Back to Journals » Clinical Ophthalmology » Volume 12
Topographic-guided treatment of hyperopic corrections with a combination of higher order aberration removal with WaveLight® Contoura and wavefront-optimized hyperopic treatment
Authors Motwani M ![]()
Received 14 January 2018
Accepted for publication 20 March 2018
Published 1 June 2018 Volume 2018:12 Pages 1021—1029
DOI https://doi.org/10.2147/OPTH.S162511
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Scott Fraser
Manoj Motwani
Motwani Lasik Institute, San Diego, CA, USA
Purpose: This pilot study was conducted to test the hypothesis that WaveLight® Contoura and wavefront-optimized (WFO) hyperopic treatment can be used together for hyperopia/hyperopic astigmatism to create more uniform corneas.
Materials and methods: A retrospective analysis was conducted in 35 consecutive hyperopic/hyperopic astigmatism eyes of 22 patients treated via LASIK on the Wavelight® EX500. Higher order aberrations and astigmatism were removed using Contoura with the Layer Yolked Reduction of Astigmatism (LYRA) Protocol, and hyperopia was treated with WFO correction. All patients had 3 months of follow-up. Outcome measures were assessed by post-operative refraction, regression, and post-operative vision. Topographic analysis showed the degree of uniformity of the cornea achieved.
Results: Average hyperopia treated was +2 diopters (D) (range +0.50 D to +7.25 D), with the average amount of astigmatism treated +1.05 D (range -0.25 D to -2.25 D). The average difference between the Contoura-measured and manifest magnitude of astigmatism was 0.674 D and the average axis difference was 5.65°. No eyes lost corrected distance visual acuity (CDVA), 22.8% of eyes gained CDVA. At 3 months, 18 (54%) eyes had regressed or not achieved their targeted goal, and the average spherical equivalent (SE) from the targeted goal was 0.973 D. Following primary procedure, 10 of these eyes had myopic SE, six had hyperopic SE, and two had SE of 0. Prior to surgery, 19 of 24 distance eyes were able to achieve 20/20 vision, and if secondary corrections are included 100% achieved 20/20 or better post-operative, and eight (42%) achieved 20/15 or better at 3 months post-operative. No eyes lost CDVA.
Conclusion: This pilot study demonstrated that more uniform corneas can be created while treating hyperopic corrections, but a high level of secondary corrections were needed.
Keywords: Contoura, topographic guided ablation, hyperopia, LYRA Protocol
Introduction
Although the US Food and Drug Administration (FDA) approval of myopia/myopic astigmatism with the Alcon WaveLight® Contoura has been available for ~2 years now (although approved by the FDA in 2013, Alcon did not begin to install and upgrade lasers in the USA until 2016), the hyperopic arm of the study was shut down due to technical issues.1 As the author has noted in the Layer Yolked Reduction of Astigmatism (LYRA) Protocol papers, virtually all eyes in the consecutive group of patients studied had significant higher order aberrations (HOAs).2–4 In our experience, very few eyes have an adequately low level of HOA that wavefront-optimized (WFO) treatment would be equivalent to topographic-guided treatment. It stands to reason that eyes with hyperopic corrections would also have the same HOA, ie, the same flaws that prevent the cornea from being uniform. Hyperopic corrections are simply caused by a cornea that is too flat for the length of the eye, or an eye length that is too short for the power of the cornea. All the flaws of biologic construction of the cornea will still be present, leaving room for us to improve our hyperopic corrections.
Hyperopic corrections via excimer laser tend to be undertreated by refractive surgeons, carrying the stigma of high levels of regression. This was true in older forms of the technology, but technical improvements involving wider ablation patterns and higher speeds of correction have dramatically changed the outcomes of even high levels of both hyperopic and myopoic corrections. However, these improvements do not appear to have changed the practice of refractive surgeons. The author undertook a retrospective study and published the results of patients from +3.0 D to +6.0 D of hyperopic correction treated with both the WaveLight® EX500 and Allegretto 400.5 That study demonstrated a far lower regression rate compared with past studies on treatment of lower amounts of hyperopia. Many practices report only 10% of their treatments are hyperopic, in our practice, it is about 30%. We feel it is an ethical dilemma to deny a third of our patient population the ability to have the improvements in vision that we have already noted utilizing WaveLight® Contoura with the LYRA Protocol on myopic patients.
For these reasons, and also due to the fact that we have no knowledge of an impending clinical trial for FDA approval of hyperopia, we undertook a pilot study to attempt to treat hyperopic patients with a combination of HOA and astigmatism treatment via WaveLight® Contoura with the LYRA Protocol, followed consecutively by WFO hyperopic treatment.
