Back to Journals » International Journal of Nanomedicine » Volume 14
The neurotoxicity induced by engineered nanomaterials
Authors Ge D, Du Q, Ran B, Liu X , Wang X, Ma X, Cheng F, Sun B
Received 30 January 2019
Accepted for publication 9 April 2019
Published 6 June 2019 Volume 2019:14 Pages 4167—4186
DOI https://doi.org/10.2147/IJN.S203352
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Linlin Sun
Dan Ge,1,2,* Qiqi Du,1,2,* Bingqing Ran,1,2 Xingyu Liu,1,2 Xin Wang,1,2 Xuehu Ma,1 Fang Cheng,1,3 Bingbing Sun1,2
1State Key Laboratory of Fine Chemicals, Dalian University of Technology, Dalian 116024, People’s Republic of China; 2Department of Chemical Engineering, Dalian University of Technology, Dalian 116024, People’s Republic of China; 3Department of Pharmaceutical Science and Technology, Dalian University of Technology, Dalian 116024, People’s Republic of China
*These authors contributed equally to this work
Abstract: Engineered nanomaterials (ENMs) have been widely used in various fields due to their novel physicochemical properties. However, the use of ENMs has led to an increased exposure in humans, and the safety of ENMs has attracted much attention. It is universally acknowledged that ENMs could enter the human body via different routes, eg, inhalation, skin contact, and intravenous injection. Studies have proven that ENMs can cross or bypass the blood–brain barrier and then access the central nervous system and cause neurotoxicity. Until now, diverse in vivo and in vitro models have been developed to evaluate the neurotoxicity of ENMs, and oxidative stress, inflammation, DNA damage, and cell death have been identified as being involved. However, due to various physicochemical properties of ENMs and diverse study models in existing studies, it remains challenging to establish the structure-activity relationship of nanomaterials in neurotoxicity. In this paper, we aimed to review current studies on ENM-induced neurotoxicity, with an emphasis on the molecular and cellular mechanisms involved. We hope to provide a rational material design strategy for ENMs when they are applied in biomedical or other engineering applications.
Keywords: engineered nanomaterials, neurotoxicity, oxidative stress, inflammation, DNA damage, cell death
Introduction
Engineered nanomaterials (ENMs) exhibit extraordinary chemical, biological, optical, mechanical, and magnetic properties due to their small size and large surface-to-volume ratio. Until now, ENMs have been successfully applied not only in traditional manufacturing, eg, catalysis, energy, electronics, and personal care products, but also in emerging biomedical fields, eg, imaging, diagnosis, drug delivery, and cancer immunology.1 With the extensive application of ENMs, their safety has raised wide public attention. In the process of ENM production, processing, transportation, usage, and disposal, people are obviously exposed to ENMs via several different routes including skin contact, inhalation, ingestion, and systemic administration.2,3 After exposure, ENMs can be distributed to various organs, eg, heart, liver, spleen, lungs, and kidneys.4–7 More importantly, more and more studies have demonstrated that ENMs could cross the blood–brain barrier (BBB) and further access the central nervous system (CNS), where ENMs could cause neurotoxicity.5,8–13
The BBB is a dynamic barrier separating blood and the CNS with the function of restricting the material exchanges between the blood and brain, and helping to maintain the homeostasis of the brain.14,15 With special physicochemical properties, ENMs could pass through BBB via different routes, eg, receptor-mediated transcytosis, adsorptive-mediated transcytosis, transcellular diffusion, paracellular diffusion, and cell-mediated transcytosis after incidental exposure or systemic administration.10,16,17 After crossing BBB, ENMs can interact with glial cells and neurons, which could potentially induce a series of disrupted outcomes in the neurological system.12 Current studies have demonstrated that the physicochemical properties of ENMs, eg, chemical composition,18 size,19 shape,20 surface coating,21 charge,22,23 and aggregation state,24 can affect their neurotoxicity in several in vivo study models, eg, zebra fish, rats, and mice (Table 1) and in various cell types, eg, microvascular endothelial cells, glial cells, and neurons (Table 2).12,25 However, ENM-induced neurotoxicity is not well understood owing to the diverse physicochemical properties of ENMs, and the complicated regulating signaling pathways in organisms.
 | Table 1 Mechanisms of ENM-induced neurotoxicity in vivo |
 |  |  |  | Table 2 Mechanisms of ENM-induced neurotoxicity in vitro |
 | Table 3 Assays for DNA damage and corresponding results |
In this paper, we aimed to review the neurotoxicity of ENMs in different study models. We focused on potential mechanisms of ENM-induced toxicity in different neurological systems. With these information, we hope to provide more knowledge for the establishment of structure-activity relationships (SARs) and safer design of ENMs.
How could ENMs access the CNS?
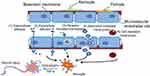
ENMs could access the blood or lymph nodes, and then translocate to different organs including lungs, liver, kidneys, heart, and even the brain which is well-protected by the BBB. It has been found that ENMs could be deposited in different regions of the brain, including olfactory bulb, hippocampus, cerebral cortex, and striatum.26,27
It was found that ENMs with specific characteristics could pass through the BBB via several different ways (Figure 1). 1) Transcellular diffusion. Solid lipid nanoparticles (NPs) with low molecular weight could pass through the BBB in this way.28 2) Paracellular diffusion. Silica NPs and reduced graphene oxide (rGO) could open paracellular spaces and access the CNS.29,30 3) Receptor-mediated transcytosis. ENMs with ligands such as transferrin, insulin, ANG, and ApoE, could be recognized by corresponding receptors on endothelial cells' surface and then cross the BBB through transcytosis. ANG conjugated carbon nanotubes could pass through BBB efficiently, probably with the help of LRP1 which is located on the brain capillary endothelium.31 4) Adsorptive-mediated transcytosis. Cationic albumin-conjugated pegylated NPs could deliver hTRAIL-encoding plasmid (pORF-hTRAIL) into the CNS through adsorptive transcytosis after intravenous administration.32 Lactoferrin-modified cationic dendrimer-based NPs could be promoted to cross BBB through both receptor- and adsorptive-mediated transcytosis.33 5) Cell-mediated transcytosis. Macrophages could phagocytize ENMs in the blood, pass through BBB, and then release ENMs into the CNS.
In addition to transpassing the BBB directly through blood circulation, studies have indicated that ENMs could translocate to the olfactory bulb of the brain through the olfactory nerve or into the CNS via the trigeminal nerve when they are inhaled or intranasally instilled.34–37 Neurons could also transport ENMs by retrograde and anterograde movement in axons and dendrites as well as perineural translocation.36 Additionally, particles can be found in the brain of offspring when animals are exposed to ENMs. For example, both 70 nm silica (SiO2) and 35 nm titanium dioxide (TiO2) particles were found in the placenta, brain, and liver of offspring when the particles were injected into pregnant mice intravenously, suggesting ENMs could cross the placenta barrier in pregnant mice and further cause damage to offspring.38
Additionally, ENMs that have been applied in medical devices could have opportunities to interact directly with glial cells or neurons and cause neurotoxicity. For example, carbon nanofibers were considered to be a potential material in neuronal implants.39 Gold (Au) NPs and silica-poly (ε-caprolactone) (Si-PCL/ICG) NPs were proposed to be used for laser-assisted tissue soldering in the brain, eg, cerebral bypass surgery.40 Nano-electromechanical systems in the brain, eg, implantable silicon-based micro-electrodes, are used in supervising and diagnoses.41–43 Once these implants that are coated with ENMs are worn, potential toxicity may be inevitable.
