Back to Journals » International Journal of Nanomedicine » Volume 13
Superparamagnetic iron oxide nanoparticles modified with polyethylenimine and galactose for siRNA targeted delivery in hepatocellular carcinoma therapy
Authors Yang Z, Duan J, Wang J, Liu Q, Shang R, Yang X, Lu P, Xia C, Wang L, Dou K
Received 30 October 2017
Accepted for publication 24 January 2018
Published 26 March 2018 Volume 2018:13 Pages 1851—1865
DOI https://doi.org/10.2147/IJN.S155537
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Linlin Sun
Zhen Yang,1,* Juanli Duan,1,* Jianlin Wang,1,* Qi Liu,1 Runze Shang,1 Xisheng Yang,1 Peng Lu,2 Congcong Xia,1 Lin Wang,1 Kefeng Dou1
1Department of Hepatobiliary Surgery, Xijing Hospital, Fourth Military Medical University, Xi’an, People’s Republic of China; 2Department of Hepatobiliary Surgery, Hainan Branch of Chinese PLA General Hospital, Sanya, People’s Republic of China
*These authors contributed equally to this work
Introduction: A safe and effective in vivo siRNA delivery system is a prerequisite for liver tumor treatment based on siRNA cancer therapeutics. Nanoparticles based on superparamagnetic iron oxide (SPIO) provide a promising delivery system. In this study, we aimed to explore a novel nanoparticle, which is composed of SPIO.
Materials and methods: The particles have a core of iron oxide that is modified by galactose (Gal) and polyethylenimine (PEI), which act as shells, providing targeted delivery of therapeutic siRNA to the liver cancer. Gal-PEI-SPIO nanoparticles were synthesized, and the characteristics of the Gal-PEI-SPIO encapsulated with siRNA were analyzed.
Results: The particles remained nanoparticles and displayed negligible cytotoxicity when loaded with siRNA. In a serum stability assay, the Gal-PEI-SPIO nanoparticles could shield the siRNA from serum degradation and prolong the half-life of the siRNA in the system. Simultaneously, we found that the mixture could be efficiently taken up by Hepa1–6 cells in a flow cytometry assay. To study the anticancer effect, quantitative polymerase chain reaction and Western blotting were used to validate the silencing efficacy of the complexes in vitro. Subsequently, the nanoparticle mixtures were administered intravenously to tumor-bearing mice to explore the tissue distribution and the effect of the siRNA against cancer. We found that the nanoparticles could provide targeted siRNA delivery, accumulate easily in orthotopic tumors, enhance siRNA accumulation in the tumor tissues for 24 h and protect the siRNA from serum nuclease degradation in comparison with the control group. After these study procedures, the mice were sacrificed, and the tumors were removed to compare the tumor size and analyze the therapeutic effect on tumor growth. The tumor volume and the liver/body weight ratio were significantly reduced in the si-c-Met therapy groups. Additionally, the mRNA levels were also lower than those observed in the controls.
Conclusion: Based on these results, we concluded that Gal-PEI-SPIO represents a promising and efficient platform for siRNA delivery in tumor therapy.
Keywords: Gal-PEI-SPIO, hepatocellular carcinoma, c-Met, nanoparticle, siRNA
Introduction
Hepatocellular carcinoma (HCC), which is the fifth most common cancer worldwide, has a high propensity toward recurrence and is a malignant tumor.1–3 More than half of these tumors occur in China.4 Because most patients have already progressed to the terminal period when they are diagnosed, they lose the opportunity for resection or transplantation. Conventional therapies, such as transcatheter arterial chemoembolization (TACE) and radiofrequency, have not had satisfying results5,6 and concomitantly induced severe adverse effects due to non-specific cytotoxicity. Therefore, the need to identify a novel treatment approach is urgent. Gene therapy, such as RNA interference (RNAi), is a promising method for targeting specific proteins7 without severe adverse effects. RNAi, which silences genes post-transcriptionally and regulates gene expression through the degradation of mRNA with a complementary sequence, has provided a promising strategy for the treatment of most cancers.8–10
c-Met is highly overexpressed in various types of human carcinomas, including papillary renal cancer and gastric and small cell lung cancer, and has been associated and connected with a “metastatic phenotype” and a poor prognosis.11,12 c-Met is the receptor for the hepatocyte growth factor (HGF) and plays important roles in the proliferation, motility, differentiation and angiogenesis of HCC.13 Consequently, c-Met has been regarded as a promising molecular target for gene therapy in HCC.14 siRNAs that target c-Met have been shown to be effective in terms of reducing c-Met expression and inhibiting tumor growth. Such siRNAs have been developed rapidly as promising candidates for cancer treatment, particularly with the assistance of a delivery system. However, the efficacy of delivery is not satisfying.
With the development of nanotechnology, nanomaterials have offered a novel and plausible strategy for siRNA delivery. Superparamagnetic iron oxide (SPIO) nanoparticles, which are nanoscale magnetic materials composed primarily of an iron-based oxide, have shown great promise in biomedical applications due to their unique magnetic properties, biocompatibility and easily modified surface for bioagents.15,16 Galactose (Gal)-modified magnetic nanoparticles can be discerned specifically by the asialoglycoprotein receptor expressed on the surface of HCC cells and will be specifically recognized by HCC cells.17 Paolini et al18 reported that nanoparticles modified by Gal could be taken up by hepatocytes. In short, due to the unique nature of the drug, the Gal-modified magnetic nanoparticles can be selectively positioned in the lesion to improve the efficacy of drugs. Polyethylenimine (PEI), which combines with the magnetic nanoparticles and binds siRNA through positive and negative interactions, is a synthetic water-soluble polymer with a strong positive charge. PEI complexes can be easily and efficiently taken up by cells via endocytosis through the cell membrane. SPIOs modified by PEI and Gal can protect the siRNA from degradation and mediate cellular uptake via osmotic rupture (“proton sponge effect”),19 which provides an intracellular escape mechanism for siRNA particles without the support of endosome-disruptive agents for lysosome escape or other mechanisms.20
In this study, we have modified PEI-SPIO with the carrier Gal, which has a specific receptor on HCC cells, to effectively target the delivery of c-Met siRNA to tumors in a rat orthotopic model. We hypothesize that Gal-PEI-SPIO with siRNA will target the tumor and deliver the siRNA intracellularly to tumor cells to inhibit tumor growth. We tested this hypothesis with the data presented subsequently.
