Back to Journals » Clinical, Cosmetic and Investigational Dermatology » Volume 14
Sequelae After Involution of Superficial Infantile Hemangioma: Early Intervention with 595-nm Pulsed Laser Combined with 755-nm Long-Pulsed Alexandrite Laser versus Wait-and-See
Authors Jiang JC , Xu Q, Fang S, Gao Y, Jin WW
Received 29 August 2020
Accepted for publication 15 October 2020
Published 12 January 2021 Volume 2021:14 Pages 37—43
DOI https://doi.org/10.2147/CCID.S279140
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Jeffrey Weinberg
Ji-Cong Jiang,1,* Qin Xu,2,* Shan Fang,3 Yu Gao,1 Wan-Wan Jin1
1Department of Dermatology, The Second Affiliated Hospital and Yuying Children’s Hospital of Wenzhou Medical University, Wenzhou 325027, Zhejiang Province, People’s Republic of China; 2The Second School of Medicine, Wenzhou Medical University, Wenzhou 325027, Zhejiang Province, People’s Republic of China; 3Institute of Photomedicine, Shanghai Skin Disease Hospital, Tongji University School of Medicine, Shanghai 200443, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Wan-Wan Jin; Yu Gao Email [email protected]; [email protected]
Background and Objective: Infantile haemangioma (IH) is the most common benign tumor in children. At present, pulsed dye laser (PDL) has made great progress in the treatment of superficial IH, showing good safety and effectiveness. But some doctors think that superficial IH should choose to wait-and-see. However, studies have reported that most of the IH after resolution still has residual disease, and thickness seems to be an important factor. Therefore, the purpose of this study is to investigate the relationship between Sequelae and thickness after superficial IH involution. In addition, compare the Sequelae difference between 595-nm pulsed laser combined with 755-nm long-pulse alexandrite laser treatment and wait-and-see.
Materials and Methods: This retrospective observational study included patients with superficial IH evaluated in the past 6 years and divided them into a laser group and an observation group.
Results: The incidence of sequelae in the laser group was 44.6%, and the incidence of sequelae in the observation group was 69.5%. The incidence of Sequelae of superficial IH in the laser group was significantly lower than that in the observation group (χ2-test, χ2=10.790, P < 0.001). In the observation group, the average A scores of the three thickness subgroups (< 2mm, 2– 5mm, and > 5mm) were 4.38, 3.39, and 1.80, and there were significant differences in the A scores between the three groups (Kruskal–Wallis, p< 0.05). There is a significant difference in the A score between the laser group and the observation group in the superficial IH with a thickness of 2– 5 mm and> 5mm (Wilcoxon rank sum test, P< 0.05).
Conclusion: This retrospective study showed that the degree of Sequelae of superficial IH after involution is related to its thickness. In addition, the early intervention of 595-nm pulsed laser combined with 755-nm long-pulse alexandrite laser can reduce the incidence and extent of sequelae.
Keywords: pulsed dye laser, alexandrite laser, infantile hemangiomas, sequelae, wait-and-see, thickness
Introduction
Infant hemangioma (IH) is the most common benign tumor in children, and the incidence in infants is about 5%.1 They are characterized by rapid proliferation in the early stages, and then spontaneous involution. Higher incidence in premature babies, women, twins and Caucasians.2 IH is usually divided into superficial, deep or mixed types, superficial is the main type.3 At present, pulsed dye laser (PDL) has made great progress in the treatment of superficial IH, showing excellent safety and effectiveness.4–6 But some doctors believe that superficial IH should choose to wait-and-see. Although the lesions are usually self-limiting, residual lesions may still occur in some cases, such as telangiectasia, residual fibers or fatty tissue, atrophy, discoloration, anatomical deformation, or hair loss.7,8 These residual lesions are permanent and will not subside spontaneously, representing aesthetic and social problems, so surgery is often required.9 It is reported that more than half of superficial IH will have permanent Sequelae after involution, and thickness seems to be the main influencing factor.7,9 Therefore, determining the relationship between thickness and the risk of permanent Sequelae after involution is crucial in treatment decisions. In addition, studies have shown that early pulsed dye laser intervention for superficial IH can improve the aesthetic effect,10 but the relevant data is very limited. Therefore, the purpose of this study is to investigate the relationship between Sequelae and thickness after superficial IH involution. Besides, compare the Sequelae difference between 595-nm pulsed laser combined with 755-nm long-pulse alexandrite laser treatment and wait-and-see.
