Back to Journals » Clinical Interventions in Aging » Volume 16
Serum sirtuin 1 is independently associated with intact PTH among patients with chronic kidney disease
Authors Bielach-Bazyluk A , Zbroch E , Czajkowska K, Koc-Zorawska E, Kakareko K, Rydzewska-Rosolowska A , Hryszko T
Received 24 November 2020
Accepted for publication 26 February 2021
Published 25 March 2021 Volume 2021:16 Pages 525—536
DOI https://doi.org/10.2147/CIA.S293665
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Prof. Dr. Nandu Goswami
Angelika Bielach-Bazyluk,1 Edyta Zbroch,2 Katarzyna Czajkowska,1 Ewa Koc-Zorawska,1 Katarzyna Kakareko,1 Alicja Rydzewska-Rosolowska,1 Tomasz Hryszko1
1 2nd Department of Nephrology and Hypertension with Dialysis Centre, Medical University, Bialystok, Poland; 2Department of Internal Medicine and Hypertension, Medical University, Bialystok, Poland
Correspondence: Angelika Bielach-Bazyluk
2nd Department of Nephrology and Hypertension with Dialysis Centre, Medical University, Sklodowskiej – Curie 24a, Bialystok, 15-276, Poland
Tel +48 858317872
Email [email protected]
Background: Sirtuin 1 is involved in the pathogenesis of age-related diseases.
Purpose: The aim of the study was to assess the clinical and diagnostic value of serum sirtuin 1 concentration in patients with CKD.
Patients and Methods: The serum sirtuin 1 level was evaluated using ELISA kit in 100 CKD patients stratified for five stages and in a control group of 24 healthy volunteers.
Results: Serum sirtuin 1 concentration was higher in the CKD group compared with the control group (p< 0.05). Sirtuin 1 correlated with conventional CKD biomarkers and eGFR equations, intact parathyroid hormone (iPTH) and age (all p< 0.05). Statins, AT1 receptor antagonists and β-blockers use were associated with decreased sirtuin concentration (p< 0.05). Sirtuin 1 was able to distinguish CKD from control group with high sensitivity and specificity (93% and 87%, respectively; AUC=0.954). Surprisingly, after adjustment only iPTH concentration was an independent predictor of sirtuin 1 level.
Conclusion: The association between sirtuin 1, eGFR equations and iPTH indicates its possible usefulness as a kidney function marker. In terms of iPTH being the only independent predictor of circulating sirtuin 1 it can be considered as an indirect cardiovascular risk biomarker regardless of renal function and provide additional information for patient management. Alternatively, sirtuin 1 is recognized as protective against vascular disease, and we demonstrated a positive correlation with iPTH, which may be related to accumulation of (7-84)-PTH having opposite biological effects to full-length PTH. Further studies are needed to explore the interplay between sirtuin 1, PTH and CKD-related vascular calcification as well as to assess its prognostic value in observational studies.
Keywords: sirtuin 1, SIRT1, CKD, chronic kidney disease, parathyroid hormone, PTH
Introduction
Chronic kidney disease (CKD) is defined as gradual loss of function of particular nephrons resulting in renal failure. The disease is characterized by accelerated vascular disease, which is the leading cause of death in this population.1 As the trend toward aging of the worldwide population is likely to continue, CKD is expected to become a major global health problem. Moreover, CKD is recognized as a novel substantial risk factor for cardiovascular disease and total risk in renal patients is greater than the one attributable to comorbidities.2 One of the possible explanations is excessive medial arterial calcification specific to renal impairment. Positive phosphate balance in the course of CKD may promote vascular calcification (VC) by depletion of sirtuin 1 expression and, in consequence, may lead to severe cardiovascular complications.3 Early diagnosis, patients’ awareness of the disease, appropriate nephroprotection and lifestyle modification are essential to halt the progression of CKD and thus reduce risk for cardiovascular events.
Sirtuin 1 (SIRT1) is the most widely studied member of a group of enzymes with NAD+- dependent deacetylase activity, detecting intracellular energy state and regulating gene expression by chromatin silencing and transcriptional repression.4 It is implicated in mitochondrial biogenesis, inflammation, cellular stress response and apoptosis, which constitute a common denominator of chronic diseases and aging. Nephroprotective and cardioprotective effects of sirtuin 1 were determined on animal and cellular models.5–11 It was found to diminish renal fibrosis, maintain podocytes’ function, augment autophagocytosis and angiogenesis, regulate blood pressure through the renin-angiotensin-aldosterone system and stimulate nitric oxide synthesis.
