR by codelivering multiple TCMs. Solid self-nanoemulsifying drug-delivery systems containing tamoxifen and Qct have also shown similar experimental results.216
PEGylated lipid bilayer–supported mesoporous silica nanoparticles
The anticancer drug axitinib (Axt) is a small-molecule tyrosine kinase–receptor inhibitor of VEGFR1, -2, and -3.217,218 Another anticancer drug, celastrol (Cst), can induce the suppression of angiogenesis219 and enhance the antitumor activity of standard chemotherapeutic drugs.220 Choi et al221 loaded Cst into an MSN carrier and subsequently coated it with a lipid bilayer containing Axt, denoted by “ACML”, to increase the synergistic efficacy of the two agents. The difference in drug loading resulted in a sequential-release pattern where Axt was released first to exert its anticancer effect, and then Cst was released to further induce a synergistic effect. The experimental results showed that the synergistic apoptotic effect of ACML against cancer cells was stronger than the Axt-Cst cocktail. Moreover, ACML had a greater tumor-inhibitory effect than either drug administered alone in a tumor-xenograft mouse model. It has been proved that PEGylated lipid bilayer–supported MSNs have the potential to be used as an effective therapeutic strategy for malignant tumors.
Micellar systems
Self-assembled polymeric micelles have been studied widely, due to their excellent role in cancer treatment. Polymeric micelles have a core–shell structure, where hydrophobic drugs are soluble and remain stable in the hydrophobic core of the micelles, and the hydrophilic shell can prolong internal circulation and improve spatial stability by reducing opsonization during blood circulation. Furthermore, polymeric micelles can selectively and effectively accumulate in tumor tissues due to the EPR effect, thus enhancing the therapeutic effects of the loaded chemotherapeutic drugs. As such, codelivery micellar systems have attracted considerable attention.222,223
Doxorubicin (Dox) has extensive antitumor activity against various solid tumors, including lung cancers, melanoma, neurological cancers, sarcoma, leukemia, lymphoma, gastrointestinal cancers, genitourinary cancers, breast cancers, and ovarian cancers.14 Due to the rapid elimination of drugs in vivo, the cocktail combination of free Dox and Cur often fails to provide enough antitumor efficacy or low systemic toxicity. Furthermore, the combination of Dox and Cur has not been realized clinically. In recent years, a few studies have proposed that codelivering Dox and Cur may result in less toxicity, good drug-release profiles, and improved drug distribution in tumor tissue.224–226 Zhang et al223 prepared dual-loaded micelles with coencapsulated Dox and Cur. The experimental results showed that Dox delivered by this method prolonged systemic circulation and increased its accumulation in the tumor, resulting in a lower level of the toxic metabolite doxorubicinol in heart tissue than free Dox alone or the cocktail combination. In addition, Gu et al225 assembled micelles loaded with Dox and Cur. The micelles prolonged the circulation of Dox or Cur when compared with the individual administration of either, and exhibited strong inhibition of tumor growth and reduced Dox side effects.
Ptx has a broad spectrum of activity against various tumors. It has been used clinically for more than two decades. However, it is poorly soluble and has considerable limitations in clinical applications. In addition, Ptx extravasation of cancer cells caused by Pgp activity is also a main factor limiting its clinical efficacy.227,228 Abouzeid et al229 prepared micelles loaded with Ptx and Cur using the thin-film hydration method. Cur–Ptx-loaded micelles released the entrapped drugs with a slow pattern, and resulted in a threefold inhibition of tumors in vitro. The combination of Cur and Ptx was shown to reverse MDR in a resistant human ovarian adenocarcinoma model. Therefore, these combinations of micelles have significant advantages in vitro and in vivo over individual drug treatment, especially in drug-resistant tumors.
Microemulsions
A microemulsion is a transparent or semitransparent oil–water system with low viscosity, isotropism, and thermodynamic stability, and is spontaneously formed by an oil phase, water phase, emulsifier, and coemulsifier. As an ideal drug carrier, it has the advantages of solubilizing components with different solubility properties, good dispersibility, excellent absorbability, and increased bioavailability.230
BA is one of the most commonly used traditional chemotherapeutic drugs for the treatment of various cancers, including HCT116 human colon cancer,31 pancreatic cancer stem cells,32 and bladder cancer.33 It is known that BA has the ability to inhibit the function of Pgp.231 Meng et al230 developed nanoemulsions (NEs) coencapsulating Ptx and BA using rotary evaporation. The research showed that compared with other Ptx preparations, Ptx-BA NEs had a better antitumor effect on MCF7/Tax cells. Studies on cellular uptake have shown that Ptx-BA NEs accumulate effectively in cancer cells. More importantly, Ptx-BA NEs have a higher antitumor effect than other Ptx formulations used in antitumor in vivo studies. The combined delivery of Ptx and BA by NEs may provide a potential combination-treatment strategy to overcome MDR.
Nanovesicles
Nanovesicles are microvesicles with a quasiliposome dual-layer structure formed by the self-assembly of synthesized or naturally modified amphiphilic polymers and cholesterol in hydrophilic media. In contrast to other micromolecular vesicles, the polymer vesicle is characterized by good molecular designability, high intensity of the vesicle, excellent stability, and strong permeability.232,233 As a TCM carrier, it can improve histocompatibility and cell permeability and encapsulate hydrophilic drugs or lipophilic drugs.
Alemi et al234 loaded both Cur and Ptx into cationic PEGylated niosomal formulations using thin-film hydration method to enhance efficacy in MCF7 human breast adenocarcinoma cells. The combination of Ptx and Cur, particularly in the nanoniosome formulations, improved the effectiveness of cancer therapy. The novel cationic PEGylated niosome delivery of combined Ptx and Cur is an effective strategy in the treatment of breast cancer.
Schematic illustrations of these nanosystems for drugs are shown in Figure 6. In addition, graphene oxide,235 carbon nanotubes,236,237 nanorods,238 nanosponges,239 solid lipid NPs,240 nanometal–organic frameworks,241 metallosupramolecular nanogels,242 and microspheres243 all provide new opportunities for the antitumor effects of TCM-combination therapy.
  | Figure 6 Schematic illustrations of several nanosystems for drugs. |
Despite the EPR effect, most nanosystems fail to find their way toward tumor sites.244 Under most circumstances, 90% or more of the administered nanosystems end up in the liver or spleen, increasing adverse systemic reactions and causing low therapeutic efficacy.245,246
Active targeted drug-delivery systems
An active targeting preparation is a drug-delivery system which can utilize the modified drug carrier as a “missile” and deliver the drug selectively to the target area to allow the drug to accumulate and exert its efficacy. The mechanism of active targeting is that after surface modification with the specific targeted antibody or ligand via covalent or noncovalent binding, the nanodelivery system can avoid recognition and phagocytosis by macrophages and change natural in vivo distribution, so as to deliver the drug to the targeted tumor site and exert its active tumor-targeting effects.247 For instance, due to the difference between tumor cells and normal cells in terms of receptor expression or other biological characteristics, the tumor-targeted drug-delivery system has been developed to ensure that the drug acts only on tumor cells248 and induces the off-target effect in normal tissue, which has become a high-profile topic in studies on drug-delivery systems.
Transferrin (Tf)-modified nanocarriers
The Tf receptor is commonly present in normal cells and tumor cells. However, expression of the Tf receptor is approximately four to five times that on the surface of tumor cells than on normal cells.249 Transferrin can bind with the Tf receptor and be internalized into the cells under mediation by the receptors to reach the targeted site.
Cui et al250 designed Tf-decorated NPs (Tf-PEG Cur–Dox NPs) to codeliver Cur and Dox for breast cancer therapy. Results showed that the combination of Tf-PEG-Cur and Dox NPs exerted higher cytotoxicity in MCF7 cells compared with Tf-PEG-Cur NPs alone. Higher accumulation of Tf-PEG Cur–Dox NPs was observed in tumors compared to the Cur-Dox injection. Therefore, Tf-PEG Cur–Dox NPs displayed higher efficiency in vitro and in vivo, and resulted in efficient tumor-targeted drug delivery, reduced cytotoxicity, and a stronger antitumor effect.
Folic acid–modified nanocarriers
Similarly to distribution of the Tf receptor on surfaces of tumor-cell membrane, folic acid receptors on tumor cells are overexpressed compared with normal cells, and their activity is also significantly higher than that on normal cells. In addition, folic acid is characterized by low immunogenicity, high modifiability, and high storability. Utilization of the difference in folic acid–receptor expression between tumor sites and normal tissue can achieve targeted delivery of a folic acid–modified drug to cancer cells.251,252
Prodrugs of Ptx and baicalein containing dual-targeted ligands of folate and hyaluronic acid have been synthesized. NPs loaded with these prodrugs (Ptx–baicalein) have also been prepared and the synergistic antitumor effect evaluated in vitro and in vivo. The results showed that the Ptx–baicalein NP drug-delivery system delivered Ptx–baicalein prodrugs to drug-resistant human lung cancer cells, and the delivery was proven to be effective. In addition, Ptx–baicalein NPs exerted an enhanced synergistic anticancer effect, which also overcame MDR to Ptx.253
Low-density lipoprotein–modified nanocarriers
The low-density lipoprotein (LDL) receptor is widely present on the surface of various cells and tissue types, but is overexpressed in tumor cells. LDL is an endogenous NP with good biocompatibility, good biodegradability, and low immunogenicity, and can avoid being recognized and cleared by the in vivo endogenous reticuloendothelial system.254,255 Therefore, LDL is an ideal potential ligand for tumor targeting.
A novel nanocarrier containing Ptx-loaded micelles and siRNA-loaded LDL has been developed. Results showed that the delivery system delivered siRNA and Ptx directly to cancer cells, enhancing the intracellular release of drugs and genes, increasing intracellular drug concentration, decreasing drug efflux, prolonging circulation, and reversing MDR.256,257
Cell-penetrating/tumor-targeting peptide–modified nanocarriers
Nanocarriers using cell-penetrating and/or tumor-targeting peptides for functionalization are a promising strategy, and have attracted the attention of researchers. In our previous report, we reviewed the classification of polypeptide- and polypeptide-modified nanocarriers in detail.258 In this report, recent research progress is summarized in the following paragraphs.
