Back to Journals » Clinical Ophthalmology » Volume 15
Stimulating Tear Production: Spotlight on Neurostimulation
Authors Yu MD, Park JK, Kossler AL
Received 22 June 2021
Accepted for publication 5 October 2021
Published 20 October 2021 Volume 2021:15 Pages 4219—4226
DOI https://doi.org/10.2147/OPTH.S284622
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Michael D Yu,1 Ji Kwan Park,2 Andrea L Kossler1
1Oculoplastic Surgery and Orbital Oncology Service, Byers Eye Institute, Stanford University, Palo Alto, CA, USA; 2Oculofacial Plastic and Orbital Surgery, Indianapolis, IN, USA
Correspondence: Andrea L Kossler
Oculoplastic Surgery and Orbital Oncology Service, Byers Eye Institute, Stanford University, 2452 Watson Ct, Palo Alto, CA 94303, USA
Tel +1 650-724-0105
Email [email protected]
Abstract: Dry eye disease (DED) affects up to one-third of the global population. Traditional therapies, including topical lubricants, have been employed with variable success in the treatment of DED. Recently, neurostimulation of the lacrimal functional unit (LFU) has emerged as a promising alternative therapy for DED. In this review, we describe the neuroanatomical and pathophysiological considerations of DED and the LFU that make neurostimulation a viable therapeutic alternative. We further detail the various neurostimulatory approaches taken thus far—from implanted stimulators to external devices to chemical neurostimulation. Existing studies reveal the strengths of the neurostimulatory approach in increasing tear volume and improving dry eye symptoms, but further studies are needed to elucidate its true potential in treatment of DED.
Keywords: oculoplastics, ocular surface, cornea, lacrimal gland, dry eye disease, tears, neurostimulation
Introduction
Dry eye disease (DED) is a common disorder of the ocular surface that affects up to one-third of the global population.1 Symptoms of DED can vary in severity, ranging from transient discomfort to disabling pain and visual impairment, with resultant impact on vision-related quality of life and workplace productivity.1 In the United States, societal costs associated with dry eye amount to approximately $55 billion annually.2 Correspondingly, DED is among the leading causes of patient visits to ophthalmologists and optometrists, resulting in an estimated $3.8 billion in direct health-care expenditures annually in the United States.2
Therapeutic strategies are guided by consideration of cause and severity of DED and have traditionally included topical lubricants like drops, gels, and ointments for symptom control. The recent identification of ocular-surface inflammation as a key component in the pathogenesis of DED has also led to the use of non-glucocorticoid immunomodulatory agents, like topical cyclosporine and lifitegrast, with demonstrated efficacy in randomized clinical trials.3 However, DED for many patients still remains refractory to conventional treatments,4 necessitating alternative therapeutic approaches.
The concept of neurostimulation for DED was introduced in 2009, when Kossler et al found direct lacrimal nerve stimulation to significantly increase natural aqueous tear production in pre-clinical studies.5 In general, neurostimulation is the application of electromagnetic energy or chemical stimulus to specific anatomic targets that in turn induce modulation of the corresponding neural circuitry. Present-day neurostimulation devices treat a vast range of neurological and psychiatric disorders, from motor cortex stimulation for central pain syndromes, to vagus nerve stimulation for treatment-resistant epilepsy.6,7 These present-day treatments represent the culmination of a decades of serendipitous discoveries and technological advances, beginning with studies in the 1960s on spinal cord stimulation for chronic pathological pain.8
Neurostimulation quickly emerged as a promising alternative therapeutic modality for DED. By 2017, TrueTear®, an intranasal neurostimulation device that delivered small electrical currents to sensory neurons of the nasal cavity, had already gained US Food and Drug Administration (FDA) approval for treatment of DED.9 Since then, interest in this realm and innovation in medical device technology has continued to accelerate the evolution of neurostimulation for DED. Additional neurostimulatory targets have been identified, and a number of devices have transitioned from theory to commercially available products.
This review will describe the pathophysiology of DED, highlighting the neural control of the tear film which makes DED amenable to neurostimulation (The Lacrimal Functional Unit and Nasolacrimal Reflex: A Physiologic Perspective), along with the evolution of neurostimulation approaches for treatment of DED from intranasal stimulation (First Forays Into Neurostimulation For Dry Eye Disease and Intranasal Neurostimulation) to external stimulation (Extranasal Neurostimulation) and chemical stimulation (Chemical Neurostimulation). Neuromodulation For Neuropathic Pain and Future Directions describe the use of neurostimulation for corneal neuropathic pain, a disease entity similar to DED, and the emerging techniques for multi-dimensional assessment of neurostimulation outcomes, respectively.
