Back to Journals » Drug Design, Development and Therapy » Volume 9
Synthesis of an anthraquinone derivative (DHAQC) and its effect on induction of G2/M arrest and apoptosis in breast cancer MCF-7 cell line
Authors Yeap S, Akhtar MN, Lim KL, Abu N, Ho WY, Zareen S, Roohani K, Ky H, Tan SW , Lajis N, Mohamed Alitheen NB
Received 3 April 2014
Accepted for publication 7 May 2014
Published 17 February 2015 Volume 2015:9 Pages 983—992
DOI https://doi.org/10.2147/DDDT.S65468
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
SweeKeong Yeap,1 Muhammad Nadeem Akhtar,2 Kian Lam Lim,3 Nadiah Abu,4,5 Wan Yong Ho,6 Seema Zareen,2 Kiarash Roohani,1 Huynh Ky,4 Sheau Wei Tan,1 Nordin Lajis,7 Noorjahan Banu Alitheen1,4
1Institute of Bioscience, Universiti Putra Malaysia, Selangor Darul Ehsan, Malaysia; 2Faculty of Industrial Sciences and Technology, Universiti Malaysia Pahang, Kuantan, Pahang, Malaysia; 3Faculty of Medicine and Health Sciences, Universiti Tunku Abdul Rahman, Selangor Darul Ehsan, Malaysia; 4Faculty of Biotechnology and Biomolecular Sciences, Universiti Putra Malaysia, Selangor Darul Ehsan, Malaysia; 5Bright Sparks Unit, University of Malaya, Kuala Lumpur, Malaysia; 6School of Biomedical Sciences, University of Nottingham Malaysia Campus, Selangor Darul Ehsan, Malaysia; 7Scientific Chairs Unit, Taibah University, Medina, Saudi Arabia
Abstract: Anthraquinones are an important class of naturally occurring biologically active compounds. In this study, anthraquinone derivative 1,3-dihydroxy-9,10-anthraquinone-2-carboxylic acid (DHAQC) (2) was synthesized with 32% yield through the Friedel–Crafts condensation reaction. The mechanisms of cytotoxicity of DHAQC (2) in human breast cancer MCF-7 cells were further investigated. Results from the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay showed that DHAQC (2) exhibited potential cytotoxicity and selectivity in the MCF-7 cell line, comparable with the naturally occurring anthraquinone damnacanthal. DHAQC (2) showed a slightly higher IC50 (inhibitory concentration with 50% cell viability) value in the MCF-7 cell line compared to damnacanthal, but it is more selective in terms of the ratio of IC50 on MCF-7 cells and normal MCF-10A cells. (selective index for DHAQC (2) was 2.3 and 1.7 for damnacanthal). The flow cytometry cell cycle analysis on the MCF-7 cell line treated with the IC50 dose of DHAQC (2) for 48 hours showed that DHAQC (2) arrested MCF-7 cell line at the G2/M phase in association with an inhibited expression of PLK1 genes. Western blot analysis also indicated that the DHAQC (2) increased BAX, p53, and cytochrome c levels in MCF-7 cells, which subsequently activated apoptosis as observed in annexin V/propidium iodide and cell cycle analyses. These results indicate that DHAQC (2) is a synthetic, cytotoxic, and selective anthraquinone, which is less toxic than the natural product damnacanthal, and which demonstrates potential in the induction of apoptosis in the breast cancer MCF-7 cell line.