Mechanisms of neurotoxicity induced by ENMs
Oxidative stress
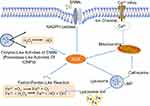
Oxidative stress is commonly caused by over-production of ROS with disturbance of redox homeostasis.44 The brain is especially vulnerable to oxidative stress due to the large amount of oxygen consumption, abundance of lipids and proteins, and low antioxidant defense activity.45 ROS are usually produced at an early stage when cells interact with ENMs and oxidative stress is regarded as one of the main mechanisms of neurotoxicity induced by ENMs.46–48 Thus far, ROS production through different sources have been documented (Figure 2).
Oxidative burst in microglia
It usually happens when microglia are activated by foreign stimuli. During this process, a large amount of superoxide anion (O2.−) is released through NADPH-oxidase mediated molecular oxygen (O2) catalysis. O2.− could be further transformed to other forms of ROS, eg, hydrogen peroxide (H2O2), hydroxyl radicals (OH.), and peroxynitrites. Phenrat et al48 found that when microglia were exposed to TiO2 NPs (Degussa P25), there was immediate (<5 minutes) concentration-dependent intracellular H2O2 production through oxidative burst. The particle size, aggregation state, surface coating, and sedimentation could influence the interaction between ENMs and cells that further affect oxidative burst-mediated ROS production. A study has demonstrated that large zero-valent iron (ZVI) NPs could agglomerate into micron-size particles and induce oxidative burst in BV-2 cells.24
Mitochondrial dysfunction
Mitochondria are a main source of ROS as a result of the electron transport chain (ETC) through which adenosine triphosphate (ATP) is generated. Mitochondrial disruption will lead to significant increase of ROS. Long et al48,49 showed the existence of O2.- in mitochondria of BV-2 cells after TiO2 (Degussa P25) exposure. Sharma et al50 showed that ZnO NPs possibly induced mitochondrial ROS, which led to mitochondrial dysfunction, eg, decreased MMP and release of cytochrome C, and further induced microglial death. In addition, Yin et al51 demonstrated that after exposure to silver (Ag) NPs, elevated calcium ions (Ca2+) influx could cause oxidative stress by promoting mitochondrial depolarization in rat cerebellar granule cells.
Fenton/Fenton-like reaction
Transition metals, eg, iron, copper, chromium, and cobalt, could mediate the formation of ROS, eg, highly reactive hydroxyl radical (OH.), through Fenton reaction. Phenrat et al showed that ZVI NPs could act as electron donors and produce ROS through Fenton reaction in microglia. They also showed that iron oxide NPs (IONPs) could induce Fenton reaction by oxidizing ferrous iron to ferric iron, which contributed to the oxidative stress in astrocytes.24,52
Enzyme-like activities of ENMs
It was demonstrated that when human glioma U251 cells were exposed to dimercaptosuccinic acid-coated Fe3O4 and γ-Fe2O3 together with H2O2, both two types of NPs could localize in lysosomes and behave as peroxidase and catalyze H2O2 into OH., which could further induce cytotoxicity.53 On the contrary, GO quantum dots (QDs) showed catalase-like activity and decreased MPP+-induced ROS generation in PC12 cells.54
Lysosome membrane permeabilization (LMP)
LMP is a known form of lysosome dysfunction and suggested as an indirect source of ROS. Firstly, LMP could produce ROS through lysosomal ferrous iron. Secondly, LMP could lead to mitochondrial membrane permeabilization and production of mitochondrial ROS by releasing proteases, eg, cathepsin B/D inside lysosomes.55 Yang et al56 found that when RAW264.7 macrophages were exposed to carbon nanohorn, LMP could be induced, which further led to the production of mitochondrial ROS.
However, it still remains unclear that how ENMs with different physicochemical properties could affect ROS generation via the previously mentioned mechanisms. Thus, more detailed studies are needed.
With the induction of oxidative stress, the subsequent consequences have been investigated. Firstly, oxidative stress is prone to enhancing the permeability of the BBB. Liu et al showed that, when brain microvessel endothelial cells (BMECs) were exposed to silica NPs, ROS were induced and considered as the mediators in increasing BBB permeability by down-regulating the tight junction proteins, eg, CLDN5 and OCLN, and further causing microtubule destabilization and triggering inflammatory response. By using N-acetyl cysteine, an antioxidant agent, ROS-induced BBB permeability was significantly inhibited.30 Furthermore, ENM-induced ROS in microglia may damage surrounding neurons.27,49,57,58 The study by Hsiao et al showed that BV-2 could produce large amounts of H2O2 after nano TiO2 exposure. Co-culture of N2a neuroblastoma cells and BV-2 cells showed that N2a cells were significantly damaged when BV-2 cells were in direct contact with nano TiO2. In contrast, direct exposure to nano-TiO2 did not reduce the viability of N2a cells.57 Another study by Long et al found that Degussa P25 induced the release of ROS and apoptosis in BV-2 microglia, but did not affect the cell viability of isolated rat dopaminergic neurons (N27). However, P25 caused neuronal loss of embryonic rat striatum containing both microglia and dopaminergic neurons, indicating the influence of microglia-generated ROS on neurons.49
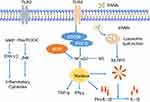
Inflammation
Inflammation is a protective response in the body with the function of eliminating xenobiotic stimuli and repairing damaged tissue.59 Neuroinflammation can be induced directly after ENMs get into the brain. It mainly occurs in microglia, the resident macrophages in the brain (Figure 3). Microglia are immune-competent cells, which usually remain quiescent and will respond to the destruction of brain homeostasis and entry of environmental toxins.11 Upon ENM exposure, microglia could be activated and change their morphology from ramified to rounded macrophage-like ones and further produce pro-inflammatory cytokines, eg, interleukins, tumour necrosis factor protein families, and chemokines.60,61 Knudsen et al found that cationic PEGylated micelles and cationic liposomes could induce the focal filtration of phagocytes and activate microglia and astrocytes in the brain after an intracerebroventricular particle injection.22
ENMs, as a kind of stimuli, can provoke TLRs, eg, TLR2 and TLR4, of microglia that reside on the cell membrane. It was reported that nano-TiO2 could induce TLR2 and TLR4 activation in the hippocampus after oral administration. The response of TLRs could further induce the activation of NF-κB through negatively regulating nucleic inhibitor of NF-κB (IκB) kinase and caused subsequent hippocampal inflammation in mice.62 Hutter et al63 showed that poly(ethylene glycol)-coated urchin/rod but not sphere Au NPs could upregulate TLR2 expression of microglia, suggesting effects of geometries on the activation of TLRs. However, detailed mechanistic studies are needed to elucidate the exact role of TLRs in neuroinflammation after ENM exposure.