The Lacrimal Functional Unit
The etiology of DED has been previously attributed to an inadequate quantity and quality of tear film. DED is now recognized also as a disease of the lacrimal function unit (LFU), a highly integrated system comprising the ocular surface (cornea, conjunctiva, and meibomian glands), lacrimal glands (main and accessory), and the nervous system that connects them.10 The primary role of the LFU is maintenance of the function and integrity of the ocular surface.10
The LFU is typically a self-regulating unit. An inadequate tear film exposes the free nerve endings richly populating the corneal surface. Stimulation of these nerve endings then generates afferent impulses along the nasociliary nerve of the ophthalmic branch of the trigeminal nerve. These signals travel through the trigeminal ganglion and synapse on the superior salivatory nucleus (SSN) within the brainstem.11 The efferent secretory parasympathetic fibers arise in the SSN and exit the pons to reach the pterygopalatine ganglion. From there, they go on to innervate the lacrimal glands, goblet cells, and Meibomian glands.12 In this way, the LFU can respond to internal and external stimuli to maintain homeostasis of the ocular surface.
However, once one or more components of the LFU are compromised, a vicious cycle of tear deficiency and inflammation can result in rapid deterioration of ocular surface homeostasis. An exposed ocular surface and resultant tear-film hyperosmolarity stimulates an inflammatory cascade that involves mitogen-activated protein (MAP) kinase and nuclear factor- κB signaling pathways.13,14 These signaling pathways subsequently produce various proinflammatory cytokines, including interleukin-1α, interleukin-1β, tumor necrosis factor α (TNF-α), as well as matrix metalloproteinase 9 (MMP9). The result is an inflammatory cycle which further inhibits lacrimal and meibomian gland function, furthering aqueous deficiency and corneal sensory nerve damage.15,16 The latter comprises the very mechanism that regulates the functionality of the LFU, perpetuating dry eye and exacerbating its sequelae. The challenge of treating DED therefore rests largely in breaking this self-propagating cycle of inflammation and tear film deficiency.17
Nasolacrimal Reflex: A Physiologic Perspective
In recent years, neurostimulation of the nasolacrimal reflex (NLR) has emerged as a novel therapeutic modality for DED. The NLR, first detailed by Wernoe in 1927,18 is a neural reflex arc that starts with chemical, mechanical, or electrical stimulation of nerve endings lining the nasal mucosa and ends with bilateral lacrimation. The afferent branch of the NLR begins at the anterior ethmoidal nerve of the nasal mucosa and travels along the nasociliary branch of the ophthalmic division of the trigeminal nerve towards the midbrain, before synapsing on the SSN of the pons. The efferent pathway emerges from the pons and ultimately synapses on the main and accessory lacrimal glands. There is growing evidence that these nerve endings similarly regulate the meibomian glands and conjunctival goblet cells, suggesting that the three major tear film components (mucin, aqueous, and lipid) can all be influenced by neurostimulation.19–21
The unique advantages to targeting the NLR are many. Because the afferent portion of the NLR neural reflex arc is external to the LFU itself, it bypasses dysfunctional intermediary elements (eg, ocular surface) that are implicated in the inflammatory cascade of DED.22 Targeting the NLR allows restoration of functionality to the LFU through a top-down approach since the direct endpoint of the NLR neural reflex arc is the stimulation of tear production by the lacrimal and meibomian glands.23 NLR stimulation, therefore, affords an opportunity to break the vicious cycle of inflammation and tear film deficiency.23
First Forays into Neurostimulation for Dry Eye Disease
In 2009, Kossler et al introduced neurostimulation for the treatment of DED by demonstrating a significant increase in aqueous tear production following direct lacrimal nerve stimulation (LNS) in anesthetized rabbits.8 Subsequent studies compared LNS, which bypasses the NLR altogether, and anterior ethmoidal nerve stimulation (AENS), which involves the NLR. Brinton et al compared acute LNS to AENS using bipolar platinum foil electrodes in rabbits and demonstrated comparable increase in Schirmer score of up to 125% from baseline tear secretion with both modalities.24 Kossler et al performed a similar comparison, albeit with implantable stimulators under the nasal mucosa and lacrimal gland for AENS and LNS, respectively. This study found AENS to be superior in efficacy to LNS, increasing tear secretion 133% above baseline compared to 32% above sham for LNS. Additionally, AENS elicited a bilateral response, as expected from the tearing reflex pathway. However, histologically, chronic AENS was poorly tolerated, resulting in nasal mucosal fibrosis and implant extrusion within 3 weeks.19,20
These clinical insights collectively ushered the advent of the non-implantable intranasal tear neurostimulator (ITN), a device designed to externally stimulate the AENS through the nasal mucosa.