Materials and methods
siRNA and cell line
All siRNA duplexes targeting c-Met (sense sequence: 5′-CCACGTGAACGCTACTTAT-3′), which were modified with 2′-ome, and the negative control siRNA (NCsiRNA: siN05815122147) were purchased from RiboBio Co. (Guangzhou, China). Fluorescent-labeled Cy5-NC siRNA was also obtained from RiboBio Co. and was synthesized via the modification of the 3′-end of the sense strand with fluorescein. Hepa1–6 cell line was purchased from American Type Culture Collection (ATCC, Manassas, VA, USA).
Synthesis and preparation of the nanocomposite
Gal-PEI-SPIO nanoparticles were synthesized using the following process. Carboxylate-capped Fe3O4 was initially synthesized via the modified oxidative coprecipitation method. Briefly, the mixture of sodium acetate trihydrate and sodium citrate and the FeCl3·6H2O were dissolved in different volumes of ethylene glycol. Subsequently, the 2 solutions were mixed, and after stirring for 30 min, the mixture was reacted at 210°C for 8 h. After cooling to room temperature, the complexes of Fe3O4-COOH were washed with ethanol and dried at 60°C.21 Subsequently, PEI was further attached to the surface of the Fe3O4-COOH via electrostatic adsorption. Briefly, to 50 mL of a 0.4 mg/mL Fe3O4-COOH aqueous solution, we added NH3 and 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide (EDC) and the mixture was left to react for 2 h. Excessed PEI was then added to the solution, and the reaction was allowed to proceed for 6 h with stirring. Finally, Gal-PEG-NH2 was added to the mixed solution and left to react for 8 h. The resulting Gal-PEI-SPIOs were purified by removing free PEI and Gal using a magnet to precipitate the complexes and then redissolved at a higher concentration.
Characterization of Gal-PEI-SPIO
The particle diameters and zeta potentials of SPIOs (Fe3O4) and Gal-PEI-SPIOs in aqueous solution were measured via dynamic light scattering (DLS) using a Beckman Coulter Delsa™ Nano Series (Brea, CA, USA). The morphology of the nanoparticles was further measured using a scanning electron microscope (SEM). The Gal-PEI-SPIOs were prepared for SEM imaging by placing a drop of the nanoparticle suspension dissolved in ethanol onto a copper sheet and drying at ambient temperature. Then, the samples were visualized using an SEM.
Agarose gel electrophoresis assay
To investigate the combined efficacy of the nanoparticles with siRNA, Gal-PEI-SPIOs complexed with siRNAs at various Fe:siRNA weight ratios were incubated for 30 min at room temperature to form the Gal-PEI-SPIO/siRNA complexes. After incubation, the complexes were analyzed via 3% agarose gel electrophoresis and visualized via ethidium bromide staining with tris-acetate (TAE) running buffer at 120 V for 15 min. The siRNA bands were visualized with the Imager System (Bio-Rad Laboratories Inc., Hercules, CA, USA).
Heparin decomplexation assay
To achieve the best heparin concentration, siRNA was mixed with Gal-PEI-SPIOs at an Fe:siRNA weight ratio of 4 for 30 min at room temperature. Varying amounts of heparin were added, and the complexes were then incubated for 30 min at 37°C. The complexes were subsequently centrifuged at 12,000 rpm for 15 min at 4°C. The supernatants were confirmed by 3% agarose gel electrophoresis, and the released siRNA was visualized via ethidium bromide staining, as described earlier.
siRNA serum stability assay
Serum stability assays were used to evaluate the ability of Gal-PEI-SPIO to protect the siRNA from serum degradation. To confirm the siRNA’s serum stability, the siRNA was mixed with Gal-PEI-SPIO at an Fe:siRNA weight ratio of 4. Subsequently, after adding an equal volume fetal bovine serum (FBS), the complexes reached a concentration of 50% and were incubated for various time periods (0 h, 3 h, 6 h, 9 h, 12 h, 24 h, and 48 h) at 37°C. The final complexes were incubated with heparin (heparin:siRNA weight ratio of 80) at room temperature for 30 min. The released siRNA fraction was then separated at 12,000 rpm and visualized as described earlier.
Cell viability assay
To assess the cytotoxicity of the complexions at various concentrations, a cell viability assay was performed. Hepa1–6 cells were seeded into 96-well plates and grown for 24 h. The cells were then exposed to various concentrations of Gal-PEI-SPIO or Gal-PEI-SPIO/siRNA for 6 h. The cytotoxicity of the nanoparticles was determined using a cell counting kit (Beyotime, Shanghai, China), following the manufacturer’s instructions.