However, to our best knowledge, only few studies have investigated the serum sirtuin 1 concentration in humans with kidney and heart disease. Doulamis et al. noted that lower sirtuin 1 concentration was associated with diabetes mellitus and a history of myocardial infarction in patients with advanced coronary disease.12 Regarding CKD, there are two papers reporting decreased sirtuin 1 levels in patients with diabetic kidney disease compared with healthy subjects.13,14 In contrast, findings from the recent work by Zbroch et al. revealed elevated sirtuin 1 concentration in hemodialysis patients and positive correlation between the concentration and duration of dialysis.15
The aim of the study was to assess the serum concentration of sirtuin 1 in conservatively treated patients with CKD stratified for five stages comparing with healthy volunteers. We also studied possible relationships between sirtuin 1, demographic data, numerous clinical measurements as well as co-existing diseases, including cardiovascular and diabetes, treatment and survival outcomes.
Patients and Methods
Study Design and Participants
We conducted a cross-sectional single-center study involving 100 inpatients suffering from chronic kidney disease and a control group of 24 healthy volunteers. The main criterion for inclusion was stable CKD according to the KDIGO (Kidney Disease Improving Global Outcome) definition. The exclusion criteria were: acute kidney injury, exacerbation of CKD, renal replacement therapy (i.e. hemodialysis, peritoneal dialysis, kidney transplantation), persons under the age of 18.
All patients gave written informed consent. Clinical data were anonymized prior to transfer to the research database. The study was approved by the Medical University Ethic Committee (approval number R-I-002/455/2016) and was in accordance with the principles of the Helsinki Declaration.
Methods
Clinical data were collected from past medical history, physical examination and laboratory tests routinely performed in the hospital laboratory. The data included age, sex, body mass index (BMI), stage and etiology of chronic kidney disease, mean blood pressure, antihypertensive therapy, statin use, history of cardiac infarction and ischemic heart disease, heart failure, diabetes, hyperlipidemia, complete blood count, calcium-phosphate metabolism, cholesterol level and proteinuria. Laboratory tests were performed using standard methods in the hospital laboratory.
Obtained fasting blood samples were centrifuged at 1300 ×g for 15 min and stored at − 80 °C until analysis. The non-routine laboratory tests were performed using commercially available immunoenzymatic (ELISA - Enzyme-Linked Immunosorbent Assay) test kits. The tests were performed following the assay instructions. The absorbance was measured by a microplate reader (MULTISCAN GO, Thermo Fisher Scientific, Finland). The concentrations were calculated based on the standard curves. The serum concentration of sirtuin 1 was assessed using kits from EIAab Science Co., Wuhan, China, with limit of detection 32 pg/mL, and intra- and interassay precision of 4.3% and 7.2%, respectively. The reference range for sirtuin 1 was 78–5000 pg/mL. Human soluble α-Klotho was measured using kits from IBL (Immuno-Biological Laboratories Co., Japan) with the limit of detection 6.15 pg/mL and intra- and interassay precision of 3.5% and 11.4%. The reference range for α-Klotho was 93.75–6000 pg/mL. Cystatin C concentrations in serum were assessed using kits from BioVendor, Brno, Czech Republic. The reference range for cystatin C was 200–10,000 ng/mL, with the limit of detection 0.25 ng/mL and intra- and interassay precision of 3.5% and 10.4%. All tests were performed by the same person in standardized laboratory settings. This study was conducted with the use of equipment purchased by Medical University of Białystok as part of the RPOWP 2007–2013 funding, Priority I, Axis 1.1, contract No. UDA- RPPD.01.01.00-20-001/15-00 dated 26.06.2015.
Statistical Analysis
The data were analyzed using Statistica 13.3 computer software (StatSoft, Poland). The significance was set at a 95% confidence limit. The Shapiro–Wilk test was used to evaluate normal distribution. Continuous variables were presented as mean and standard deviation, when normal distribution or median and interquartile range, when non-Gaussian and categorical variables as percentage. The comparison between studied and control groups was performed by the use of Mann–Whitney U test. We used Kruskal–Wallis non-parametric test to compare sirtuin-1 serum concentration between particular stages of CKD. The association between sirtuin 1 and clinical data was determined by Spearman correlation test. Multivariable conditional logistic regression was used to assess association between examined protein and each of significantly correlated cofounders. The overall survival of subjects after 24-months follow-up stratified for terciles of sirtuin 1 concentration was measured by chi-squared test. Receiver operating characteristic (ROC) and precision-recall curves analysis were performed to assess the utility of serum sirtuin 1 for diagnosing CKD.