Materials and methods
This was a retrospective analysis of the first 35 consecutive hyperopic eyes that were chosen to have primary laser correction (of 22 patients) using Contoura-measured astigmatism and axis (as per the LYRA protocol)2,4 within the FDA indications of astigmatism up to −3.00 D, followed by WFO treatment of hyperopia. These were performed by one surgeon (MM) at one center (San Diego, CA, USA).
The protocol used was correction via Contoura with LYRA Protocol for the HOA and astigmatism,2–4 equalization of c4/c12 as necessary, and then consecutive treatment of the residual hyperopic refraction utilizing WFO. In one patient, the Contoura-measured astigmatism exceeded 3 D, so the residual astigmatism was treated as part of the hyperopic WFO treatment. No changes were made to the amount of spherical correction treated via a nomogram, and no other nomogram was used as this was a pilot study and no nomogram exists for this type of procedure.
All patients had LASIK performed, and corneal flaps were made with either a femtosecond laser, WaveLight® FS200 (Wavelight GmBH, Erlangen, Germany), or Moria M2 (Moria Surgical, Antony, France) microkeratome. Target flap thickness was between 100 and 120 μm.
All excimer laser treatments were performed utilizing the Alcon WaveLight® EX500 laser (WaveLight GmbH, Erlangen, Germany), WaveNet planning software, and the LYRA Protocol.2–4 The Contoura treatment optical zone was 6.5 mm on all patients, as was the WFO hyperopia treatment optical zone.
We monitored all eyes for at least 3 months’ post-LASIK via visual acuity, refraction, and Topolyzer Vario HOAs to determine if the eye was accurately treated. We noted any patients that complained of increased night glare and halos over pre-operative.
Retreatments (secondary corrections) were performed at 3 months or later as necessary. Due to this being a pilot study without an existing nomogram to refine the correction accuracy, we mainly tabulated vision results including retreatments.
Preoperative evaluation included corrected distance visual acuity (CDVA), cyclopleged manifest refraction, anterior exam, posterior dilated exam, tonometry, pachymetry via Pentacam, auto-refraction and wavefront analysis with the Nidek OPD, and topographic analysis with the Topolyzer Vario. All pre-operative manifest refractions were performed with tropicamide 0.5% for cycloplegia. Although not as effective as cyclopentalate for cycloplegia, the latter is poorly tolerated by patients in our Southern California elective surgery population. Using tropicamide in conjunction with careful refractive techniques such as blurring has, in our experience, been effective in refracting adult hyperopic patients.
Specific attention was paid to obtaining high-quality reproducible scans with the Topolyzer Vario. Each patient had 8–12 scans taken per eye, and at least four accurate similar scans with appropriate iris registration and complete data (as indicated by the Topolyzer Vario) were necessary to proceed into surgical planning. Great care was taken not to induce astigmatism when holding the eyelids open for scans, and blinks were allowed to prevent the corneal surface from drying out.
Patients were not included if they had prior refractive surgery, had anterior segment abnormalities or findings that could affect the outcome such as keratoconus or corneal ectasia, recurring eye disease such as iritis or hepatitis, uncontrolled diabetes or hypertension, severe dry eye, or pregnancy. Patients were included if they could not achieve 20/20 as long as there was no other active ocular pathology.
All patients consented to being included as part of this study and understood that they were undergoing a combination of two procedures that were both FDA approved. They also were told that using the procedures together in a novel way could be considered an off-label procedure. They understood that there may be inaccuracies or secondary corrections involved. However, there was the promise of a more uniform cornea, which had in our past clinical and study experience led to better overall quantitative and qualitative results with myopic patients. Each patient consented as part of their formal written consent to allow their data to be used as part of this study and for manuscript publication with anonymization of their personal details.
This study conformed with the exemption under Health and Human Services Policy for Protection of Human Research Subjects 45 CFR 46.101 (b) for retrospective studies and therefore did not require institutional review board approval.
Results
Results were evaluated for amount of correction performed, regression, or change that occurred by 3 months, final vision, improvement or loss of lines of CDVA, and anecdotal complaints of increased night glare/halos/night vision issues. This latter category was strictly emphasized as anecdotal; no standardized subjective patient survey was utilized. For purposes of this manuscript, all manifest refractions were dilated refractions using tropicamide 1% to reduce the impact of accommodation. All refractions were in minus cylinder.
This study included 35 eyes from 22 patients (14 men and 8 women). The average age of the patients was 48.6 years.