Epigallocatechin-3-gallate (EGCG), a major polyphenol in green tea, has been widely studied as a potential anticancer drug. Narayanan et al259 prepared targeted drug-loaded core–shell NPs using anti-EGFR and anti-HER2 antibodies, and entrapped a combination of Ptx and EGCG at different doses in the core and shell, respectively, using emulsion precipitation. Cellular uptake in MDA-MB231 cells was higher for targeted NPs than untargeted NPs at 24 hours. The sequential release of EGCG followed by Ptx from this core–shell nanocarrier sensitized Ptx-resistant MDA-MB231 cells to Ptx, induced their apoptosis, and inhibited NFκB activation. In addition, EGFR-peptide (GE11)-targeted, pH-sensitive docetaxel Dtx–Cur NPs260 and arginylglycylaspartic acid–modified lipid-coated PLGA NPs targeting delivery of both sorafenib and Qct261 achieved significant inhibition of tumor growth in vitro and in vivo.
In addition, galactosamine can recognize and bind to the asialoglycoprotein receptor on the surface of hepatocellular carcinoma cells, and a galactosamine-mediated drug-delivery carrier was significant for targeted liver cancer therapy.262,263 Glycyrrhizin, glycyrrhetinic acid, and mannose can serve as the guiding group in liver-targeted drug-delivery systems, with good potential.264–270 As natural endogenous ligands, bile acids have good biocompatibility and are ideal routes for targeting hepatocellular cancer.271 In addition, sialic acid,272 human Nanog,273 and hyaluronic acid274 are excellent targets in cancer therapy. Vapreotide is a somatostatin analogue and can be also used as a ligand for targeted drug delivery based on its high affinity to somatostatin receptors, which are overexpressed in many tumor cells.275 Several studies have shown that double-modified nanocarriers have also attracted considerable attention in anticancer drug research.276,277 Dual or multiple targeting also provides a new approach for antitumor therapy.
Physicochemical targeted drug-delivery systems
Physicochemical targeting refers to the binding of magnetic, pH-sensitive, temperature-sensitive, or electromagnetic wave–responsive materials onto the surface of drug-delivery systems (such as NPs and liposomes) to make them respond to various stimuli in vitro and in vivo (such as pH, temperature, applied magnetic fields, ultrasonic waves, infrared rays, and electromagnetic radiation) to ensure that the drug acts directly on the target area, increases drug concentration at the lesion site, and reduces adverse reactions. Most studies have used magnetic NPs, temperature-sensitive NPs, and pH-sensitive NPs.
NPs that are pH-sensitive have been designed to promote uptake in tumor cells278 and accelerate drug release at tumor sites, as the extracellular pH (6.5–7.2) of the tumor is different to that of normal tissue.216 Zhang et al278 developed a codelivery system for Dox and Cur using pH-sensitive NPs. Enhanced release in the acidic environment of cancer cells and enhanced cellular internalization of the cargoes delivered from Dox–Cur NPs were observed in SMMC7721 cells and human umbilical vein endothelial cells compared to the free drugs. Therefore, pH-sensitive NPs can provide a promising strategy for the effective inhibition of cancer in a synergistic manner. Danafar et al279 achieved codelivery of Cur and sulforaphane (SF) with PEGylated gold-coated Fe3O4 magnetic NPs as an effective and promising antitumor agent. Results showed that SF–Cur coloaded Fe3O4@Au NPs caused a decrease in cell viability and induced apoptosis by increasing the Bax:Bcl2 ratio. Moreover, photosensitizer NPs,280 thermosensitive NPs,281 and redox-sensitive NPs282 also provide new opportunities for nanosystems with antitumor TCM combinations.
Conclusion
The significant challenge posed by cancers, as well as adverse reactions and drug resistance induced by long-term treatment of a single drug, compels us to change our focus from a single target to the regulation of networks in vivo. Many complex factors cause cancer; therefore, it is rational that treatment should involve multiple components, genes, systems, and target pathways. The combination of drugs has resulted in a new approach to cancer treatment. Rational combinations of drugs not only result in synergy but also reduce the occurrence of drug resistance and adverse reactions, which has resulted in combination therapy, thus becoming a significant antitumor treatment in the clinic and in research. As such, this significant research direction may allow medical researchers to identify a chemotherapeutic combination regimen with high efficacy and low toxicity.
The combination of TCMs for clinical therapy has increased. TCM combinations can exert improved synergistic antitumor effects by adjusting the multiple signaling pathways of tumor cells. Compared with single-drug therapy, combinations of TCMs can reduce the toxicity and side effects of chemotherapy drugs and increase the antitumor effect of drugs. Simultaneously, TCM combinations and chemical drugs can improve immunofunction, relieve clinical symptoms, improve patient survival and quality of life, and improve the efficacy of chemotherapy drugs.
It should be noted that there are usually three approaches in targeted nanotechnology-based TCM-combination-therapy: nanodrugs combined with conventional preparations, coloading of two or more anticancer drugs in a single nanocarrier system (recorded as codelivery), and combined administration of different nanosystems. The loading of two drugs into a single well-designed nanocarrier synchronizes the pharmacokinetic and biological distribution of different drugs to achieve a synergistic effect, which has distinct advantages. This method has been summarized in detail. An increasing number of studies have shown that dual nanosystems have distinct advantages in antitumor treatment and can provide drugs for different targets or sites of action, as they are administered flexibly using different dose/time schedules. Consequently, the remaining two methods urgently require further investigation. In addition, with further comparative analyses of these three research methods, the most suitable form of drug use for cancers can be identified to provide basic considerations in terms of design principles and management progress.
Acknowledgment
This study was financially supported by the National Natural Science Foundation of China (81673605).
Disclosure
The authors report no conflicts of interest in this work.
References
Chen W, Zheng R, Baade PD, et al. Cancer statistics in China, 2015. CA Cancer J Clin. 2016;66(2):115–132. | ||
Bray F, Ferlay J, Soerjomataram I, et al. GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;2018(68):394–424. | ||
Ferlay J, Colombet MI. Global and regional estimates of the incidence and mortality for 38 cancers: GLOBOCAN 2018. Lyon: International Agency for Research on Cancer/World Health Organization; 2018. | ||
Li FM, Wang XQ. Identifying anticancer peptides by using improved hybrid compositions. Sci Rep. 2016;6:33910. | ||
Zheng PP, Li J, Kros JM. Breakthroughs in modern cancer therapy and elusive cardiotoxicity: critical research-practice gaps, challenges, and insights. Med Res Rev. 2018;38(1):325–376. | ||
Wu Q, Yang Z, Nie Y, Shi Y, Fan D. Multi-drug resistance in cancer chemotherapeutics: mechanisms and lab approaches. Cancer Lett. 2014;347(2):159–166. | ||
Li S, Zhang B, Jiang D, Wei Y, Zhang N. Herb network construction and co-module analysis for uncovering the combination rule of traditional Chinese herbal formulae. BMC Bioinformatics. 2010;11(S11):S6. | ||
Heath JR. Nanotechnologies for biomedical Science and translational medicine. Proc Natl Acad Sci U S A. 2015;112(47):14436–14443. | ||
Ho BN, Pfeffer CM, Singh AT. Update on Nanotechnology-based drug delivery systems in cancer treatment. Anticancer Res. 2017;37(11):5975–5981. | ||
Wang K, Kievit FM, Zhang M. Nanoparticles for cancer gene therapy: recent advances, challenges, and strategies. Pharmacol Res. 2016;114:56–66. | ||
Yang Y, Yu C. Advances in silica based nanoparticles for targeted cancer therapy. Nanomedicine. 2016;12(2):317–332. | ||
Grover JK, Yadav SP. Pharmacological actions and potential uses of Momordica charantia: a review. J Ethnopharmacol. 2004;93(1):123–132. | ||
Cyranoski D. Why Chinese medicine is heading for clinics around the world. Nature. 2018;561(7724):448–450. | ||
Anand P, Sundaram C, Jhurani S, Kunnumakkara AB, Aggarwal BB. Curcumin and cancer: an “old-age” disease with an “age-old” solution. Cancer Lett. 2008;267(1):133–164. | ||