NLRP3 inflammasome belongs to the NLR family, and is a multiprotein complex that could respond to exogenous stimuli. NLRP3 inflammasome could be directly activated by ENMs, and modulate the release of pro-inflammatory cytokines, eg, IL-1β.64 Moquin et al65 showed that lipopolysaccharides-QDs (LPS-QDs) could potentially induce mitochondrial ROS and lead to lysosome rupture, followed by NLRP3 inflammasome activation and cleavage of IL-1β in microglia. In addition, the uptake of large LPS-QDs contributed to the rupture of lysosomes. As many studies have already shown, ENMs could be taken up by microglia through macropinocytosis, phagocytosis, and clathrin-mediated endocytosis, and then translocate to lysosomes through endo-lysosome pathway for the activation of NLRP3 inflammasome.57,66,67
Neuroinflammation occurs not only in immune cells,64 eg, microglia68,69 in the brain, but also in microvascular endothelial cells,19,70,71 astrocytes,57 and neurons.57 When encountering ENMs, common in vitro BBB models, BMECs or co-culture of BMECs and astrocytes/pericytes could produce pro-inflammatory cytokines. Trickler et al71-73 found that CuO NPs and Ag NPs induced an increase of pro-inflammatory mediators IL-1β, TNF-α, and PGE2 in rat BMECs and porcine BMECs, which affected the integrity of the BBB. ENM-induced pro-inflammatory cytokines and chemokines in BMECs could recruit immune cells in the blood and also promote the expression of CAMs, eg, E-selectin and ICAM-1, to facilitate the adhesion and transmigration of leukocytes through the BBB.30,74 In addition, there was an immuno-regulatory loop between BMECs and astrocytes after exposure of ENMs.74 In short, astrocytes could integrate the paracrine inflammatory signals from BMECs, amplify them, and then return them to BMECs, thereby contributing to the destruction of the BBB.30,75
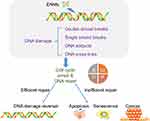
DNA damage
ENM exposure could induce DNA damage that could potentially lead to carcinogenesis or even impact offspring (Figure 4).76 Thus far, different types of DNA damage have been identified, including DNA cross-links, single/double strand breaks, and DNA adducts (Table 3).77
Studies have found that ENMs could induce DNA damage in two different ways, ie, primary DNA damage and secondary DNA damage. The former refers to the fact that ENMs could enter the cells and subsequently the nuclei, and further directly interact with DNA or DNA-related proteins.78 For example, Ren et al79 showed that GO sheet could translocate into cell nuclei of diencephalon in zebra fish larvae brain from water. Even if ENMs could not penetrate the nucleus, there still remains the opportunity that they could affect DNA in the process of mitosis. As to secondary DNA damage, it usually refers to the impact induced by ROS and inflammation. Shah et al80 showed that nano-alumina could induce oxidative DNA damage in both in vitro models, ie, mouse hippocampal HT22 cells and human neuroblastoma SHSY5Y cells, and in vivo model, ie, brain of mouse models. Valdiglesias et al81 showed that ZnO NPs could induce oxidative DNA damage and H2AX phosphorylation representing DNA double strand breaks in human SHSY5Y cells. El-Ghor et al82 found that nano-TiO2 induced both chromosomal damage and DNA damage in brain tissue due to ROS. In addition, Hawkins et al83 found that cobalt and chromium (CoCr) NPs triggered release of IL-6 in a placental barrier model, and IL-6 led to DNA double strand breaks in differentiating neural progenitor cells.
In addition, studies have demonstrated that DNA damage could lead to cell cycle arrest to provide enough time for DNA repair, while inefficient DNA repair could induce apoptosis.84 Kang et al85 found that GO and rGO exposure could lead to two or more nuclei in PC12 cells and G0/G1 cell cycle arrest, further leading to apoptosis. Valdiglesias et al86 found that TiO2 NPs induced micronuclei together with DNA strand breaks in SHSY5Y cells, and further, apoptosis was induced.
ENM induces differential cell death
Studies have suggested that different types of cell death are involved in the neurotoxicity of ENMs, such as apoptosis, necrosis, and autophagy (Figure 5).
Apoptosis and necrosis
Apoptosis is programmed cell death with the function of eliminating damaged cells and further maintaining the organism's homeostasis. It is the most common cell death found in ENM-induced neurotoxicity with the characteristics of cell shrinkage, nuclear condensation, and fragments.87,88 In general, apoptosis can be induced by mitochondrial dysfunction (intrinsic pathway), death receptor (extrinsic pathway), and endoplasmic reticulum (ER) stress. Different caspases, eg, CASP8, 9, 3, are the primary molecular signals of apoptosis.
The underlying mechanisms of ENM-induced intrinsic apoptosis have been widely studied. MAPK molecules including p38 MAPK, JNK, and ERK are usually associated with intrinsic apoptosis.89 In addition, P53 can be activated by ROS and DNA damage which may lead to apoptosis by inducing the transcription of pro-apoptotic genes like Bax. Xu et al72 found that Ag NPs induced apoptosis through activating MAPK pathway, reducing anti-apoptotic Bcl-2 expression, and altering activity of mTOR in primary astrocytes. Wang et al90 showed that ZnO NPs induced phosphorylation of JNK, ERK, and p38 MAPK in astrocytes, and that JNK played the major role in apoptosis. Wu et al91 discovered that P53 promoted apoptosis together with JNK pathway in PC12 cells while they were treated with TiO2 NPs. Zhao et al92 found that ZnO NPs could lead to apoptosis in zebra fish embryos characterized by reduction in the Bcl-2/Bax ratio, decrease of MMP, release of cytochrome C, and activation of CASP9 and 3. In addition, Hadrup et al93 showed that Ag NPs could induce both extrinsic and intrinsic apoptosis in PC12 cells based on the activation of CASP8 and 9. Another study also found that ZnO NPs induced the phosphorylation of ERK and p38, which further activated CASP9 and 8 respectively, and then apoptosis was induced.50
The ER is responsible for the synthesis, processing, and trafficking of protein, and the regulation of Ca2+. ER stress could be induced by the change of Ca2+ homeostasis and misfolded proteins.94 Ducray et al95 found that two types of silica NPs, ICG/polycaprolactone (PCL)-rhodamine-doped NPs and Si-ICG/polycaprolactone-polylactic acid-rhodamine-doped NPs could be taken up by microglia and translocate in the ER when they were exposed to hippocampal slice cultures. This could cause a potential risk of inducing protein misfolding. Parveen et al96 demonstrated that silica NPs could induce ER stress in the corpus striatum of young male Wistar rats. They showed significantly increased ER stress markers including CHOP, also known as GADD153, and CASP12, and further, Bax/Bcl-2 homeostasis was disrupted and apoptosis was induced.
In contrast, necrosis is caspase-independent with the characteristics of cytoplasmic swelling, expansion of organelles, and rupture of cell membrane. It further causes the outflow of intracellular content and induces inflammatory response.97 Zhang et al98 found that both necrosis and apoptosis were induced in the hippocampal cells in ICR mice when nano-alumina NPs were intranasally injected. More necrotic than apoptotic cell death was observed in hippocampal cells, and necrosis was further considered as a type of primary cell death contributing to the neurobehavioral defects in ICR mice.
Current studies have demonstrated that necrosis is not as common as apoptosis in the neurotoxicity induced by ENMs, however, more studies are needed to clarify the effects of necrosis induced by ENMs and the underlying mechanisms.
Autophagy
Autophagy is an intracellular mechanism for the degradation and reuse of cellular components, elimination of aberrant materials, and maintaining cellular homeostasis.55 Autophagy occurs when ENMs get into endosomes in cells and form double-membrane autophagosomes, which fuse with lysosomes to produce autolysosomes for the degradation of ENMs.
Xue et al99 demonstrated that TiO2 NPs and oleic acid-coated IONPs could induce autophagy in human cerebral endothelial cells, which may involve oxidative stress and aggregations of ENMs. Xie et al100 found that SiO2 NPs induced autophagy in PC12 cells with elevated LC3-II and BECN1 levels. Further more, a mechanistic study showed that the autophagy was induced by inhibiting PI3K-protein kinase-B (Akt)-mTOR pathway. In addition, Wang et al101 showed that cationic polyamidoamine (PAMAM) dendrimers could induce autophagy via Akt-mTOR pathway in human gloma cell lines.