Results
Studied Population
Clinical and biochemical characteristics of patients and controls are presented in Tables 1 and 2. The study included 100 patients (49 men and 51 women) with mean age of 69 (23–91) and compared them with 24 age-matched and sex-matched healthy volunteers with no history of chronic kidney disease, diabetes, cardiovascular disease and medication use. The creatinine and cystatin C level in all controls was within the normal range.
 |
Table 1 Basal Characteristics of Patients Group and Controls Group |
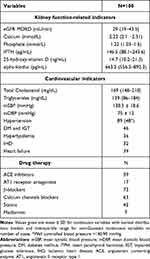 |
Table 2 Basal Clinical and Biochemical Characteristics of the Studied Group |
In the patients group the most prevalent comorbidities were hypertension, followed by diabetes mellitus and heart failure; 54% of hypertensive patients had well controlled blood pressure (<140/90 mmHg). The most widely used drugs were β-blockers, calcium channel blockers and statins.
Sirtuin 1 Concentrations in Studied Groups
Serum sirtuin 1 concentration was significantly higher (2.23 [1.57–3.24] vs 0.44 [0.19–0.64] ng/mL, p<0.0001) in the studied group compared with the control group (Figure 1).
 |
Figure 1 The comparison of serum sirtuin 1 concentration between studied and control group. |
The levels of sirtuin 1, cystatin C, mineral and bone disorder-related parameters stratified by CKD stage are reported in Table 3. Sirtuin 1 concentration was moderately (R=0.54) and cystatin C was strongly (R=0.84) associated with CKD stage, while α-klotho correlation did not reach statistical significance (p=0.09). From the group of measurements related to mineral and bone disorder, only iPTH and phosphate level correlated significantly with the advancement of the disease (R=0.62, p<0.001, R=0.4 p<0.001, respectively). As illustrated in Figure 2, a considerable increase in the median sirtuin 1 concentration between stage 1 CKD and control group was noted. Further gradual increase across CKD stages was accompanied by greater range of concentrations in particular subgroups. Differences between particular CKD stages were significant (p<0.001).
 |
Table 3 Sirtuin 1, Cystatin C and Mineral Bone Metabolism-Related Measurements According to the Stage of Chronic Kidney Disease |
 |
Figure 2 Serum sirtuin 1 concentration according to KDIGO stage. |
Sirtuin 1 Correlation Analyses and Multiple Regression Analysis
Square root transformation of sirtuin 1 concentration was made to compare normal distribution. Then, the association between the concentration and examined biochemical measurements, demographic and clinical data was performed (Table 4). We found sirtuin 1 significantly correlated with conventional CKD biomarkers and eGFR equations, with the strongest association with CKD-EPIcystatin C formula (R=0.61, p<0.001). There was a positive correlation with PTH level (R=0.39, p<0.001) and age (R=0.27, p=0.002), see Figure 3.
 |
Table 4 The Relationship Between Serum Sirtuin 1 and Studied Parameters |
 |
Figure 3 The relationship between sirtuin 1 and eGFR CKD-EPI based on cystatin C concentration, iPTH level and age. |
Statins, AT1 receptor antagonists and β-blockers use was associated with decreased sirtuin concentration (p<0.001, p=0.02 and p=0.04, respectively). There was no significant correlation between sirtuin 1 and α-klotho, BMI, blood lipids, proteinuria and blood pressure. Sirtuin 1 concentration was not related to sex and comorbidities.
CKD-EPIcystatin C filtration rate and cofounders significantly correlated with sirtuin 1 were included to a model of multiple regression analysis (Table 5). Other kidney function parameters associated with sirtuin 1 (creatine, cystatin C) were excluded due to multicollinearity. After adjustment only PTH concentration (beta = 0.3, p = 0.009) was an independent predictor of sirtuin 1 level. The equation explained 52% of the variation of serum sirtuin 1 concentration in the CKD group (F = 6.74, p<001).
 |
Table 5 Regression Models Related to Sirtuin 1 Concentration |
Association with CKD Etiology
Sirtuin 1 concentration was not related to the etiology of CKD. In contrast, cystatin C level was statistically higher in patients with diabetic kidney disease compared with hypertensive nephropathy. At the same time, median creatinine concentration between both etiologic groups was comparable (2.21 vs 2.40 mg/dL).