Assessment of the efficiency of cellular uptake via flow cytometry in vitro
The cellular uptake efficiency of complexes of Gal-PEI-SPIO with siRNA was evaluated via flow cytometry. Hepa1–6 cells were seeded into 6-well plates at a density of 2×105 cells/well and grown overnight. After the culture medium was replaced with a serum-free medium solution, medium containing Gal-PEI-SPIO/Cy5-siRNA or Gal-PEI-SPIO/siRNA as a control group was added to the wells, followed by incubation for 6 h at 37°C. Subsequently, the transfection medium was replaced with serum-containing medium, and the cells were incubated at 37°C for 24 h. Then, the cells were trypsinized into a single-cell suspension with 0.25% trypsin, harvested via centrifugation, washed 3 times with cold PBS and resuspended in 500 μL of PBS. The resulting cell suspensions were analyzed by flow cytometry.
In vitro silencing efficacy of si-c-Met
Hepa1–6 cells were seeded in a 6-well plate and incubated overnight at 37°C. Then, the cells were transfected with Gal-PEI-SPIO with the si-c-Met and si-NC complexes, as described earlier. Subsequently, total RNA was obtained from the cells using RNAiso Plus (Takara Biotechnology (Dalian) Co. Ltd, Dalian, China) according to the manufacturer’s instructions. The RNA was reverse transcribed into cDNA with the PrimeScript™ RT Reagent Kit (Takara Biotechnology (Dalian) Co. Ltd). To assess the silencing efficacy of si-c-Met, the c-Met mRNA was quantified using quantitative polymerase chain reaction (PCR) with the following primers: 5′ ATCTCGGAGCCACAAACTACA 3′ (forward) and 5′ CAGTCCCGACAAGGTAAACAAT 3′ (reverse). c-Met mRNA levels were normalized according to β-actin levels using the following primers: 5′ CATCCGTAAAGACCTCTATGCCAAC 3′ (forward) and 5′ ATGGAGCCACCGATCCACA 3′ (reverse). The PCR reaction was performed according to the manufacturer’s instructions.
To further evaluate the silencing efficacy, we performed a Western blotting analysis. Hepa1–6 cells that were transfected for 48 h as described earlier were washed twice with cold PBS and lysed in RIPA lysis buffer (Beyotime, Shanghai, China) containing fresh protease and phosphatase inhibitor cocktails (Sigma-Aldrich Co., St Louis, MO, USA). The concentration was measured using the Bio-Rad assay system (Bio-Rad Laboratories Inc.). Equal amounts of protein sample were separated via 8% sodium dodecyl sulfate-polyacrylamide gel (SDS-PAGE) and transferred to polyvinylidene difluoride membranes (EMD Millipore, Billerica, MA, USA). Subsequently, the membranes were incubated with a mouse anti-β-actin antibody (1:2,000; Cell Signaling Technology, Danvers, MA, USA) and mouse anti-c-Met antibody (1:1,000; Cell Signaling Technology) overnight at 4°C. The membranes were incubated with a horseradish peroxidase-conjugated secondary antibody (1:5,000; Abcam, Cambridge, UK). Finally, the membranes were developed using enhanced chemiluminescence detection reagents (Pierce Biotechnology Inc., Rockford, IL, USA) and visualized using a molecular imager system (Bio-Rad Laboratories Inc.).
Proliferation inhibition assay in vitro
A proliferation inhibition assay was performed in Hepa1–6 cells using the thymidine analog 5-ethynyl-2′-deoxyuridine (EdU), which is incorporated into DNA during active DNA synthesis (during the S phase of the cell cycle).22 Thus, the cell proliferation inhibition assay was analyzed with the Cell-Light™ EdU Apollo® 488 In Vitro Imaging Kit (RiboBio Co.) according to the manufacturer’s instructions. In brief, the cells were transfected for 48 h, and the medium was replaced with medium supplemented with optimized EdU at a concentration of 50 μM for 2 h. Subsequently, the cells were fixed with 4% paraformaldehyde, and a glycine solution was added to neutralize the excess paraformaldehyde. The cells were then permeabilized with 0.5% TritonX-100 in PBS and incubated with 1×Apollo reaction cocktail for 30 min in the dark. Finally, the cells were incubated with 1×Hoechst 33342 reaction buffer for 30 min at room temperature and simultaneously protected from light. The cells were then visualized via fluorescence microscopy (Olympus Corporation, Tokyo, Japan).
In situ hepatic tumor model in C57BL/6 mice
All animal experimental protocols were approved by the ethics committee of Xijing Hospital (the Fourth Military Medical University). All animal experiments were performed following a guideline from the Animal Experiment Administration Committee of the Fourth Military Medical University and conducted in accordance with ARRIVE guidelines.39 Adult male C57BL/6 mice were purchased from the animal center of the Fourth Military Medical University and aged ~8 weeks. The orthotopic hepatic tumor models were established via direct intrahepatic injection of the luciferase-Hepa1–6 (luc-Hepa1–6) cell line. The mice were anesthetized with pentobarbital, and a midline incision of the abdomen below the sternum was made carefully to expose the liver. The left lobe was lightly extruded to expose the lobe fully. The luc-Hepa1–6 cells were mixed with Matrigel (Discovery Labware Inc., Bedford, MA, USA) to form complexes that contained 2×106 cells in 50 μL. The cell complexes were slowly injected into the left hepatic lobe using a 30-G needle until we observed a whitish color on the hepatic surface at the point of injection. Then, the injection was blocked with gel sponge to avoid bleeding and backflow of the cells. Subsequently, the abdomen was closed carefully with a silk suture, and the luc-Hepa1–6 in situ liver tumor model was created. Three days post-surgery, the tumor-bearing mice were randomly divided into 3 groups (n=6) and treated with Gal-PEI-SPIO/si-NC (0.5 mg siRNA/kg) or si-c-Met, which were administered via the tail vein every 3 days. The mice in one of the c-Met groups had a permanent magnet (10 mm diameter and 8 mm thick, with a central hole 9 mm in diameter and a surface magnetic field of 200 mT) placed on the skin over the liver tumor site for 2 h post-injection. The groups of mice were anesthetized with pentobarbital during the magnetic exposure. The entire therapy was repeated 4 times during a 16-day treatment period.