Intranasal Neurostimulation
The first iteration of the non-implantable ITN was designed by Oculeve (South San Francisco, CA) and consisted of a reusable base unit that produced electrical microcurrents and a disposable hydrogel tip assembled over the base unit and intended for insertion into the nasal cavity. The device was validated by Friedman et al in a prospective, open-label, non-randomized clinical trial of 40 patients with mild-to-severe DED.25 The study demonstrated an immediate improvement in Schirmer score with stimulation, ranging from 55% to 138%, at all follow-up assessments up to the study end point of day 180. Importantly, the device also resulted in an enduring improvement in unstimulated Schirmer score by approximately 30% from baseline (mean Schirmer score 9.4 ± 13 mm and 10.5 ± 1.1 mm in right and left eyes, respectively) that stabilized at 2 weeks post-initiation of treatment and was maintained at day 180 (mean Schirmer score 12.7 ± 1.7 mm and 13.2 ± 1.6 mm in right and left eyes, respectively). Device use resulted in improvement in dry eye symptoms, as assessed by Ocular Surface Disease Index (OSDI) scores, that persisted for an average 3.0 hours following use. Corneal and conjunctival staining scores measured using lissamine green and fluorescein staining graded by the modified Oxford scale from baseline to day 180 were also significantly reduced.25 A similar statistically significant increase in acute tear production was found when assessing tear volume secretion before and after ITN use using tear meniscus height captured by anterior segment optical coherence tomography.26
Several studies have investigated the effect of ITN on non-aqueous components of the tear film. Gumus et al examined impression cytology from goblet cells of bulbar conjunctiva of normal and dry eye patients. The results indicated that the ratio of degranulated goblet cells increased significantly following ITN use in both normal and dry eye patients, suggesting ITN could stimulate mucin secretion from goblet cells in addition to aqueous tear production.27 Dieckmann et al further supported enhanced merocrine and apocrine secretions from conjunctival goblet cells by showing reduced mean areas and perimeters by in vivo confocal microscopy.28
Despite a few study findings that suggest meibomian gland expression, the effects of ITN on protein and lipid component remain unknown.29,30
Oculeve Inc. (Oculeve, South San Francisco, CA) was later acquired by Allergan Plc. (Dublin, Ireland), and the ITN was branded TrueTear® (Figure 1) for commercialization following Federal Drug Administration (FDA)-approval for tear production in adults in April 2017.9 Despite demonstrated clinical efficacy, the prohibitive cost to market and subsequent low sales volumes led Allergan to discontinue the product in 2020 upon Allergan’s merger with AbbVie Inc.31 Patients who previously purchased the product were offered a refund upon return of TrueTear® device.9
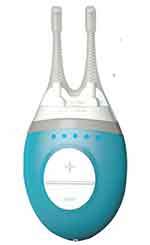 |
Figure 1 Allergan’s handheld TrueTear® intranasal tear neurostimulator device features two prongs covered in disposable hydrogel tips. The tips are inserted into the nasal cavity and provide electrical stimulation to the trigeminal nerve. Adapted from US Food & Drug Administration. De Novo Classification Request for Intranasal Tear Neurostimulator. Available from: https://www.accessdata.fda.gov/cdrh_docs/reviews/DEN160030.pdf.55 |
Extranasal Neurostimulation
The iTEAR device (Olympic Ophthalmics Inc., Issaquah, WA), which gained FDA approval for tear production in adults in May 2020, is the sole commercially available extranasal neurostimulation device. Its primary target is the external nasal nerve, which was regarded until recently as a sensory cutaneous nerve lacking any interface with the NLR. The clinical efficacy of the iTEAR device, however, demonstrates that such an interface between the external nasal and nasociliary nerves and NLR does exist and can result in a robust lacrimal response.32
The device is a palm-sized electromechanical nerve stimulator containing a single unidirectional tip (Figure 2). The tip is intended to rest against the skin of the nose at the junction between the nasal cartilage and nasal bone where the external nasal nerve exits superficially. Upon patient activation of the device, the unidirectional tip oscillates at a frequency between 220 and 270 Hz and an amplitude of between 0.5 and 1 mm for up to 30 seconds per side. Patients are instructed to use the device for 30 seconds at a time, at least twice daily or more, on an as needed basis daily.32
 |