Studies have demonstrated that autophagy plays both protective and detrimental roles in ENM-induced cytotoxicity. Wei et al102 showed that ZnO NPs induced mitophagy, a form of autophagy which degrades dysfunctional mitochondria in BV-2 cells. They proved that mitophagy played a protective role in ENM-induced cytotoxicity. Hu et al103 suggested that when zebra fish were exposed to GO, autophagy in the offspring played an important role in clearing α-synuclein and damaged ER. However, ENM-induced autophagy could also lead to toxicity.104 Zhou et al105 found that TiO2 NPs could induce excessive autophagy in offspring of mice after prenatal exposure, based on the findings that there were significant increases of PI3K3C, BECN1, c-Jun, p-JNK, and microtubule-associated protein 1A/1B-light chain 3 (LC3), which led to the inhibition of dendritic outgrowth of hippocampal neurons. Wang et al101 found that cationic PAMAM dendrimers induced autophagic flux in human gloma cell lines, which was suggested as the main source of their cytotoxicity. In addition, Li et al106 also showed that the inhibition of PAMAM dendrimers-induced autophagy could improve the viability of SHSY-5Y cells.
Conclusion
We have comprehensively reviewed ENM-induced oxidative stress, inflammation, DNA damage, and cell death, all of which are considered as potential mechanisms of neurotoxicity. These mechanisms may work independently or they could interact with each other. For example, oxidative stress, as one of the most common mechanisms, is considered to trigger inflammation, DNA damage, apoptosis, and autophagy in some cases. DNA damage could also result in apoptosis. In addition, different types of cell death may share the same characteristics. For example, ATP depletion, ROS increase, and MMP decrease are associated with both apoptosis and necrosis. However, the specific roles of these mechanisms and their relationship with ENM-induced neurotoxicity remain elusive. Deeper exploration of the underlying mechanisms requires more efforts, and effective methodologies are required to establish a complete neurotoxicity evaluation system. More importantly, it is critical to establish SARs between ENMs and their biological effects. However, we still face the dilemma of lack of enough experimental data and sufficient knowledge about the interaction of nanomaterials and the biological system.107–109 In addition, the neurotoxicity evaluations are mostly focused on inorganic nanomaterials, eg, metallic nanomaterials and carbon-based nanomaterials, and the studies on organic nanomaterials, eg, polymeric NPs and liposomes are limited.110–112 Thus, more detailed studies are warranted for a better understanding of the interaction of ENMs with neurological systems for safer and effective applications of ENMs.
Acknowledgments
This work was supported by the National Natural Science Foundation of China (31870919, 31300809, and 21773022), Natural Science Foundation of Liaoning Province (20180550597), LiaoNing Revitalization Talents Program (XLYC1807113) and Fundamental Research Funds for the Central Universities (DUT17RC(3)021). Bingbing Sun would also like to thank the Recruitment Program of Global Youth Experts for support.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Morris JE. Nanoparticle properties. In: Morris JE, editor. Nanopackaging: Nanotechnologies and Electronics Packaging. Boston, MA: Springer US; 2008:93–107.
2. Shakeel M, Jabeen F, Shabbir S, Asghar MS, Khan MS, Chaudhry AS. Toxicity of nano-titanium dioxide (TiO2-NP) through various routes of exposure: a review. Biol Trace Elem Res. 2016;172(1):1–36. doi:10.1007/s12011-015-0550-x
3. Song B, Liu J, Feng X, Wei L, Shao L. A review on potential neurotoxicity of titanium dioxide nanoparticles. Nanoscale Res Lett. 2015;10(1):342. doi:10.1186/s11671-015-1042-9
4. Bakand S, Hayes A. Toxicological considerations, toxicity assessment, and risk management of inhaled nanoparticles. Int J Mol Sci. 2016;17(6):929. doi:10.3390/ijms17060929
5. Feng X, Chen A, Zhang Y, Wang J, Shao L, Wei L. Application of dental nanomaterials: potential toxicity to the central nervous system. Int J Nanomed. 2015;10:3547–3565.
6. Recordati C, De Maglie M, Bianchessi S, et al. Tissue distribution and acute toxicity of silver after single intravenous administration in mice: nano-specific and size-dependent effects. Part Fibre Toxicol. 2016;13:12. doi:10.1186/s12989-016-0124-x
7. Kafa H, Wang JT, Rubio N, et al. The interaction of carbon nanotubes with an in vitro blood-brain barrier model and mouse brain in vivo. Biomaterials. 2015;53:437–452. doi:10.1016/j.biomaterials.2015.02.083
8. Feng XL, Chen A, Zhang Y, Wang J, Shao L, Wei L. Central nervous system toxicity of metallic nanoparticles. Int J Nanomed. 2015;10:4321–4340.
9. Cupaioli FA, Zucca FA, Boraschi D, Zecca L. Engineered nanoparticles. How brain friendly is this new guest? Prog Neurobiol. 2014;119–120:20–38. doi:10.1016/j.pneurobio.2014.05.002
10. Win-Shwe T, Fujimaki H. Nanoparticles and neurotoxicity. Int J Mol Sci. 2011;12(9):6267–6280. doi:10.3390/ijms12096267
11. Hu Y, Gao J. Potential neurotoxicity of nanoparticles. Int J Pharmaceut. 2010;394(1–2):115–121. doi:10.1016/j.ijpharm.2010.04.026
12. Wang Y, Xiong L, Tang M. Toxicity of inhaled particulate matter on the central nervous system: neuroinflammation, neuropsychological effects and neurodegenerative disease. J Appl Toxicol. 2017;37(6):644–667. doi:10.1002/jat.3451
13. Ou L, Song B, Liang H, et al. Toxicity of graphene-family nanoparticles: a general review of the origins and mechanisms. Part Fibre Toxicol. 2016;13(1):57. doi:10.1186/s12989-016-0168-y
14. Cardoso FL, Brites D, Brito MA. Looking at the blood–brain barrier: molecular anatomy and possible investigation approaches. Brain Res Rev. 2010;64(2):328–363. doi:10.1016/j.brainresrev.2010.05.003
15. Saraiva C, Praça C, Ferreira R, Santos T, Ferreira L, Bernardino L. Nanoparticle-mediated brain drug delivery: overcoming blood–brain barrier to treat neurodegenerative diseases. J Control Release. 2016;235:34–47. doi:10.1016/j.jconrel.2016.05.044
16. Wong HL, Wu XY, Bendayan R. Nanotechnological advances for the delivery of CNS therapeutics. Adv Drug Deliv Rev. 2012;64(7):686–700. doi:10.1016/j.addr.2011.10.007
17. Yang Z, Liu ZW, Allaker RP, et al. A review of nanoparticle functionality and toxicity on the central nervous system. J R Soc Interface. 2010;7(Suppl_4):S411–S422. doi:10.1098/rsif.2010.0158.focus
18. Adams DH, Joyce G, Richardson VJ, Ryman BE, Wiśniewski HM. Liposome toxicity in the mouse central nervous system. J Neurol Sci. 1977;31(2):173–179.