The Discriminative and Prognostic Value of Sirtuin 1 in CKD Population
The ROC and precision recall curves were constructed to measure the usefulness of sirtuin 1 level as a potential biomarker of CKD (Figure 4). The analysis demonstrated that sirtuin 1 is able to distinguish CKD from the control group (cut-off: 0.96 ng/mL) with high sensitivity and specificity (93% and 87%, respectively; AUC=0.954, 95% CI 0.919–0.989).
 |
Figure 4 Receiver operating characteristic (ROC) curve of serum sirtuin 1 between CKD and control group. |
No relationship was found between terciles of sirtuin 1 concentration and death outcomes.
Discussion
High global burden of chronic kidney disease and related accelerated development of cardiovascular disease resulting in decline in life expectancy puts an emphasis on early detection and identification of new targets for more specific therapies. Sirtuin 1 may convey the impression of being a novel target related to kidney function. A number of studies addressed its protective role in systemic metabolism, cardiometabolic and age-related diseases as well as its relevance in vascular dysfunction. However, the majority of available literature’s data derive from molecular and cellular models. We aimed to verify if clinical findings could be translated into practical patient care using easily accessible serum samples for the measurement of sirtuin 1 concentration.
To our best knowledge, it is the first study assessing the serum sirtuin 1 concentration in humans with respect to association with different stages of chronic kidney disease, its etiology and clinical measurements. As has been previously reported, the expression of SIRT1 in tissue kidney specimens correlates with renal impairment and strategies up-regulating SIRT1 expression are able to mitigate kidney injury.7,16,17 It could be hypothesized, that serum sirtuin 1 concentration would be an equivalent for tissue levels and would decrease correspondingly with eGFR decline. Based on the available literature we found two studies investigating serum sirtuin 1 in diabetic kidney disease and one in maintenance dialysis patients.13–15 Regarding diabetic kidney disease the authors of both studies showed significantly lower sirtuin 1 concentration in patients compared with healthy controls.13,14 However, findings on the relationship between the sirtuin concentration and the degree of albuminuria revealed discrepancies. This conflicting data may be partially explained by distinctive characteristics of enrolled patients, which included only those with eGFR above 60 mL/min in the latter study.14 On the contrary, substantially elevated sirtuin 1 concentration was found in patients with end-stage renal disease undergoing hemodialysis.15 These findings support the notion that sirtuin 1 may be influenced to greater extent by diabetes-related factors if only mild or moderate renal impairment is present. In line with this idea, in our study, we demonstrated raised serum sirtuin concentration in patients with CKD compared with healthy subjects. We observed a gradual increase of the concentration in a stage-dependent manner and its negative correlation with eGFR equation. The elevated sirtuin 1 concentration in CKD patients could be explained by its accumulation due to decreased filtration rate or its increased secretion from kidney cells as a result of nephrons’ apoptosis. However, this cannot explain the greater range of concentration in particular stages detected in our study. Interestingly, after adjustment for other cofounders including age, iPTH level, β-blockers and statin use, iPTH was the only independent determinant of sirtuin 1 concentration in our population.
The influence of PTH on the cardiovascular system has gained much importance in the past years. PTH has a detrimental influence on cardiac function and vasculature and it is associated with left ventricular hypertrophy, hypertension and arrhythmias.18–20 Moreover, individuals with higher plasma PTH are at risk of cardiovascular mortality,21 even if PTH remains within laboratory reference range.22 Although prior research generally confirms that PTH concentrations become apparent when eGFR declines below 60mL/min only a few studies have examined PTH in subjects with normal kidney function.23 For example, in a cross-sectional study on individuals without CKD with the mean eGFR=105 mL/min based on CKD-EPICr-Cys, the authors demonstrated that PTH concentration began to rise much earlier when eGFR drops below 126 mL/min.24 From this standpoint, sirtuin 1 similarly to PTH can be considered as a cardiovascular risk biomarker regardless of renal function and provide additional information for patient management.
Disturbances of mineral and bone metabolism related to CKD are widely considered as the major cause of extraskeletal calcification, including vascular calcification (VC). Previous studies on young dialysis-dependent patients, lacking classical cardiovascular risk factors, brought clear evidence of VC contribution in increased cardiovascular risk.25,26 Despite ample data regarding association of PTH with VC and poor cardiac outcomes, its role as an independent contributor to vascular smooth muscle cells’ (VSMCs) phenotypic change remains uncertain. A reason for conflicting results may be due to difficulties with their interpretation due to distinctive methodologies applied in individual studies. It is of particular importance in the context of variable response to intermittent and continuous PTH exposure.27 What is more, in the circulation, there are different fragments of PTH encompassing full-length (1-84)-PTH, middle-PTH, amino- and carboxyl-terminal PTH, some of which remains biologically inactive. The classical actions of PTH on bones and kidney accounts for 1–84 fragment and could be antagonized by (7-84)-PTH.28 It should be stressed that routinely used second generation assay (iPTH) detects both (1-84)-PTH and (7-84)-PTH.29 According to previous data, (7-84)-PTH accumulates progressively in the circulation along with eGFR decline,30 therefore it may overestimate secondary hyperparathyroidism. Hence, iPTH assay may not sufficiently reflect PTH-related VC and cardiovascular risk.
