Back to Journals » International Journal of Women's Health » Volume 12
The Effect of Umbilical Cord Milking Compared with Delayed Cord Clamping in Term Neonates: A Randomized Controlled Trial
Authors Panburana P , Odthon T, Pongmee P, Hansahiranwadee W
Received 5 October 2019
Accepted for publication 18 March 2020
Published 17 April 2020 Volume 2020:12 Pages 301—306
DOI https://doi.org/10.2147/IJWH.S233487
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Everett Magann
Panyu Panburana,1 Theethuch Odthon,1 Pharuhad Pongmee,2 Wirada Hansahiranwadee1
1Division of Maternal Fetal Medicine, Department of Obstetrics and Gynecology, Faculty of Medicine Ramathibodi Hospital, Mahidol University, Bangkok, Thailand; 2Division of Neonatology, Department of Pediatrics, Faculty of Medicine Ramathibodi Hospital, Mahidol University, Bangkok, Thailand
Correspondence: Panyu Panburana
Division of Maternal Fetal Medicine, Department of Obstetrics and Gynaecology, Faculty of Medicine, Ramathibodi Hospital, Mahidol University, Bangkok 10400, Thailand
Tel/Fax +66 818469343
Email [email protected]
Objective: To study the effect of intact umbilical cord milking (I-UCM) procedure in comparison with the procedure of delayed cord clamping (DCC) in term neonates.
Study Design: A randomized controlled trial.
Setting: Department of Obstetrics and Gynecology and Department of Pediatrics, Faculty of Medicine Ramathibodi Hospital, Mahidol University.
Materials and Methods: The study was performed from June 2017 to March 2018. Singleton term pregnant women (37– 42 weeks’ gestation) were recruited. Neonates were randomized into DCC and I-UCM groups. Umbilical cord was 3 times milked toward the neonate in 25 cm length from umbilical stump which was still attached to the placenta before cutting the cord in I-UCM group. Delay in cord clamping for 60 seconds was performed before the cord cutting in DCC group. Neonatal and maternal outcomes were recorded.
Results: Of 168 neonates included in this trial, 84 cases were randomized into each group. The baseline characteristics of both groups were comparable. No statistical difference was found in terms of the mean of hemoglobin levels in the DCC and I-UCM groups which were 16.9 (± 1.6) g/dl and 17.0 (± 1.9) g/dl, respectively (P-value 0.75). There was no difference in terms of adverse neonatal and maternal outcomes in both groups.
Conclusion: Both I-UCM and DCC revealed a comparable effect on hematologic status without deleterious effects on neonatal and maternal outcomes at the age of 48– 72 hours in term neonates.
Keywords: delayed cord clamping, intact umbilical cord milking, term neonates
Introduction
One of the worldwide public health obstacles in developing countries is anemia which is commonly found in young children and pregnant women. The evidences reveal that long-term ramification in cognitive brain effect comes from anemia during infancy and young children. The iron deficiency anemia during infancy results from the iron stored during the period of delivery.1–3
When focusing on the blood transfer from the placenta to the term neonates, approximately 80 mL of blood flow from the placenta to neonates occurs during 1 minute after delivery and reaches 100 mL within 3 minutes.4,5 This phenomenon supplies the term neonates with 40–50 mg/kg of iron, the effect of which, can prevent iron anemia during the age of one year.6
There are currently two interventions, which transfer blood from cords or placentae toward the neonates. Delayed umbilical cord clamping (DCC) is the method of umbilical cord clamping from 30 to 180 seconds after delivery which is considered as a passive transfer method. While the umbilical cord milking (UCM) is performed by squeezing blood from a cord toward a newborn. This is called an active transfer method.7
The neonate’s hemoglobin at birth as well as iron storage in the first few months is believed to influence the favorable developmental outcomes.8 However, 30–60 seconds’ cord clamping delay seems too long for some emergency conditions such as nonreassuring fetal status and maternal hemodynamic vulnerability. Therefore, UCM has been pondered as the procedure to achieve a rapid time for placenta-to-neonate blood transfusion within less than 10–15 seconds.9 However, there are no published data comparing the effects of I-UCM and DCC in term infants. One trial compared cut cord milking (C-UCM) and DCC in term neonates, which did not demonstrate any difference.10
The purpose of this study was to compare the consequences of I-UCM and DCC procedures in terms of term neonates’ hematologic status at birth as well as the adverse outcomes.