19. Trickler WJ, Lantz SM, Murdock RC, et al. Silver nanoparticle induced blood-brain barrier inflammation and increased permeability in primary rat brain microvessel endothelial cells. Toxicol Sci. 2010;118(1):160–170. doi:10.1093/toxsci/kfq244
20. Yin Y, Qiang L, Sun H, et al. Cytotoxic effects of ZnO hierarchical architectures on RSC96 Schwann cells. Nanoscale Res Lett. 2012;7(1):439. doi:10.1186/1556-276X-7-439
21. Zeng Y, Kurokawa Y, Win-Shwe TT, et al. Effects of PAMAM dendrimers with various surface functional groups and multiple generations on cytotoxicity and neuronal differentiation using human neural progenitor cells. J Toxicol Sci. 2016;41(3):351. doi:10.2131/jts.41.13
22. Knudsen KB, Northeved H, Pramod Kumar EK, et al. Differential toxicological response to positively and negatively charged nanoparticles in the rat brain. Nanotoxicology. 2014;8(7):764–774. doi:10.3109/17435390.2013.829589
23. Calienni MN, Feas DA, Igartúa DE, Chiaramoni NS, Sdv A, Prieto MJ. Nanotoxicological and teratogenic effects: A linkage between dendrimer surface charge and zebrafish developmental stages. Toxicol Appl Pharmacol. 2017;337:1. doi:10.1016/j.taap.2017.10.003
24. Phenrat T, Long TC, Lowry GV, Veronesi B. Partial oxidation (“Aging”) and surface modification decrease the toxicity of nanosized zerovalent iron. Environ Sci Technol. 2009;43(1):195–200.
25. Karmakar A, Zhang Q, Zhang Y. Neurotoxicity of nanoscale materials. J Food Drug Anal. 2014;22(1):147–160. doi:10.1016/j.jfda.2014.01.012
26. Zhang L, Bai R, Li B, et al. Rutile TiO2 particles exert size and surface coating dependent retention and lesions on the murine brain. Toxicol Lett. 2011;207(1):73–81. doi:10.1016/j.toxlet.2011.08.001
27. Wu J, Wang C, Sun J, Xue Y. Neurotoxicity of silica nanoparticles: brain localization and dopaminergic neurons damage pathways. ACS Nano. 2011;5(6):4476–4489. doi:10.1021/nn103530b
28. Neves AR, Queiroz JF, Lima SAC, Reis S. Apo E-functionalization of solid lipid nanoparticles enhances brain drug delivery: uptake mechanism and transport pathways. Bioconjugate Chem. 2017;28(4):995–1004. doi:10.1021/acs.bioconjchem.6b00705
29. Mendonca MCP, Soares ES, de Jesus MB, et al. Reduced graphene oxide induces transient blood-brain barrier opening: an in vivo study. J Nanobiotechnol. 2015;13(1):78. doi:10.1186/s12951-015-0143-z
30. Liu X, Sui B, Sun J. Blood-brain barrier dysfunction induced by silica NPs in vitro and in vivo: involvement of oxidative stress and Rho-kinase/JNK signaling pathways. Biomaterials. 2017;121:64–82. doi:10.1016/j.biomaterials.2017.01.006
31. Kafa H, Wang JT, Rubio N, et al. Translocation of LRP1 targeted carbon nanotubes of different diameters across the blood–brain barrier in vitro and in vivo. J Control Release. 2016;225:217–229. doi:10.1016/j.jconrel.2016.01.031
32. Lu W, Sun Q, Wan J, She Z, Jiang XG. Cationic albumin-conjugated pegylated nanoparticles allow gene delivery into brain tumors via intravenous administration. Cancer Res. 2006;66(24):11878–11887. doi:10.1158/0008-5472.CAN-06-2354
33. Huang R, Ke W, Han L, et al. Brain-targeting mechanisms of lactoferrin-modified DNA-loaded nanoparticles. J Cereb Blood Flow Metab. 2009;29(12):1914–1923. doi:10.1038/jcbfm.2009.104
34. Kreyling WG. Discovery of unique and ENM—specific pathophysiologic pathways: comparison of the translocation of inhaled iridium nanoparticles from nasal epithelium versus alveolar epithelium towards the brain of rats. Toxicol Appl Pharm. 2016;299:41–46. doi:10.1016/j.taap.2016.02.004
35. Elder A, Gelein R, Silva V, et al. Translocation of inhaled ultrafine manganese oxide particles to the central nervous system. Environ Health Persp. 2006;114(8):1172–1178. doi:10.1289/ehp.9030
36. Oberdorster G, Elder A, Rinderknecht A. Nanoparticles and the brain: cause for concern? J Nanosci Nanotechnol. 2009;9(8):4996–5007.
37. Mistry A, Stolnik S, Illum L. Nanoparticles for direct nose-to-brain delivery of drugs. Int J Pharmaceut. 2009;379(1):146–157. doi:10.1016/j.ijpharm.2009.06.019
38. Kohei Y, Yasuo Y, Kazuma H, et al. Silica and titanium dioxide nanoparticles cause pregnancy complications in mice. Nat Nanotechnol. 2011;6(5):321. doi:10.1038/nnano.2011.41
39. Webster TJ, Waid MC, McKenzie JL, Price RL, Ejiofor JU. Nano-biotechnology: carbon nanofibres as improved neural and orthopaedic implants. Nanotechnology. 2004;15(1):48–54. doi:10.1088/0957-4484/15/1/009
40. Koch F, Möller A, Frenz M, Pieles U, Kuehni-Boghenbor K, Mevissen M. An in vitro toxicity evaluation of gold-, PLLA- and PCL-coated silica nanoparticles in neuronal cells for nanoparticle-assisted laser-tissue soldering. Toxicol In Vitro. 2014;28(5):990–998. doi:10.1016/j.tiv.2014.04.010
41. Wei X, Luan L, Zhao Z, et al. Nanofabricated ultraflexible electrode arrays for high-density intracortical recording. Adv Sci. 2018;5(6):1700625. doi:10.1002/advs.201700625
42. Biran R, Martin DC, Tresco PA. Neuronal cell loss accompanies the brain tissue response to chronically implanted silicon microelectrode arrays. Exp Neurol. 2005;195(1):115–126. doi:10.1016/j.expneurol.2005.04.020
43. Zhang S, Yen S, Xiang Z, Liao L, Kwong D, Lee C. Development of silicon probe with acute study on in vivo neural recording and implantation behavior monitored by integrated Si-nanowire strain sensors. J Microelectromech Syst. 2015;24(5):1303–1313. doi:10.1109/JMEMS.2015.2417678
44. Droge W. Free radicals in the physiological control of cell function. Physiol Rev. 2002;82(1):47. doi:10.1152/physrev.00018.2001
45. Cui K, Luo X, Xu K, Ven Murthy MR. Role of oxidative stress in neurodegeneration: recent developments in assay methods for oxidative stress and nutraceutical antioxidants. Prog Neuropsychopharmacol Biol Psychiatry. 2004;28(5):771–799. doi:10.1016/j.pnpbp.2004.05.023
46. Ze Y, Zheng L, Zhao X, et al. Molecular mechanism of titanium dioxide nanoparticles-induced oxidative injury in the brain of mice. Chemosphere. 2013;92(9):1183–1189. doi:10.1016/j.chemosphere.2013.01.094
47. Shrivastava R, Raza S, Yadav A, Kushwaha P, Flora SJS. Effects of sub-acute exposure to TiO2, ZnO and Al2O3 nanoparticles on oxidative stress and histological changes in mouse liver and brain. Drug Chem Toxicol. 2013;37(3):336–347. doi:10.3109/01480545.2013.866134
48. Long TC, Saleh N, Tilton RD, Lowry GV, Veronesi B. Titanium dioxide (P25) produces reactive oxygen species in immortalized brain microglia (BV2): implications for nanoparticle neurotoxicity†. Environ Sci Technol. 2006;40(14):4346–4352.