The average of preoperative spherical equivalent of hyperopia treated was 2.01 D. The range of hyperopia treated was +0.50 D to +7.25 D. The average of astigmatism treated was −1.05 D. The range of astigmatism treated was −0.25 D to −2.25 D. The Contoura-measured astigmatism average before treatment was −1.03 D. The range was −0.14 D to −3.67 D.
The average difference between manifest astigmatic axis and Contoura-measured astigmatic axis was 5.65°, with the range of difference from 0° to 112°. The average amount of difference between magnitude of astigmatism from manifest to Contoura-measured was 0.674 D, with a range of 0.08 D −2.18 D. This number differs from the average amount of correction above as some eyes had no astigmatism present on manifest refraction but was present on Contoura-measured refraction. Every patient had their astigmatism treated, even the three eyes that did not have astigmatism on manifest refraction but did have astigmatism on Contoura-measured refraction.
No eyes lost CDVA, and eight eyes (22.8%) gained lines of visual acuity. Seven eyes gained one line of CDVA and one eye gained two lines (Figure 1).
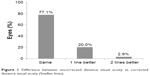  | Figure 1 Difference between uncorrected distance visual acuity vs corrected distance visual acuity (Snellen lines). |
Eighteen eyes did not achieve goal and had residual refraction that was either enhanced as a secondary correction (16) or followed (2). The average spherical equivalent of the amount from target goal was 0.973 D (range of sphere +1.50 D to −2.25 D; range of cylinder −0.50 D to −1.75 D). Of these eyes, 10 ended up with myopic spherical equivalents, six with hyperopic spherical equivalents, and two with a spherical equivalent of 0. Only five of the eyes showed spherical “regression” 2 myopic (−1 D, −0.75 D) and 3 hyperopic (1.0 D, 1.0 D, 0.50 D).
Of the eyes that demonstrated a hyperopic deviation from goal, the average spherical equivalent was 0.826 D, and those that experienced a myopic deviation from goal the average spherical equivalent was 1.19 D.
Out of 24 distances eyes (11 were monovision eyes), only 19 (79.17%) could achieve 20/20 vision preoperatively (Figure 2). With at least 3 months of postoperative follow-up and including any enhancements that were performed, all 19 (100%) achieved 20/20 vision or better, eight (42%) achieved 20/15 vision or better, and one (5.2%) achieved 20/10 vision. Of the other five eyes, two achieved 20/25 vision, two achieved 20/30 vision, and one achieved 20/40 vision. Two of these eyes had a residual refraction of −1.00 D and +0.75 D, including one that was only 1 week out from enhancement. Of these five eyes, two had an increase of one line of CDVA. No eyes had a loss of CDVA.
  | Figure 2 CDVA. Cumulative Snellen visual acuity (20/× or better). Twenty-four distance eyes. |
Of the 11 monovision eyes, all achieved J3 vision or better. Nine of these eyes were able to achieve 20/20 preoperatively, and these all achieved J2 or J1 vision Figure 3. Of these nine eyes, the distance vision was 20/25 in two eyes, 20/30 in four eyes, and 20/40 in three eyes. The other two eyes had CDVA of 20/25 and 20/40 preoperatively and achieved 20/70 and 20/100 vision at distance postoperatively.
  | Figure 3 CDVA. Cumulative Snellen visual acuity (20/× or better). Eleven monovision eyes. |
Sample cases
All sample cases have imaging that shows preoperative topography, pre-operative HOA (performed by zeroing out sphere and cylinder on the Contoura surgical planning page), post-operative topography, pre-operative Pentacam anterior elevation map, and pre-operative elevation treatment map (performed by zeroing out sphere and entering Contoura-measured astigmatism and axis on the Contoura surgical planning page) (Figures 4–6). In case 3 where a secondary WFO treatment was performed, postenhancement topography maps are also shown.
Discussion
Patients with hyperopic corrections have been notoriously undertreated with excimer laser. Our experience has been that many patients have been told by ophthalmologists and optometrists that even low to moderate amounts of hyperopia should not be treated. In a past study, we showed that technological changes to the speed of the machine and the shape of the ablation had demonstrated a dramatic positive change in the ability to stably treat even high hyperopia patients.5
It has been well documented in previous papers that “regression of laser effect” usually is caused by epithelial changes, as obviously stromal tissue cannot regrow. In our almost 2 years of experience with Contoura patients, both primary and secondary repair patients, it is the author’s opinion that corneal irregularity increases the chances of regression.6 In past studies by the authors, it was noted that most “regression” post-Contoura with LYRA Protocol treatment was mainly astigmatic, which would likely indicate further aberration that was being hidden by epithelial hyperplasia/compensation.2–4 Apart from this, even in our study of high myopic patients (−9.0 D to −16 D), it was noted that many high myopic treatments had no regression at all even though large changes to cornea were made.7
With this background, it was theorized by the author that Hyperopic Contoura may not only be used to make more uniform corneas but also possibly increase the stability of the hyperopic correction long term. The latter premise was not the subject of this paper, but the ability to make a more uniform cornea was the desired goal. Looking at the large, smooth hyperopic correction topographies in our sample cases, it is easy to see how this might be a desirable outcome for good optical qualities of vision that we have noted in myopic cases.