Figure 2 The commercial iTEAR100 device applies repetitive electromechanical stimulation to the external nasal nerve via an oscillating tip (black arrow). Reproduced from Ji MH, Moshfeghi DM, Periman L, et al. Novel Extranasal Tear Stimulation: Pivotal Study Results. Transl Vis Sci Technol. 2020;9(12):23. Available from: https://tvst.arvojournals.org/article.aspx?articleid=2771969&resultClick=1.32 |
The TEAR2 randomized, controlled study of 59 subjects with DED demonstrated a significant change in Schirmer score (average 22 mm) immediately following treatment with the iTEAR device compared to no change with sham treatment.33 The subsequent TEAR1 multicenter, open-label, single-arm trial evaluated 101 subjects with DED at a primary endpoint of 30 days.33 Subjects demonstrated a significant improvement in mean Schirmer score from a baseline of 6.0 mm to 9.4 mm at day 30. Schirmer score improvement extended to a mean of 10.9 mm by day 180 of use. Given a non-significant dropout rate of 42%, this latter result might reflect a selection bias. OSDI similarly improved from a baseline mean of 40.3 to a 30-day mean of 27.1. The device was well-tolerated with 2–3% incidence of dizziness and headache during the study.32,33
Other pre-clinical studies have evaluated additional modalities of external neurostimulation for treatment of DED. Pedrotti et al investigated transcutaneous electrostimulation with electrodes placed onto the periorbital skin.34 Twenty-seven patients underwent 12 twenty-minute sessions over a span of 2 months. Their data reflect a significant increase in unstimulated Schirmer scores from a mean of 5.8 mm at pre-treatment baseline to 10.1 mm at 12 months, with a commiserate significant improvement in mean OSDI from 43.0 at baseline to 25.3 at the end of treatment.34 The modality of stimulation was found to be safe and effective, with no reported adverse events. A separate trial which randomized 52 patients 1:1 to either transcutaneous stimulation with artificial tears versus artificial tears alone similarly demonstrated improved Schirmer and OSDI scores in the stimulation group.35
Chemical Neurostimulation
Chemical neurostimulation of the NLR has been documented as early as the 1950s.36 Historically, this revelation served a diagnostic rather than therapeutic purpose. Patients were asked to sniff a trigeminal stimulant, such as ammonia, and monitored for lacrimation, the absence of which suggested an otoneurological lesion affecting the reflex arc.
Chemical neurostimulation has gained traction as a potential therapeutic target in the treatment of DED. In December 2020, Oyster Point Pharma, Inc (Princeton, NJ) submitted a new drug application to the FDA for its candidate OC-01, an intranasal formulation of varenicline, developed for DED treatment by way of NLR chemical neurostimulation.37 Varenicline is a highly selective nicotinic acetylcholine receptor (nAChR) agonist with existing therapeutic use in smoking cessation and nicotine addiction (Chantix, Pfizer, New York, NY).38,39 The spray stimulates the nasociliary nerve within the nasal mucosa, activating the NLR and resulting in lacrimation. Varenicline was chosen given the high density of nicotinic acetylcholine receptors on nasal trigeminal nerve endings.38,39
OC-01 was evaluated in the recently concluded ONSET-2 Phase 3 trial.39 The multicenter, randomized, double-masked, vehicle-controlled study enrolled 758 subjects with a pre-treatment Schirmer score < 10 mm and OSDI > 23. Subjects were randomized into two treatment arms (candidate drug at 0.6 mg/mL or 1.2 mg/mL) or placebo, with a primary endpoint of Schirmer score improvement to >10 mm at 4 weeks. The results demonstrated a statistically significant improvement in Schirmer score in 44% and 47% of study patients in both treatment arms compared to 26% in the control group. The most commonly reported mild adverse effects were sneezing (62–84%) or coughing (9–25%) after instillation, with prevalence in direct correlation to the drug concentration.39
Neuromodulation for Neuropathic Pain
Corneal neuropathic pain is often conflated with conventional DED because the presentation manifests often with severe dry eye-like symptoms, including hyperalgesia, photoallodynia, and blepharospasm. However, corneal neuropathic pain can typically be differentiated by the minimal to absent ocular surface findings.39 The recent use of in vivo confocal microscopy, which can reveal evidence of nerve injury, also aids in the diagnosis of corneal neuropathic pain.40,41 Risk factors for development of neuropathic pain include young age, clinical depression or anxiety, and corneal nerve injury as a side effect of laser in situ keratomileusis (LASIK).42