49. Long TC, Tajuba J, Sama P, et al. Nanosize titanium dioxide stimulates reactive oxygen species in brain microglia and damages neurons in vitro. Environ Health Perspect. 2007;115(11):1631–1637. doi:10.1289/ehp.10216
50. Sharma AK, Singh V, Gera R, Purohit MP, Ghosh D. Zinc oxide nanoparticle induces microglial death by NADPH-oxidase-independent reactive oxygen species as well as energy depletion. Mol Neurobiol. 2017;54(8):6273–6286. doi:10.1007/s12035-016-0133-7
51. Yin N, Liu Q, Liu J, et al. Silver nanoparticle exposure attenuates the viability of rat cerebellum granule cells through apoptosis coupled to oxidative stress. Small. 2013;9(9–10):1831–1841. doi:10.1002/smll.201202732
52. Geppert M, Hohnholt MC, Nürnberger S, Dringen R. Ferritin up-regulation and transient ROS production in cultured brain astrocytes after loading with iron oxide nanoparticles. Acta Biomater. 2012;8(10):3832–3839. doi:10.1016/j.actbio.2012.06.029
53. Chen Z, Yin J, Zhou Y, et al. Dual enzyme-like activities of iron oxide nanoparticles and their implication for diminishing cytotoxicity. ACS Nano. 2012;6(5):4001–4012. doi:10.1021/nn300291r
54. Ren C, Hu X, Zhou Q. graphene oxide quantum dots reduce oxidative stress and inhibit neurotoxicity in vitro and in vivo through catalase-like activity and metabolic regulation. Adv Sci. 2018;5(5):1700595. doi:10.1002/advs.v5.5
55. Stern ST, Adiseshaiah PP, Crist RM. Autophagy and lysosomal dysfunction as emerging mechanisms of nanomaterial toxicity. Part Fibre Toxicol. 2012;9:20. doi:10.1186/1743-8977-9-20
56. Yang M, Zhang M, Tahara Y, et al. Lysosomal membrane permeabilization: carbon nanohorn-induced reactive oxygen species generation and toxicity by this neglected mechanism. Toxicol Appl Pharm. 2014;280(1):117–126. doi:10.1016/j.taap.2014.07.022
57. Hsiao I, Chang C, Wu C, et al. Indirect effects of TiO2 nanoparticle on neuron-glial cell interactions. Chem-Biol Interact. 2016;254(C):34–44. doi:10.1016/j.cbi.2016.05.024
58. Hsiao I, Hsieh Y, Chuang C, Wang C, Huang Y. Effects of silver nanoparticles on the interactions of neuron- and glia-like cells: toxicity, uptake mechanisms, and lysosomal tracking. Environ Toxicol. 2017;32(6):1742–1753. doi:10.1002/tox.22397
59. Weiss U. Inflammation. Nature. 2002;420(6917):845. doi:10.1038/nature01131
60. Li X, Zheng H, Zhang Z, et al. Glia activation induced by peripheral administration of aluminum oxide nanoparticles in rat brains. Nanomedicine. 2009;5(4):473–479. doi:10.1016/j.nano.2009.01.013
61. Stansley B, Post J, Hensley K. A comparative review of cell culture systems for the study of microglial biology in Alzheimer‘s disease. J Neuroinflamm. 2012;9:115. doi:10.1186/1742-2094-9-115
62. Ze Y, Sheng L, Zhao X, et al. TiO2 nanoparticles induced hippocampal neuroinflammation in mice. PLoS One. 2014;9(3):e92230. doi:10.1371/journal.pone.0092230
63. Hutter E, Boridy S, Labrecque S, et al. Microglial response to gold nanoparticles. ACS Nano. 2010;4(5):2595–2606. doi:10.1021/nn901869f
64. Sun B, Wang X, Ji Z, Li R, Xia T. NLRP3 inflammasome activation induced by engineered nanomaterials. Small. 2013;9(9–10):1595–1607. doi:10.1002/smll.201201962
65. Moquin A, Hutter E, Choi AO, et al. Caspase-1 activity in microglia stimulated by pro-inflammagen nanocrystals. ACS Nano. 2013;7(11):9585–9598. doi:10.1021/nn404473g
66. Luther EM, Petters C, Bulcke F, et al. Endocytotic uptake of iron oxide nanoparticles by cultured brain microglial cells. Acta Biomater. 2013;9(9):8454–8465. doi:10.1016/j.actbio.2013.05.022
67. Ye D, Raghnaill MN, Bramini M, Mahon E. Nanoparticle accumulation and transcytosis in brain endothelial cell layers. Nanoscale. 2013;5(22):11153–11156. doi:10.1039/c3nr02905k
68. Xue Y, Wu J, Sun J. Four types of inorganic nanoparticles stimulate the inflammatory reaction in brain microglia and damage neurons in vitro. Toxicol Lett. 2012;214(2):91–98. doi:10.1016/j.toxlet.2012.08.009
69. Choi J, Zheng Q, Katz HE, Guilarte TR. Silica-based nanoparticle uptake and cellular response by primary microglia. Environ Health Perspect. 2010;118(5):589–595. doi:10.1289/ehp.0901534
70. Chen I, Hsiao I, Lin H, Wu C, Chuang C, Huang Y. Influence of silver and titanium dioxide nanoparticles on in vitro blood-brain barrier permeability. Environ Toxicol Pharm. 2016;47:108–118. doi:10.1016/j.etap.2016.09.009
71. Trickler WJ, Lantz SM, Schrand AM, et al. Effects of copper nanoparticles on rat cerebral microvessel endothelial cells. (Report). Nanomedicine-Uk. 2012;7(6):835. doi:10.2217/nnm.11.154
72. Xu L, Dan M, Shao A, et al. Silver nanoparticles induce tight junction disruption and astrocyte neurotoxicity in a rat blood-brain barrier primary triple coculture model. Int J Nanomedicine. 2015;10:6105.