Keywords: cytotoxic, selective index, cell cycle
Introduction
Cancer is one of the most major health problems, according to any frame of reference, and, unfortunately, is not likely to lose that qualification any time soon. The breast is one of the four major cancer sites, and, of all cancers, breast cancer has the highest incidence rate and is the second highest cause of death in females.1 Thus, research in medicinal chemistry has been focused on the discovery of novel anticancer compounds, including natural compounds that can specifically target cancer cells and minimize the possible side effects on normal cells.2 Anthraquinones are potential natural anticancer compounds that can be isolated from Damnacanthus and Morinda spp. Among the natural anthraquinones, damnacanthal has been shown to possess potential cytotoxic, immunomodulatory,3 and anticancer activities.4 These cytotoxic and apoptotic activities against breast cancer cell lines were found to be regulated by damnacanthal via activation of p53 and p21 genes.5 Other naturally occurring anthraquinones have also been shown to exhibit similar anti-inflammatory, antibiotic, antiviral, and antineoplastic activities.6–8 However, due to the lack of accessibility, the time and cost of preparation, and the limited quantity of naturally occurring anthraquinones, chemically synthesized compounds have garnered a great deal of attention as a means of complementing the natural isolated compounds in anticancer drug discovery.9 Based on this connection, we synthesized the compound 1,3-dihydroxy-9,10-anthraquinone-2-carboxylic acid (DHAQC) (2) by Jones oxidation10 similar to the previously synthesized compound, 2-hydroxymethyl-1,3-dimethoxy-9,10-anthraquinone (1).
One of the major obstacles in anticancer compound discovery is that the candidate compounds could have nonspecific toxicity against both cancerous and normal cells.11 Thus, identification of a novel compound with high selectivity against cancerous cells rather than normal cells is one of the major objectives of this type of study. The current study aimed to evaluate and compare the selectivity of DHAQC (2) to damnacanthal on cancerous and normal cell lines. Moreover, the cell cycle arrest and apoptotic effects of DHAQC (2) on MCF-7 cells was also evaluated by quantitative polymerase chain reaction (PCR), Western blot, and flow cytometry analysis.
Materials and methods
Synthesis of DHAQC (2)
Damnacanthal was synthesized according to the methods of Akhtar et al.12 Compound (1) (2-hydroxymethyl-1,3-dimethoxy-9,10-anthraquinone) was synthesized as described in our previous publication.12 Briefly, the phthalic anhydride and 1,3-dihydroxy-2-methylbenzene were mixed in a molten mixture of AlCl3/NaCl. Then, the product was acetylated with acetic anhydride and potassium carbonate. Next, the product underwent methylation with K2CO3/(CH3)2SO4, which was further brominated with Wohl-Ziegler’s reaction. Finally, the product was hydrolyzed in acetic acid and water (8:2) to yield compound (1). For the synthesis of compound (2), DHAQC (2), compound (1) (500 mg, 1.7 mmol) was dissolved in 30 mL of acetone in a round bottle flask equipped with a CaCl2 drying tube. The reaction mixture was stirred at 0°C–5°C in an ice bath. Jones reagent was prepared by mixing CrO3 + H2SO4 at 0°C in acetone. After 2 hours of storage in a refrigerator, 5 mL of the Jones reagent was diluted with water and added slowly under nitrogen atmosphere until the yellow solution turned greenish. The product was treated with 5% solution of sodium bisulfite, extracted with ethyl acetate, and purified by column chromatography. The compound DHAQC (2) was analyzed by electron ionization mass spectrometry (EI-MS), infrared (IR), and nuclear magnetic resonance spectroscopic (NMR) techniques. Compound (2) was obtained as a yellow-green amorphous powder and purified by column chromatography. The yield was 32.0%. IR (cm−1) in KBr disk: 3450-, 3230 (OH), 2973 (CH), 1646 (H-C=O), 1670 (non-chelated C=O), 1466 (C=C< aromatic), 1244, 1262, 1230, 1132, 710. 1H-NMR (500 MHz, acetone-d6):δ 12.21 (s, OH), 8.22 (d, 1H, J=7.5 Hz, H-8), 8.09 (d, 1H, J=7.5 Hz, H-5), 7.75–7.72 (m, 2H, H-6, and H-7), 7.70 (s, 1H, H-4), MS m/z (relative intensity):284.20 (M+, 242), 281 (45), 226 (56), 254 (79), 206 (7), 196 (24), 180 (57), 77, 65, 54.