In vivo distribution of nanoparticles encapsulated with siRNA
To assess the distribution of the siRNA in vivo, Cy5-siRNA (0.5 mg siRNA/kg) was incubated with Gal-PEI-SPIO as described earlier. The mice bearing orthotopic tumors were then injected intravenously with the complexes. At different time points (2 h, 8 h, and 24 h), the mice were anesthetized via the inhalation of pure oxygen mixed with 2% isoflurane throughout the imaging process. Subsequently, the mice were placed into the dark box of the IVIS Lumina II system (PerkinElmer, Waltham, MA, USA), and the distribution in the liver and the tumor was observed under the laser (excitation 640 nm). The other parts of the mouse were covered with black tape to avoid interference with other parts of the mouse body.
Live imaging
The tumor-bearing mice were administered Gal-PEI-SPIO/si-NC (0.5 mg siRNA/kg) or si-c-Met every 3 days. To validate the therapeutic effect of Gal-PEI-SPIO/si-c-Met, which was evaluated based on the fluorescence intensity of the tumor in the orthotopic tumor-bearing mice; the mice were injected intraperitoneally with a corresponding volume of D-luciferin on day 18. Fifteen minutes after the injection, the mice were anesthetized via the inhalation of pure oxygen mixed with 2% isoflurane, as described earlier. The mice were observed with the IVIS Lumina II imaging system (PerkinElmer) for fluorescence imaging of the orthotopic liver tumor. After imaging, the rats were anesthetized with pentobarbital and perfused with saline from the apex cordis. After perfusion, the tumors were harvested and measured. The tumor and the major organs (kidney, spleen, and liver) were immediately fixed in 10% formalin and embedded in paraffin. Subsequently, 4 μm tissue sections were cut and stained with hematoxylin-eosin (H&E) and observed under a 200× magnification. Prussian blue staining was performed simultaneously to validate the distribution of Gal-PEI-SPIO with the siRNA under a 400× magnification in the tumor and the normal organ.23
Immunohistochemistry
To explore tissue cell proliferation, murine liver cancer tissues were embedded in paraffin and cut into 4 μm thick sections. Subsequently, the tumor tissues were immunostained with an anti-Ki67 (Abcam) antibody, as described in the manual. The positive cells in 10 randomly selected fields were observed under a 400× magnification. Simultaneously, to explore the inhibition of c-Met in the tissue further, we used a c-Met antibody (Abcam) to stain the liver cancer tissue embedded in paraffin. The positively stained cells from 4 tumor tissues per group were counted in 10 randomly selected fields under a 200× magnification.
Murine tumor quantitation of c-Met mRNA
The mice bearing orthotopic tumors were sacrificed on the 18th day, and the tumor tissues were then resected and stored at −80°C. The total RNA was obtained from the tumor tissue using the RNAiso Plus according to the manufacturer’s instructions (n=6 per group). The quantitation of tumor tissue c-Met mRNA was executed as described earlier.
Statistics
All results were expressed as mean ± SD. The P-value was analyzed using Student’s t-test and one-way analysis of variance (ANOVA). A value of P<0.05 was considered to be significant.
Results
Characterization of Gal-PEI-SPIO
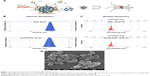
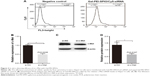
The surface structure of the synthesized nanoparticles was modified by Gal and PEI, as shown in Figure 1A. The particle diameters and zeta potentials of the SPIO (Fe3O4) solution were 95.0±1.2 nm and −25.98±0.3 mV, respectively, while those of the Gal-PEI-SPIO were 98.2±2.3 nm and +28.51±0.4 mV, respectively (Figure 1B and C). These results of increasing the effective diameter of SPIO illustrated that PEI and Gal were associated with the surface of the nanocomplexes. Moreover, the particle diameter of Gal-PEI-SPIO still classifies the particles as nanoscale particles. Simultaneously, this finding was shown by SEM, which demonstrated that Gal-PEI-SPIO was ~108 nm (Figure 1D) and showed a uniform particle size. In an agarose gel electrophoresis assay (Figure 2A), Gal-PEI-SPIO with siRNA formed a mixture at an Fe:siRNA weight ratio of over 4 and showed absent bands in an agarose gel, which indicated that the siRNA duplexes were tightly bound to Gal-PEI-SPIO. To achieve a more suitable ratio, we explored the cytotoxicity of various concentrations of the particles using a cell viability assay. The results revealed that the cell viability at a ratio over 4 (Figure 2B and C) declined quickly, indicating cell death. Therefore, we selected an Fe: siRNA ratio of 4 as the final ratio for the following series of experiments.
Since the capacity of nanoparticles protecting cargo siRNA from nuclease degradation is crucial during in vivo siRNA delivery, we first confirmed the heparin displacement assays and found that the complex stabilities changed at a ratio of 80 (Figure 2D). Subsequently, we further assessed the complexes of Gal-PEI-SPIO encapsulated with siRNA during incubation with 50% FBS at 37°C. As shown in the image (Figure 2E), the naked siRNA was quickly and completely degraded within 3 h, while the complexes survived beyond 48 h. Notably, the mixtures of nanoparticles with siRNA protected the siRNA from nuclease degradation in comparison with siRNA alone and probably due to the successful encapsulation of the siRNA into Gal-PEI-SPIO nanoparticles.
