Back to Journals » Therapeutics and Clinical Risk Management » Volume 12
The effect of interferon gamma on conventional fractionated radiation-induced damage and fibrosis in the pelvic tissue of rabbits
Authors Yang Y ![]() , liu Z, Wang J, Chai YL
, liu Z, Wang J, Chai YL ![]() , Su J, Shi F, Wang J, Che SM
, Su J, Shi F, Wang J, Che SM
Received 26 November 2015
Accepted for publication 30 January 2016
Published 11 May 2016 Volume 2016:12 Pages 755—762
DOI https://doi.org/10.2147/TCRM.S101346
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor De Yun Wang
Yunyi Yang, Zi Liu, Juan Wang, Yanlan Chai, Jin Su, Fan Shi, Jiquan Wang, Shao Min Che
Department of Radiation Oncology, The First Affiliated Hospital of Medical College, Xi’an Jiaotong University, Xi’an, Shanxi, People’s Republic of China
Abstract: We aim to investigate the effect of interferon gamma (IFN-γ) on conventional fractionated radiation–induced damage and fibrosis in ureter and colorectal mucosa. Fifty-two rabbits were randomly divided into three groups comprising a conventional radiation group, an IFN-γ group, and a control group. X-rays were used to irradiate the pelvic tissues of the rabbits in the IFN-γ and conventional radiation groups. Five days after radiation exposure, the rabbits in the IFN-γ group were administered 250,000 U/kg IFN-γ intramuscularly once a week for 5 weeks. The rabbits in the conventional radiation group received 5.0 mL/kg saline. The rabbits were sacrificed at 4, 8, 12, and 16 weeks postradiation, and the rectal and ureteral tissues within the radiation areas were collected. The results showed that the morphology of rectal and ureteral tissues was changed by X-ray radiation. The degree of damage at 4, 8, and 12 weeks, but not at 16 weeks, postradiation was significantly different between the IFN-γ and conventional radiation groups. The expression of transforming growth factor beta 1 mRNA in the ureter and colorectal mucosa of the IFN-γ group was significantly lower than that in the conventional radiation group at 4, 8, 12, and 16 weeks postradiation, but it was still higher than that in the control group. There were significant differences in the expression of collagen III among the three groups. IFN-γ can inhibit the radiation-induced upregulation of transforming growth factor beta 1 mRNA and collagen III protein in the ureter and colorectal mucosa and attenuate radiation-induced damage and fibrosis.
Keywords: IFN-γ, pelvic irradiation damage, TGF-β1 mRNA, collagen III, rabbit
Introduction
Radiotherapy will inevitably induce a radiation injury and damage the tissues and/or organs within the radiation field. With increases in the use of radiotherapy in pelvic cancer patients, the incidence of radiation damage also increases. Radiation injury, such as damage to the rectum and ureter, is the main factor that limits the radiation dose used to treat pelvic tumors. The progress in radiotherapy and the quality of life of pelvic cancer patients are significantly affected by the high dose of radiation and the high incidence of side effects, such as bone marrow suppression, proctitis, and pelvic fibrosis. Moreover, a ureteral stricture can also occur in some patients exposed to pelvic radiotherapy, which, in turn, induces hydronephrosis, renal dysfunction, and other side effects severe enough that the patient needs dialysis treatment. All these conditions are related to pressure on the ureter or an obstruction induced by pelvic fibrosis, which not only affects the quality of life of patients but also increases the treatment costs. Fibrosis is a common complication of radiotherapy, but there is no effective treatment. Studies have shown that interferon gamma (IFN-γ) is a multifunctional cytokine that plays an important role in antifibrotic therapy. For example, studies have shown that IFN-γ produces antifibrotic effects on keloids and hypertrophic scars. IFN-γ has an inhibitory effect on fibroblast proliferation and collagen synthesis and on collagenous protein production and cell proliferation in vitro.1–3 Based on these findings, this study aimed to investigate the effect of IFN-γ on the prevention and treatment of radiation-induced fibrosis.