Cell culture
Human erythromyeloblastoid leukemia K562 cells, estrogen-dependent breast cancer MCF-7 cells, estrogen-independent breast cancer MDA-MB-231 cells, and normal breast MCF-10A cells were purchased from American Type Culture Collection ([ATCC] Manassas, VA, USA). K562 and MCF-7 cells were cultured in Roswell Park Memorial Institute (RPMI)-1640 (Sigma-Aldrich Co, St Louis, MO, USA) and supplemented with 10% fetal bovine serum (FBS) (GE Healthcare, Little Chalfont, UK), while MDA-MB-231 cells were cultured in Dulbecco’s Modified Eagle’s Medium (DMEM) (Sigma-Aldrich Co) supplemented with 10% FBS. MCF-10A cells were cultured in DMEM/F-12 (Sigma-Aldrich Co) supplemented with 10% FBS, hydrocortisone (0.5 μg/mL), insulin (10 μg/mL), and human epidermal growth factor (20 ng/mL) (Sigma-Aldrich Co). All cell lines were kept in a 37°C incubator with 5% CO2.
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) cell viability assay
MCF-7, MDA-MB-231, and MCF-10A cells were seeded in 96-well plates (0.8×105 cells/mL) and cultured overnight. Then, starting at 30 μg/mL, serial 1:1 dilutions of the examined compounds were added to triplicate wells on the seeded plates. The nonadherent K562 cells were loaded (0.8×105 cells/mL) on the 96-well plate, which contained the appropriate dilutions of compounds. All plates were incubated for 48 hours at 37°C, 5% CO2. After the incubation period, 20 μL of MTT (5 mg/mL) solution (Sigma-Aldrich Co) was added to all wells and incubated in darkness for 3 hours. Then, 170 μL of solution was discarded and 20 μL of dimethyl sulfoxide (DMSO) was added to each well to solubilize the purple crystals. The plates were read using a microplate reader at 570 nm (BioTek Instruments, Inc., Winooski, VT, USA). IC50 (inhibitory concentration with 50% cell viability) values, indicating the concentration of DHAQC (2) or damnacanthal that inhibited 50% of cell viability compared to untreated control, were obtained, and selective index was calculated by dividing the IC50 value of MCF-10A cells by the IC50 value of MCF-7 or MDA-MB-231 cells.
Cell preparation
MCF-7 cells were seeded overnight and treated with the IC50 value of DHAQC (2) for 24 hours for quantitative real-time PCR (qRT-PCR) and with IC25 (inhibitory concentration that inhibits 25% of cell viability) (3.50 μg/mL) and IC50 (7.20 μg/mL) of DHAQC (2) for 48 hours for flow cytometry, Western blot, and caspase-9 analysis. Untreated control cells were prepared simultaneously. After the treatment period, cells were harvested, washed, and subjected to the following assays.
Flow cytometry cell cycle PI (propidium iodide) assay
Treated and untreated MCF-7 cells were stained with BD CycletestTM Plus DNA reagent kit (BD, Franklin Lakes, NJ, USA) according to the manufacturer’s protocol. The cell cycle phase was analyzed using BD CellQuest Pro software by FACSCalibur™ (BD, Franklin Lakes, NJ, USA).
Flow cytometry annexin V/PI assay
Treated and untreated MCF-7 cells were mixed with 100 μL of 1× binding buffer. Then, 5 μL of annexin V and PI, each from the Annexin V-FITC apoptosis detection kit (BD Biosciences, San Jose, CA, USA), were added to the cells and incubated for 15 minutes. Afterwards, stained cells were washed and mixed with 500 μL of 1× binding buffer, and the apoptosis of the cells was analyzed using BD CellQuest Pro software by FACSCalibur.