Duvoix A, Blasius R, Delhalle S, et al. Chemopreventive and therapeutic effects of curcumin. Cancer Lett. 2005;223(2):181–190. | ||
Zhang TD, Chen GQ, Wang ZG, Wang ZY, Chen SJ, Chen Z. Arsenic trioxide, a therapeutic agent for APL. Oncogene. 2001;20(49):7146–7153. | ||
Dogra S, Bandi S, Viswanathan P, Gupta S. Arsenic trioxide amplifies cisplatin toxicity in human tubular cells transformed by HPV-16 E6/E7 for further therapeutic directions in renal cell carcinoma. Cancer Lett. 2015;356(2 Pt B):953–961. | ||
Ji H, Li Y, Jiang F, et al. Inhibition of transforming growth factor beta/SMAD signal by MiR-155 is involved in arsenic trioxide-induced anti-angiogenesis in prostate cancer. Cancer Sci. 2014;105(12):1541–1549. | ||
Wang X, Jiang F, Mu J, et al. Arsenic trioxide attenuates the invasion potential of human liver cancer cells through the demethylation-activated microRNA-491. Toxicol Lett. 2014;227(2):75–83. | ||
Jiang X, Chen C, Zhao W, Zhang Z. Sodium arsenite and arsenic trioxide differently affect the oxidative stress, genotoxicity and apoptosis in A549 cells: an implication for the paradoxical mechanism. Environ Toxicol Pharmacol. 2013;36(3):891–902. | ||
Walker AM, Stevens JJ, Ndebele K, Tchounwou PB. Evaluation of arsenic trioxide potential for lung cancer treatment: assessment of apoptotic mechanisms and oxidative damage. J Cancer Sci Ther. 2016;08(01):1–9. | ||
Alkhalaf M, Jaffal S. Potent antiproliferative effects of resveratrol on human osteosarcoma SJSA1 cells: novel cellular mechanisms involving the ERKs/p53 cascade. Free Radic Biol Med. 2006;41(2):318–325. | ||
Zhu W, Qin W, Zhang K, et al. Trans-resveratrol alters mammary promoter hypermethylation in women at increased risk for breast cancer. Nutr Cancer. 2012;64(3):393–400. | ||
Patel KR, Brown VA, Jones DJ, et al. Clinical pharmacology of resveratrol and its metabolites in colorectal cancer patients. Cancer Res. 2010;70(19):7392–7399. | ||
Rauf A, Imran M, Butt MS, Nadeem M, Peters DG, Mubarak MS. Resveratrol as an anti-cancer agent: a review. Crit Rev Food Sci Nutr. 2018;58(9):1428–1447. | ||
Cai H, Scott E, Kholghi A, et al. Cancer chemoprevention: evidence of a nonlinear dose response for the protective effects of resveratrol in humans and mice. Sci Transl Med. 2015;7(298):298ra117. | ||
Oi N, Yuan J, Malakhova M, et al. Resveratrol induces apoptosis by directly targeting Ras-GTPase-activating protein SH3 domain-binding protein 1. Oncogene. 2015;34(20):2660–2671. | ||
Zou M, Wang J, Gao J, Han H, Fang Y. Phosphoproteomic analysis of the antitumor effects of ginsenoside Rg3 in human breast cancer cells. Oncol Lett. 2018;15(3):2889–2898. | ||
Yang X, Zou J, Cai H, et al. Ginsenoside Rg3 inhibits colorectal tumor growth via down-regulation of C/EBPβ/NF-κB signaling. Biomed Pharmacother. 2017;96:1240–1245. | ||
Liu T, Zhao L, Hou H, Ding L, Chen W, Li X. Ginsenoside 20(S)-Rg3 suppresses ovarian cancer migration via hypoxia-inducible factor 1 alpha and nuclear factor-kappa B signals. Tumour Biol. 2017;39(5):101042831769222. | ||
Su MQ, Zhou YR, Rao X, et al. Baicalein induces the apoptosis of HCT116 human colon cancer cells via the upregulation of DEPP/Gadd45a and activation of MAPKs. Int J Oncol. 2018;53(2):750–760. | ||
Song L, Chen X, Wang P, Gao S, Qu C, Liu L. Effects of baicalein on pancreatic cancer stem cells via modulation of sonic hedgehog pathway. Acta Biochim Biophys Sin. 2018;50(6):586–596. | ||
Chao JI, Su WC, Liu H-F. Baicalein induces cancer cell death and proliferation retardation by the inhibition of CDC2 kinase and survivin associated with opposite role of p38 mitogen-activated protein kinase and Akt. Mol Cancer Ther. 2007;6(11):3039–3048. | ||
Zhou Q, Wang S, Zhang H, et al. The combination of baicalin and baicalein enhances apoptosis via the ERK/p38 MAPK pathway in human breast cancer cells. Acta Pharmacol Sin. 2009;30(12):1648–1658. | ||
Yan XI, Rui X, Zhang KAI. Baicalein inhibits the invasion of gastric cancer cells by suppressing the activity of the p38 signaling pathway. Oncol Rep. 2015;33(2):737–743. | ||
Wang YF, Li T, Tang ZH, et al. Baicalein triggers autophagy and inhibits the protein kinase B/Mammalian target of rapamycin pathway in hepatocellular carcinoma HepG2 cells. Phytother Res. 2015;29(5):674–679. | ||
Wu JY, Tsai KW, Li YZ, et al. Anti-Bladder-Tumor effect of baicalein from Scutellaria baicalensis Georgi and its application in vivo. Evid Based Complement Alternat Med. 2013;2013:579751. | ||
Guo Z, Hu X, Xing Z, et al. Baicalein inhibits prostate cancer cell growth and metastasis via the caveolin-1/AKT/mTOR pathway. Mol Cell Biochem. 2015;406(1–2):111–119. | ||
Peng Y, Guo C, Yang Y, et al. Baicalein induces apoptosis of human cervical cancer HeLa cells in vitro. Mol Med Rep. 2015;11(3):2129–2134. | ||
Chandrashekar N, Selvamani A, Subramanian R, Pandi A, Thiruvengadam D. Baicalein inhibits pulmonary carcinogenesis-associated inflammation and interferes with COX-2, MMP-2 and MMP-9 expressions in-vivo. Toxicol Appl Pharmacol. 2012;261(1):10–21. | ||
Yu J, Guo QL, You QD, et al. Gambogic acid-induced G2/M phase cell-cycle arrest via disturbing CDK7-mediated phosphorylation of CDC2/p34 in human gastric carcinoma BGC-823 cells. Carcinogenesis. 2007;28(3):632–638. | ||
Wang X, Deng R, Lu Y, et al. Gambogic acid as a non-competitive inhibitor of ATP-binding cassette transporter B1 reverses the multidrug resistance of human epithelial cancers by promoting ATP-binding cassette transporter B1 protein degradation. Basic Clin Pharmacol Toxicol. 2013;112(1):25–33. | ||
Wu ZQ, Guo QL, You QD, Zhao L, Gu H-Y. Gambogic acid inhibits proliferation of human lung carcinoma SPC-A1 cells in vivo and in vitro and represses telomerase activity and telomerase reverse transcriptase mRNA expression in the cells. Biol Pharm Bull. 2004;27(11):1769–1774. | ||
Wang C, Zhang H, Chen Y, Shi F, Chen B. Gambogic acid-loaded magnetic Fe(3)O(4) nanoparticles inhibit Panc-1 pancreatic cancer cell proliferation and migration by inactivating transcription factor ETS1. Int J Nanomedicine. 2012;7:781–787. | ||
Guo QL, You QD, Wu ZQ, Yuan ST, Zhao L. General gambogic acids inhibited growth of human hepatoma SMMC-7721 cells in vitro and in nude mice. Acta Pharmacol Sin. 2004;25(6):769–774. | ||
Zhu X, Zhang H, Lin Y, et al. Mechanisms of gambogic acid-induced apoptosis in non-small cell lung cancer cells in relation to transferrin receptors. J Chemother. 2009;21(6):666–672. | ||
Nie F, Zhang X, Qi Q, et al. Reactive oxygen species accumulation contributes to gambogic acid-induced apoptosis in human hepatoma SMMC-7721 cells. Toxicology. 2009;260(1–3):60–67. | ||
Yan F, Wang M, Chen H, et al. Gambogenic acid mediated apoptosis through the mitochondrial oxidative stress and inactivation of Akt signaling pathway in human nasopharyngeal carcinoma CNE-1 cells. Eur J Pharmacol. 2011;652(1–3):23–32. | ||
Chen HB, Zhou LZ, Mei L, et al. Gambogenic acid-induced time- and dose-dependent growth inhibition and apoptosis involving Akt pathway inactivation in U251 glioblastoma cells. J Nat Med. 2012;66(1):62–69. | ||
Wang K, Tang Y, Sun M, et al. The mechanism of neogambogic acid-induced apoptosis in human MCF-7 cells. Acta Biochim Biophys Sin. 2011;43(9):698–702. | ||
Mei W, Dong C, Hui C, et al. Gambogenic acid kills lung cancer cells through aberrant autophagy. PLoS One. 2014;9(1):e83604. | ||
Li Q, Cheng H, Zhu G, et al. Gambogenic acid inhibits proliferation of A549 cells through apoptosis-inducing and cell cycle arresting. Biol Pharm Bull. 2010;33(3):415–420. | ||
Cheng H, Su JJ, Peng JY, et al. Gambogenic acid inhibits proliferation of A549 cells through apoptosis inducing through up-regulation of the p38 MAPK cascade. J Asian Nat Prod Res. 2011;13(11):993–1002. | ||
Zhang X, Xu Q, Saiki I. Quercetin inhibits the invasion and mobility of murine melanoma B16-BL6 cells through inducing apoptosis via decreasing Bcl-2 expression. Clin Exp Metastasis. 2000;18(5):415–421. | ||
Tsai PH, Cheng CH, Lin C-Y, et al. Dietary flavonoids luteolin and quercetin suppressed cancer stem cell properties and metastatic potential of isolated prostate cancer cells. Anticancer Res. 2016;36(12):6367–6380. | ||
Jia L, Huang S, Yin X, Zan Y, Guo Y, Han L. Quercetin suppresses the mobility of breast cancer by suppressing glycolysis through Akt-mTOR pathway mediated autophagy induction. Life Sci. 2018;208:123–130. | ||
Calgarotto AK, Maso V, Junior GC, et al. Antitumor activities of quercetin and green tea in xenografts of human leukemia HL60 cells. Sci Rep. 2018;8(1):3459. | ||
Anand David AV, Arulmoli R, Parasuraman S. Overviews of biological importance of quercetin: a bioactive flavonoid. Pharmacogn Rev. 2016;10(20):84–89. | ||