The question arises whether distinctive response to PTH in VSMCs could also be expected. Several former studies on PTH-induced VSMCs transition indicated that (1-34)-PTH may inhibit medial artery calcification.31,32 More recent reports have demonstrated independent regulation of arterial medial calcification by PTH and hyperphosphatemia.33,34 As an illustration, Neves et al. noted that continuous PTH overstimulation triggers VC regardless of serum calcium and phosphate concentration in rodents.33 Similar findings were recently obtained from a rodent model,34 in which (1-34)-PTH augmented VC by binding to its principal receptor PTH1R, without regard to phosphate and α-klotho level. To sum up, low level of sirtuin 1 is associated with exacerbated VC and full-length PTH seems to act as an independent promoter of VC via PTH1R signaling. However, in our study on renal patients prone to VC, increased iPTH concentrations were correlated with high levels of sirtuin 1. Furthermore, the extent of detected sirtuin levels continued to grow with advancing CKD stage, which rather follows PTH variability than reflects eGFR decline. It should be underlined that we used a second generation PTH assay, so we can suspect that (7-84)-PTH might have accounted for even up to 50% of detected PTH.34 In other words, we can assume the growth in sirtuin 1 concentration corresponded with the rise in (7-84)-PTH. On the basis of previous findings confirming antagonistic effects of (7–84) and full-length PTH, we can hypothesize that (7-84)-PTH may be able to attenuate the process of calcification, speculatively via sirtuins. The main argument in favor of such a possibility is that (7-84)-PTH in contrast to full-length PTH predominantly operates through a C-terminal PTH receptor and may involve another downstream pathway.34 Nevertheless, we must acknowledge inability to determine a cause-effect relationship, which renders our conclusions of limited value. Whether sirtuin 1 expression in VSMCs is regulated independently by (7-84)-PTH needs further explanatory research. Even though, the proportion of PTH fragments definitely could not be translated into VC risk burden. One should bear in mind that in renal patients high serum calcification propensity derives from alteration in the whole mineral metabolism. For instance, it is more pronounced in maintenance dialysis patients owing to the use of calcium-containing phosphate binders.35
The positive correlation between sirtuin 1 and age, demonstrated in our study, is consistent with a report presented by Kilic et al.,36 who noted significantly higher serum sirtuin 1 levels in older people compared with both children and adults. However, the study did not assess kidney function, hence a lack of relation after adjustment for kidney function cannot be ruled out. The authors also suggested that raised protein level of SIRT1 parallel to decreased protein activity in older people serves as a compensatory mechanism to declined NAD+ levels associated with aging and accumulation of oxygen reactive species.36–39 Thus, sirtuin 1 level may better reflect biological than chronological age, which is supported by the disappearance of the correlation after adjustment in our study. We also demonstrated that sirtuin 1 concentration was significantly lower in patients treated with several drugs. In line with our findings, a study conducted on patients with coronary artery disease showed initially increased sirtuin 1 expression, reduced after statin therapy implementation.40 In contrast, Yamaç et al. reported an increase in circulating sirtuin 1 expression after initiation of statin therapy in patients after myocardial infarction.41 To our knowledge, only one paper investigated the impact of AT1-receptor blockers on sirtuin 1 levels.42 The study was performed on middle-aged, healthy men, who started the treatment with valsartan or the combination of valsartan and fluvastatin. One month after commencement of the therapy serum sirtuin 1 concentration increased in both subgroups compared with baseline. Regarding the effect of β-blockers use on sirtuin 1 levels in humans the data are lacking, hence we refer to preclinical studies. Metoprolol, similarly to valsartan, was found to activate in vitro SIRT1/AMPK pathway and subsequently ameliorate arterial remodeling.43,44
With regard to sirtuin 1 concentration, our results did not reveal any association with left over clinical measurements, the etiology of CKD, concomitant diseases and survival probability during follow-up. In our study, sirtuin 1 was able to discriminate with high sensitivity and specificity between CKD and control group.