qRT-PCR assay
RNA from the treated and untreated MCF-7 cells was extracted using the RNeasy Plus Mini Kit (Qiagen NV, Venlo, the Netherlands) according to the manufacturer’s protocol. The RNA quality was quantified using a NanoDrop spectrophotometer (Thermo Fisher Scientific, Waltham, MA, USA). The RNA was converted to complementary (c)DNA using an iScriptTM cDNA synthesis kit (Bio-Rad Laboratories Inc., Hercules, CA, USA). Expression of p21, PLK1, and FOXM1 were evaluated by qRT-PCR with an SYBR® Select Master Mix (Thermo Fisher Scientific, Waltham, MA, USA) on an iQ-5 Real Time PCR (Bio-Rad Laboratories Inc.) using the primers as stated in Table 1 and the following PCR conditions: one cycle of 50°C for 2 minutes for UDG (uracil-DNA glycosylase) activation; one cycle of 95°C for 2 minutes for DNA polymerase activation; 40 cycles of 95°C for 2 seconds for denature; and 60°C for 30 seconds for annealing and extension. Standard curves for housekeeping genes (β-actin, 18srRNA, and GAPDH) and target genes (p21, PLK1, and FOXM1) were generated from the amplification of the serially diluted RNA samples isolated from the control MCF-7 cell line to optimize the assay. Each of the, treated and untreated samples were run in triplicate and non-template control was included. Normalization of reference genes was done using geNorm.13
Western blot analysis
Protein was extracted from IC25 and IC50 DHAQC (2)-treated and untreated control cells using radioimmunoprecipitation assay buffer, and the protein concentration was determined using Bradford protein assay (Thermo Fisher Scientific). The cell homogenates were electrophoresed by sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) (Bio-Rad Laboratories, Inc., Hercules, CA, USA), transferred onto nitrocellulose membrane, blocked with Tris-buffered saline–Tween buffer containing 5% skimmed milk powder for 1 hour, and incubated with primary antibodies (anti-p53, anti-BAX, anti-Bcl-2, anti-cytochrome c, and anti-β-actin at dilution 1:1000) (Abcam plc, Cambridge, UK) for another hour. Then, membranes were washed and incubated with the anti-rabbit secondary antibodies-conjugated with alkaline phosphatase (1:5000 dilution) for 1 hours, followed by washing with Tris-buffered saline–Tween buffer, and finally visualized by chemiluminescence with CDP-Star® reagent (NEB [UK], Hitchin, UK) using a BioSpectrum® system (UVP, Upland, CA, USA). The size of the targeted proteins was determined by protein marker (PageRuler Prestained Protein Ladder; Thermo Fisher Scientific), and the density results obtained from this targeted protein were analyzed using VisionWorks®LS analysis software (Version 7.1 RC3.54; UVP) by normalizing against β-actin.
Caspase-9 fluorescence assay and viability in the presence of general caspase inhibitor
MCF-7 cells were pretreated with or without cell permeable general caspase inhibitor (Z-VAD-FMK) (Promega Corporation, Fitchburg, WI, USA) at 10 μM before treatment with IC25 and IC50 of DHAQC (2) for 48 hours. After the treatment period, viability of DHAQC (2)-treated MCF-7 cells with or without Z-VAD-FMK was quantified using MTT assay as described above. Then, protein was extracted from IC25 and IC50 DHAQC (2)-treated and untreated control cells with or without Z-VAD-FMK using radioimmunoprecipitation assay buffer, and the protein concentration was determined using Bradford protein assay. Levels of activated caspase-9 were detected using CaspGLOW Red Active Caspase-9 Staining Kit (BioVision, Inc., Milpitas, CA, USA) according to the manufacturer’s protocol. The fluorescence intensity in DHAQC (2)-treated and untreated MCF-7 cells was measured at excitation (Ex)/emission (Em) =540/570 nm using a microplate fluorometer (Arzoskan Flash; Thermo Fisher Scientific).
Statistical analysis
Data were presented as mean ± standard deviation from three experiments in three different biological replicates, and the significance (P<0.05) was evaluated by one-way analysis of variance (ANOVA) with Duncan test as post hoc analysis using SPSS software (v 14; SPSS Inc., Chicago, IL, USA).
Results and discussion
Chemical modifications in the side chain are one common approach in drug discovery for improving the therapeutic potential of a certain compound. Our main interest was to improve the yield of damnacanthal using different kinds of oxidizing agents, such as AgO/HNO3, Vilsmeier reagent, and Lewis acid.14 None of the methods gave a reasonable yield and most gave oxidative demethylated products.14 However, the oxidation approach of a previously synthesized compound, 2-hydroxymethyl-1,3-dimethoxy-9,10-anthraquinone (1),12 with Jones reagents10 resulted in a 32% yield of DHAQC (2) (Figure 1).