Lugli E, Ferraresi R, Roat E, et al. Quercetin inhibits lymphocyte activation and proliferation without inducing apoptosis in peripheral mononuclear cells. Leuk Res. 2009;33(1):140–150. | ||
Zhang H, Li H, Liu Z, et al. Triptolide inhibits the proliferation and migration of medulloblastoma DAOY cells by upregulation of microRNA-138. J Cell Biochem. 2018;119(12):9866–9877. | ||
Mao X, Tong J, Wang Y, Zhu Z, Yin Y, Wang Y. Triptolide exhibits antitumor effects by reversing hypermethylation of WIF1 in lung cancer cells. Mol Med Rep. 2018;18(3):3041–3049. | ||
Zhou ZL, Yang YX, Ding J, Li YC, Miao Z-H. Triptolide: structural modifications, structure-activity relationships, bioactivities, clinical development and mechanisms. Nat Prod Rep. 2012;29(4):457–475. | ||
Li SG, Shi QW, Yuan LY, et al. C-Myc-dependent repression of two oncogenic miRNA clusters contributes to triptolide-induced cell death in hepatocellular carcinoma cells. J Exp Clin Cancer Res. 2018;37:51. | ||
Wang Y, Liu T, Li H. Enhancement of triptolide-loaded micelles on tumorigenicity inhibition of human ovarian cancer. J Biomater Sci Polym Ed. 2016;27(7):545–556. | ||
Hu G, Gong X, Wang L, et al. Triptolide promotes the clearance of α-synuclein by enhancing autophagy in neuronal cells. Mol Neurobiol. 2017;54(3):2361–2372. | ||
Kim SH, Kang JG, Kim CS, et al. Synergistic cytotoxicity of BIIB021 with triptolide through suppression of PI3K/AKT/mTOR and NF-κB signal pathways in thyroid carcinoma cells. Biomed Pharmacother. 2016;83:22–32. | ||
Li W, Yang Y, Hu Z, Ling S, Fang M. Neuroprotective effects of DAHP and triptolide in focal cerebral ischemia via apoptosis inhibition and PI3K/Akt/mTOR pathway activation. Front Neuroanat. 2015;9:48. | ||
Yao Z, Wan Y, Li B, et al. Berberine induces mitochondrialmediated apoptosis and protective autophagy in human malignant pleural mesothelioma NCIH2452 cells. Oncol Rep. 2018;40(6):3603–3610. | ||
Fukuda K, Hibiya Y, Mutoh M, Koshiji M, Akao S, Fujiwara H. Inhibition by berberine of cyclooxygenase-2 transcriptional activity in human colon cancer cells. J Ethnopharmacol. 1999;66(2):227–233. | ||
Wang Z, Wang Y-S, Chang Z-M, et al. Berberine-loaded Janus nanocarriers for magnetic field-enhanced therapy against hepatocellular carcinoma. Chem Biol Drug Des. 2017;89(3):464–469. | ||
Kuo HP, Chuang TC, Yeh MH, et al. Growth suppression of HER2-overexpressing breast cancer cells by berberine via modulation of the HER2/PI3K/AKT signaling pathway. J Agric Food Chem. 2011;59(15):8216–8224. | ||
Kim S, Han J, Lee SK, et al. Berberine suppresses the TPA-induced MMP-1 and MMP-9 expressions through the inhibition of PKC-α in breast cancer cells. J Surg Res. 2012;176(1):e21–e29. | ||
Lin CC, Yang JS, Chen JT, et al. Berberine induces apoptosis in human HSC-3 oral cancer cells via simultaneous activation of the death receptor-mediated and mitochondrial pathway. Anticancer Res. 2007;27(5A):3371–3378. | ||
Mantena SK, Sharma SD, Katiyar SK. Berberine inhibits growth, induces G1 arrest and apoptosis in human epidermoid carcinoma A431 cells by regulating Cdki-Cdk-cyclin cascade, disruption of mitochondrial membrane potential and cleavage of caspase 3 and PARP. Carcinogenesis. 2006;27(10):2018–2027. | ||
Shin DS, Kim HN, Shin KD, et al. Cryptotanshinone inhibits constitutive signal transducer and activator of transcription 3 function through blocking the dimerization in DU145 prostate cancer cells. Cancer Res. 2009;69(1):193–202. | ||
Chen W, Luo Y, Liu L, et al. Cryptotanshinone inhibits cancer cell proliferation by suppressing mammalian target of rapamycin-mediated cyclin D1 expression and RB phosphorylation. Cancer Prev Res. 2010;3(8):1015–1025. | ||
Chen L, Wang HJ, Xie W, Yao Y, Zhang YS, Wang H. Cryptotanshinone inhibits lung tumorigenesis and induces apoptosis in cancer cells in vitro and in vivo. Mol Med Rep. 2014;9(6):2447–2452. | ||
Xiao W, Zhu MX, Zhu MX, Pan XJ, Yang ZH, Zhou S-Y. Inhibition of cyclooxygenase-2 by tetramethylpyrazine and its effects on A549 cell invasion and metastasis. Int J Oncol. 2012;40(6):2029–2037. | ||
Wang Y, Fu Q, Zhao W. Tetramethylpyrazine inhibits osteosarcoma cell proliferation via downregulation of NF-κB in vitro and in vivo. Mol Med Rep. 2013;8(4):984–988. | ||
Wang SJ, Gao Y, Chen H, et al. Dihydroartemisinin inactivates NF-kappaB and potentiates the anti-tumor effect of gemcitabine on pancreatic cancer both in vitro and in vivo. Cancer Lett. 2010;293(1):99–108. | ||
Ji Y, Zhang YC, Pei LB, Shi LL, Yan JL, Ma XH. Anti-tumor effects of dihydroartemisinin on human osteosarcoma. Mol Cell Biochem. 2011;351(1–2):99–108. | ||
Gao X, Luo Z, Xiang T, Wang K, Li J, Wang P. Dihydroartemisinin induces endoplasmic reticulum stress-mediated apoptosis in HepG2 human hepatoma cells. Tumori. 2011;97(6):771–780. | ||
Lai H, Singh NP. Selective cancer cell cytotoxicity from exposure to dihydroartemisinin and holotransferrin. Cancer Lett. 1995;91(1):41–46. | ||
Lai H, Sasaki T, Singh NP, Messay A. Effects of artemisinin-tagged holotransferrin on cancer cells. Life Sci. 2005;76(11):1267–1279. | ||
Chen H, Gu S, Dai H, Li X, Zhang Z. Dihydroartemisinin sensitizes human lung adenocarcinoma A549 cells to arsenic trioxide via apoptosis. Biol Trace Elem Res. 2017;179(2):203–212. | ||
Oh M, Choi YH, Choi S, et al. Anti-proliferating effects of ginsenoside Rh2 on MCF-7 human breast cancer cells. Int J Oncol. 1999;14(5):869–875. | ||
Nakata H, Kikuchi Y, Tode T, et al. Inhibitory effects of ginsenoside Rh2 on tumor growth in nude mice bearing human ovarian cancer cells. Jpn J Cancer Res. 1998;89(7):733–740. | ||
Xie X, Eberding A, Madera C, et al. Rh2 synergistically enhances paclitaxel or mitoxantrone in prostate cancer models. J Urol. 2006;175(5):1926–1931. | ||
Xia T, Wang YN, Zhou CX, et al. Ginsenoside Rh2 and Rg3 inhibit cell proliferation and induce apoptosis by increasing mitochondrial reactive oxygen species in human leukemia Jurkat cells. Mol Med Rep. 2017;15(6):3591–3598. | ||
Yang J, Yuan D, Xing T, et al. Ginsenoside Rh2 inhibiting HCT116 colon cancer cell proliferation through blocking PDZ-binding kinase/T-LAK cell-originated protein kinase. J Ginseng Res. 2016;40(4):400–408. | ||
Yang Z, Zhao T, Liu H, Zhang L. Ginsenoside Rh2 inhibits hepatocellular carcinoma through β-catenin and autophagy. Sci Rep. 2016;6(1):19383. | ||
Huang J, Peng K, Wang L, et al. Ginsenoside Rh2 inhibits proliferation and induces apoptosis in human leukemia cells via TNF-α signaling pathway. Acta Biochim Biophys Sin. 2016;48(8):750–755. | ||
Li S, Gao Y, Ma W, et al. EGFR signaling-dependent inhibition of glioblastoma growth by ginsenoside Rh2. Tumor Biol. 2014;35(6):5593–5598. | ||
Han S, Jeong AJ, Yang H, et al. Ginsenoside 20(S)-Rh2 exerts anti-cancer activity through targeting IL-6-induced JAK2/STAT3 pathway in human colorectal cancer cells. J Ethnopharmacol. 2016;194:83–90. | ||
Guan N, Huo X, Zhang Z, Zhang S, Luo J, Guo W. Ginsenoside Rh2 inhibits metastasis of glioblastoma multiforme through Akt-regulated MMP13. Tumor Biol. 2015;36(9):6789–6795. | ||
Abedinpour P, Baron VT, Chrastina A, et al. Plumbagin improves the efficacy of androgen deprivation therapy in prostate cancer: a pre-clinical study. Prostate. 2017;77(16):1550–1562. | ||
Kawiak A, Domachowska A. Plumbagin suppresses the invasion of HER2-overexpressing breast cancer cells through inhibition of IKKα-Mediated NF-κB activation. PLoS One. 2016;11(10):e0164064. | ||
Oh TI, Yun JM, Park EJ, et al. Plumbagin suppresses α-MSH-Induced melanogenesis in B16F10 mouse melanoma cells by inhibiting tyrosinase activity. Int J Mol Sci. 2017;18(2):320. | ||
Liu Y, Cai Y, He C, Chen M, Li H. Anticancer properties and pharmaceutical applications of plumbagin: a review. Am J Chin Med. 2017;45(3):423–441. | ||
Pan ST, Li ZL, He ZX, Qiu JX, Zhou S-F. Molecular mechanisms for tumour resistance to chemotherapy. Clin Exp Pharmacol Physiol. 2016;43(8):723–737. | ||
Crona DJ, Faso A, Nishijima TF, Mcgraw KA, Galsky MD, Milowsky MI. A systematic review of strategies to prevent cisplatin-induced nephrotoxicity. Oncologist. 2017;22(5):609–619. | ||
Zhu J, Carozzi VA, Reed N, et al. Ethoxyquin provides neuroprotection against cisplatin-induced neurotoxicity. Sci Rep. 2016;6(1):28861. | ||
Karasawa T, Steyger PS. An integrated view of cisplatin-induced nephrotoxicity and ototoxicity. Toxicol Lett. 2015;237(3):219–227. | ||
Hu S, Li X, Xu R, et al. The synergistic effect of resveratrol in combination with cisplatin on apoptosis via modulating autophagy in A549 cells. Acta Biochim Biophys Sin. 2016;48(6):528–535. | ||