The limitations of our study include a relatively small number of patients, enrolled exclusively in the Nephrology Department, which resulted in predominance of subjects with average and severely impaired kidney function, who may not be representative of kidney disease in the general population. The causality of demonstrated associations cannot be determined due to the cross-sectional nature of the study. Since this study primarily was designed to investigate circulating sirtuin 1 in relation to kidney function, we were not able to assess its association to VC. Given the complexity of sirtuin 1 biology its level may be influenced by genetic variation and various individual factors, including coexisting conditions and use of drugs, therefore studies on more homogenous cohorts are needed.
Conclusions
In summary, our findings showed substantially elevated sirtuin 1 concentration in CKD patients versus control group. Association between sirtuin, eGFR equations and abnormal mineral metabolism indicates a possible usefulness of sirtuin 1 as a kidney function or indirectly cardiovascular marker. Accumulation of sirtuin 1 may be related to impaired kidney function, however in terms of iPTH being the only independent predictor of circulating sirtuin 1 it can be considered similarly to PTH as a cardiovascular risk biomarker regardless of renal function and provide additional information for patient management. Despite the limitations it makes interesting points in the discussion about sirtuin 1 and CKD-related VC and alternative explanations of our results should be considered. To a great extent, sirtuin 1 is recognized as protective against vascular disease, and in our study on patients prone to VC we showed a positive correlation between sirtuin 1 and iPTH, which may be related to accumulation of (7-84)-PTH having opposite biological effects to full-length PTH. However, further studies are needed for expanding the knowledge on circulating sirtuin 1 in regard to VC and its predictive value for related cardiovascular risk and mortality.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agreed to be accountable for all aspects of the work.
Funding
This study was supported by a study grant from the Medical University of Bialystok (Project No: N/ST/MN/18/001/1186).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Liu M, Li XC, Lu L, et al. Cardiovascular disease and its relationship with chronic kidney disease. Eur Rev Med Pharmacol Sci. 2014;18(19):2918–2926.
2. Go AS, Chertow GM, Fan D, McCulloch CE, Hsu C. Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization. N Engl J Med. 2004;351(13):1296–1305. doi:10.1056/NEJMoa041031
3. Gao D, Zuo Z, Tian J, et al. Activation of SIRT1 attenuates klotho deficiency-induced arterial stiffness and hypertension by enhancing AMP-activated protein kinase activity. Hypertension. 2016;68(5):1191–1199. doi:10.1161/HYPERTENSIONAHA.116.07709
4. Haigis MC, Guarente LP. Mammalian sirtuins–emerging roles in physiology, aging, and calorie restriction. Genes Dev. 2006;20(21):2913–2921. doi:10.1101/gad.1467506
5. Hasegawa K, Wakino S, Simic P, et al. Renal tubular Sirt1 attenuates diabetic albuminuria by epigenetically suppressing Claudin-1 overexpression in podocytes. Nat Med. 2013;19(11):1496–1504. doi:10.1038/nm.3363
6. Tagawa A, Yasuda M, Kume S, et al. Impaired podocyte autophagy exacerbates proteinuria in diabetic nephropathy. Diabetes. 2016;65(3):755–767. doi:10.2337/db15-0473
7. Li J, Qu X, Ricardo SD, Bertram JF, Nikolic-Paterson DJ. Resveratrol inhibits renal fibrosis in the obstructed kidney: potential role in deacetylation of Smad3. Am J Pathol. 2010;177(3):1065–1071. doi:10.2353/ajpath.2010.090923
8. Hariharan N, Maejima Y, Nakae J, Paik J, Depinho RA, Sadoshima J. Deacetylation of FoxO by Sirt1 plays an essential role in mediating starvation-induced autophagy in cardiac myocytes. Circ Res. 2010;107(12):1470–1482. doi:10.1161/CIRCRESAHA.110.227371
9. Zhang QJ, Wang Z, Chen HZ, et al. Endothelium-specific overexpression of class III deacetylase SIRT1 decreases atherosclerosis in apolipoprotein E-deficient mice. Cardiovasc Res. 2008;80(2):191–199. doi:10.1093/cvr/cvn224
10. Mattagajasingh I, Kim CS, Naqvi A, et al. SIRT1 promotes endothelium-dependent vascular relaxation by activating endothelial nitric oxide synthase. Proc Natl Acad Sci. 2007;104(37):14855 LP– 14860. doi:10.1073/pnas.0704329104
11. Gao P, Xu TT, Lu J, et al. Overexpression of SIRT1 in vascular smooth muscle cells attenuates angiotensin II-induced vascular remodeling and hypertension in mice. J Mol Med. 2014;92(4):347–357. doi:10.1007/s00109-013-1111-4
12. Doulamis IP, Tzani AI, Konstantopoulos PS, et al. A sirtuin 1/MMP2 prognostic index for myocardial infarction in patients with advanced coronary artery disease. Int J Cardiol. 2017;230:447–453. doi:10.1016/j.ijcard.2016.12.086
13. Fathy SA, Ibrahim DM, Elkhayat WA, Ahmed HS. Association between serum sirt 1 and advanced glycation end products levels in type 2 diabetic nephropathy patients. Int J Biosci. 2017;10:398–404.