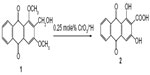  | Figure 1 Synthesis of DHAQC (2) from 2-hydroxymethyl-1,3-dimethoxy-9,10-anthraquinone (1). |
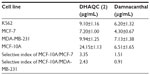
Both damnacanthal and DHAQC (2) contain the anthraquinone planer basic skeleton, which is the major contributor to the cytotoxicity properties. The planar tricyclic structure of anthraquinone is essential for cytotoxic activities and has been widely investigated for cancer therapy.15 Anthraquinone derivatives such as doxorubicin, daunorubicin, and carminomycin are highly effective in the treatment of tumors.16 Cytotoxicity of DHAQC (2) on K562, MCF-7, and MCF-10A cell lines was evaluated using the MTT assay, and IC50 value of these cell lines after 48 hours of treatment were obtained (Table 2). The selectivity of DHAQC (2) and damnacanthal on normal and cancerous cell lines was calculated by dividing the IC50 on normal MCF-10A cells by that of either estrogen-dependent MCF-7 or estrogen-independent MDA-MB-231 cells. Both DHAQC (2) and damnacanthal showed the highest cytotoxicity against MCF-7 compared to the other cell lines. Further, the IC50 values of DHAQC (2) and damnacanthal in MCF-7 and K562 were lower compared to the precursor, compound (1) with IC50 values of 12.80 and 14.00 μg/mL, respectively.12 Previously, we demonstrated the contribution of methoxy, CHO groups, and OH group to the cytotoxicity of anthraquinones against tested cancerous cell lines.12 DHAQC (2) possesses two OH groups and one carboxylic acid (COOH) group, which gives it a greater hydrophilic character compared to damnacanthal. The presence of hydroxyl and acidic groups in DHAQC (2) increases the solubility and might facilitate absorption or diffusion across the cellular membrane, as revealed by the theoretical membrane-interaction quantitative structure–activity relationship (MI-QSAR) studies of 188 drug-like compounds.17,18 The better selectivity of DHAQC (2) toward cancer cell lines is possibly a result of its hydrophilic character, which might fulfill Lipinski’s rule of five and have drug-like properties.19 Many drugs, for instance, mitoxantrone, possessing 1,4 hydroxyl groups have demonstrated potent antitumor activity and have been used widely in clinic since 1980.20,21 Moreover, the hydrophilic property of DHAQC (2) may also be involved in it’s chelating with magnesium and zinc ions in DNA synthesis and the repair process.15 Added to this, the hydrophilicity or introduction of acidic groups increased the hydrophilicity of the rings and facile to bind with receptor. It is well known that the polycyclic (aromatic) structure of anthraquinone can intercalate with DNA in between base pairs, either covalently or electrostatically. This DNA intercalation with anthraquinone compounds possibly inhibits the DNA replication in cancer cell lines.22 Previous research found that the position but not the total amount of hydroxyl in anthraquinone played an important role in determining its toxicity against normal and cancerous cell lines in vitro. Thus, modification of the hydroxyl group in DHAQC (2) might reduce its toxicity to normal cells while maintaining its apoptosis-inducing effect on cancer cells, thus leading to greater selectivity of DHAQC (2) compared to that of damnacanthal.23 As a result, the slight modification in natural compounds, as in DHAQC (2), could be useful for the development of new prototypes of anthraquinone-type cytotoxic agents.