In vitro silencing efficacy of siRNA with nanoparticles
The presence of more siRNA in the cytoplasm promotes a stronger silencing efficiency.24 It is critical to assess the cellular uptake efficiency of siRNA when complexes are formed via flow cytometry using Cy5-siRNA. As shown in Figure 3A, the flow cytometry assay illustrated that ~68.6% of the cells engulfed siRNA in comparison with ~8.69% in the control group.
Subsequently, to examine the RNAi efficiency of siRNA with Gal-PEI-SPIO nanoparticles in vitro, Hepa1–6 cells were used to conduct a transfection experiment. The RNAi efficiency of Gal-PEI-SPIO with siRNA complexes was analyzed by measuring the amount of c-Met protein and mRNA transcript using Western blotting and quantitative PCR, respectively. The results showed that Gal-PEI-SPIO with siRNA complexes significantly downregulated c-Met mRNA (Figure 3B) and protein expression (Figure 3C and D) in Hepa1–6 cells in comparison with Gal-PEI-SPIO/si-NC.
c-Met inhibited cell proliferation in vitro
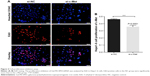
c-Met plays important roles in the proliferation, motility, differentiation and angiogenesis of HCC cells and inhibits HCC cell growth via the downregulation of c-Met, with an RNAi function.25 Therefore, we further explored the role of si-c-Met tumor suppressor activity, namely, the ability to inhibit cell proliferation. As shown in Figure 4A and B, the percentage of EdU-positive cells was significantly larger in the negative control (NC) group than in the c-Met group and illustrated that the Gal-PEI-SPIO encapsulated with si-c-Met inhibited cell growth.
In vivo distribution of nanoparticles encapsulated with siRNA
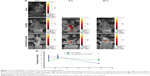
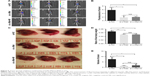
To further investigate the anticancer therapeutic capability of the mixture of Gal-PEI-SPIO encapsulated with siRNA in vivo, we first studied the biological activity of the nanoparticles with siRNA. To explore the tumor tissue distribution of Gal-PEI-SPIO/siRNA in mice, Cy5-siRNA, which was encapsulated by Gal-PEI-SPIO or without nanoloading, was injected into C57BL/6 tumor-bearing mice via the tail vein, and simultaneously the in situ fluorescence distributions was monitored via fluorescence imaging at various time periods. As shown in Figure 5A, the Cy5-siRNA rapidly accumulated in the liver and tumor within 8 h, and the fluorescence intensity was obviously higher than that observed with nanoencapsulated Cy5-siRNA. While Cy5-siRNA was rapidly eliminated from mice, the fluorescence intensity was observed and declined significantly in the animals at 24 h. However, the fluorescence intensity of the Cy5-siRNA-coated Gal-PEI-SPIO nanoparticles decreased gradually after 8 h and was still observed after 24 h, but was stronger than that of the control group (Figure 5B).
Tumor growth inhibition mediated by Gal-PEI-SPIO/siRNA in vivo
The tumor growth inhibition of the nanoparticles with siRNA was further illustrated in C57BL/6 mice, which were subjected to an orthotopic liver tumor. After 18 days, the tumor-bearing mice were examined by D-luciferin by using the fluorescence intensity to compare the effects of tumor inhibition (Figure 6A). As shown in Figure 6B, the mice treated with Gal-PEI-SPIO/si-NC exhibited stronger fluorescence intensity than the other 2 groups treated with Gal-PEI-SPIO/si-c-Met. However, si-c-Met and si-c-Met in combination with the magnet did not differ significantly from each other. To further elucidate the therapeutic effect of the nanoparticles with siRNA, the mice were sacrificed after fluorescence imaging, and the livers were removed. As shown in Figure 6C and E, the tumors treated with Gal-PEI-SPIO/si-c-Met displayed an obvious volume reduction, and the fluorescence intensity results were similar. Furthermore, the liver/body weight ratio was significantly decreased in comparison with the controls (Figure 6D). However, there was no significant difference between the remaining 2 groups, and the result was consistent with the fluorescence intensity.
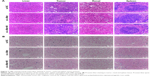
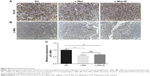
To investigate the effect of the mixture on mouse organs, we extracted the kidney, spleen tumor and liver organs and carried out H&E staining to observe morphological changes. Histological examination of the tumor, liver, spleen and kidney with staining (Figure 7A), in which images were taken at a 200× magnification, revealed no obvious pathological damage in the liver and kidney, while the spleen showed an accumulation of lymph nodes. Additionally, we simultaneously examined the distribution of Gal-PEI-SPIO with siRNA based on the evaluation of Prussian blue staining in tumor, renal and spleen tissue sections. As shown in Figure 7B, Gal-PEI-SPIO, which appeared as blue spots, accumulated in the tumor and the spleen but not in the kidney. This finding suggested that Gal-PEI-SPIO, which was modified by Gal and PEI, could selectively enter the tumor tissue through specific binding to the tumor, as well as by being engulfed by macrophages through non-specific phagocytosis into the spleen, which could be regarded as a kind of elimination of the nanoparticles. It was reported previously that iron nanoparticles would be degraded in the endosomes and lysosomes of macrophages.26 It is feasible to anticipate that Gal-PEI-SPIO nanoparticles in the liver and spleen can be degraded inside macrophages. No nanoparticles were observed in the renal tissue, which means that nanoparticles would not retain in the kidney without recognizing the specific receptor. Based on the immunohistochemistry assay, Ki-67-based quantification of cell proliferation in the tumor tissues showed that there are fewer Ki-67-positive cells in the c-Met-treated group than in the NC group (Figure 8A). Meanwhile, to further explore the inhibition of si-c-Met in mouse liver tumors (Figure 8B), we found that the expression of c-Met was significantly higher in the NC group than in the other 2 groups. These results suggested that c-Met packaged with Gal-PEI-SPIO nanoparticles could significantly inhibit HCC tumor growth in vivo. No differences were observed between the si-c-Met groups and the groups that received si-c-Met with the magnet.