73. Trickler WJ, Lantz-McPeak SM, Robinson BL, et al. Porcine brain microvessel endothelial cells show pro-inflammatory response to the size and composition of metallic nanoparticles. Drug Metab Rev. 2014;46(2):224–231. doi:10.3109/03602532.2013.873450
74. Brun E, Carrière M, Mabondzo A. In vitro evidence of dysregulation of blood–brain barrier function after acute and repeated/long-term exposure to TiO2 nanoparticles. Biomaterials. 2012;33(3):886–896. doi:10.1016/j.biomaterials.2011.10.025
75. Raghnaill MN, Bramini M, Ye D, et al. Paracrine signalling of inflammatory cytokines from an in vitro blood brain barrier model upon exposure to polymeric nanoparticles. Analyst. 2014;139(5):923–930. doi:10.1039/c3an01621h
76. Song B, Zhang Y, Liu J, Zhou T. Unraveling the neurotoxicity of titanium dioxide nanoparticles: focusing on molecular mechanisms. Beilstein J Nanotech. 2016;7(1):645–654. doi:10.3762/bjnano.7.57
77. Singh N, Nelson B, Scanlan L, Coskun E, Jaruga P, Doak S. Exposure to engineered nanomaterials: impact on DNA repair pathways. Int J Mol Sci. 2017;18(7):1515. doi:10.3390/ijms18071515
78. Golbamaki N, Rasulev B, Cassano A, et al. Genotoxicity of metal oxide nanomaterials: review of recent data and discussion of possible mechanisms. Nanoscale. 2015;7(6):2154–2198. doi:10.1039/c4nr06670g
79. Ren C, Hu X, Li X, Zhou Q. Ultra-trace graphene oxide in a water environment triggers Parkinson‘s disease-like symptoms and metabolic disturbance in zebrafish larvae. Biomaterials. 2016;93:83–94. doi:10.1016/j.biomaterials.2016.03.036
80. Shah SA, Yoon GH, Ahmad A, Ullah F, Amin FU, Kim MO. Nanoscale-alumina induces oxidative stress and accelerates amyloid beta (Aβ) production in ICR female mice. Nanoscale. 2015;7(37):15225–15237. doi:10.1039/c5nr03598h
81. Valdiglesias V, Costa C, Kiliç G, et al. Neuronal cytotoxicity and genotoxicity induced by zinc oxide nanoparticles. Environ Int. 2013;55(C):92–100. doi:10.1016/j.envint.2013.02.013
82. El-Ghor A, Noshy MM, Galal A, Mohamed HRH. Normalization of nano-sized TiO2-induced clastogenicity, genotoxicity and mutagenicity by chlorophyllin administration in mice brain, liver, and bone marrow cells. Toxicol Sci. 2014;142(1):21–32. doi:10.1093/toxsci/kfu157
83. Hawkins SJ, Crompton LA, Sood A, et al. Nanoparticle-induced neuronal toxicity across placental barriers is mediated by autophagy and dependent on astrocytes. Nat Nanotechnol. 2018;13(5):427–433. doi:10.1038/s41565-018-0085-3
84. Singh N, Manshian B, Jenkins GJS, et al. NanoGenotoxicology: the DNA damaging potential of engineered nanomaterials. Biomaterials. 2009;30(23–24):3891–3914. doi:10.1016/j.biomaterials.2009.04.009
85. Kang Y, Liu J, Wu J, et al. Graphene oxide and reduced graphene oxide induced neural pheochromocytoma-derived PC12 cell lines apoptosis and cell cycle alterations via the ERK signaling pathways. Int J Nanomed. 2017;12:5501–5510. doi:10.2147/IJN.S141032
86. Valdiglesias V, Costa C, Sharma V, et al. Comparative study on effects of two different types of titanium dioxide nanoparticles on human neuronal cells. Food Chem Toxicol. 2013;57(C):352–361. doi:10.1016/j.fct.2013.04.010
87. Ganguly P, Breen A, Pillai SC. Toxicity of nanomaterials: exposure, pathways, assessment, and recent advances. Acs Biomater Sci Eng. 2018;4(7):2237–2275. doi:10.1021/acsbiomaterials.8b00068
88. Márquez-Ramírez SG, Delgado-Buenrostro NL, Chirino YI, Iglesias GG, López-Marure R. Titanium dioxide nanoparticles inhibit proliferation and induce morphological changes and apoptosis in glial cells. Toxicology. 2012;302(2–3):146–156. doi:10.1016/j.tox.2012.09.005
89. Valko M, Rhodes CJ, Moncol J, Izakovic M, Mazur M. Free radicals, metals and antioxidants in oxidative stress-induced cancer. Chem-Biol Interact. 2006;160(1):1–40. doi:10.1016/j.cbi.2005.12.009
90. Wang J, Deng X, Zhang F, Chen D, Ding W. ZnO nanoparticle-induced oxidative stress triggers apoptosis by activating JNK signaling pathway in cultured primary astrocytes. Nanoscale Res Lett. 2014;9(1):117. doi:10.1186/1556-276X-9-117
91. Wu J, Sun J, Xue Y. Involvement of JNK and P53 activation in G2/M cell cycle arrest and apoptosis induced by titanium dioxide nanoparticles in neuron cells. Toxicol Lett. 2010;199(3):269–276. doi:10.1016/j.toxlet.2010.09.009
92. Zhao X, Ren X, Zhu R, Luo Z, Ren B. Zinc oxide nanoparticles induce oxidative DNA damage and ROS-triggered mitochondria-mediated apoptosis in zebrafish embryos. Aquat Toxicol. 2016;180:56–70. doi:10.1016/j.aquatox.2016.09.013
93. Hadrup N, Loeschner K, Mortensen A, et al. The similar neurotoxic effects of nanoparticulate and ionic silver in vivo and in vitro. Neurotoxicology. 2012;33(3):416–423. doi:10.1016/j.neuro.2012.04.008
94. Rao RV, Ellerby HM, Bredesen DE. Coupling endoplasmic reticulum stress to the cell death program. Cell Death Differ. 2004;11(4):372–380. doi:10.1038/sj.cdd.4401378
95. Ducray AD, Stojiljkovic A, Möller A, et al. Uptake of silica nanoparticles in the brain and effects on neuronal differentiation using different in vitro models. Nanomedicine. 2017;13(3):1195–1204. doi:10.1016/j.nano.2016.11.001
96. Parveen A, Rizvi S, Mahdi F, et al. Silica nanoparticles mediated neuronal cell death in corpus striatum of rat brain: implication of mitochondrial, endoplasmic reticulum and oxidative stress. J Nanopart Res. 2014;16(11):1–15. doi:10.1007/s11051-014-2664-z
97. Majno G, Joris I. Apoptosis, oncosis, and necrosis. An overview of cell death. Am J Pathol. 1995;146(1):3–15.
98. Zhang QL, Li MQ, Ji JW, et al. In vivo toxicity of nano-alumina on mice neurobehavioral profiles and the potential mechanisms. Int J Immunopath Ph. 2011;24(1 Suppl):23S.
99. Halamoda Kenzaoui B, Chapuis Bernasconi C, Guney-Ayra S, Juillerat-Jeanneret L. Induction of oxidative stress, lysosome activation and autophagy by nanoparticles in human brain-derived endothelial cells. Biochem J. 2012;441(3):813. doi:10.1042/BJ20111252
100. Xie H, Wu J. Silica nanoparticles induce alpha-synuclein induction and aggregation in PC12-cells. Chem-Biol Interact. 2016;258:197–204. doi:10.1016/j.cbi.2016.09.006
101. Wang S, Li Y, Fan J, et al. The role of autophagy in the neurotoxicity of cationic PAMAM dendrimers. Biomaterials. 2014;35(26):7588–7597. doi:10.1016/j.biomaterials.2014.05.029
102. Wei L, Wang J, Chen A, Liu J, Feng X, Shao L. Involvement of PINK1/parkin-mediated mitophagy in ZnO nanoparticle-induced toxicity in BV-2 cells. Int J Nanomed. 2017;12:1891–1903.