Transforming growth factor beta 1 (TGF-β1) is currently recognized as an important cytokine that is involved in the development of radiation-induced and other types of fibrosis. TGF-β1 plays an integral role in fibrosis formation by promoting the chemoattraction of fibroblasts and their conversion to myofibroblasts.4 Halofuginone has been shown to protect against radiation-induced soft tissue fibrosis by the virtue of inhibiting various members of TGF-β signaling pathway.5 It is one of the first discovered cytokines in irradiated tissues and has been demonstrated to be the most potent profibrosis factor; therefore, TGF-β1 is a key target for the development of antifibrotic drugs. It has been shown that IFN-γ can decrease TGF-β1 levels. When mouse liver cell lines were transfected with the IFN gene in vitro and transplanted into mice, TGF-β1 expression was inhibited markedly in the liver, indicating that intrasplenic transplantation of IFN-γ gene-modified hepatocyte can be a candidate approach to treat hepatic fibrosis.6 In this study, the effect of IFN-γ on conventional fractionated radiation-induced pelvic tissue damage and fibrosis was investigated to help provide targeted and potential drugs for the prevention and treatment of radiation-induced tissue damage and fibrosis.
Materials and methods
Materials
The New Zealand white rabbits (Animal Center, School of Medicine, Xi’an Jiaotong University, Xi’an, People’s Republic of China) were housed in standard conditions. Mouse antirabbit collagen III monoclonal antibody was purchased from Merck & Co., Inc. (Whitehouse Station, NJ, USA). A TGF-β1 (messenger RNA [mRNA]) ISH DIG Kit was purchased from Tianjin Haoyang Biological Manufacture Co., Ltd. (Tianjin, People’s Republic of China). Recombinant human IFN-γ was purchased from Shanghai Kelong Biotechnology Co., Ltd. (Shanghai, People’s Republic of China). The streptavidin–biotin complex immunohistochemistry kit and 3,3′-diaminobenzidine kit were purchased from Wuhan Boster Bio-Engineering Co., Ltd. (Wuhan, People’s Republic of China). The animals were kept in Xi’an Jiaotong University animal experiment center. The study was approved by the Xi’an Jiaotong University ethics committee.
Methods
Animal group, model establishment, and treatment
Fifty-two rabbits were randomly divided into three groups comprising a control group (n=4), conventional radiation group (n=24), and IFN-γ group (n=24). Each rabbit from the IFN-γ and conventional radiation groups received radiation treatment. During treatment, the rabbits were supine on a mounting plate and awake. They received the 6.0 MV X-ray five times per week, at a test dose of 2.0 Gy per time to the pelvic region from a Varian accelerator (Varian Medical Systems, Inc., Palo Alto, CA, USA). The radiation area was 5×4 cm with the pubic symphysis as the axis center. An area of 1.5 cm under the abdominal wall was considered as the reference plane, where the dose was 400 cGy/min, the source-skin distance was 100 cm, and the total test dose was 50 Gy. Rabbits in the IFN-γ group were administered 250,000 U/kg of IFN-γ intramuscularly once a week for five times after the first 5 days of radiation, and rabbits in the control group received 5.0 mL/kg of saline once a week for 5 weeks postradiation. Rabbits were sacrificed at 4, 8, 12, and 16 weeks postradiation, and rectal and ureteral tissues within the radiation areas were harvested for hematoxylin and eosin (HE) staining, in situ hybridization, and immunohistochemical staining.
HE staining of rectal and ureteral tissues
Rectal and ureteral tissues were fixed with 4.0% formalin, embedded in paraffin, sliced to 5.0 μm, and fixed and stained with HE. Each specimen was graded as follows: 0, normal or minor alterations, which cannot be ascribed with certainty to radiation; 1, slight radiation damage (mild inflammation and/or slight crypt change); 2, mild damage (more significant inflammation and/or crypt damage); 3, moderate damage (must have prominent loss of epithelium and degree of inflammation variable); and 4, severe damage (ulcers and necrosis).7
In situ hybridization for TGF-β1 in rectal and ureteral tissues
Samples were fixed in 4.0% formalin containing 1.0‰ diethylpyrocarbonate for 6.0 hours and then dehydrated, waxed, embedded, and sliced. After dewaxing with water, the sections were incubated with proteinase K at 37°C for 15 minutes and then incubated with the probe (sequences were designed based on the mRNA sequence in the NCBI database, 5′-AATTAACCCTCACTAAAGG-3′), which was diluted in prehybridization solution (50% deionized formamide, 5× saline sodium citrate, 5× Denhardt’s salmon sperm DNA, and yeast tRNA) for 24 hours. After washing and staining, a dark brown tissue indicated a positive result for TGF-β1. The negative control was set without adding the probe.
RNase-free solutions and RNase-free certified, disposable plasticware and filter tips were used, whenever possible. A separate area was maintained for RNA work. The surfaces were carefully cleaned to avoid RNA enzyme contamination. Testing results fall into three categories: positive (3+), equivocal (2+), and negative (1+ and 0).