Gao LP, Li Z, Guo ZY, Zhao Y-M. The effects of vitamin C on DDP-induced anemia in rats. Toxicol Mech Methods. 2013;23(6):383–388. | ||
Lu DY, Chen EH, Wu HY, et al. Anticancer drug combinations, how far we can go through? Anticancer Agents Med Chem. 2017;17(1):21–28. | ||
Webster RM. Combination therapies in oncology. Nat Rev Drug Discov. 2016;15(2):81–82. | ||
Zhang W, Shi H, Chen C, et al. Curcumin enhances cisplatin sensitivity of human NSCLC cell lines through influencing Cu-Sp1-CTR1 regulatory loop. Phytomedicine. 2018;48:51–61. | ||
Mohapatra P, Satapathy SR, Siddharth S, Das D, Nayak A, Kundu CN. Resveratrol and curcumin synergistically induces apoptosis in cigarette smoke condensate transformed breast epithelial cells through a p21(Waf1/Cip1) mediated inhibition of Hh-Gli signaling. Int J Biochem Cell Biol. 2015;66:75–84. | ||
Xia C, Bai X, Hou X, et al. Cryptotanshinone reverses cisplatin resistance of human lung carcinoma A549 cells through down-regulating Nrf2 pathway. Cell Physiol Biochem. 2015;37(2):816–824. | ||
Li CJ, Chu CY, Huang LH, et al. Synergistic anticancer activity of triptolide combined with cisplatin enhances apoptosis in gastric cancer in vitro and in vivo. Cancer Lett. 2012;319(2):203–213. | ||
Che JB, Liu ZH, Ma HB, et al. Influence of As2O3 combined with ginsenosides Rg3 on inhibition of lung cancer NCI-H1299 cells and on subsistence of nude mice bearing hepatoma. Asian Pac J Trop Med. 2014;7(10):772–775. | ||
Yuan Z, Jiang H, Zhu X, Liu X, Li J. Ginsenoside Rg3 promotes cytotoxicity of paclitaxel through inhibiting NF-κB signaling and regulating Bax/Bcl-2 expression on triple-negative breast cancer. Biomed Pharmacother. 2017;89:227–232. | ||
Chang L, Huo B, Lv Y, Wang Y, Liu W. Ginsenoside Rg3 enhances the inhibitory effects of chemotherapy on esophageal squamous cell carcinoma in mice. Mol Clin Oncol. 2014;2(6):1043–1046. | ||
Tang Q, Ji F, Sun W, et al. Combination of baicalein and 10-hydroxy camptothecin exerts remarkable synergetic anti-cancer effects. Phytomedicine. 2016;23(14):1778–1786. | ||
Zhen YS. Anticancer Drug research and development. Beijing, China: Chemical Industry Press; 2004. | ||
Zhan Y, Chen Y, Liu R, Zhang H, Zhang Y. Potentiation of paclitaxel activity by curcumin in human breast cancer cell by modulating apoptosis and inhibiting EGFR signaling. Arch Pharm Res. 2014;37(8):1086–1095. | ||
Liu N, Huang H, Xu L, et al. The combination of proteasome inhibitors bortezomib and gambogic acid triggers synergistic cytotoxicity in vitro but not in vivo. Toxicol Lett. 2014;224(3):333–340. | ||
Majumdar AP, Banerjee S, Nautiyal J, et al. Curcumin synergizes with resveratrol to inhibit colon cancer. Nutr Cancer. 2009;61(4):544–553. | ||
Masuelli L, Marzocchella L, Focaccetti C, et al. Resveratrol and diallyl disulfide enhance curcumin-induced sarcoma cell apoptosis. Front Biosci. 2012;17(1):498–508. | ||
Sánchez Y, Simón GP, Calviño E, de Blas E, Aller P. Curcumin stimulates reactive oxygen species production and potentiates apoptosis induction by the antitumor drugs arsenic trioxide and lonidamine in human myeloid leukemia cell lines. J Pharmacol Exp Ther. 2010;335(1):114–123. | ||
Fehl DJ, Ahmed M. Curcumin promotes the oncoltyic capacity of vesicular stomatitis virus for the treatment of prostate cancers. Virus Res. 2017;228:14–23. | ||
Zhang JY, Lin MT, Zhou MJ, et al. Combinational treatment of curcumin and quercetin against gastric cancer MGC-803 cells in vitro. Molecules. 2015;20(6):11524–11534. | ||
Li Y, Feng L, Li Y, Jiang W, Shan N, Wang X. Artesunate possesses anti-leukemia properties that can be enhanced by arsenic trioxide. Leuk Lymphoma. 2014;55(6):1366–1372. | ||
Tomuleasa C, Soritau O, Fischer-Fodor E, et al. Arsenic trioxide plus cisplatin/interferon α-2b/doxorubicin/capecitabine combination chemotherapy for unresectable hepatocellular carcinoma. Hematol Oncol Stem Cell Ther. 2011;4(2):60–66. | ||
Wang W, Qin SK, Chen BA, Chen HY. Experimental study on antitumor effect of arsenic trioxide in combination with cisplatin or doxorubicin on hepatocellular carcinoma. World J Gastroenterol. 2001;7(5):702–705. | ||
Zhao YY, Yu L, Liu BL, He XJ, Zhang B-Y. Downregulation of P-gp, Ras and p-ERK1/2 contributes to the arsenic trioxide-induced reduction in drug resistance towards doxorubicin in gastric cancer cell lines. Mol Med Rep. 2015;12(5):7335–7343. | ||
Yu Q, Chen B, Zhang X, Qian W, Ye B, Zhou Y. Arsenic trioxide-enhanced, matrine-induced apoptosis in multiple myeloma cell lines. Planta Med. 2013;79(09):775–781. | ||
Rai G, Mishra S, Suman S, Shukla Y. Resveratrol improves the anticancer effects of doxorubicin in vitro and in vivo models: a mechanistic insight. Phytomedicine. 2016;23(3):233–242. | ||
Jiang Q, Yang M, Qu Z, Zhou J, Zhang Q. Resveratrol enhances anticancer effects of paclitaxel in HepG2 human liver cancer cells. BMC Complement Altern Med. 2017;17(1):477. | ||
Kong F, Zhang R, Zhao X, Zheng G, Wang Z, Wang P. Resveratrol raises in vitro anticancer effects of paclitaxel in NSCLC cell line A549 through COX-2 expression. Korean J Physiol Pharmacol. 2017;21(5):465–474. | ||
Yuan Y, Xue X, Guo RB, Sun XL, Hu G. Resveratrol enhances the antitumor effects of temozolomide in glioblastoma via ROS-dependent AMPK-TSC-mTOR signaling pathway. CNS Neurosci Ther. 2012;18(7):536–546. | ||
Tomas-Hernández S, Blanco J, Rojas C, et al. Resveratrol potently counteracts quercetin starvation-induced autophagy and sensitizes HepG2 cancer cells to apoptosis. Mol Nutr Food Res. 2018;62(5):1700610. | ||
Zhao XY, Yang S, Chen YR, Li PC, Dou MM, Zhang J. Resveratrol and arsenic trioxide act synergistically to kill tumor cells in vitro and in vivo. PLoS One. 2014;9(6):e98925. | ||
Kim SM, Lee SY, Yuk DY, et al. Inhibition of NF-kappaB by ginsenoside Rg3 enhances the susceptibility of colon cancer cells to docetaxel. Arch Pharm Res. 2009;32(5):755–765. | ||
Pan Q, Xue M, Xiao S-S, Wan Y-J, Xu D-B. A combination therapy with baicalein and taxol promotes mitochondria-mediated cell apoptosis: involving in Akt/β-Catenin signaling pathway. DNA Cell Biol. 2016;35(11):646–656. | ||
Wang LH, Li Y, Yang SN, et al. Gambogic acid synergistically potentiates cisplatin-induced apoptosis in non-small-cell lung cancer through suppressing NF-κB and MAPK/HO-1 signalling. Br J Cancer. 2014;110(2):341–352. | ||
Xia G, Wang H, Song Z, Meng Q, Huang X, Huang X. Gambogic acid sensitizes gemcitabine efficacy in pancreatic cancer by reducing the expression of ribonucleotide reductase subunit-M2 (RRM2). J Exp Clin Cancer Res. 2017;36(1):107. | ||
Jiang XL, Zhang Y, Luo CL, Wu XH. Targeting renal cell carcinoma with gambogic acid in combination with sunitinib in vitro and in vivo. Asian Pac J Cancer Prev. 2012;13(12):6463–6468. | ||
He Y, Ding J, Lin Y, et al. Gambogenic acid alters chemosensitivity of breast cancer cells to adriamycin. BMC Complement Altern Med. 2015;15(1):181. | ||
Chen R, Zhang H, Liu P, Wu X, Chen B. Gambogenic acid synergistically potentiates bortezomib-induced apoptosis in multiple myeloma. J Cancer. 2017;8(5):839–851. | ||
Ma ZS, Huynh TH, Ng CP, do PT, Nguyen TH, Huynh H. Reduction of CWR22 prostate tumor xenograft growth by combined tamoxifen-quercetin treatment is associated with inhibition of angiogenesis and cellular proliferation. Int J Oncol. 2004;24(5):1297–1304. | ||
Sun S, Gong F, Liu P, Miao Q. Metformin combined with quercetin synergistically repressed prostate cancer cells via inhibition of VEGF/PI3K/Akt signaling pathway. Gene. 2018;664:50–57. | ||
Chan ST, Chuang CH, Lin YC, Liao JW, Lii CK, Yeh S-L. Quercetin enhances the antitumor effect of trichostatin A and suppresses muscle wasting in tumor-bearing mice. Food Funct. 2018;9(2):871–879. | ||
Tang Q, Ji F, Wang J, Guo L, Li Y, Bao Y. Quercetin exerts synergetic anti-cancer activity with 10-hydroxy camptothecin. Eur J Pharm Sci. 2017;109:223–232. | ||
Lei CS, Hou YC, Pai MH, Lin MT, Yeh S-L. Effects of quercetin combined with anticancer drugs on metastasis-associated factors of gastric cancer cells: in vitro and in vivo studies. J Nutr Biochem. 2018;51:105–113. | ||
Alsaied OA, Sangwan V, Banerjee S, et al. Sorafenib and triptolide as combination therapy for hepatocellular carcinoma. Surgery. 2014;156(2):270–279. | ||
Matsui Y, Watanabe J, Ikegawa M, Kamoto T, Ogawa O, Nishiyama H. Cancer-specific enhancement of cisplatin-induced cytotoxicity with triptolide through an interaction of inactivated glycogen synthase kinase-3beta with p53. Oncogene. 2008;27(33):4603–4614. | ||