14. Shao Y, Ren H, Lv C, Ma X, Wu C, Wang Q. Changes of serum Mir-217 and the correlation with the severity in type 2 diabetes patients with different stages of diabetic kidney disease. Endocrine. 2017;55(1):130–138. doi:10.1007/s12020-016-1069-4
15. Zbroch E, Bazyluk A, Malyszko J, et al. The serum concentration of anti-aging proteins, sirtuin1 and αklotho in patients with end-stage kidney disease on maintenance hemodialysis. Clin Interv Aging. 2020;15:387–393. doi:10.2147/CIA.S236980
16. Ugur S, Ulu R, Dogukan A, et al. The renoprotective effect of curcumin in cisplatin-induced nephrotoxicity. Ren Fail. 2015;37(2):332–336. doi:10.3109/0886022X.2014.986005
17. He W, Wang Y, Zhang MZ, et al. Sirt1 activation protects the mouse renal medulla from oxidative injury. J Clin Invest. 2010;120(4):1056–1068. doi:10.1172/JCI41563
18. Laflamme MH, Mahjoub H, Mahmut A, et al. Parathyroid hormone is associated with the LV mass after aortic valve replacement. Heart. 2014;100(23):1859–1864. doi:10.1136/heartjnl-2014-305837
19. Schiffl H, Lang SM. Hypertension secondary to PHPT: cause or coincidence? Int J Endocrinol. 2011;2011:974647. doi:10.1155/2011/974647
20. Pepe J, Cipriani C, Curione M, et al. Reduction of arrhythmias in primary hyperparathyroidism, by parathyroidectomy, evaluated with 24-h ECG monitoring. Eur J Endocrinol. 2018;179(2):117–124. doi:10.1530/EJE-17-0948
21. Fujii H. Association between parathyroid hormone and cardiovascular disease. Ther Apher Dial. 2018;22(3):236–241. doi:10.1111/1744-9987.12679
22. Hagström E, Hellman P, Larsson TE, et al. Plasma parathyroid hormone and the risk of cardiovascular mortality in the community. Circulation. 2009;119(21):2765–2771. doi:10.1161/CIRCULATIONAHA.108.808733
23. Yuen NK, Ananthakrishnan S, Campbell MJ. Hyperparathyroidism of renal disease. Perm J. 2016;20(3):15–127. doi:10.7812/TPP/15-127
24. Dhayat NA, Ackermann D, Pruijm M, et al. Fibroblast growth factor 23 and markers of mineral metabolism in individuals with preserved renal function. Kidney Int. 2016;90(3):648–657. doi:10.1016/j.kint.2016.04.024
25. Weaver DJ, Mitsnefes M. Cardiovascular disease in children and adolescents with chronic kidney disease. Semin Nephrol. 2018;38(6):559–569. doi:10.1016/j.semnephrol.2018.08.002
26. Civilibal M, Caliskan S, Adaletli I, et al. Coronary artery calcifications in children with end-stage renal disease. Pediatr Nephrol. 2006;21(10):1426–1433. doi:10.1007/s00467-006-0159-6
27. Smajilovic S, Schaal-Jensen R, Jabbari R, Smajilovic U, Haunso S, Tfelt-Hansen J. Effect of intermittent versus continuous parathyroid hormone in the cardiovascular system of rats. Open Cardiovasc Med J. 2010;4(1):110–116. doi:10.2174/1874192401004010110
28. Nakajima K, Nohtomi K, Sato M, Takano K, Sato K. PTH(7-84) inhibits PTH(1-34)-induced 1,25-(OH)2D3 production in murine renal tubules. Biochem Biophys Res Commun. 2009;381(2):283–287. doi:10.1016/j.bbrc.2009.02.023
29. Kakuta T, Ishida M, Fukagawa M. Critical governance issue of parathyroid hormone assays and its selection in the management of chronic kidney disease mineral and bone disorders. Ther Apher Dial. 2018;22(3):220–228. doi:10.1111/1744-9987.12690
30. Souberbielle JC, Brazier F, Piketty ML, Cormier C, Minisola S, Cavalier E. How the reference values for serum parathyroid hormone concentration are (or should be) established? J Endocrinol Invest. 2017;40(3):241–256. doi:10.1007/s40618-016-0553-2