Immunohistochemistry for collagen III in rectal and ureteral tissues
Frozen sections were rinsed with phosphate buffer saline, treated with 5.0% H2O2 for blocking peroxidase, and incubated at 4.0°C overnight with collagen III antibody (1:200). Tissues were then transferred to biotinylated secondary IgG complex (1:200) for 2 hours, followed by exposure to an avidin–biotin horseradish peroxidase complex. Yellow tissues indicated a positive result for collagen II. Testing results fall into three categories: positive (3+), equivocal (2+), and negative (1+ and 0).
For both in situ hybridization and immunohistochemistry experiments, the results were observed using the double-blind method, and the images were taken using a Nikon Spot on ballistics program (Nikon, Tokyo, Japan) and analyzed using IMAGE-Pro Plus 5.0.
Statistical analyses
Statistical analyses of the data were performed using Statistical Package for the Social Sciences 13.0 using the analysis of variance (ANOVA) test among the groups and Dunnett’s t-test between the groups. A P-value of <0.05 was considered as statistically significant.
Results
Changes in tissue pathological morphology
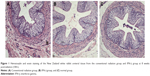
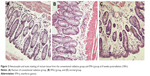
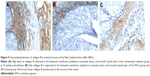
HE staining showed that congestion, edema, and inflammatory cells were present in the rectal and ureteral mucosa at 4 weeks postradiation. Inflammatory cell (including macrophages and neutrophils) infiltration was observed most obviously at 8 weeks postradiation, when necrotic cells were found in the rectal and ureteral mucosa. At 12 weeks postradiation, the morphology of part of the rectal and ureteral mucosa was disorganized, and there was fibrosis in the rectal and ureteral connective tissues; however, the tissue damage was partly attenuated at 16 weeks postradiation. There were significant differences in congestion, edema, fibrosis, and inflammatory cell infiltration at 4, 8, and 12 weeks postradiation between the IFN-γ group and the conventional radiation or control group (P<0.05) but no significant difference at 16 weeks postradiation (P>0.05; Tables 1 and 2 and Figures 1 and 2).
Expression of TGF-β1 mRNA in rectal and ureteral tissues
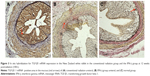
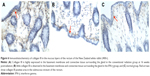
There was little expression of TGF-β1 mRNA in the rectal and ureteral mucosa of the control group; however, its expression was observed at the beginning of 4 weeks postradiation in both the IFN-γ and conventional radiation groups, significantly increased at 12 weeks postradiation, and was still obviously present at 16 weeks postradiation. The positively stained TGF-β1 mRNA was mainly present in the rectum, ureteral mucosa, muscle layer, and areas of infiltrated inflammatory cells. The expression of TGF-β1 mRNA was significantly increased in the conventional radiation group compared to that in the IFN-γ group, especially in the ureteral mucosa (Tables 3 and 4 and Figures 3 and 4).
Expression of collagen III in rectal and ureteral tissues
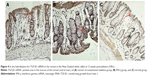
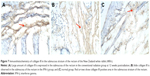
The results showed that the expression of collagen III was significantly higher in the conventional radiation group than in the control group and was mainly present in the rectum and in the submucosal basal layer, smooth muscle layer, and peripheral connective tissue of the ureter. The expression of collagen III in the IFN-γ group was dramatically lower than that in the conventional radiation group (Figures 5–7).
Discussion
Radiotherapy inevitably damages tissues and/or organs that are within the radiation field. Radiation-induced damage in patients who receive pelvic radiotherapy includes rectal mucositis and fibrosis, as well as ureteral damage and fibrosis. Common symptoms of acute radiation proctopathy include diarrhea, irregular bowel habits, hematochezia, cramping, and tenesmus. These symptoms correlate with loss of rectal mucosal cells, acute inflammation in the rectal lamina propria, and endothelial swelling.8,9 Acute radiation proctopathy occurs within 3 months of radiation therapy. It has been estimated that >75% of patients who receive pelvic radiation develop acute anorectal symptoms.10 The most important manifestation of ureteral exposure to radiation is fibrosis, which may lead to ureteral stenosis.
These side effects decrease the quality of life of these patients and increase the treatment costs; therefore, it is very important to develop new approaches to prevent and treat radiation-induced damage. IFN-γ has been shown to display potent antifibrotic effects in vitro and in animal models and has also been explored as a potential treatment for renal and idiopathic fibrosis.11,12 In fibrotic skin diseases, TGF-β plays a key role in activating fibroblast proliferation and stimulating the production of extracellular matrix proteins. The role of TGF-β in the pathogenesis of skin cancer and fibrotic diseases has been extensively studied. In addition to the direct effect of TGF-β on keratinocytes and fibroblasts, TGF-β-mediated inflammation, angiogenesis, and regulation of immune cells have additional impacts on the development of skin cancer and fibrosis.13 It has been shown that TGF-β is the master switch cytokine, which once activated after radiation promotes a train of cellular events that result in radiation-induced fibrosis.