Liu Y, Xiao E, Yuan L, Li G. Triptolide Synergistically Enhances Antitumor Activity of Oxaliplatin in Colon Carcinoma In Vitro and In Vivo. DNA Cell Biol. 2014;33(7):418–425. | ||
Tang XY, Zhu YQ, Tao WH, Wei B, Lin XL. Synergistic effect of triptolide combined with 5-fluorouracil on colon carcinoma. Postgrad Med J. 2007;83(979):338–343. | ||
Qiao Z, He M, He MU, et al. Synergistic antitumor activity of gemcitabine combined with triptolide in pancreatic cancer cells. Oncol Lett. 2016;11(5):3527–3533. | ||
Pan Y, Zhang F, Zhao Y, et al. Berberine enhances chemosensitivity and induces apoptosis through Dose-orchestrated AMPK signaling in breast cancer. J Cancer. 2017;8(9):1679–1689. | ||
Zhu T, Li LL, Xiao GF, et al. Berberine increases doxorubicin sensitivity by suppressing STAT3 in lung cancer. Am J Chin Med. 2015;43(7):1487–1502. | ||
du J, Sun Y, Lu YY, et al. Berberine and evodiamine act synergistically against human breast cancer MCF-7 cells by inducing cell cycle arrest and apoptosis. Anticancer Res. 2017;37(11):6141–6151. | ||
Ren K, Zhang W, Wu G, et al. Synergistic anti-cancer effects of galangin and berberine through apoptosis induction and proliferation inhibition in oesophageal carcinoma cells. Biomed Pharmacother. 2016;84:1748–1759. | ||
Wen C, Wu L, Fu L, Zhang X, Zhou H. Berberine enhances the antitumor activity of tamoxifen in drugsensitive MCF7 and drugresistant MCF7/TAM cells. Mol Med Rep. 2016;14(3):2250–2256. | ||
Huang Y, Wang K, Gu C, et al. Berberine, a natural plant alkaloid, synergistically sensitizes human liver cancer cells to sorafenib. Oncol Rep. 2018;40(3):1525–1532. | ||
Wang CJ, Wang C, Han J, et al. Effect of combined treatment with recombinant interleukin-2 and allicin on pancreatic cancer. Mol Biol Rep. 2013;40(12):6579–6585. | ||
Zou X, Liang J, Sun J, et al. Allicin sensitizes hepatocellular cancer cells to anti-tumor activity of 5-fluorouracil through ROS-mediated mitochondrial pathway. J Pharmacol Sci. 2016;131(4):233–240. | ||
Dai B, Ma Y, Wang W, et al. Dihydroberberine exhibits synergistic effects with sunitinib on NSCLC NCI-H460 cells by repressing MAP kinase pathways and inflammatory mediators. J Cell Mol Med. 2017;21(10):2573–2585. | ||
Liu Y, Bi T, Wang Z, et al. Oxymatrine synergistically enhances antitumor activity of oxaliplatin in colon carcinoma through PI3K/Akt/mTOR pathway. Apoptosis. 2016;21(12):1398–1407. | ||
Song MQ, Zhu JS, Chen JL, et al. Synergistic effect of oxymatrine and angiogenesis inhibitor NM-3 on modulating apoptosis in human gastric cancer cells. World J Gastroenterol. 2007;13(12):1788–1793. | ||
Byun JM, Jeong DH, Lee DS, et al. Tetraarsenic oxide and cisplatin induce apoptotic synergism in cervical cancer. Oncol Rep. 2013;29(4):1540–1546. | ||
Tang JH, Zhang HM, Zhang ZH, Zhang X-L. Effect of tetramethylpyrazine combined with cisplatin on VEGF, KLF4 and ADAMTS1 in Lewis lung cancer mice. Asian Pac J Trop Med. 2017;10(8):813–818. | ||
Castillo-Pichardo L, Dharmawardhane SF. Grape polyphenols inhibit Akt/mammalian target of rapamycin signaling and potentiate the effects of gefitinib in breast cancer. Nutr Cancer. 2012;64(7):1058–1069. | ||
Stearns ME, Amatangelo MD, Varma D, Sell C, Goodyear SM. Combination therapy with epigallocatechin-3-gallate and doxorubicin in human prostate tumor modeling studies: inhibition of metastatic tumor growth in severe combined immunodeficiency mice. Am J Pathol. 2010;177(6):3169–3179. | ||
Liang G, Tang A, Lin X, et al. Green tea catechins augment the antitumor activity of doxorubicin in an in vivo mouse model for chemoresistant liver cancer. Int J Oncol. 2010;37(1):111–123. | ||
Brodsky FM. Monoclonal antibodies as magic bullets. Pharm Res. 1988;05(1):1–9. | ||
Ye BL, Ruan XJ, Zheng ZH, Cai HJ. Chitosan-coated doxorubicin nano-particles drug delivery system inhibits cell growth of liver cancer via p53/PRC1 pathway. Biochem Biophys Res Commun. 2018;495(1):414–420. | ||
Gao GH, Li Y, Lee DS. Environmental pH-sensitive polymeric micelles for cancer diagnosis and targeted therapy. J Control Release. 2013;169(3):180–184. | ||
Zhang X, Achazi K, Steinhilber D, Kratz F, Dernedde J, Haag R. A facile approach for dual-responsive prodrug nanogels based on dendritic polyglycerols with minimal leaching. J Control Release. 2014;174:209–216. | ||
Zhang H, Wang K, Zhang P, He W, Song A, Luan Y. Redox-sensitive micelles assembled from amphiphilic mPEG-PCL-SS-DTX conjugates for the delivery of docetaxel. Colloids Surf B Biointerfaces. 2016;142:89–97. | ||
Sun YW, Xu J, Zhou J, Liu W-J. Targeted drugs for systemic therapy of lung cancer with brain metastases. Oncotarget. 2018;9(4):5459–5472. | ||
Wick MR. Metastases of malignant neoplasms: Historical, biological, & clinical considerations. Semin Diagn Pathol. 2018;35(2):112–122. | ||
Obinu A, Gavini E, Rassu G, Maestri M, Bonferoni MC, Giunchedi P. Lymph node metastases: importance of detection and treatment strategies. Expert Opin Drug Deliv. 2018;15(5):459–467. | ||
Hull LC, Farrell D, Grodzinski P. Highlights of recent developments and trends in cancer nanotechnology research – View from NCI Alliance for nanotechnology in cancer. Biotechnol Adv. 2014;32(4):666–678. | ||
Yallapu MM, Khan S, Maher DM, et al. Anti-cancer activity of curcumin loaded nanoparticles in prostate cancer. Biomaterials. 2014;35(30):8635–8648. | ||
Zhang Z, Qian H, Yang M, et al. Gambogic acid-loaded biomimetic nanoparticles in colorectal cancer treatment. Int J Nanomedicine. 2017;12:1593–1605. | ||
Zhang YQ, Shen Y, Liao MM, et al. Galactosylated chitosan triptolide nanoparticles for overcoming hepatocellular carcinoma: enhanced therapeutic efficacy, low toxicity, and validated network regulatory mechanisms. Nanomedicine. 2019;15(1):86–97. | ||
Jabr-Milane LS, van Vlerken LE, Yadav S, Amiji MM. Multi-functional nanocarriers to overcome tumor drug resistance. Cancer Treat Rev. 2008;34(7):592–602. | ||
Narayanan S, Pavithran M, Viswanath A, et al. Sequentially releasing dual-drug-loaded PLGA-casein core/shell nanomedicine: design, synthesis, biocompatibility and pharmacokinetics. Acta Biomater. 2014;10(5):2112–2124. | ||
Gowda R, Kardos G, Sharma A, Singh S, Robertson GP. Nanoparticle-based celecoxib and plumbagin for the synergistic treatment of melanoma. Mol Cancer Ther. 2017;16(3):440–452. | ||
Torchilin V. Tumor delivery of macromolecular drugs based on the EPR effect. Adv Drug Deliv Rev. 2011;63(3):131–135. | ||
Gaumet M, Vargas A, Gurny R, Delie F. Nanoparticles for drug delivery: the need for precision in reporting particle size parameters. Eur J Pharm Biopharm. 2008;69(1):1–9. | ||
Yu C, Zhou Q, Xiao F, et al. Enhancing doxorubicin delivery toward tumor by hydroxyethyl Starch-g-Polylactide partner nanocarriers. ACS Appl Mater Interfaces. 2017;9(12):10481–10493. | ||
Hafeez BB, Zhong W, Fischer JW, et al. Plumbagin, a medicinal plant (Plumbago zeylanica)-derived 1,4-naphthoquinone, inhibits growth and metastasis of human prostate cancer PC-3M-luciferase cells in an orthotopic xenograft mouse model. Mol Oncol. 2013;7(3):428–439. | ||
Li YC, He SM, He ZX, et al. Plumbagin induces apoptotic and autophagic cell death through inhibition of the PI3K/Akt/mTOR pathway in human non-small cell lung cancer cells. Cancer Lett. 2014;344(2):239–259. | ||
Lee JH, Yeon JH, Kim H, et al. The natural anticancer agent plumbagin induces potent cytotoxicity in MCF-7 human breast cancer cells by inhibiting a PI-5 kinase for ROS generation. PLoS One. 2012;7(9):e45023. | ||
Youm I, West MB, Li W, du X, Ewert DL, Kopke RD. siRNA-loaded biodegradable nanocarriers for therapeutic MAPK1 silencing against cisplatin-induced ototoxicity. Int J Pharm. 2017;528(1–2):611–623. | ||
Kuhad A, Pilkhwal S, Sharma S, Tirkey N, Chopra K. Effect of curcumin on inflammation and oxidative stress in cisplatin-induced experimental nephrotoxicity. J Agric Food Chem. 2007;55(25):10150–10155. | ||
Al Moundhri MS, Al-Salam S, Al Mahrouqee A, Beegam S, Ali BH. The effect of curcumin on oxaliplatin and cisplatin neurotoxicity in rats: Some behavioral, biochemical, and histopathological studies. J Med Toxicol. 2013;9(1):25–33. | ||
Zhang J, Liu J, Xu X, Li L. Curcumin suppresses cisplatin resistance development partly via modulating extracellular vesicle-mediated transfer of MEG3 and miR-214 in ovarian cancer. Cancer Chemother Pharmacol. 2017;79(3):479–487. | ||
Hu Q, Sun W, Wang C, Gu Z. Recent advances of cocktail chemotherapy by combination drug delivery systems. Adv Drug Deliv Rev. 2016;98:19–34. | ||
Cheng Y, Zhao P, Wu S, et al. Cisplatin and curcumin co-loaded nano-liposomes for the treatment of hepatocellular carcinoma. Int J Pharm. 2018;545(1–2):261–273. | ||