Materials and Methods
This randomized controlled trial was performed from June 2017 to March 2018 at Department of Obstetrics and Gynecology and Department of Pediatrics, Ramathibodi Hospital, Mahidol University, Bangkok, Thailand. It was approved by the Ethical Clearance Committee on Human Rights Related to Researches Involving Human subjects (Protocol number ID 01-60-01), Faculty of Medicine, Ramathibodi Hospital. This research was conducted in accordance with the principles of the Declaration of Helsinki.
The written informed consent from the eligible pregnant women before delivery was obtained. The pregnant women who delivered only at Ramathibodi Hospital were enrolled. The inclusion criteria were living singleton term pregnancies (37–42 weeks of gestation). The exclusion criteria were umbilical cord length less than 25 cm or cord abnormality (such as true knots or cord prolapse), multiple gestation, maternal Rh-negative blood group, positive anti-HIV, positive HBsAg, and syphilis infection during pregnancy, antenatal diagnosed major congenital anomalies of fetus or apparent at birth, fetal hydrops and fetal growth restriction, intrapartum fetal nonreassuring or fetal distress, non-vigorous neonates, unstable maternal hemodynamic condition, placenta abruption, placenta previa, uterine rupture and denied to participate.
The primary outcome of the study was hemoglobin at the age of 48–72 hours’ term neonates. While the secondary outcomes were as the followings (A) neonatal outcomes: Apgar score at one and five minutes, hemoglobin and hematocrit at 6–24 hours of life, total serum bilirubin at 48–72 hours of life, polycythemia, jaundice requiring phototherapy or exchange transfusion and length of hospitalization and (B) maternal outcomes: estimated blood loss after delivery and postpartum hemorrhage requiring blood transfusion.
Randomization was performed right after fetal delivery and when the adequate umbilical cord length requirement and all pregnant women as well as neonates met criteria. All subjects were divided into two groups: The intact umbilical cord was 3 times milked toward the neonate in 25-cm length in I-UCM group and the 60-minute cord clamping delay was performed in DCC group. Randomization was performed by blocks-of-4 computer-generated random numbers. Serially numbered concealed envelopes were allocated for each group. The on-call staffs who did not involve in the study were requested to open the envelopes.
Regarding the position of neonates right after birth, they were placed on the maternal abdomen when delivered via vaginal route and placed over mothers’ thigh in cesarean section. Standard early neonatal cares in both I-UCM and DCC groups were the same. In I-UCM group, an umbilical cord was 3 times milked toward a neonate in 25 cm length from umbilical stump which was still attached to the placenta by a delivering obstetrician at the speed of 10 cm/second with 2-second interval to allow blood refilling from the placenta and then clamped 2–3 cm from the umbilical stump. In DCC group, an umbilical cord was also clamped 2–3 cm from the umbilical stump by a delivering obstetrician at 60 seconds after delivery of a neonate. Neonates from either group were kept with mothers and underwent routine standard management except for neonatal care requirement in NICU. Encouraging breastfeeding for all neonates was done as per our standard guidelines. Blood samples were for hemoglobin and hematocrit at 6–24 and 48–72 hours of life and in serum separator tubes for total serum bilirubin levels at 48–72 hours of life. Hemoglobin and hematocrit were measured by complete blood count test (CBC). Data collection was composed of maternal and neonatal demographic data, estimated blood loss after delivery, Apgar score, hemoglobin, hematocrit, total serum bilirubin and length of stay in the hospital.
Laboratory investigators were blinded with the intervention. The neonatologists were unable to be blinded owing to our regulation that required neonatologists to be present in the delivery room.
Sample Size and Statistical Analysis
The sample size was reckoned on the Jaiswal’s research result.10 This study revealed hemoglobin standard deviation in UCM and DCC groups was 2.4 and 2.2 g/dl, respectively. Regarding 80% power and 0.05 two tailed α value, there would be 84 neonates required in each group to study the discrepancy of 1gm/dL hemoglobin levels of term neonates. STATA 14.1 software was used for statistical analysis. Continuous data were analyzed by Mann–Whitney test or independent t-test. Chi square or Fischer exact test was used for categorical data analysis. Less than 0.05 of P value was significant.
Results
Total of 852 pregnancies were assessed for eligibility. There were 684 cases excluded for various reasons. A number of 168 neonates were enrolled and divided by half into 84 cases in both I-UCM and DCC groups (Figure 1). The characteristic baselines were comparable in both groups (Table 1).