In this study, HE staining showed that there was necrosis in the rectal and ureteral mucosa and a great number of infiltrated inflammatory cells in the mucosal layers at 4 weeks postradiation. With the increase in radiation time, the structure of rectal and ureteral mucosa was rescued to some extent; however, there was less necrosis in rectal and ureteral mucosa and less infiltrated inflammatory cells in the mucosal layers at 4 weeks postradiation in the IFN-γ group compared to those in the conventional radiation group, suggesting that IFN-γ can inhibit radiation-induced infiltration of inflammatory cells and attenuate acute radiation-induced damage to the rectal and ureteral mucosa. Based on these findings, we speculate that IFN-γ can to some extent alleviate the side effects of radiation in mucosa and might prevent mucosal damage from clinical radiotherapy; however, further studies are needed to examine whether IFN-γ can prevent acute radiation-induced mucositis in the oral cavity, intestines, bladder, and ureter. In situ hybridization of TGF-β1 showed that the expression of TGF-β1 mRNA was enhanced with an increase in radiation time. In some animal experiments, there was a sustained increase in TGF-β immunoreactivity in the small intestines after radiation.14 TGF-β acts as a potent fibrogenic and proinflammatory cytokine. Hyperplasia of connective tissue mast cells and increased leukocyte migration result from the activation of TGF-β in the intestine wall.15
Previous studies also demonstrated that TGF-β1 plays an important role in radiation-induced development of fibrosis.16,17 Together with these findings, we believe that increased TGF-β1 postradiation might be closely related to radiation-induced fibrosis. We further found that the expression of TGF-β1 mRNA was significantly decreased in the IFN-γ group compared to that in the conventional radiation group, suggesting that IFN-γ inhibits radiation-induced increases in TGF-β1 mRNA in rectal and ureteral tissues; however, the expression of TGF-β1 mRNA in the IFN-γ group was still higher than that in the control group, suggesting that IFN-γ cannot completely inhibit radiotherapy-induced upregulation of TGF-β1 mRNA. These findings suggest that IFN-γ can reduce the expression of TGF-β1, thus decreasing the development of fibrosis; however, the underlying mechanisms are still unknown. Immunohistochemistry for collagen III showed that the expression of this protein gradually increased with the increase in radiation time in the rectal tissues, as well as in the submucosal basal layer, smooth muscle layer, and peripheral connective tissue of the ureter, suggesting the gradual formation of radiation-induced fibrosis in the rectum and ureter. These findings suggest that fibrosis in the rectum, peritoneum, and the connective tissues of the ureter develops gradually following pelvic radiation, thus inducing various complications in pelvic tissues. The expression of collagen III was significantly decreased in the IFN-γ group compared to that in the conventional radiation group, suggesting that IFN-γ inhibits radiation-induced increases in collagen III in the rectal and ureteral tissues. Combined with the findings that IFN-γ inhibits radiation-induced upregulation of TGF-β1 mRNA, these findings imply that IFN-γ might inhibit the expression of collagen III by downregulating TGF-β1 mRNA, thus decreasing fibrosis in the rectum and ureter.
With the increased incidence of tumors, the use of radiotherapy for pelvic tumors also increases. The direction for studies on tumor therapy is to develop approaches that not only enhance treatment efficacy but also reduce radiotherapy-induced complications, thus enhancing the long-term survival and quality of life of cancer patients. Acute and chronic radiation-induced damage seriously affects the quality of life of cancer patients, and there are no effective drugs that can prevent this damage; therefore, there remains the critical issue of preventing and treating radiotherapy-induced damage and fibrosis in clinics. In this study, our findings show that IFN-γ attenuates radiotherapy-induced damage and fibrosis in rectal and ureteral tissues, which has positive significance for developing effective drugs that can prevent and treat these side effects; however, clinical applications of IFN-γ also have side effects, and more studies are needed to clarify whether IFN-γ can be used to prevent and treat radiation-induced damage and fibrosis in rectal and ureteral tissues.