Since DHAQC (2) showed the highest sensitivity and selectivity on hormone-dependent breast cancer MCF-7 cells, further evaluation on cell cycle regulation, apoptosis, gene expression, and apoptosis-related protein levels were evaluated. The cell cycle regulation and apoptotic effects were evaluated using flow cytometry after 48 hours of treatment. Many cytotoxic agents arrest the cell cycle of cancerous cells at the S and G2/M phases, followed by the induction of apoptosis.24 DHAQC (2) arrested MCF-7 at the S and G2/M phases (21.33%±1.74% and 37.66%±4.11%, respectively) as compared to the control (11.76%±1.31% and 4.48%±%3.72%, respectively) after 48 hours of incubation. DHAQC (2) also induced 20.48%±2.91% of the MCF-7 cell population to enter the subG0 phase, which indicated the occurrence of DNA fragmentation in the DHAQC (2)-treated cells. This result was further supported by the annexin V/PI apoptosis study, wherein DHAQC (2) was able to induce 16.36%±3.72% of cells to undergo late apoptosis and 29.35%±3.81% to undergo early apoptosis (Figure 2). These cell cycle and apoptosis results were similar to those of our previous study, wherein damnacanthal was found to induce G2/M arrest, DNA fragmentation, and apoptosis toward MCF-7 cells.5
Since cell cycle arrest was observed in DHAQC (2)-treated MCF-7 cells, expression of p21, PLK1, and FOXM1 genes involved in G2/M arrest was evaluated using qRT-PCR. PLK1 is a key regulator of G2/M phase progression, while forkhead box M1 (FOXM1) is a direct binding partner of PLK1, FOXM1 plays an important role in regulating the mitotic entry and expression of clusters of G2/M target genes.25 Downregulation of PLK1 and FOXM1 expression are always associated with G2/M arrest and apoptosis (Kim et al, 2012).26 On the other hand, upregulation of p21 was found to arrest the cell cycle in the G2/M phase as reported in damnacanthal-treated MCF-7 cells.5 Based on the qRT-PCR results, DHAQC (2) was able to inhibit the expression of PLK1 significantly. Further, expression of p21 and FOXM1 were up- and downregulated, respectively, although it is not significant (Figure 3). Depletion of PLK1 gene was also reported to subsequently induce apoptosis in cancer cells (Liu and Erikson, 2003).27 Thus, significant PLK1 downregulation by DHAQC (2) contributed to the G2/M arrest and apoptosis in MCF-7 cells as observed in the flow cytometry analysis.
Besides regulation of G2/M arrest-related genes, effects of DHAQC (2) on apoptosis-related proteins, including Bcl-2, BAX, p53, and cytochrome c, were also evaluated by Western blot analysis. After 48 hours of treatment, both IC25 and IC50 of DHAQC (2) were able to enhance the levels of p53, BAX, and cytochrome c, but had no dose-dependent effect on Bcl-2 (Figure 4). The ability of DHAQC (2), as an anthraquinone, to bind and chelate the DNA may contribute to enhancement of p53 levels.28 Furthermore, this DNA binding ability may be further enhanced by the increased hydrophilic property of DHAQC (2). p53 is a tumor suppressor protein that plays an important role in regulating G2/M arrest and apoptosis via upregulation of p21, transcriptional target of p53, and multidomain proapoptotic protein BAX. BAX is essential for the onset of mitochondrial permeabilization and apoptosis via the intrinsic pathway. The balance between expression levels of antiapoptotic Bcl-2 and proapoptotic BAX is crucial for cell survival or death, wherein increase of the BAX/Bcl-2 ratio will activate the release of cytochrome c from mitochondria and subsequently activate the intrinsic apoptotic pathway.29 Induction of BAX increased the BAX/Bcl-2 ratio, which subsequently enhanced the release of cytochrome c from the mitochondria (Figure 4) and increased the level of activated caspase-9 (Figure 5), which indicates the activation of the intrinsic apoptotic pathway.24 Similarly to another anthraquinone derivative, dantron,30 stimulation of activated caspase-9 and reduction of cell viability was suppressed in the presence of general caspase inhibitor Z-VAD-FMK. These results indicate that activation of caspase-9 is crucial for induction of apoptosis in MCF-7 by DHAQC (2). Furthermore, activation of p53 that induced G2/M arrest and the intrinsic apoptotic pathway was similar to the effects induced by damnacanthal.5 Additionally, regulation of these apoptosis-related proteins was in a dose-dependent manner, wherein upregulation of BAX, p53, cytochrome c, and caspase-9 were found to be higher in MCF-7 cells treated with IC50 of DHAQC (2) than in those treated with IC25 of DHAQC (2).