The transcutaneous electrical nerve stimulation (TENS) therapy is a nonpharmacological treatment that has shown promise in addressing ocular neuropathic pain by delivering alternating current via cutaneous electrodes placed in the proximity to the terminal cutaneous trigeminal nerve branches over the forehead and temple.43 TENS therapy blocks or changes a patient’s perception of pain. A small device delivers a current at or near the target nerves, typically set at 5000/5100 Hz frequencies generating a 100 Hz beat frequency per treatment. The amplitude can be manually determined by the patient depending on the level of comfort. The treatment can be repeated up to 3 times a day.43
In 2015, Hayek et al reported a case of intractable post-LASIK corneal neuropathic pain successfully managed with electrical stimulation of the trigeminal ganglion via an implanted electrode array.44 A larger subsequent series of 10 patients demonstrated a mean post-treatment reduction in subjective pain intensity by 27.4% following periocular TENS treatment. Study patients were provided a TENS device for home use up to three times daily for three months. Prior to use, patients were instructed to place four electrodes over the forehead and temple, in close proximity to terminal cutaneous trigeminal nerve branches over the forehead and temple.45 Pain was surveyed on a 0–10 scale at an initial visit and every week thereafter. Common comorbid ocular conditions in this cohort included a history of refractive surgery (40%), aqueous tear deficiency by Schirmer (30%), and evaporative dry eye with increased tear breakup time (60%).45
While the exact mechanisms of pain reduction with TENS are unclear, there is a biologic plausibility that TENS should modulate eye pain. This plausibility rests on the gate control theory of pain, which was introduced in 1965, and asserts that stimulation of large diameter, low threshold mechanoreceptive (touch-related) nerve fibers could inhibit the transmission of action potentials from small diameter, higher threshold nociceptive (pain-related) fibers.42 Since then, researchers have elucidated specific mechanisms of actions of TENS and identified anatomic pathways, neurotransmitters, and the types of neurons involved in pain signal inhibition.46
Specifically, it is now understood that the pathway of noxious sensation from the cornea to the brain begins along primary afferent unmyelinated C and lightly myelinated A-delta fibers of the nasociliary branch of V1 to the trigeminal ganglion.37 These are ultimately connected centrally to higher-order somatosensory pain pathways and the thalamus, where pain is perceived. TENS likely interacts with these pathways at multiple points. In regard to ocular pain, TENS could stimulate deep A-beta afferent fibers within frontal branches of the V1, blocking pain signals arising from unmyelinated C and lightly myelinated A-delta fibers in the long ciliary nerves.47 High-frequency TENS can also modify release of gamma-aminobutyric (GABA) and enkephalins at the level of the spinal cord, which have an inhibitory effect on interneurons of the trigeminal-thalamic tract, thereby preventing propagation of pain signals.48
Future Directions
Recent advancements in the diagnosis of DED might offer additional insights in accurate measurement of outcomes in the development of neurostimulation devices. Significant advances in ocular imaging technology have enabled improvement in objective and reproducible evaluation of ocular surface change, tear film parameters, and optical quality associated with DED, offering a multi-faceted view of DED not previously permitted by traditional metrics, such as TBUT or Schirmer’s.49 Optical coherence tomography, for instance, has been employed to quantify tear meniscus parameters.50 Infrared meibography and interferometry can be used to evaluate meibomian gland dropout and the tear lipid layer.51,52 Tear film imaging (TFI) with spectral interference technology enables noninvasive measurement of the mucoaqueous and lipid layers, and the thickness change rate of each over time.53 Studies have also used proteomic analysis to characterize the proteins constituting the tear film and lacrimal fluid.54
Employing these new modalities to evaluate emerging technologies in neurostimulation would not only demonstrate efficacy more holistically but may also yield new insights into the unique strengths of each neurostimulatory approach. This could ultimately lead to a customized approach to treating DED with neurostimulation, where, for instance, a diagnosis of lipid layer deficiency would be matched with the neurostimulatory technique that offers the best restoration of the lipid layer.