31. Shuichi J, Yoshiki N, Atsushi S, Hirotoshi M. Parathyroid hormone–related peptide as a local regulator of vascular calcification. Arterioscler Thromb Vasc Biol. 1997;17(6):1135–1142. doi:10.1161/01.ATV.17.6.1135
32. Shao JS, Cheng SL, Charlton-Kachigian N, Loewy AP, Towler DA. Teriparatide (human parathyroid hormone (1-34)) inhibits osteogenic vascular calcification in diabetic low density lipoprotein receptor-deficient mice. J Biol Chem. 2003;278(50):50195–50202. doi:10.1074/jbc.M308825200
33. Neves KR, Graciolli FG, Reis LM, et al. Vascular calcification: contribution of parathyroid hormone in renal failure. Kidney Int. 2007;71(12):1262–1270. doi:10.1038/sj.ki.5002241
34. Carrillo-López N, Panizo S, Alonso-Montes C, et al. High-serum phosphate and parathyroid hormone distinctly regulate bone loss and vascular calcification in experimental chronic kidney disease. Nephrol Dial Transplant. 2018;34(6):934–941. doi:10.1093/ndt/gfy287
35. Di Iorio B, Bellasi A, Russo D. Mortality in kidney disease patients treated with phosphate binders: a randomized study. CJASN. 2012;7(3):487–493. doi:10.2215/CJN.03820411
36. Kilic U, Gok O, Erenberk U, et al. A remarkable age-related increase in SIRT1 protein expression against oxidative stress in elderly: SIRT1 gene variants and longevity in human. PLoS One. 2015;10(3):e0117954–e0117954. doi:10.1371/journal.pone.0117954
37. Braidy N, Guillemin GJ, Mansour H, Chan-Ling T, Poljak A, Grant R. Age related changes in NAD+ metabolism oxidative stress and Sirt1 activity in wistar rats. PLoS One. 2011;6(4):e19194–e19194. doi:10.1371/journal.pone.0019194
38. Ramsey KM, Mills KF, Satoh A, Imai SI. Age-associated loss of Sirt1-mediated enhancement of glucose-stimulated insulin secretion in beta cell-specific Sirt1-overexpressing (BESTO) mice. Aging Cell. 2008;7(1):78–88. doi:10.1111/j.1474-9726.2007.00355.x
39. Kim MY, Zhang T, Kraus WL. Poly(ADP-ribosyl)ation by PARP-1: ‘PAR-laying’ NAD+ into a nuclear signal. Genes Dev. 2005;19:(17):1951–1967. doi:10.1101/gad.1331805
40. Kilic U, Gok O, Elibol-Can B, Uysal O, Bacaksiz A. Efficacy of statins on sirtuin 1 and endothelial nitric oxide synthase expression: the role of sirtuin 1 gene variants in human coronary atherosclerosis. Clin Exp Pharmacol Physiol. 2015;42(4):321–330. doi:10.1111/1440-1681.12362
41. Yamaç AH, Kılıç Ü. Effect of statins on sirtuin 1 and endothelial nitric oxide synthase expression in young patients with a history of premature myocardial infarction. Turk Kardiyol Dern Ars. 2018;46(3):205–215. doi:10.5543/tkda.2018.32724
42. Janić M, Lunder M, Novaković S, Škerl P, Šabovič M. Expression of longevity genes induced by a low-dose fluvastatin and valsartan combination with the potential to prevent/treat aging-related disorders. Int J Mol Sci. 2019;20(8):1844. doi:10.3390/ijms20081844
43. Sun L, Yan S, Wang X, et al. Metoprolol prevents chronic obstructive sleep apnea-induced atrial fibrillation by inhibiting structural, sympathetic nervous and metabolic remodeling of the atria. Sci Rep. 2017;7(1):14941. doi:10.1038/s41598-017-14960-2
44. Ha YM, Park EJ, Kang YJ, Park SW, Kim HJ, Chang KC. Valsartan independent of AT1 receptor inhibits tissue factor, TLR-2 and −4 expression by regulation of Egr-1 through activation of AMPK in diabetic conditions. J Cell Mol Med. 2014;18(10):2031–2043. doi:10.1111/jcmm.12354
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