Hyperopic patients have a different profile from most myopic patients. They tend to skew older, some have some lens changes but not enough to need cataract surgery, and the younger patients who come for treatment usually have much higher levels of hyperopia that they cannot accommodate for, and many have some level of amblyopia. These patients likely will not have as a high a 20/15 rate as we see in myopic patients as a result of these factors.
The results of this pilot study are mixed. There was a very high percentage of patients that needed a second procedure, but interestingly the majority of these procedures were undertaken for myopic spherical equivalent amounts. This is important, as we noted part of the issue, as in sample case 1, was that “myopic” Contoura removal of HOA flattened the central area, and then we steepened the peripheral area of the cornea. We saw evidence as in sample case 1 that peri-pupillary coma may cause epithelial hyperplasia in the pupillary area leading to manifest refraction of a higher amount of spherical hyperopia. This patient ended up with significant myopia after his treatment, myopia that never subsided or changed over time. We have noticed the same effect on other topographic-guided repair patients that have peri-pupillary irregularity. Since the first epithelial thickness mapping system was only approved in August 2017 (Optovue), we are now in the process of collecting data to directly demonstrate how the epithelium interacts with corneal HOA. This may be the reason that we had so many myopic end results.
In the current study, there was also some selection bias involved, as patients were not just taken in a controlled consecutive manner, but were at times added to this study because of a significant amount of difference in between Contoura (topography)-measured astigmatism and manifest astigmatism. This may have also contributed to a higher “regression,” as we have already theorized that epithelial compensation may mask the ability to fully measure the full magnitude of HOA, an effect that would occur with greater frequency in those patients with a higher level of HOA.
On the other hand, we had excellent visual results in many patients, with anecdotal reporting of excellent vision by many patients. The vision in the distance eyes was especially noted by many patients. Monovision eyes mainly achieved 20/25 to 20/40 even at distance. With WFO treatment of hyperopia, it is not uncommon to see a −1.50 D monovision eye with distance vision of 20/60–20/100. This is similar to what we have noted in monovision in myopic patients also. These patients also seem to complain less about night vision or halos with monovision, although this is far too small a sample to declare that a trend exists.
In sample case 1, we had a bilateral LASIK that was performed on a young engineer whose goal was to fly fighter jets and was having laser correction to enable him to achieve this goal. His preoperative correction showed a significant degree of anisometropia, but he was able to refract to 20/20 in both eyes. He did remarkably well very rapidly in the right eye, achieving 20/10 vision, but his OS eye showed some myopia that subsided over the course of 3 months leaving a slight amount of astigmatism. With this correction, he was able to achieve 20/15 vision. The smoothness of his corneal topography is notable in both eyes, and his best corrected visual acuity improved by 2 lines in the OD, and 1 line in the OS vs preoperative. His high hyperopia correction shows a very smooth refractive shape to the steepened cornea. A remarkable result for a young man who may make use of this increased quantity of vision in his choice of career.
In sample case 2, we had a 54-year-old woman who had a moderate amount of hyperopia and elected to have monovision for reading. Her postoperative results showed initial myopia that faded across a few weeks to the specified goal. It is notable that she achieved 20/30 distance vision even in her monovision eye which had a −1.50 D refraction, an effect that we also noticed in myopic patients whom we treated with Contoura with the LYRA Protocol. This patient also had gained a line of vision in her distance eye.
Sample case 3 was a patient who had high amounts of hyperopia in both eyes, and upon treatment immediately experienced significant myopia, and the patient eventually had treatment for mainly spherical myopia. Cyclopleged refraction revealed the same amount of refraction, so this was not due to accommodation. It was noted that this patient had peri-pupillary coma in both eyes, and it was theorized that the patient may have had epithelial hyperplasia in the area of the corneal vertex as compensation to smooth the irregularity of the coma, resulting in a more hyperopic refraction. Treatment of the coma centrally would have flattened the raised area, thereby eliminating the cause of the epithelial hyperplasia. We also noticed the same effect in treating irregular corneas caused by past surgery, such as radial keratotomy or de-centered LASIK laser correction. Eliminating the irregularity eliminates some of the hyperopic correction, but this was the first time that we had encountered this in a primary laser correction. The patient at 6 months’ post-operative demonstrated excellent vision, with a gain of one line of vision in one eye.