To further study the inhibition of tumor growth that was observed in the tumor tissues upon treatment with c-Met, quantitative PCR was used in the tumor to assess the inhibition of tumor growth. We examined the c-Met mRNA levels in the tumor following treatment. The results showed that the expression of c-Met in the group treated with Gal-PEI-SPIO/si-c-Met was significantly reduced in comparison with the negative control Gal-PEI-SPIO/NC siRNA group (Figure 8C). Interestingly, there was no obvious difference between the si-c-Met group and the group that received si-c-Met in combination with the magnet.
Discussion
In recent years, RNAi-based therapies that mediate the silencing of gene expression have given rise to productive research in the context of numerous cancers.27–30 Although RNAi has strong potential for use in anticancer therapy, its application is limited due to its short half-life in vivo, its lack of target cell specificity and its poor transport across the cell membrane. Several gene or drug delivery carriers have been used for therapy for liver cancer,26,31,32 including liposomes and biodegradable polymers. The purpose of the present study is to investigate a novel and integrative gene delivery system for delivering siRNA into HCC tumors and inhibiting tumor growth. In this study, we developed a novel therapy nanosystem based on Gal- and PEI-coated SPIO nanoparticles, which could target siRNA delivery into tumors. Although SPIO nanoparticles are usually modified by PEI33 and polyamidoamine dendrimers34 and have been investigated for non-viral gene delivery, these carriers were not targeted to reach the tumor. To address this problem, we modified the SPIO with Gal, which can be recognized specifically by the asialoglycoprotein receptor on the surface of HCC cells. Therefore, we investigated the SPIOs that were modified with Gal and PEI for systemic siRNA delivery in liver cancer therapy. Based on the results obtained using a transmission electron microscope, nanoparticles modified with Gal and PEI remained nanoparticles and could be taken up by cells. In addition, the particle diameters and zeta potentials of the nanosolution were consistent with the SEM results. To investigate the ratio of nanoparticles to siRNA using agarose gel electrophoresis, Gal-PEI-SPIO with siRNA formed at an Fe:siRNA weight ratio of over 4 showed absent bands on agarose gels, which indicated that the siRNA duplexes were tightly bound to the Gal-PEI-SPIO nanoparticles. Moreover, we explored the cytotoxicity of various concentrations of the nanoparticles using a cell viability assay and discovered that the cell viability declined quickly at a ratio over 4, indicating cell death. This effect may be due to a strong positive charge or the toxicity of PEI, which leads to cell death. Alternatively, the cytotoxicity of the nanoparticles may be size-dependent. Hence, we selected an Fe:siRNA ratio of 4 as the final ratio for the following series of experiments.
c-Met is strongly expressed in liver cancer35,36 and has been regarded as a promising and potential molecular target for gene therapy in HCC. SPIO modified with PEI and Gal was easily encapsulated with siRNA, as visualized by agarose gel electrophoresis. Through a serum stability assay, Gal-PEI-SPIO could protect the siRNA from nuclease degradation and extend its half-life. In addition, nanoparticles of Gal-PEI-SPIO could encapsulate the siRNA and deliver the siRNA into HCC with high efficiency by flow cytometer. This finding indicates that Gal-PEI-SPIO can act as an effective siRNA delivery. Meanwhile, our study also demonstrated that Gal-PEI-SPIO/si-c-Met could reduce c-Met mRNA levels and protein expression in Hepa1–6 cells. In addition, we also tested the ability of si-c-Met to inhibit cell proliferation and found that Gal-PEI-SPIO encapsulated with si-c-Met significantly inhibited the proliferation of Hepa1–6 cells. Therefore, siRNA-based gene therapy is a promising approach for the treatment of cancer.10 Nevertheless, identifying a strategy to avoid off-target effects via efficient delivery to target cells remains an urgent problem.37 In this study, Gal-PEI-SPIO encapsulated with siRNA could attach to and accumulate in orthotopic tumors in C57BL/6 mice. In addition, the nanoparticles could also protect siRNA from nuclease degradation and extend the siRNA’s half-life in the tumor in comparison with the NC free siRNA. Based on these results, we demonstrated that nanoparticles could enhance siRNA accumulation in the tumor tissues and protect the siRNA from serum nuclease degradation, which is consistent with the serum stability results.