103. Hu X, Wei Z, Mu L. Graphene oxide nanosheets at trace concentrations elicit neurotoxicity in the offspring of zebrafish. Carbon. 2017;117:182–191. doi:10.1016/j.carbon.2017.02.092
104. Zhong W, Lü M, Liu L, et al. Autophagy as new emerging cellular effect of nanomaterials. Chin Sci Bull. 2013;58(33):4031–4038. doi:10.1007/s11434-013-6058-x
105. Zhou Y, Hong F, Tian Y, et al. Nanoparticulate titanium dioxide-inhibited dendritic development is involved in apoptosis and autophagy of hippocampal neurons in offspring mice. Toxicol Res-Uk. 2017;6(6):889–901. doi:10.1039/C7TX00153C
106. Li Y, Zhu H, Wang S, et al. Interplay of oxidative stress and autophagy in PAMAM dendrimers-induced neuronal cell death. Theranostics. 2015;5(12):1363–1377. doi:10.7150/thno.13181
107. Haiyuan Z, Zhaoxia J, Tian X, et al. Use of metal oxide nanoparticle band gap to develop a predictive paradigm for oxidative stress and acute pulmonary inflammation. ACS Nano. 2012;6(5):4349. doi:10.1021/nn3010087
108. Tomasz P, Bakhtiyor R, Agnieszka G, et al. Using nano-QSAR to predict the cytotoxicity of metal oxide nanoparticles. Nat Nanotechnol. 2011;6(3):175–178. doi:10.1038/nnano.2011.10
109. Oksel C, Cai YM, Jing JL, Wilkins T, Xue ZW. (Q)SAR modelling of nanomaterial toxicity: acritical review. Particuology. 2015;21(4):1–19. doi:10.1016/j.partic.2014.12.001
110. Vidal F, Vásquez P, Cayumán F, et al. Prevention of synaptic alterations and neurotoxic effects of PAMAM dendrimers by surface functionalization. Nanomaterials-Basel. 2018;8(1):7. doi:10.3390/nano8010007
111. Tianyao H, Barth RF, Weilian Y, et al. Preparation, biodistribution and neurotoxicity of liposomal cisplatin following convection enhanced delivery in normal and F98 glioma bearing rats. PLoS One. 2012;7(11):e48752. doi:10.1371/journal.pone.0048752
112. Zeng Y, Kurokawa Y, Zeng Q, et al. Effects of polyamidoamine dendrimers on a 3-D neurosphere system using human neural progenitor cells. Toxicol Sci. 2016;152(1):w68. doi:10.1093/toxsci/kfw068
113. Imam SZ, Lantz-McPeak SM, Cuevas E, et al. Iron oxide nanoparticles induce dopaminergic damage: in vitro pathways and in vivo imaging reveals mechanism of neuronal damage. Mol Neurobiol. 2015;52(2):913–926. doi:10.1007/s12035-015-9259-2
114. Yang X, Liu X, Zhang Y, et al. Cognitive deficits and decreased locomotor activity induced by single-walled carbon nanotubes and neuroprotective effects of ascorbic acid. Int J Nanomed;2014:823. doi:10.2147/IJN.S56339.
115. Park E, Bae E, Yi J, et al. Repeated-dose toxicity and inflammatory responses in mice by oral administration of silver nanoparticles. Environ Toxicol Pharm. 2010;30(2):162–168. doi:10.1016/j.etap.2010.05.004
116. Cui Y, Chen X, Zhou Z, et al. Prenatal exposure to nanoparticulate titanium dioxide enhances depressive-like behaviors in adult rats. Chemosphere. 2014;96:99–104. doi:10.1016/j.chemosphere.2013.07.051
117. Miao W, Zhu B, Xiao X, et al. Effects of titanium dioxide nanoparticles on lead bioconcentration and toxicity on thyroid endocrine system and neuronal development in zebrafish larvae. Aquat Toxicol. 2015;161:117–126. doi:10.1016/j.aquatox.2015.02.002
118. Oliveira GMTD, Kist LW, Pereira TCB, et al. Transient modulation of acetylcholinesterase activity caused by exposure to dextran-coated iron oxide nanoparticles in brain of adult zebrafish. Comp Biochem Physiol C Toxicol Pharmacol. 2014;162(1):77–84. doi:10.1016/j.cbpc.2014.03.010
119. Oberdorster E. Manufactured nanomaterials (fullerenes, C60) induce oxidative stress in the brain of juvenile largemouth bass. Environ Health Perspect. 2004;112(10):1058–1062. doi:10.1289/ehp.7021
120. Bulcke F, Thiel K, Dringen R. Uptake and toxicity of copper oxide nanoparticles in cultured primary brain astrocytes. Nanotoxicology. 2014;8(7):775–785. doi:10.3109/17435390.2013.829591
121. Huang CL, Hsiao IL, Lin HC, Wang CF, Huang YJ, Chuang CY. Silver nanoparticles affect on gene expression of inflammatory and neurodegenerative responses in mouse brain neural cells. Environ Res. 2015;136:253–263. doi:10.1016/j.envres.2014.11.006
122. Huerta-García E, Pérez-Arizti JA, Márquez-Ramírez SG, et al. Titanium dioxide nanoparticles induce strong oxidative stress and mitochondrial damage in glial cells. Free Radical Bio Med. 2014;73:84–94. doi:10.1016/j.freeradbiomed.2014.04.026
123. Wilson CL, Natarajan V, Hayward SL, Khalimonchuk O, Kidambi S. Mitochondrial dysfunction and loss of glutamate uptake in primary astrocytes exposed to titanium dioxide nanoparticles. Nanoscale. 2015;7(44):18477–18488. doi:10.1039/c5nr03646a
124. Coccini T, Grandi S, Lonati D, Locatelli C, De Simone U. Comparative cellular toxicity of titanium dioxide nanoparticles on human astrocyte and neuronal cells after acute and prolonged exposure. Neurotoxicology. 2015;48:77–89. doi:10.1016/j.neuro.2015.03.006
125. Xu P, Xu J, Liu S, Ren G, Yang Z. In vitro toxicity of nanosized copper particles in PC12 cells induced by oxidative stress. J Nanopart Res. 2012;14(6):1–9. doi:10.1007/s11051-011-0686-3
126. Zhang Y, Ali SF, Dervishi E, et al. Cytotoxicity effects of graphene and single-wall carbon nanotubes in neural phaeochromocytoma-derived PC12 cells. ACS Nano. 2010;4(6):3181–3186. doi:10.1021/nn1007176
127. Hussain SM, Javorina AK, Schrand AM, Duhart HM, Ali SF, Schlager JJ. The interaction of manganese nanoparticles with PC-12 cells induces dopamine depletion. Toxicol Sci. 2006;92(2):456–463. doi:10.1093/toxsci/kfl020
128. Yang X, He CE, Li J, et al. Uptake of silica nanoparticles: neurotoxicity and Alzheimer-like pathology in human SK-N-SH and mouse neuro2a neuroblastoma cells. Toxicol Lett. 2014;229(1):240–249. doi:10.1016/j.toxlet.2014.05.009
129. Chorley B, Ward W, Simmons SO, Vallanat B, Veronesi B. The cellular and genomic response of rat dopaminergic neurons (N27) to coated nanosilver. Neurotoxicology. 2014;45:12–21. doi:10.1016/j.neuro.2014.08.010
130. Bonaventura G, Cognata V, Iemmolo R, et al. Ag-NPs induce apoptosis, mitochondrial damages and MT3/OSGIN2 expression changes in an in vitro model of human dental-pulp-stem-cells-derived neurons. Neurotoxicology. 2018. doi:10.1016/j.neuro.2018.04.014
131. Zhang Y, Xu Y, Li Z, et al. Mechanistic toxicity evaluation of uncoated and PEGylated single-walled carbon nanotubes in neuronal PC12 cells. ACS Nano. 2011;5(9):7020–7033. doi:10.1021/nn2016259
132. Hu Q, Guo F, Zhao F, Fu Z. Effects of titanium dioxide nanoparticles exposure on parkinsonism in zebrafish larvae and PC12. Chemosphere. 2017;173:373–379. doi:10.1016/j.chemosphere.2017.01.063
133. Liu S, Xu L, Zhang T, Ren G, Yang Z. Oxidative stress and apoptosis induced by nanosized titanium dioxide in PC12 cells. Toxicology. 2010;267(1–3):172–177. doi:10.1016/j.tox.2009.11.012
134. Liu J, Kang Y, Yin S, et al. Zinc oxide nanoparticles induce toxic responses in human neuroblastoma SHSY5Y cells in a size-dependent manner. Int J Nanomed. 2017;12:8085–8099. doi:10.2147/IJN.S149070.
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.