It is notable that no lines of CDVA were lost in any of these patients. The optics of these patients were improved in some patients, but not worsened in any of the eyes where lines of vision were lost.
The amount of deviation in magnitude of astigmatism (0.674 D) is similar to other cohorts we have examined and seems to be a natural average number of deviation between manifest astigmatism and Contoura-measured astigmatism.3
Treatment of hyperopia with incorporation of HOA removal via topographic-guided ablation has enormous potential. Making a more uniform cornea may well increase overall visual stability, and when combined with the already noted accuracy and stability in our published hyperopia paper,5 may make hyperopic excimer laser treatment more widespread and trusted among refractive surgeons. We already have seen an increase in quantity and quality of vision documented in the LYRA Protocol manuscripts,2–4 and further refinement of the nomograms may make this procedure more accurate. It may well be that the technical handling of aberration removal with Hyperopic Contoura may also increase accuracy.
Much work remains to be done, as we have not yet attempted to use our data to create a nomogram to meld the Contoura with LYRA Protocol and WFO hyperopia procedures together. Certainly, having FDA approved one-step Hyperopic Contoura would be the best-case scenario, but in the meantime, it is important for us as surgeons to have a tool in hyperopic patients with significant HOAs and significant differences in Contoura-measured and manifest astigmatism.
The main issue with using this paradigm changing technology to help these patients has been the limited FDA approval obtained by Alcon Surgical (Fort Worth, TX, USA) on the WaveLight® Contoura approval. The mass adoption of topographic-guided ablation has been slow, as it is incurring a complete paradigm shift in thinking and theory about how the eye refracts, the nature of HOA, and how to improve the quantity and quality of vision. It is fundamentally important that hyperopic treatment approval is obtained for Contoura, as these patients are only under-represented in the excimer laser correction procedures due to refractive surgeons incorrectly believing that outcomes are subpar. If the work on myopic treatments is any indication, the ability to create more uniform corneas in hyperopic treatment may not only increase quantity, quality, and stability of vision but it may also increase the number of hyperopic procedures that refractive surgeons are willing to perform. Unexplored in this current manuscript are the effects that a more uniform cornea may have on corneal inlays and intraocular lenses, including multi focal and accommodative lenses. We are currently conducting a study on topographic-guided ablation with corneal inlays, which will also result in a paper, and there is a glaring need for hyperopic correction in these patients. Although there have been no further FDA approval studies begun to our knowledge on expanding the approval ranges for Contoura, the authors hope that Alcon Surgical will consider doing so in the future.
Acknowledgments
The author would like to acknowledge Ron Pei, OD for conception of this study and Sissimos Lemonis for inspiration.
Disclosure
The author has received an IIT grant for an unrelated Contoura study from Alcon. The author advises no other conflicts of interest in this work.
References
Alcon introduces Contoura Vision as first personalized LASIK procedure at American Academy of Ophthalmology Annual Meeting [press release]. Novartis. Basel, Switzerland. November 15, 2015. | ||
Motwani M. The use of WaveLight® Contoura to create a uniform cornea: the LYRA Protocol. Part 1: the effect of higher-order corneal aberrations on refractive astigmatism. Clin Ophthalmol. 2017;11:897–905. | ||
Motwani M. The use of WaveLight® Contoura to create a uniform cornea: the LYRA Protocol. Part 2: the consequences of treating astigmatism on an incorrect axis via excimer laser. Clin Ophthalmol. 2017;11:907–913. | ||
Motwani M. The use of WaveLight® Contoura to create a uniform cornea: the LYRA Protocol. Part 3: the results of 50 treated eyes. Clin Ophthalmol. 2017;11:915–921. | ||
Motwani M, Pei R. Treatment of moderate-to-high hyperopia with the WaveLight Allegretto 400 and EX500 excimer laser systems. Clin Ophthalmol. 2017;11:999–1007. | ||
Motwani M. A protocol for topographic-guided corneal repair utilizing the US Food and Drug Administration-approved Wavelight Contoura. Clin Ophthalmol. 2017;11:573–581. | ||
Motwani M. Treatment of high myopia/myopic astigmatism with a combination of WaveLight® Contoura with LYRA Protocol and wavefront-optimized treatment. Clin Ophthalmol. 2018;12:875–883. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.