For liver cancer therapy, the siRNA retention period in the tumor plays an important role. Many factors influence the retention period of siRNA.38 For instance, some of the siRNA is transported into the secondary organs, and some remains in the circulation. We found that Prussian blue spots of Gal-PEI-SPIO accumulated in the tumor and the spleen but rarely accumulated in the kidney, which indicated that Gal-PEI-SPIO with siRNA would not harm the renal tissue and damage kidney function. Based on the fluorescence intensity, siRNA labeled with Cy5 could accumulate rapidly in the liver and tumor within 8 h and significantly declined in the animals after 24 h. In contrast, the siRNA with nanoparticles survived for a longer time than the control. Thus, combining the siRNA with nanoparticles protected the siRNA from degradation and transported it to the liver and the tumor, which is rich in blood flow. We further confirmed the therapeutic anticancer efficacy of the mixture of Gal-PEI-SPIO with siRNA in orthotopic tumors administered via intravenous injection in C57BL/6 mice. Tumor growth was obviously inhibited in the Gal-PEI-SPIO/si-c-Met and Gal-PEI-SPIO/si-c-Met plus magnet groups in comparison with the Gal-PEI-SPIO/NC siRNA group. To explore the growth inhibition of si-c-Met in tumor tissues, the immunohistochemistry assay showed significantly fewer Ki-67-positive cells in the NC group than in the other 2 groups that were treated with si-c-Met. Meanwhile, the expression levels of c-Met were significantly higher in the NC group than in the other 2 groups. These results indicated that Gal-PEI-SPIO/si-c-Met could inhibit HCC tumor growth in vivo. However, there was no significant difference in tumor growth inhibition between the Gal-PEI-SPIO/si-c-Met group and the Gal-PEI-SPIO/si-c-Met plus magnet group. This result was also validated by the fluorescence intensity of the tumors and the liver:body weight ratios. We suspected that the targeting effect of Gal may effectively deliver the mixture of Gal-PEI-SPIO with siRNA into the tumor, thereby playing an important role in inhibiting tumor growth.
Conclusion
In summary, our results demonstrated that SPIO nanoparticles modified with Gal and PEI can tightly bind siRNA, protect siRNA from nuclease degradation in the system, extend the siRNA half-life and transfect siRNA encapsulated with nanoparticles into tumor cells without exhibiting cellular toxicity in vitro. Furthermore, the Gal-PEI-SPIO encapsulated with siRNA could simultaneously accumulate siRNA, specifically target the siRNA to the target tissue, extend the siRNA’s time in the tumor and inhibit tumor growth. Therefore, our study provided a new targeting strategy for liver cancer treatment, and Gal-PEI-SPIO nanoparticles have great promise for tumor gene therapy.
Acknowledgments
This study was funded by the National Natural Science Foundation of China (Project number 81401940, 81671838 and 81370512) and the Outstanding Youth Science Foundation of China (Project number 81422009), as well as the Hainan Province Natural Science Foundation (Project number 817357) and the Sanya Medical Technology Innovation Project (2016YW13). The authors are also grateful to Dr Qing Zhou (Department of Pharmaceutical Analysis, School of Pharmacy, Fourth Military Medicine of University) for assistance with nanomaterial synthesis.
Disclosure
The authors report no conflicts of interest in this work.
References
Bruix J, Gores GJ, Mazzaferro V. Hepatocellular carcinoma: clinical frontiers and perspectives. Gut. 2014;63(5):844–855. | ||
Yang JD, Roberts LR. Hepatocellular carcinoma: A global view. Nat Rev Gastroenterol Hepatol. 2010;7(8):448–458. | ||
Venook AP, Papandreou C, Furuse J, de Guevara LL. The incidence and epidemiology of hepatocellular carcinoma: A global and regional perspective. Oncologist. 2010;15 Suppl 4:5–13. | ||
Guo Y, Wang J, Zhang L, et al. Theranostical nanosystem-mediated identification of an oncogene and highly effective therapy in hepatocellular carcinoma. Hepatology. 2016;63(4):1240–1255. | ||
Henry JC, Malhotra L, Khabiri H, et al. Best radiological response to trans-arterial chemoembolization for hepatocellular carcinoma does not imply better outcomes. HPB (Oxford). 2013;15(3):196–202. | ||
Avila MA, Berasain C, Sangro B, Prieto J. New therapies for hepatocellular carcinoma. Oncogene. 2006;25(27):3866–3884. | ||
Coburn GA, Cullen BR. siRNAs: A new wave of RNA-based therapeutics. J Antimicrob Chemother. 2003;51(4):753–756. | ||
Castanotto D, Rossi JJ. The promises and pitfalls of RNA-interference-based therapeutics. Nature. 2009;457(7228):426–433. | ||
Pecot CV, Calin GA, Coleman RL, Lopez-Berestein G, Sood AK. RNA interference in the clinic: Challenges and future directions. Nat Rev Cancer. 2011;11(1):59–67. | ||
Shu Y, Pi F, Sharma A, et al. Stable RNA nanoparticles as potential new generation drugs for cancer therapy. Adv Drug Deliv Rev. 2014;66:74–89. | ||
Ma PC, Maulik G, Christensen J, Salgia R. c-Met: Structure, functions and potential for therapeutic inhibition. Cancer Metastasis Rev. 2003;22(4):309–325. | ||