Conclusion
Although the animal experiments showed that IFN-γ can inhibit the radiation-induced damage and fibrosis, there are still many problems to be solved. At present, there are few clinical studies available for the treatment of radiation-induced damage and fibrosis with IFN-γ. The major problem hampering a wider application of IFN-γ in treatment is its adverse effects. Although different routes and delivery systems have been used to deliver IFN-γ for the treatment of a variety of viral and neoplastic diseases, little has been reported on the most efficient and least toxic routes and drug delivery modes required to achieve these goals. One important thing is the concentration of IFN-γ that is necessary to achieve an optimal therapeutic response. Unfortunately, we do not know much about the specific mechanisms involved in the control of radiation-induced damage and fibrosis development during the initial and the latency phases of damage and fibrosis. More research is needed in this area. In this study, treating radiation-induced damage and fibrosis using IFN-γ for is tested. Further testing of IFN-γ in treating radiation-induced damage and fibrosis is needed.
Disclosure
The authors report no conflicts of interest in this work.
References
McCauley RL, Chopra V, Li YY, Hendersen DN, Robson MC. Altered cytokine production in Black patients with keloids. J Clin Immunol. 1992;12:300–308. | ||
Young-Chun L, Min-Hyoung P, Nam-Ho B. Effect of g-interferon on fibroblasts proliferation and collagen synthesis after glaucoma filtering surgery in white rabbits. Korean J Ophthalmol. 1991;5:59–67. | ||
Nguyen KD, Hoang AT, Lee DA. Transcriptional control of human Tenon’s capsule fibroblasts collagen-synthesis in vitro by gamma interferon. Invest Ophthalmol Vis Sci. 1994;35:3064–3070. | ||
Graves PR, Siddiqui F, Anscher MS, Movsas B. Radiation pulmonary toxicity: from mechanisms to management. Semin Radiat Oncol. 2010;20:201–207. | ||
Xavier S, Piek E, Fujii M, et al. Amelioration of radiation-induced fibrosis: inhibition of transforming growth factor-beta signaling by halofuginone. J Biol Chem. 2004;279:15167–15176. | ||
Zhang L, Mi J, Yu Y, et al. IFN-gene therapy by intrasplenic hepatocyte transplantation: a novel strategy for reversing hepatic fibrosis in Schistosoma japonicum-in-fested mice. Parasite Immunol. 2001;23:11–17. | ||
Northway MG, Scobey MW, Geisinger KR. Radiation proctitis in the rat. Sequential changes and effects of anti-inflammatory agents. Cancer. 1988;62(9):1962–1969. | ||
Johnson MJ, Robertson GM, Frizelle FA. Management of late complications of pelvic radiation in the rectum and anus: a review. Dis Colon Rectum. 2003;46:247–259. | ||
Ajlouni M. Radiation-induced proctitis. Curr Treat Options Gastroenterol. 1999;2:20–26. | ||
Anscher MS, Chen L, Rabbani Z, et al. Recent progress in defining mechanisms and potential targets for prevention of normal tissue injury after radiation therapy. Int J Radiat Oncol Biol Phys. 2005;62:255–259. | ||
Bouros D, Antoniou KM, Tzouvelekis A, Siafakas NM. Interferon-gamma 1b for the treatment of idiopathic pulmonary fibrosis. Expert Opin Biol Ther. 2006;6:1051–1060. | ||
Oldroyd SD, Thomas GL, Gabbiani G, El Nahas AM. Interferon-gamma inhibits experimental renal fibrosis. Kidney Int. 1999;56:2116–2127. | ||
Lagares D, Garcia-Fernandez RA, Jimenez CL, et al. Endothelin 1 contributes to the effect of transforming growth factor β1 on wound repair and skin fibrosis. Arthritis Rheum. 2010;62:878–889. | ||
Langberg CW, Hauer-Jensen M, Sung CC, Kane CJ. Expression of fibrogenic cytokines in rat small intestines after fractionated irradiation. Radiother Oncol. 1994;32:29–36. | ||
Richter KK, Langberg CW, Sung CC, Hauer-Jensen M. Increased transforming growth factor beta immunoreactivity is independently associated with chronic injury in both consequential and primary radiation enteropathy. Int J Radiat Oncol Biol Phys. 1997;39:187–195. | ||
Martin M, Delanian S, Sivan V, et al. TGF-β1 and radiation fibrosis. Cancer Radiother. 2000;4(5):369–384. | ||
Martin M, Lefaix J, Delanian S. TGF-beta1 and radiation fibrosis: a master switch and a specific therapeutic target? Int J Radiat Oncol Biol Phys. 2000;47(2):277–290. |
 © 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.