Conclusion
The past decade has witnessed the advent of new neurostimulatory modalities, each targeting various aspects of the lacrimal functional unit with demonstrated efficacy by traditional metrics, like fluorescein tear film break-up time (TBUT) and Schirmer’s test. Despite these rapid initial strides, neurostimulation for DED is still in its early phases. Many questions remain regarding the specific physiological changes within the tear film elicited by various neurostimulatory techniques and the differences between them. Long-term durability of any changes must also be demonstrated. While neurostimulation represents a promising novel modality for treatment of DED, further studies are needed to elucidate its true potential.
Disclosure
The authors report no conflicts of interest.
References
1. The epidemiology of dry eye disease: report of the Epidemiology Subcommittee of the International Dry Eye WorkShop (2007). Ocul Surf. 2007;5(2):93–107.
2. Yu JH, Asche CV, Fairchild CJ. The economic burden of dry eye disease in the United States: a decision tree analysis. Cornea. 2007;30:379–387. doi:10.1097/ICO.0b013e3181f7f363
3. Sall K, Stevenson OD, Mundorf TK, et al. Two multicenter, randomized studies of the efficacy and safety of cyclosporine ophthalmic emulsion in moderate to severe dry eye disease. Ophthalmol. 2000;107:631–639. doi:10.1016/S0161-6420(99)00176-1
4. Clayton JA. Dry eye. N Engl J Med. 2018;378:2212–2223. doi:10.1056/NEJMra1407936
5. Kossler AL, Wang JH, Feuer W, et al. Lacrimal nerve stimulation by a neurostimulator for tear production. Invest Ophthalmol Vis Sci. 2009;50:4244.
6. Rokyta R, Fricova J. Neurostimulation methods in the treatment of chronic pain. Physiol Res. 2012;61:S23–S31. doi:10.33549/physiolres.932392
7. Ben-Menachem E. Nerve stimulation for the treatment of epilepsy. Lancet Neurol. 2002;1:477–482. doi:10.1016/S1474-4422(02)00220-X
8. Shealy CN, Mortimer JT, Reswick JB. Electrical inhibition of pain by stimulation of the dorsal columns: preliminary clinical report. Anesth Analg. 1967;46:489–491.
9. Allergan TrueTear. Available from: https://www.truetear.com.
10. Stern ME, Beuerman RW, Fox RI, et al. The pathology of dry eye; the interaction between the ocular surface and lacrimal glands. Cornea. 1998;17:584–589. doi:10.1097/00003226-199811000-00002
11. Dartt D. Neural regulation of lacrimal gland secretory processes: relevance in dry eye diseases. Prog Retin Eye Res. 2009;28:155–177. doi:10.1016/j.preteyeres.2009.04.003
12. LeDoux MS, Zhou QH, Murphy RB, et al. Parasympathetic innervation of the meibomian glands in rats. Invest Ophthalmol Vis Sci. 2001;42:2434–2441.
13. Li DQ, Zhuo C, Xiu JS, et al. Stimulation of matrix metalloproteinases by hyperosmolarity via a JNK pathway in human corneal epithelial cells. Invest Ophthalmol Vis Sci. 2004;45:4302–4311. doi:10.1167/iovs.04-0299
14. Luo LH, Li DQ, Corrlaes RM, et al. Hyperosmolar saline is a proinflammatory stress on the mouse ocular surface. Eye Contact Lens. 2005;31:186–193. doi:10.1097/01.ICL.0000162759.79740.46
15. De Paiva CS, Corrales RM, Villarreal AL, et al. Corticosteroid and doxycycline suppress MMP-9 and inflammatory cytokine expression, MAPK activation in the corneal epithelium in experimental dry eye. Exp Eye Res. 2006;83:526–535. doi:10.1016/j.exer.2006.02.004
16. Luo L, Li DQ, Doshi A, et al. Experimental dry eye stimulates production of inflammatory cytokines and MMP-9 and activates MAPK signaling pathways on the ocular surface. Invest Ophthalmol Vis Sci. 2004;2:124–130.