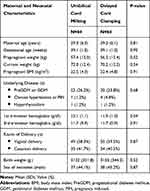 |
Table 1 Baseline Characteristics |
 |
Figure 1 Flow diagram of study participants. |
The mean (SD) of hemoglobin at 48–72 hours of life in I-UCM group was 17.0 (±1.9) g/dl and in DCC group was 16.9 (±1.6) g/dl, which was no statistical difference between both groups (P-value 0.75) (Table 2, Figure 2). Neonatal outcomes consisting of hemoglobin, hematocrit, total serum bilirubin, Apgar score, polycythemia, jaundice requiring phototherapy or exchange transfusion and length of hospitalization of both groups were found without any statistical differences (Table 2). However, one neonate in I-UCM group (1.2%) was diagnosed with polycythemia but did not require partial exchange transfusion. Complete recovery was achieved by giving an intravenous fluid and was discharged after 4 days’ hospitalization. There was also no statistical difference between these two groups in terms of maternal outcomes regarding estimated blood loss after delivery, postpartum hemorrhage and requiring blood transfusion (Table 3).
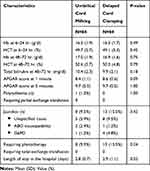 |
Table 2 Neonatal Outcomes |
 |
Table 3 Maternal Outcomes |
 |
Figure 2 Box and Whisker plot of hemoglobin (g/dl) at 48–72 hours. |
Discussion
This research focused on full-term neonates. The I-UCM was defined as 3 times umbilical cord milking toward the neonate in 25 cm from umbilical stump which was still attached to the placenta and then the umbilical cord was clamped right after completely milking the umbilical cord and the DCC was defined as the umbilical cord clamp at 60 seconds after birth. There was no hematologic effect difference between I-UCM and DCC groups. These two interventions demonstrated comparable hemoglobin levels at 6–24 and 48–72 hours of life without statistically significant difference in adverse outcomes during initial 72 hours.
The benefits of DCC on the hematological status in term infants have been reported. Cernadas et al demonstrated that DCC after birth increased neonatal hematocrit at the age of 6 hours.11 Jaleel et al also demonstrated that higher neonatal hemoglobin levels were found in DCC when comparing to early cord clamping (ECC).12 Rheenen et al demonstrated the benefit of DCC over ECC in terms of infant hemoglobin levels and incidence of lower iron deficiency anemia.13 Chaparro et al evaluated the effect of DCC on term neonates regarding higher mean corpuscular volume, ferritin, body iron and stored iron until 6 months of age.14 The Cochrane database systematic review of 15 trials on the effect of clamping the umbilical cord in term neonates regarding maternal and fetal outcomes showed no significant effect on postpartum hemorrhage in patients with DCC. Higher hemoglobin levels at 24–48 hours were found in the neonates in DCC group, when comparing with ECC group. The incidence of iron deficiency anemia of infants with ECC was found over twice higher than in infants with DCC.8
The American College of Obstetricians and Gynecologists (ACOG) and the American Academy of Pediatrics (AAP) suggested that delayed umbilical cord clamping should be performed by at least 60 seconds in both term and preterm neonates except for emergency situation that requires resuscitation.7,15
UCM seems to overcome the DCC in terms of neonatal or maternal emergency intervention requirement because the UCM can increase blood transfusion from the placenta to the neonate within the short period of time.
A randomized controlled trial on 200 term and near-term neonates by Upadhyay et al reported that infants with UCM have higher hemoglobin and iron storage than infants with ECC.16 The systematic review and meta-analysis of 7 trials on the efficacy and safety of UCM at birth in term and preterm infants revealed that UCM was associated with higher hemoglobin levels in the first 48 hours and at 6 weeks of life.9
Many literatures focus on comparing either DCC or UCM to early umbilical cord clamping (EEC) in preterm neonates. By contrast, only limited data were found when comparing DCC and UCM in term neonates. Jaiswal et al performed a randomized controlled trial comparing C-UCM and DCC on 200 term infants. There was no statistically significant difference in terms of hematologic status including serum ferritin and 6-week infants’ hemoglobin levels.10
Our I-UCM methodology was unique and different from the previous studies. Our I-UCM procedure demonstrated 3 times milking the cord which still adhered to the placenta, toward the neonates with 2 seconds’ interval to permit blood perfusion from placenta. Regarding the vaginal delivery, a neonate was held at the mother’s introitus level in our study while a neonate was placed on the maternal abdomen in Jaiswal et al’s study.