Zhang YD. Nanobiotechnology. Science Press; Beijing, China, 2005. | ||
Moghimi SM, Hunter AC, Murray JC. Longcirculating and target specific nanoparticles: theory to practice. Pharmacol Rev. 2001;53:283–318. | ||
Pasut G, Veronese FM. State of the art in pegylation: the great versatility achieved after forty years of research. J Control Release. 2012;161(2):461–472. | ||
Suk JS, Xu Q, Kim N, Hanes J, Ensign LM. PEGylation as a strategy for improving nanoparticle-based drug and gene delivery. Adv Drug Deliv Rev. 2016;99:28–51. | ||
Fatma S, Talegaonkar S, Iqbal Z, et al. Novel flavonoid-based biodegradable nanoparticles for effective oral delivery of etoposide by P-glycoprotein modulation: an in vitro, ex vivo and in vivo investigations. Drug Deliv. 2016;23(2):500–511. | ||
Schmid D, Jarvis GE, Fay F, et al. Nanoencapsulation of ABT-737 and camptothecin enhances their clinical potential through synergistic antitumor effects and reduction of systemic toxicity. Cell Death Dis. 2014;5(10):e1454. | ||
Xu Y, Wang C, Ding Y, et al. Nanoparticles with optimal ratiometric co-delivery of docetaxel with gambogic acid for treatment of multidrug-resistant breast cancer. J Biomed Nanotechnol. 2016;12(9):1774–1781. | ||
Jain AK, Thanki K, Jain S. Co-encapsulation of tamoxifen and quercetin in polymeric nanoparticles: implications on oral bioavailability, antitumor efficacy, and drug-induced toxicity. Mol Pharm. 2013;10(9):3459–3474. | ||
Liu Q, Zhao D, Zhu X, et al. Coloaded nanoparticles of paclitaxel and Piperlongumine for enhancing synergistic antitumor activities and reducing toxicity. J Pharm Sci. 2017;106(10):3066–3075. | ||
Katiyar SS, Muntimadugu E, Rafeeqi TA, Domb AJ, Khan W. Co-delivery of rapamycin- and piperine-loaded polymeric nanoparticles for breast cancer treatment. Drug Deliv. 2016;23:2608–2616. | ||
Chen Y, Zheng XL, Fang DL, et al. Dual agent loaded PLGA nanoparticles enhanced antitumor activity in a multidrug-resistant breast tumor eenograft model. Int J Mol Sci. 2014;15(2):2761–2772. | ||
Hadinoto K, Sundaresan A, Cheow WS. Lipid-polymer hybrid nanoparticles as a new generation therapeutic delivery platform: a review. Eur J Pharm Biopharm. 2013;85(3 Pt A):427–443. | ||
Mandal B, Bhattacharjee H, Mittal N, et al. Core–shell-type lipid–polymer hybrid nanoparticles as a drug delivery platform. Nanomedicine. 2013;9(4):474–491. | ||
Li C, Ge X, Wang L. Construction and comparison of different nanocarriers for co-delivery of cisplatin and curcumin: a synergistic combination nanotherapy for cervical cancer. Biomed Pharmacother. 2017;86:628–636. | ||
Zhu B, Yu L, Yue Q. Co-delivery of vincristine and quercetin by nanocarriers for lymphoma combination chemotherapy. Biomed Pharmacother. 2017;91:287–294. | ||
Ruttala HB, Ko YT. Liposomal co-delivery of curcumin and albumin/paclitaxel nanoparticle for enhanced synergistic antitumor efficacy. Colloids Surf B Biointerfaces. 2015;128:419–426. | ||
Mendes LP, Gaeti MP, de Ávila PH, et al. Multicompartimental nanoparticles for co-encapsulation and multimodal drug delivery to tumor cells and neovasculature. Pharm Res. 2014;31(5):1106–1119. | ||
Jiang H, Geng D, Liu H, Li Z, Cao J. Co-delivery of etoposide and curcumin by lipid nanoparticulate drug delivery system for the treatment of gastric tumors. Drug Deliv. 2016;23(9):3665–3673. | ||
Tang F, Li L, Chen D. Mesoporous silica nanoparticles: synthesis, biocompatibility and drug delivery. Adv Mater. 2012;24(12):1504–1534. | ||
Zhang XG, Miao J, Dai YQ, du YZ, Yuan H, Hu F-Q. Reversal activity of nanostructured lipid carriers loading cytotoxic drug in multi-drug resistant cancer cells. Int J Pharm. 2008;361(1–2):239–244. | ||
Jia L, Li Z, Shen J, et al. Multifunctional mesoporous silica nanoparticles mediated co-delivery of paclitaxel and tetrandrine for overcoming multidrug resistance. Int J Pharm. 2015;489(1–2):318–330. | ||
Jain AK, Thanki K, Jain S. Solidified self-nanoemulsifying formulation for oral delivery of combinatorial therapeutic regimen: Part II in vivo pharmacokinetics, antitumor efficacy and hepatotoxicity. Pharm Res. 2014;31(4):946–958. | ||
Pemovska T, Johnson E, Kontro M, et al. Axitinib effectively inhibits BCR-ABL1(T315I) with a distinct binding conformation. Nature. 2015;519(7541):102–105. | ||
Escudier B, Gore M. Axitinib for the management of metastatic renal cell carcinoma. Drugs R D. 2011;11(2):113–126. | ||
Pang X, Yi Z, Zhang J, et al. Celastrol suppresses angiogenesis-mediated tumor growth through inhibition of Akt/mammalian target of rapamycin pathway. Cancer Res. 2010;70(5):1951–1959. | ||
Sethi G, Ahn KS, Pandey MK, Aggarwal BB. A novel triterpene, potentiates TNF-induced apoptosis and suppresses invasion of tumor cells by inhibiting NF-kappaB-regulated gene products and TAK1-mediated NF-kappaB activation. Blood. 2007;109:2727–2735. | ||
Choi JY, Ramasamy T, Kim SY, et al. PEGylated lipid bilayer-supported mesoporous silica nanoparticle composite for synergistic co-delivery of axitinib and celastrol in multi-targeted cancer therapy. Acta Biomater. 2016;39:94–105. | ||
Ramasamy T, Ruttala HB, Chitrapriya N, et al. Engineering of cell microenvironment-responsive polypeptide nanovehicle co-encapsulating a synergistic combination of small molecules for effective chemotherapy in solid tumors. Acta Biomater. 2017;48:131–143. | ||
Zhang D, Xu Q, Wang N, et al. A complex micellar system co-delivering curcumin with doxorubicin against cardiotoxicity and tumor growth. Int J Nanomedicine. 2018;13:4549–4561. | ||
Ma W, Guo Q, Li Y, Wang X, Wang J, Tu P. Co-assembly of doxorubicin and curcumin targeted micelles for synergistic delivery and improving anti-tumor efficacy. Eur J Pharm Biopharm. 2017;112:209–223. | ||
Gu Y, Li J, Li Y, et al. Nanomicelles loaded with doxorubicin and curcumin for alleviating multidrug resistance in lung cancer. Int J Nanomedicine. 2016;11:5757–5770. | ||
Wang J, Ma W, Tu P. Synergistically improved anti-tumor efficacy by co-delivery doxorubicin and curcumin polymeric micelles. Macromol Biosci. 2015;15(9):1252–1261. | ||
Shapira A, Livney YD, Broxterman HJ, Assaraf YG. Nanomedicine for targeted cancer therapy: towards the overcoming of drug resistance. Drug Resist Updat. 2011;14(3):150–163. | ||
Sreekanth CN, Bava SV, Sreekumar E, Anto RJ. Molecular evidences for the chemosensitizing efficacy of liposomal curcumin in paclitaxel chemotherapy in mouse models of cervical cancer. Oncogene. 2011;30(28):3139–3152. | ||
Abouzeid AH, Patel NR, Torchilin VP. Polyethylene glycol-phosphatidylethanolamine (PEG-PE)/vitamin E micelles for co-delivery of paclitaxel and curcumin to overcome multi-drug resistance in ovarian cancer. Int J Pharm. 2014;464(1–2):178–184. | ||
Meng L, Xia X, Yang Y, et al. Co-encapsulation of paclitaxel and baicalein in nanoemulsions to overcome multidrug resistance via oxidative stress augmentation and P-glycoprotein inhibition. Int J Pharm. 2016;513(1–2):8–16. | ||
Ishii K, Tanaka S, Kagami K, et al. Effects of naturally occurring polymethyoxyflavonoids on cell growth, P-glycoprotein function, cell cycle, and apoptosis of daunorubicin-resistant T lymphoblastoid leukemia cells. Cancer Invest. 2010;28(3):220–229. | ||
Letchford K, Burt H. A review of the formation and classification of amphiphilic block copolymer nanoparticulate structures: micelles, nanospheres, nanocapsules and polymersomes. Eur J Pharm Biopharm. 2007;65(3):259–269. | ||
Singh R, Lillard JW. Nanoparticle-based targeted drug delivery. Exp Mol Pathol. 2009;86(3):215–223. | ||
Alemi A, Zavar Reza J, Haghiralsadat F, et al. Paclitaxel and curcumin coadministration in novel cationic PEGylated niosomal formulations exhibit enhanced synergistic antitumor efficacy. J Nanobiotechnol. 2018;16(1):28. | ||
Han C, Zhang C, Ma T, et al. Hypericin-functionalized graphene oxide for enhanced mitochondria-targeting and synergistic anticancer effect. Acta Biomater. 2018;77:268–281. | ||
Pereira S, Lee J, Rubio N, et al. Cationic Liposome- multi-walled carbon nanotubes hybrids for dual siPLK1 and doxorubicin delivery in vitro. Pharm Res. 2015;32(10):3293–3308. | ||
Martincic M, Tobias G. Filled carbon nanotubes in biomedical imaging and drug delivery. Expert Opin Drug Deliv. 2015;12(4):563–581. | ||
Zhu F, Tan G, Jiang Y, Yu Z, Ren F. Rational design of multi-stimuli-responsive gold nanorod–curcumin conjugates for chemo-photothermal synergistic cancer therapy. Biomater Sci. 2018;6(11):2905–2917. | ||
Lockhart JN, Stevens DM, Beezer DB, Kravitz A, Harth E. Dual drug delivery of tamoxifen and quercetin: regulated metabolism for anticancer treatment with nanosponges. J Control Release. 2015;220(Pt B):751–757. | ||