Materials and Methods
In this retrospective observational study, we included patients with superficial IH who were evaluated in the past 6 years (from January 2014 to January 2020). These patients were evaluated in the department of Dermatology, the Second Affiliated Hospital and Yuying Children’s Hospital of Wenzhou Medical University.
Patients
The inclusion criteria of the observation group are as follows: (i) patients with superficial IH diagnosed as uncomplicated within 12 months of age (based on clinical history, physical examination and imaging); (ii) not receiving any treatment; (iii) Complete available information. The exclusion criteria are as follows: (i) patients with IH ulcers before treatment; (ii) patients who received other treatments later.
The inclusion criteria of the laser treatment group are as follows: (i) patients with superficial IH diagnosed as uncomplicated within 12 months of age (based on clinical history, physical examination and imaging); (ii) did not receive any treatment and selected Receive laser treatment; (iii) Complete available information. The exclusion criteria are as follows: (i) patients with IH ulcers before treatment; (ii) patients who received other treatments later.
We extract data on demographics, thickness and location of IH, management, follow-up, and results from each eligible case (Table 1).
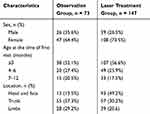 |
Table 1 Baseline Characteristics at Time of Inclusion |
Procedures
This study was approved by the independent ethics committee of the Second Affiliated Hospital of Wenzhou Medical University and Yuying Children’s Hospital. We have obtained the prior consent of all patient guardians to keep the photos in our files for medical purposes. And we have obtained written informed consent from all patient guardians before participating in the study.
Observation group: Follow-up was conducted by first reaching out to patients’ parents via telephone interview. Patient’s basic information such as gender, birthday, lesion type, and lesion site were confirmed. Requests for a follow-up visit to the hospital were initiated and obtain follow-up images of the lesion.
Laser group: Before treatment, the family members explained the risks of treatment, expected results and postoperative care information, and obtained written informed consent. Collected data about infant patients, including name, age, gender, time of first visit, home address and contact number, and previous treatment history. Use the same camera to take pictures for comparison before and after treatment. Before treatment, 5% lidocaine was applied to the infant for local anesthesia for 0.5–2.0 hours. According to the patient’s age and IH position, color, thickness, select the appropriate parameters of the treatment device, pulse length, energy, spot and interval time. During treatment, a brief gray or dark purple is considered the best treatment endpoint and can be observed 1 or 2 minutes after the start of treatment. Give cold compresses (ice cubes) for 15–30 minutes to reduce pain and swelling after surgery. Then, fusidic acid cream is used to prevent infection and adverse reactions for 1 week, with a 4-week interval between treatments. Instruct patients to avoid exposure to water and sunlight within 1 week after surgery, and to reduce strenuous exercise. Before each treatment, a photo was taken to obtain an image of the lesion.
All images were independently evaluated by three individual surgeons, blinded to this study. The image of the lesion taken at the first visit was used as a control.
Assessment
Superficial IH with a size reduction of 95–100% is classified as complete involution. In superficial IH with complete involution, the following aesthetic scores (A score) are obtained according to the degree of Sequelae: No obvious Sequelae, the A score is rated as 5; Mild telangiectasia, pigmentation or hypopigmentation, the A score is rated as 4; Obvious telangiectasia, hyperpigmentation or hypopigmentation, the A score is rated as 3; Mild fibrofatty residue, anetoderma or scar, the A score is rated as 2; Obvious fibrofatty residue, anetoderma or scar, the A score is rated as 1 (Figure 1).
Statistical Analysis
Use SPSS Statistics (version23.0; IBM Company, Armonk NY, USA) to analyze the data. Chi-square test was used to compare the incidence of Sequelae between the laser group and the treatment group, and the significance level was P <0.05; the Kruskal–Wallis test was used to compare the A score between the thickness subgroups of the observation group, and the significance level was P <0.05; Wilcoxon rank sum test was used to compare the A score between the laser treatment group and the observation group, and the significance level was P <0.05.