The mean hemoglobin and hematocrit levels at the age of 48 hours in both I-UCM and DCC groups were comparable to Jaiswal et al’s study. Although the mean total serum bilirubin levels at the initial 48 hours’ age in both groups were higher in our study than Jaiswal et al’s study, but the incidence of jaundice requiring phototherapy and/or exchange transfusion seemed to be the same. Moreover, we demonstrated no significant adverse effect on maternal outcomes such as estimated blood loss after delivery, postpartum hemorrhage and blood transfusion requirement in both groups. The concept of I-UCM seems to be more relevant in terms of faster blood refilling phenomenon than C-UCM method. The I-UCM can achieve placental-to-neonate transfusion within 15 seconds in our study. It is more compatibly used not only in emergency neonatal resuscitation but also in maternal instable hemodynamic status. Short duration of follow-up was our limitation. A 6–12 months’ follow-up is our further ongoing research to study the hematologic status as well as advantage and adverse outcomes during infant and early childhood periods.
Conclusion
Both intact UCM and DCC revealed the comparable effect on hematologic status without deleterious effects on both neonatal and maternal outcomes at the age of 48–72 hours in term neonates.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Allen LH, Gillespie SR. What Works? A Review of the Efficacy and Effectiveness of Nutrition Interventions. Manila: Asian Development Bank; 2001:43–54.
2. McGregor SG, Ani C. A review of the studies of iron deficiency on cognitive development in children. J Nutr. 2001;131:649S–66S. doi:10.1093/jn/131.2.649S
3. Haneef SM, Maqbool S, Arif MA, eds. Text Book of Paediatrics. International Book Bank; 2004:545–585.
4. Yao AC, Moinian M, Lind J. Distribution of blood between infant and placenta after birth. Lancet. 1969;25:871–873. doi:10.1016/S0140-6736(69)92328-9
5. Linderkamp O. Placental transfusion: determinants and effects. Clin Perinatol. 1982;9:559–592. doi:10.1016/S0095-5108(18)31013-3
6. Pisacane A. Neonatal prevention of iron deficiency. BMJ. 1996;312(7024):136–137. doi:10.1136/bmj.312.7024.136
7. The American College of Obstetricians and Gynecologists’ Committee on Obstetric Practice. Committee opinion No.684: delayed umbilical cord clamping after birth. Obstet Gynecol. 2017;129(1):e5–e10. doi:10.1097/AOG.0000000000001860
8. McDonald SJ, Middleton P, Dowswell T, Morris PS. Effect of timing of umbilical cord clamping of term infants on maternal and neonatal outcomes. Cochrane Database Syst Rev. 2013;11(2):CD004074.
9. Al-Wassia H, Shah PS. Efficacy and safety of umbilical cord milking at birth: a systematic review and meta-analysis. JAMA Pediatr. 2015;169(1):18–25. doi:10.1001/jamapediatrics.2014.1906
10. Jaiswal P, Upadhyay A, Gothwal S, et al. Comparison of two types of intervention to enhance placental redistribution in term infants: randomized control trial. Eur J Pediatr. 2015;174(9):1159–1167. doi:10.1007/s00431-015-2511-y
11. Cernadas C, Carroli G, Pellegrini L, et al. The effect of timing of cord clamping on neonatal venous hematocrit values and clinical outcome at term: a randomized, controlled trial. Pediatrics;2006. e 779–86. doi:10.1542/peds.2005-1156
12. Jaleel R, Deeba F, Khan A. Timing of umbilical cord clamping and neonatal haematological status. J Pak Med Assoc. 2009;59(7):468–470.
13. Rheenen PV, Moor LD, Eschbach S, de Grooth H, Brabin B. Delayed cord clamping and haemoglobin levels in infancy: a randomised controlled trial in term babies. Trop Med Int Health. 2007;12(5):603–616. doi:10.1111/j.1365-3156.2007.01835.x
14. Chaparro CM, Neufeld LM, Alavez GT, Cedillo RE, Dewey KG. Effect of timing of umbilical cord clamping on iron status in Mexican infants: a randomised controlled trial. Lancet. 2006;367(9527):1997–2004. doi:10.1016/S0140-6736(06)68889-2
15. Wyckoff MH, Aziz K, Escobedo MB, et al. Part 13: neonatal resuscitation: 2015 American Heart Association guidelines update for cardiopulmonary resuscitation and emergency cardiovascular care. Pediatrics. 2015;136(Supplement):S196–S218. doi:10.1542/peds.2015-3373G
16. Upadhyay A, Gothwal S, Parihar R, et al. Effect of umbilical cord milking in term and near term infants: randomized control trial. Am J Obstet Gynecol. 2013;120:e1–e6.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