Conclusion
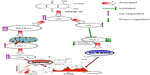
This study indicates that the production of DHAQC (2) warranted a better yield (32%) than the synthesis of damnacanthal. The synthesized DHAQC (2) also possessed better selectivity against estrogen-dependent breast cancer MCF-7 cells compared to damnacanthal. The better selectivity of DHAQC (2) compared with damnacanthal may have resulted from the increase of hydrophilicity or introduction of acidic groups. Induction of cell death by DHAQC (2) was contributed by G2/M cell cycle arrest and the downregulation of the PLK1 gene induces the intrinsic pathway of apoptosis via upregulation of p53, BAX/Bcl-2 ratio, and release of cytochrome c (Figure 6). Caspase inhibitor Z-VAD-FMK prevented the cell death induced by DHAQC (2) and inhibited the increase of active caspase-9 levels, suggesting that caspase-9 might be involved in DHAQC (2)-induced apoptosis of MCF-7 cells. Besides cell cycle arrest and apoptosis, damnacanthal demonstrated other bioactivities, including inhibition of cell migration and invasion via inhibition of kinases.31 Thus, further evaluation and comparison of the mechanisms of in vitro and in vivo antitumor and antimetastatic effects via inhibition of protein tyrosine kinase between DHAQC (2) and damnacanthal should be carried out.
Acknowledgment
This study was supported by Universiti Malaysia Pahang (internal grant number RDU 120373).
Author contributions
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data. All authors took part in either drafting the article or revising it critically for important intellectual content. All authors gave final approval of the version to be published.
Disclosure
The authors report no conflicts of interest in this work.
References
Siegel R, Naishadham D, Jemal A. Cancer statistics, 2013. CA Cancer J Clin. 2013;63:11–30. | |
Newman DJ. Natural products as leads to potential drugs: an old process or the new hope for drug discovery? J Med Chem. 2008;51:2589–2599. | |
Alitheen NB, Manaf AA, Yeap SK, Shuhaimi M, Nordin L, Mashitoh AR. Immunomodulatory effects of damnacanthal isolated from roots of Morinda elliptica. Pharm Biol. 2010;48:446–452. | |
Nualsanit T, Rojanapanthu P, Gritsanapan W, Lee SH, Lawson D, Baek SJ. Damnacanthal, a noni component, exhibits antitumorigenic activity in human colorectal cancer cells. J Nutr Biochem. 2012;23:915–923. | |
Aziz MY, Omar AR, Subramani T, et al. Damnacanthal is a potent inducer of apoptosis with anticancer activity by stimulating p53 and p21 genes in MCF-7 breast cancer cells. Oncol Lett. 2014;7:1479–1484. | |
Fuzellier M, Mortier F, Girard T, Payen J. [Study of antibiotic properties of anthraquinones using chromatographic microplates (author’s transl)]. Ann Pharm Fr. 1981;39(4):313–318. French. | |
Kuo YC, Meng HC, Tsai WJ. Regulation of cell proliferation, inflammatory cytokine production and calcium mobilization in primary human T lymphocytes by emodin from Polygonum hypoleucum Ohwi. Inflamm Res. 2001;50:73–82. | |
Cha TL, Qiu L, Chen CT, Wen Y, Hung MC. Emodin down-regulates androgen receptor and inhibits prostate cancer cell growth. Cancer Res. 2005;65:2287–2295. | |
Li JW, Vederas JC. Drug discovery and natural products: end of an era or an endless frontier? Science. 2009;325:161–165. | |
Harding KE, May LM, Dick KF. Selective oxidation of allylic alcohols with chromic acid. J Org Chem. 1975;40:1664–1665. | |
Abu N, Akhtar MN, Ho WY, Yeap SK, Alitheen NB. 3-Bromo-1-hydroxy-9,10-anthraquinone (BHAQ) inhibits growth and migration of the human breast cancer cell lines MCF-7 and MDA-MB231. Molecules. 2013;18:10367–10377. | |