Results
In the observation group, 73 patients participated in the follow-up and sent follow-up images of the lesions, 19 of which had multiple superficial IH. In the end, 96 cases of superficial IH and follow-up imaging were included in the study. The ratio of men to women is 1:1.81. There were 13 (13.5%) lesions on the head and neck, 55 (57.5%) lesions on the trunk, and 28 (29.2%) lesions on the limbs. Among 96 lesions, 59 superficial IH (61.5%) completely involution. Among them, 18 superficial IH (30.5%) had a A score of 5; 15 superficial IH (25.4%) had a A score of 4; 13 superficial IH (22.0%) had a A score of 3; 10 superficial IH (16.9%) had a A score of 2; 3 superficial IH (5.1%) had a A score of 1. In the laser treatment group, 147 patients participated in the follow-up, 34 of whom had multiple IHs. In the end, 189 cases of IH and subsequent images were included in the study. The ratio of men to women is 1:2.77. There were 93 (49.2%) lesions on the head and neck, 57 (30.2%) lesions on the trunk, and 39 (20.6%) lesions on the limbs. Among 189 lesions, 168 superficial IH (88.9%) completely involution. Among them, 93 superficial IH (55.4%) had a A score of 5; 40 superficial IH (23.8%) had a A score of 4; 22 superficial IH (13.1%) had a A score of 3; 8 superficial IH (4.8%) had a A score of 2; 5 superficial IH (3.0%) had a A score of 1. We summarize the A score of the observation group and laser group (Table 2).
 |
Table 2 The A Score of the Observation Group and Laser Group |
The incidence of Sequelae of superficial IH in the laser group was significantly lower than that in the observation group (χ2-test, χ2=10.790, P <0.001). In the observation group, the average A scores of the three thickness subgroups (<2mm, 2–5mm, >5mm) were 4.38, 3.39, 1.80, and there were significant differences in the A scores between the three groups (Kruskal–Wallis, p<0.05). There is no significant difference between the laser group and the observation group in the superficial IH with thickness <2mm (Wilcoxon rank sum test, P>0.05). There is a significant difference in the A score between the laser group and the observation group in the superficial IH with a thickness of 2–5 mm (Wilcoxon rank sum test, P<0.05). There is a significant difference in the A score between the laser group and the observation group in superficial IH (Wilcoxon rank sum test, P<0.05). We summarize the mean value of A score is in different thickness subgroups (Table 3).
 |
Table 3 The Mean Value of A Score is in Different Thickness Subgroups |
Discussion
Infantile hemangioma (IH) is the most common benign tumor in children, and superficial IH is the main type of IH. For superficial IH, the current practice recommends for a wait-and-see approach, and actually are those that might lead to permanent lesions.7,11 Studies have reported that more than half of untreated IH has not completely involution, and there residual Sequelae lesions in IH after involution.8,9,12 These Sequelae lesions include telangiectasia, skin atrophy, excess skin, scars or fibrofatty tissue. This retrospective study showed that 39.5% of superficial IH in the observation group did not completely involution. Among superficial IH that completely resolved, 69.5% had at least one residual lesion. The most common residual lesions are telangiectasia, excess fibrofatty tissue and skin. Usually, residual lesions show a combination of multiple characteristics. Therefore, the aesthetic score is used to evaluate the degree of Sequelae. Thickness is an important factor affecting the degree of Sequelae. In the observation group, the average A scores of the three thickness subgroups (<2mm, 2–5mm, >5mm) were 4.38, 3.39, 1.80, and there were significant differences. (Kruskal–Wallis test, P <0.001). How to reduce the incidence of Sequelae and improve the degree of Sequelae to obtain a higher aesthetic score is a problem we need to face. And pulsed dye laser seems to be a good choice.13 At present, the main treatment methods for infantile hemangioma include systemic or local external use of propranolol, local injection of drugs, surgery and laser treatment, etc.14 Oral propranolol is currently the first-line treatment for IH,15 but it is not the best treatment for local superficial IH. The external use of propranolol can be used for superficial and thin IH, but has limited effect on some thicker IH.16 Surgical treatment will inevitably leave scars, and the recurrence rate is not low, so it is rarely used at present. Local injection of drugs is more suitable for deeper local IH, because it may cause skin or soft tissue atrophy.17,18 Laser therapy is suitable for local superficial IH, and has good safety and effectiveness.19 It has become a research hotspot. In 1983, Anderson proposed the selective light theory,20 which is a precedent for the use of lasers to treat skin vascular diseases. In the field of IH therapy, the most widely used laser device is the pulsed dye laser (PDL), which has a wavelength between 585 and 595 nm. Many studies have confirmed the efficacy of 595-nm PDL, especially for the treatment of superficial IH.21,22 However, it is reported that it is not very effective for deep or thick IH, and deep components may continue to proliferate.23 Because the penetration depth of PDL is only 1.2 mm, it may only be 1/10 of the thickness of some IH.24 In order to overcome this shortcoming, the 755 nm long pulse alexandrite laser has been proved to be effective in the treatment of refractory port-wine stain (PWS). Because it can penetrate deeper skin than PDL.24 Recent studies have shown that it has shown good safety and effectiveness in the treatment of IH.25,26 YAG laser can also be used to treat relatively thick or deep hemangioma,27 but its treatment range is narrow, its operation is not easy to control, it is easy to damage the deep dermal tissue and form scars. Therefore, for thicker superficial IH, the 755-nm long pulse alexandrite laser may be a more ideal choice.28
This retrospective study laser group used 595 nm pulsed dye laser combined with 755 nm long pulse alexandrite laser therapy6 In this study, 88.9IH% of the laser group completely involution, and 45.2% of the completely involution IH had at least one residual lesion. The incidence of Sequelae in the laser group was 45.2%, significantly lower than the incidence of Sequelae in the observation group (χ2-test, χ2=10.790, P <0.001). This shows that laser can reduce the incidence of Sequelae. In addition, the average AR score of the laser group is also higher than that of the observation group, which shows that the laser can reduce the degree of Sequelae.