17. Baudouin C, Messmer EM, Aragona P, et al. Revisiting the vicious circle of dry eye disease: a focus on the pathophysiology of meibomian gland dysfunction. Br J Ophthalmol. 2016;100(3):300–306. doi:10.1136/bjophthalmol-2015-307415
18. Wernoe TB. [The vasodilatory naso-ocular reflex and its diagnostic value]. Acta Psychiatr Scand. 1927;2:385–398. doi:10.1111/j.1600-0447.1927.tb07448.x
19. Kossler AL Chronic electrical stimulation for tear & secretion: lacrimal vs. anterior ethmoid nerve.
20. Brinton M, Kossler AL, Patel ZM, et al. Enhanced tearing by electrical & stimulation of the anterior ethmoid nerve. Invest Ophthalmol Vis Sci. 2017;58:2341–2348. doi:10.1167/iovs.16-21362
21. Gumus K, Pflugfelder SC. Intranasal tear neurostimulation: an emerging concept in the treatment of dry eye. Int Ophthalmol Clin. 2016;57:101–108. doi:10.1097/IIO.0000000000000163
22. Baudouin C, Aragona P, Messmer EM, et al. Role of hyperosmolarity in the pathogenesis and management of dry eye disease: proceedings of the OCEAN group meeting. Ocul Surf. 2013;11:246–258. doi:10.1016/j.jtos.2013.07.003
23. Baudouin C, Messmer EM, Aragona P, et al. [A new approach for better comprehension of diseases of the ocular surface]. J Fr Ophthalmol. 2007;30:239–246. French. doi:10.1016/S0181-5512(07)89584-2
24. Brinton M, Chung JL, Kossler A, et al. Electronic enhancement of tear secretion. J Neural Eng. 2016;13(1):016006. doi:10.1088/1741-2560/13/1/016006
25. Friedman NJ, Butron K, Robledo N, et al. A nonrandominized, open-label study to evaluate the effect of nasal stimulation on tear production in subjects with dry eye disease. Clin Ophthalmol. 2016;10:795–804.
26. Orrick B, Watson M, Angjeli E, et al. Quantitation of tear production by tear meniscus height following acute use of the intranasal tear neurostimulator. Invest Ophthalmol Vis Sci. 2016;58:2692.
27. Woodward A, Senchyna M, Franke M, et al. Effect of intranasal neurostimulation on tear protein content in patients with dry eye. Invest Ophthalmol Vis Sci. 2017;58(8):2673.
28. Dieckmann G, Kataguiri P, Pondelis N, et al. In vivo confocal microscopy demonstrates intranasal neurostimulation-induced goblet cell alterations. Invest Ophthalmol Vis Sci. 2017;58:2694.
29. Green KB, Kamat M, Franke M, et al. Tear total lipid concentration in patients with dry eye following intranasal neurostimulation. Invest Ophthalmol Vis Sci. 2017;58(8):2693.
30. Cohn GS, Corbett D, Tenen A, et al. Randomized, controlled, double-masked, multicenter, pilot study evaluating safety and efficacy of intranasal neurostimulation for dry eye disease. Invest Ophthalmol Vis Sci. 2019;60(1):147–153. doi:10.1167/iovs.18-23984
31. Abbvie News Center. AbbVie to Acquire Allergan in transformative move for both companies. Available from: https://news.abbvie.com/news/press-releases/abbvie-to-acquire-allergan-in-transformative-move-for-both-companies.htm.
32. Ji MH, Moshfeghi MD, Periman L, et al. Novel extranasal tear stimulation: pivotal study results. Transl Vis Sci Technol. 2020;9(12):23. doi:10.1167/tvst.9.12.23
33. PR Newswire. Olympic ophthalmics presents clinical evidence for iTEAR100.
34. Pedrotti E, Bosello F, Fasolo A, et al. Transcutaneous periorbital electrical stimulation in the treatment of dry eye. Br J Ophthalmol. 2016;101(6):814–819.