Kaposi-Novak P, Lee JS, Gòmez-Quiroz L, Coulouarn C, Factor VM, Thorgeirsson SS. Met-regulated expression signature defines a subset of human hepatocellular carcinomas with poor prognosis and aggressive phenotype. J Clin Invest. 2006;116(6):1582–1595. | ||
Furge KA, Zhang YW, Vande WG. Met receptor tyrosine kinase: Enhanced signaling through adapter proteins. Oncogene. 2000;19(49):5582–5589. | ||
Corso S, Comoglio PM, Giordano S. Cancer therapy: Can the challenge be MET? Trends Mol Med. 2005;11(6):284–292. | ||
Mok H, Zhang M. Superparamagnetic iron oxide nanoparticle-based delivery systems for biotherapeutics. Expert Opin Drug Deliv. 2013;10(1):73–87. | ||
Midoux P, Breuzard G, Gomez JP, Pichon C. Polymer-based gene delivery: A current review on the uptake and intracellular trafficking of polyplexes. Curr Gene Ther. 2008;8(5):335–352. | ||
Sato A, Takagi M, Shimamoto A, Kawakami S, Hashida M. Small interfering RNA delivery to the liver by intravenous administration of galactosylated cationic liposomes in mice. Biomaterials. 2007;28(7):1434–1442. | ||
Paolini M, Poul L, Berjaud C, et al. Nano-sized cytochrome P450 3A4 inhibitors to block hepatic metabolism of docetaxel. Int J Nanomedicine. 2017;12:5537–5556. | ||
Akinc A, Thomas M, Klibanov AM, Langer R. Exploring polyethylenimine-mediated DNA transfection and the proton sponge hypothesis. J Gene Med. 2005;7(5):657–663. | ||
ur Rehman Z, Hoekstra D, Zuhorn IS. Mechanism of polyplex- and lipoplex-mediated delivery of nucleic acids: Real-time visualization of transient membrane destabilization without endosomal lysis. ACS Nano. 2013;7(5):3767–3777. | ||
Bromberg L, Diao Y, Wu H, Speakman SA, Hatton TA. Chromium(III) terephthalate metal organic framework (MIL-101): HF-free synthesis, structure, polyoxometalate composites, and catalytic properties. Chem Mater. 2012;24(9):1664–1675. | ||
Okada M, Shi YB. Cell proliferation analysis during xenopus metamorphosis: Using 5-ethynyl-2-deoxyuridine (EdU) to stain proliferating intestinal cells. Cold Spring Harb Protoc. 2017;(9):696–700. | ||
Cole AJ, David AE, Wang J, Galb NCJ, Yang VC. Magnetic brain tumor targeting and biodistribution of long-circulating PEG-modified, cross-linked starch-coated iron oxide nanoparticles. Biomaterials. 2011;32(26):6291–6301. | ||
Liu X, Sun C, Yang X, Wang J. Polymeric-micelle-based nanomedicine for siRNA delivery. Part Part Syst Char. 2013;30(3):211–228. | ||
Xie B, Xing R, Chen P, et al. Down-regulation of c-Met expression inhibits human HCC cells growth and invasion by RNA interference. J Surg Res. 2010;162(2):231–238. | ||
Shao D, Li J, Zheng X, et al. Janus “nano-bullets” for magnetic targeting liver cancer chemotherapy. Biomaterials. 2016;100:118–133. | ||
Amjad MW, Amin MCIM, Katas H, Butt AM, Kesharwani P, Iyer AK. In vivo antitumor activity of folate-conjugated cholic acid-polyethylenimine micelles for the codelivery of doxorubicin and siRNA to colorectal adenocarcinomas. Mol Pharm. 2015;12(12):4247–4258. | ||
Arami S, Mahdavi M, Rashidi MR, Fathi M, Hejazi M, Samadi N. Novel polyacrylate-based cationic nanoparticles for survivin siRNA delivery combined with mitoxantrone for treatment of breast cancer. Biologicals. 2016;44(6):487–496. | ||
Lipka J, Semmler-Behnke M, Wenk A, Burkhardt J, Aigner A, Kreyling W. Biokinetic studies of non-complexed siRNA versus nano-sized PEI F25-LMW/siRNA polyplexes following intratracheal instillation into mice. Int J Pharm. 2016;500(1–2):227–235. | ||
Ji M, Li P, Sheng N, et al. Sialic acid-targeted nanovectors with phenylboronic acid-grafted polyethylenimine robustly enhance siRNA-based cancer therapy. ACS Appl Mater Interfaces. 2016;8(15):9565–9576. | ||
Chen Y, Zhu X, Zhang X, Liu B, Huang L. Nanoparticles modified with tumor-targeting scFv deliver siRNA and miRNA for cancer therapy. Mol Ther. 2010;18(9):1650–1656. | ||
Liu J, Chiang T, Liu C, et al. Delivery of siRNA using CXCR4-targeted nanoparticles modulates tumor microenvironment and achieves a potent antitumor response in liver cancer. Mol Ther. 2015;23(11):1772–1782. | ||
Chertok B, David AE, Yang VC. Polyethyleneimine-modified iron oxide nanoparticles for brain tumor drug delivery using magnetic targeting and intra-carotid administration. Biomaterials. 2010;31(24):6317–6324. | ||
Pan B, Cui D, Sheng Y, et al. Dendrimer-modified magnetic nanoparticles enhance efficiency of gene delivery system. Cancer Res. 2007;67(17):8156–8163. | ||
Suzuki K, Hayashi N, Yamada Y, et al. Expression of the c-met protooncogene in human hepatocellular carcinoma. Hepatology. 1994;20(5):1231–1236. | ||
Daveau M, Scotte M, François A, et al. Hepatocyte growth factor, transforming growth factor alpha, and their receptors as combined markers of prognosis in hepatocellular carcinoma. Mol Carcinog. 2003;36(3):130–141. | ||
Apparailly F, Jorgensen C. siRNA-based therapeutic approaches for rheumatic diseases. Nat Rev Rheumatol. 2013;9(1):56–62. | ||
Opitz L, Salinas-Riester G, Grade M, et al. Impact of RNA degradation on gene expression profiling. BMC Med Genomics. 2010;3:36. | ||
Kilkenny C, Browne WJ, Cuthill IC, Emerson M, Altman DG. Improving bioscience research reporting: the ARRIVE guidelines for reporting animal research. PLoS Biol. 2010;8(6):e1000412. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.