However, we have observed that the degree of reduction in Sequelae is different in different thickness groups. In the thickness group of <2mm, the average AR score of the observation group is 4.38, and the average score of the laser group A is 4.64. Although the average score of the laser group A has improved, there is no significant difference (Wilcoxon rank sum test, P >0.05). This indicates that in superficial IH with a thickness of less than 2mm, there is no significant difference in A score between the observation group and the laser group. This may be because the Sequelae of superficial IH with a thickness of less than 2mm are rarely left or the Sequelae are relatively minor, so the effect of laser intervention is not obvious. However, laser treatment can help IH complete the involution early29 We need to note that IH may increase the psychosocial morbidity of parents and children, especially hemangioma located on the face.30 Therefore, it may be necessary to help the hemangioma to involution before the child enters school, which may be helpful to the child’s psychology. In superficial IH with a thickness of 2–5mm, the average AR score in the laser group was 4.31, and the average A score in the observation group was 3.39. The A score of the laser group was significantly higher than that of the observation group. This shows that in superficial IH with a thickness of 2–5mm, laser intervention can significantly reduce the degree of Sequelae (Wilcoxon rank sum test, P <0.05). This may be that in superficial IH with a thickness of 2–5mm, the probability of Sequelae is increased, and the degree of Sequelae is more serious. And laser intervention can not only reduce telangiectasia,31 but also promote the deposition of adipose tissue and correct anatomical aberrations through the thermally induced lysis of collagen, thereby promoting tissue remodeling.32–34 In superficial IH with thickness >5mm, the mean A score of the laser group was also significantly higher than the mean AR score of the observation group (Wilcoxon rank sum test, P<0.05). But the average AR score of the laser group was only 2.59. This shows that in superficial IH with thickness >5mm, although laser intervention can reduce the degree of Sequelae, it still inevitably leaves more obvious Sequelae.
The above data indicate that we can choose wait-and-see for superficial IH with a thickness of less than 2mm, because the final aesthetic score is not much different from laser intervention. However, laser intervention can help IH involution early. Therefore, the superficial IH located on the face can choose laser intervention to help IH involution early and avoid psychological problems caused to children and parents. For superficial IH with a thickness of 2 to 5 mm, laser intervention is necessary, not only to promote the early involution of IH but also to significantly reduce the Sequelae after involution and improve the aesthetic score. Laser intervention is equally effective for superficial IH thicker than 5mm, but Sequelae are inevitable.
Limitations
There are several limitations to this study. First of all, compared with the treatment group, the number of cases in the Observation group is limited. There are numeric differences between the two groups. Second, the visual assessment of thickness may not always be accurate. Finally, since this is a retrospective study, there is no systematic follow-up, so some IH may continue to degenerate after we consider it to be the final photo.
Conclusions
This retrospective study showed that the degree of Sequelae of superficial IH after involution is related to its thickness. In addition, the early intervention of 595-nm pulsed laser combined with 755-nm long-pulse alexandrite laser can reduce the incidence of Sequelae of superficial IH regression. Besides can reduce the degree of Sequelae, especially in superficial IH thicker than 2mm. The information provided by our study may help clinicians and families in predicting the risk of Sequelae and in the treatment decision-making process.
Acknowledgment
This study is supported by the National Natural Science Foundation of China (81801862).
Disclosure
The authors report no conflicts of interest for this work.