35. Cai MM, Zhang J. Effectiveness of transcutaneous electrical stimulation combined with artificial tears for the treatment of dry eye: a randomized controlled trial. Exp Ther Med. 2020;20(6):175. doi:10.3892/etm.2020.9305
36. Zilstorff-Pedersen K. Quantitative measurements of the nasolacrimal reflex. Acta Otolaryngol. 1959;50:501–506. doi:10.3109/00016485909129225
37. EyeWire News. Oyster point announces results of Phase 3 Trial of OC-01 nasal spray for treatment of signs and symptoms of dry eye disease.
38. Keiger CJH, Case LD, Kendal-Reed M, et al. Nicotinic cholinergic receptor expression in the human nasal mucosa. Ann Otol Rhinol Laryngol. 2003;112(1):7–84. doi:10.1177/000348940311200115
39. Alimohammadi H, Silver WL. Evidence for nicotinic acetylcholine receptors on nasal trigeminal nerve endings of the rat. Chem Senses. 2000;25(1):61–66. doi:10.1093/chemse/25.1.61
40. Galor A, Moein H-R, Lee C, et al. Review: neuropathic pain and dry eye. Ocul Surf. 2018;16(1):31–44. doi:10.1016/j.jtos.2017.10.001
41. Dieckmann G, Goyal S, Hamrah P. Neuropathic corneal pain: approaches for management. Ophthalmol. 2017;124(11):S34–S47. doi:10.1016/j.ophtha.2017.08.004
42. Rosenthal P, Borsook D. The corneal pain system. Part I: the Missing Piece of the Dry Eye Puzzle. Ocul Surf. 2012;10:2–14. doi:10.1016/j.jtos.2012.01.002
43. Vance CG, Dailey DL, Rakel BA, Sluka KA. Using TENS for pain control: the state of the evidence. Pain Manag. 2014;4(3):197–209. doi:10.2217/pmt.14.13
44. Hayek SM, Sweet JA, Miller JP, Sayegh RR. Successful management of corneal neuropathic pain with intrathecal targeted drug delivery. Pain Med. 2016;17(7):1302–1307. doi:10.1093/pm/pnv058
45. Zayan K, Aggarwal S, Felix E, et al. Transcutaneous electrical nerve stimulation for the long-term treatment of ocular pain. Neuromodulation. 2020;23(6):781–787. doi:10.1111/ner.13146
46. Melzack R, Wall PD. Pain Mechanisms: a New Theory. Science. 1965;150:3699. doi:10.1126/science.150.3699.971
47. Radhakrishnan R, Sluka K. Deep tissue afferents, but not cutaneous afferents, mediate transcutaneous electrical nerve stimulation-induced antihyperalgesia. J Pain. 2016;6:673–680. doi:10.1016/j.jpain.2005.06.001
48. Jiang L, Yuan DL, Li ML, et al. Combination of flunarizine and transcutaneous supraorbital neurostimulation improves migraine prophylaxis. Acta Neurol Scand. 2019;139:276–283.
49. Han SB, Liu YC, Mohamed-Noriega K, et al. Objective imaging diagnostics for dry eye disease. J Ophthalmol. 2020;2020:1–11. doi:10.1155/2020/3509064
50. Ibrahim OMA, Dogru M, Takano Y, et al. Application of visante optical coherence tomography tear meniscus height measurement in the diagnosis of dry eye disease. Ophthalmol. 2010;117(10):1923–1929. doi:10.1016/j.ophtha.2010.01.057
51. Ban Y, Shimazaki-den S, Tsubota K, Shimazaki J. Morphological evaluation of meibomian glands using noncontact infrared meibography. Ocul Surf. 2013;11(1):47–53. doi:10.1016/j.jtos.2012.09.005
52. Arita R, Morishige N, Fujii T, et al. Tear interferometric patterns reflect clinical tear dynamics in dry eye patients. Invest Ophthalmol Vis Sci. 2016;57(8):3928–3934. doi:10.1167/iovs.16-19788
53. Cohen Y, Epshtein S, Harris A, et al. Tear film imager for dynamic mapping of the human tear film. Appl Opt. 2019;58(29):7987–7995. doi:10.1364/AO.58.007987
54. Jung JH, Ji YW, Hwang HS, et al. Proteomic analysis of human lacrimal and tear fluid in dry eye disease. Nature. 2017;7:1.
55. US Food & Drug Administration. De Novo Classification Request for Intranasal Tear Neurostimulator. Available from: https://www.accessdata.fda.gov/cdrh_docs/reviews/DEN160030.pdf. Accessed October 13, 2021.
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