Liu B, Han L, Liu J, Han S, Chen Z, Jiang L. Co-delivery of paclitaxel and TOS-cisplatin via TAT-targeted solid lipid nanoparticles with synergistic antitumor activity against cervical cancer. Int J Nanomedicine. 2017;12:955–968. | ||
He C, Lu K, Liu D, Lin W. Nanoscale Metal–Organic frameworks for the co-delivery of cisplatin and pooled siRNAs to enhance therapeutic efficacy in drug-resistant ovarian cancer cells. J Am Chem Soc. 2014;136(14):5181–5184. | ||
Yao X, Chen L, Chen X, et al. pH-responsive metallo-supramolecular nanogel for synergistic chemo-photodynamic therapy. Acta Biomater. 2015;25:162–171. | ||
Gaspar VM, Moreira AF, Costa EC, et al. Gas-generating TPGS-PLGA microspheres loaded with nanoparticles (NIMPS) for co-delivery of minicircle DNA and anti-tumoral drugs. Colloids Surf B Biointerfaces. 2015;134:287–294. | ||
Wilhelm S, Tavares AJ, Dai Q, et al. Analysis of nanoparticle delivery to tumours. Nat Rev Mat. 2016;1(5). | ||
Albanese A, Tang PS, Chan WCW. The effect of nanoparticle size, shape, and surface chemistry on biological systems. Annu Rev Biomed Eng. 2012;14(1):1–16. | ||
Zhang XQ, Xu X, Bertrand N, Pridgen E, Swami A, Farokhzad OC. Interactions of nanomaterials and biological systems: implications to personalized nanomedicine. Adv Drug Deliv Rev. 2012;64(13):1363–1384. | ||
Bertrand N, Wu J, Xu X, Kamaly N, Farokhzad OC. Cancer nanotechnology: the impact of passive and active targeting in the era of modern cancer biology. Adv Drug Deliv Rev. 2014;66:2–25. | ||
Gabizon A, Horowitz AT, Goren D, Tzemach D, Shmeeda H, Zalipsky S. In vivo fate of folate-targeted polyethylene-glycol liposomes in tumor-bearing mice. Clin Cancer Res. 2003;9:6551–6559. | ||
Calzolari A, Oliviero I, Deaglio S, et al. Transferrin receptor 2 is frequently expressed in human cancer cell lines. Blood Cells Mol Dis. 2007;39(1):82–91. | ||
Cui T, Zhang S, Sun H. Co-delivery of doxorubicin and pH-sensitive curcumin prodrug by transferrin-targeted nanoparticles for breast cancer treatment. Oncol Rep. 2017;37(2):1253–1260. | ||
Low PS, Antony AC. Folate receptor-targeted drugs for cancer and inflammatory diseases. Adv Drug Deliv Rev. 2004;56(8):1055–1058. | ||
Wang Y, Wang Y, Xiang J, Yao K. Target-specific cellular uptake of taxol-loaded heparin-PEG-folate nanoparticles. Biomacromolecules. 2010;11(12):3531–3538. | ||
Wang W, Xi M, Duan X, Wang Y, Kong F. Delivery of baicalein and paclitaxel using self-assembled nanoparticles: synergistic antitumor effect in vitro and in vivo. Int J Nanomedicine. 2015;10:3737–3750. | ||
Firestone RA. Low-density lipoprotein as a vehicle for targeting antitumor compounds to cancer cells. Bioconjug Chem. 1994;5(2):105–113. | ||
Sudhof T, Goldstein J, Brown M, Russell D. The LDL receptor gene: a mosaic of exons shared with different proteins. Science. 1985;228(4701):815–822. | ||
Zhu WJ, Yang SD, Qu CX, et al. Low-density lipoprotein-coupled micelles with reduction and pH dual sensitivity for intelligent co-delivery of paclitaxel and siRNA to breast tumor. Int J Nanomedicine. 2017;12:3375–3393. | ||
Yang SD, Zhu WJ, Zhu QL, et al. Binary-copolymer system base on low-density lipoprotein-coupled N-succinyl chitosan lipoic acid micelles for co-delivery MDR1 siRNA and paclitaxel, enhances antitumor effects via reducing drug. J Biomed Mater Res. 2017;105(5):1114–1125. | ||
Kebebe D, Liu Y, Wu Y, Vilakhamxay M, Liu Z, Li J. Tumor-targeting delivery of herb-based drugs with cell-penetrating/tumor-targeting peptide-modified nanocarriers. Int J Nanomedicine. 2018;13:1425–1442. | ||
Narayanan S, Mony U, Vijaykumar DK, Koyakutty M, Paul-Prasanth B, Menon D. Sequential release of epigallocatechin gallate and paclitaxel from PLGA-casein core/shell nanoparticles sensitizes drug-resistant breast cancer cells. Nanomedicine. 2015;11(6):1399–1406. | ||
Yan J, Wang Y, Jia Y, et al. Co-delivery of docetaxel and curcumin prodrug via dual-targeted nanoparticles with synergistic antitumor activity against prostate cancer. Biomed Pharmacother. 2017;88:374–383. | ||
Wang C, Su L, Wu C, Wu J, Zhu C, Yuan G. RGD peptide targeted lipid-coated nanoparticles for combinatorial delivery of sorafenib and quercetin against hepatocellular carcinoma. Drug Dev Ind Pharm. 2016;42(12):1938–1944. | ||
Zhao R, Li T, Zheng G, Jiang K, Fan L, Shao J. Simultaneous inhibition of growth and metastasis of hepatocellular carcinoma by co-delivery of ursolic acid and sorafenib using lactobionic acid modified and pH-sensitive chitosan-conjugated mesoporous silica nanocomplex. Biomaterials. 2017;143:1–16. | ||
Shen Z, Wei W, Tanaka H, et al. A galactosamine-mediated drug delivery carrier for targeted liver cancer therapy. Pharmacol Res. 2011;64(4):410–419. | ||
Yan T, Li D, Li J, et al. Effective co-delivery of doxorubicin and curcumin using a glycyrrhetinic acid-modified chitosan-cystamine-poly(ε-caprolactone) copolymer micelle for combination cancer chemotherapy. Colloids Surf Biointerfaces. 2016;145:526–538. | ||
Tsuji H, Osaka S, Kiwada H. Targeting of liposomes surface-modified with glycyrrhizin to the liver. I. Preparation and biological disoposition. Chem Pharm Bull. 1991;39(4):1004–1008. | ||
Ishida S, Sakiya Y, Ichikawa T, Taira Z. Uptake of glycyrrhizin by isolated rat hepatocytes. Biol Pharm Bull. 1993;16(3):293–297. | ||
Negishi M, Irie A, Nagata N, Ichikawa A. Specific binding of glycyrrhetinic acid to the rat liver membrane. Biochim Biophys Acta. 1991;1066(1):77–82. | ||
Lin A, Liu Y, Huang Y, et al. Glycyrrhizin surface-modified chitosan nanoparticles for hepatocyte-targeted delivery. Int J Pharm. 2008;359(1–2):247–253. | ||
Lee SJ, Zheng NY, Clavijo M, Nussenzweig MC. Normal host defense during systemic candidiasis in mannose receptor-deficient mice. Infect Immun. 2003;71(1):437–445. | ||
Hirata K, Maruyama T, Watanabe H, et al. Genetically engineered mannosylated-human serum albumin as a versatile carrier for liver-selective therapeutics. J Control Release. 2010;145(1):9–16. | ||
Zhang J, Yu C, Jiang G. Synthesis of cholic-acid-carrying polymer and in-vitro evaluation of hepatoma-targeting nanoparticles decorated with the polymer. J Biomater Sci Polym Ed. 2016;27(9):865–879. | ||
Elgohary MM, Helmy MW, Abdelfattah E-ZA, et al. Targeting sialic acid residues on lung cancer cells by inhalable boronic acid-decorated albumin nanocomposites for combined chemo/herbal therapy. J Control Release. 2018;285:230–243. | ||
Xia Y, Xu T, Wang C, et al. Novel functionalized nanoparticles for tumor-targeting co-delivery of doxorubicin and siRNA to enhance cancer therapy. Int J Nanomedicine. 2018;13:143–159. | ||
Wan L, Jiao J, Cui Y, et al. Hyaluronic acid modified mesoporous carbon nanoparticles for targeted drug delivery to CD44-overexpressing cancer cells. Nanotechnology. 2016;27(13):135102. | ||
Feng Q, Yu MZ, Wang JC, et al. Synergistic inhibition of breast cancer by co-delivery of VEGF siRNA and paclitaxel via vapreotide-modified core–shell nanoparticles. Biomaterials. 2014;35(18):5028–5038. | ||
Chen J, Li S, Shen Q. Folic acid and cell-penetrating peptide conjugated PLGA–PEG bifunctional nanoparticles for vincristine sulfate delivery. Eur J Pharm Sci. 2012;47(2):430–443. | ||
Gao Z, You C, Wu H, Wang M, Zhang X, Sun B. FA and cRGD dual modified lipid-polymer nanoparticles encapsulating polyaniline and cisplatin for highly effective chemo-photothermal combination therapy. J Biomater Sci Polym Ed. 2018;29(4):397–411. | ||
Zhang J, Li J, Shi Z, et al. pH-sensitive polymeric nanoparticles for co-delivery of doxorubicin and curcumin to treat cancer via enhanced pro-apoptotic and anti-angiogenic activities. Acta Biomater. 2017;58:349–364. | ||
Danafar H, Sharafi A, Kheiri S, Kheiri Manjili H. Co-delivery of sulforaphane and curcumin with pegylated iron Oxide-Gold core shell nanoparticles for delivery to breast cancer cell line. Iran J Pharm Res. 2018;17(2):480–494. | ||
Chang JE, Cho HJ, Yi E, Kim DD, Jheon S. Hypocrellin B and paclitaxel-encapsulated hyaluronic acid-ceramide nanoparticles for targeted photodynamic therapy in lung cancer. J Photochem Photobiol B. 2016;158:113–121. | ||
Yin Y, Hu Q, Xu C, et al. Co-delivery of doxorubicin and interferon-γ by thermosensitive nanoparticles for cancer immunochemotherapy. Mol Pharm. 2018;15(9):4161–4172. | ||
Yin T, Wang L, Yin L, Zhou J, Huo M. Co-delivery of hydrophobic paclitaxel and hydrophilic AURKA specific siRNA by redox-sensitive micelles for effective treatment of breast cancer. Biomaterials. 2015;61:10–25. |
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.