Akhtar MN, Zareen S, Yeap SK, et al. Total synthesis, cytotoxic effects of damnacanthal, nordamnacanthal and related anthraquinone analogues. Molecules. 2013;18:10042–10055. | |
Vandesompele J, De Preter K, Pattyn F, et al. Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol. 2002;3:RESEARCH0034. | |
Jin GZ, Song GY, Zheng XG, Kim Y, Sok DE, Ahn BZ. 2-(1-Oxyalkyl)-1,4-dioxy-9,10-anthraquinones: synthesis and evaluation of antitumor activity. Arch Pharm Res. 1998;21:198–206. | |
Hsin LW, Wang HP, Kao PH, et al. Synthesis, DNA binding, and cytotoxicity of 1,4-bis(2-amino-ethylamino)anthraquinone-amino acid conjugates. Bioorg Med Chem. 2008;16:1006–1014. | |
Preobrazhenskaya MN, Shchekotikhin AE, Shtil AA, Huang H. Antitumor anthraquinone analogues for multidrug resistant tumor cells. J Med Sci. 2006;26:1–4. | |
Iyer M, Tseng YJ, Senese CL, Liu J, Hopfinger AJ. Prediction and mechanistic interpretation of human oral drug absorption using MI-QSAR analysis. Mol Pharm. 2007;4:218–231. | |
Zhao YH, Le J, Abraham MH, et al. Evaluation of human intestinal absorption data and subsequent derivation of a quantitative structure-activity relationship (QSAR) with the Abraham descriptors. J Pharm Sci. 2001;90:749–784. | |
Shah SP, Shah MD, Agrawal YK. Self-micro emulsifying drug delivery system: a novel approach for enhancement of oral bioavailability of poorly soluable drugs. American Journal of PharmTech Research. 2012;2:193–215. | |
Willette RE. Analgesic agents. In: Block JH, Beale JM Jr, editors. Wilson and Gisvold’s Textbook of Organic Medicinal and Pharmaceutical Chemistry. 12th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2010:687–708. | |
Lown JW, editor. Anthracycline and Anthracenedione–Based Anticancer Agents. Amsterdam: Elsevier Science Publishers; 1988. | |
Pors K, Paniwnyk Z, Teesdale-Spittle P, et al. Alchemix: a novel alkylating anthraquinone with potent activity against anthracycline- and cisplatin-resistant ovarian cancer. Mol Cancer Ther. 2003;2:607–610. | |
Bondy GS, Armstrong CL, Dawson BA, HÉroux-Metcalf C, Neville GA, Rogers CG. Toxicity of structurally related anthraquinones and anthrones to mammalian cell in vitro. Toxicol In Vitro. 1994;8:329–335. | |
Ghate NB, Chaudhuri D, Sarkar R, et al. An antioxidant extract of tropical lichen, Parmotrema reticulatum, induces cell cycle arrest and apoptosis in breast carcinoma cell line MCF-7. PLoS One. 2013;8:e82293. | |
Fu Z, Malureanu L, Huang J, et al. Plk1-dependent phosphorylation of FoxM1 regulates a transcriptional programme required for mitotic progression. Nat Cell Biol. 2008;10:1076–1082. | |
Su-Hwa Kim, Sanghoon Lee, Stephen R. Piccolo, et al. Menthol induces cell-cycle arrest in PC-3 cells by down-regulating G2/M genes, including polo-like kinase 1. Biochemical and Biophysical Research Communications. 2012;422(3):436–441. | |
Liu X and Erikson RL (2003). Polo-like kinase 1 in the life and death of cancer cells. Cel l Cycle. 2(5):424–425. | |
Friedmann C. Structure-activity relationships of anthraquinones in some pathological conditions. Pharmacology. 1980;20 Suppl 1:113–122. | |
Wu S, Liu B, Zhang Q, et al. Dihydromyricetin reduced Bcl-2 expression via p53 in human hepatoma HepG2 cells. PLoS One. 2013;8:e76886. | |
Chiang JH, Yang JS, Ma CY, et al. Danthron, an anthraquinone derivative, induces DNA damage and caspase cascades-mediated apoptosis in SNU-1 human gastric cancer cells through mitochondrial permeability transition pores and Bax-triggered pathways. Chem Res Toxicol. 2011;24:20–29. | |
Ohashi K, Sampei K, Nakagawa M, et al. Damnacanthal, an effective inhibitor of LIM-kinase, inhibits cell migration and invasion. Mol Biol Cell. 2014;25:828–840. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.