References
1. Munden A, Butschek R, Tom W, et al. Prospective study of infantile haemangiomas: incidence, clinical characteristics and association with placental anomalies. Br J Dermatol. 2014;170(4):907–913. doi:10.1111/bjd.12804
2. Smith C, Friedlander S, Guma M, et al. Infantile hemangiomas: an updated review on risk factors, pathogenesis, and treatment. Birth Defects Res. 2017;109(11):809–815. doi:10.1002/bdr2.1023
3. Chang L, Haggstrom A, Drolet B, et al. Growth characteristics of infantile hemangiomas: implications for management. Pediatrics. 2008;122(2):360–367. doi:10.1542/peds.2007-2767
4. Hartmann F, Lockmann A, Grönemeyer L, et al. Nd: YAG and pulsed dye laser therapy in infantile haemangiomas: a retrospective analysis of 271 treated haemangiomas in 149 children. J Eur Acad Dermatol Venereol. 2017;31(8):1372–1379. doi:10.1111/jdv.14074
5. Chen W, Yang C, Liu S, et al. Curative effect study of pulsed dye laser in the treatment of 43 patients with hand infantile hemangioma. Eur J Dermatol. 2014;24(1):76–79. doi:10.1684/ejd.2013.2223
6. Jin W, Tong Y, Wu J, et al. Observation on the effects of 595- nm pulsed dye laser and 755- nm long-pulsed alexandrite laser on sequential therapy of infantile hemangioma. J Cosmet Laser Ther. 2020;22(3):159–164. doi:10.1080/14764172.2020.1783452
7. Bauland C, Lüning T, Smit J, et al. Untreated hemangiomas: growth pattern and residual lesions. Plast Reconstr Surg. 2011;127(4):1643–1648. doi:10.1097/PRS.0b013e318208d2ac
8. Chang S, Qiao C, Chang L, et al. A 7-year follow-up study on untreated deep or mixed facial infantile hemangioma in East-Asian patients: when propranolol was not yet an option. J Dermatol. 2019;46(11):962–966. doi:10.1111/1346-8138.15080
9. Baselga E, Roe E, Coulie J, et al. Risk factors for degree and type of sequelae after involution of untreated hemangiomas of infancy. JAMA Dermatol. 2016;152(11):1239–1243. doi:10.1001/jamadermatol.2016.2905
10. Kessels J, Hamers E, Ostertag J. Superficial hemangioma: pulsed dye laser versus wait-and-see. Dermatol Surg. 2013;39(3pt1):414–421. doi:10.1111/dsu.12081
11. Chelleri C, Monzani N, Gelmetti C, et al. Residual lesions after pharmacological and dye-laser treatment of infantile hemangiomas: critical review of 432 cases. Lasers Surg Med. 2020;52(7):597–603. doi:10.1002/lsm.23205
12. Tognetti L, Pianigiani E, Ierardi F, et al. A new clinical and dermoscopic monitoring of infantile hemangiomas treated with oral propranolol. Dermatol Ther. 2020:e14283. doi:10.1111/dth.14283.
13. Valdebran M, Martin B, KJSicm K, et al. State-of-the-art lasers and light treatments for vascular lesions: from red faces to vascular malformations. Semin Cutan Med Surg. 2017;36(4):207–212. doi:10.12788/j.sder.2017.044
14. Krowchuk D, Frieden I, Mancini A, et al. Clinical practice guideline for the management of infantile hemangiomas. Pediatrics. 2019;143(1):e20183475. doi:10.1542/peds.2018-3475
15. Solman L, Glover M, Beattie P, et al. Oral propranolol in the treatment of proliferating infantile haemangiomas: British society for paediatric dermatology consensus guidelines. Br J Dermatol. 2018;179(3):582–589. doi:10.1111/bjd.16779
16. Price A, Rai S, Mcleod R, et al. Topical propranolol for infantile haemangiomas: a systematic review. J Eur Acad Dermatol Venereol. 2018;32(12):2083–2089. doi:10.1111/jdv.14963
17. Qiu Y, Lin X, Ma G, et al. Eighteen cases of soft tissue atrophy after intralesional bleomycin a5 injections for the treatment of infantile hemangiomas: a long-term follow-up. Pediatr Dermatol. 2015;32(2):188–191. doi:10.1111/pde.12422
18. Couto J, Greene A. Management of problematic infantile hemangioma using intralesional triamcinolone: efficacy and safety in 100 infants. J Plast Reconstr Aesthet Surg. 2014;67(11):1469–1474. doi:10.1016/j.bjps.2014.07.009
19. Chinnadurai S, Sathe N, Surawicz T, et al. Laser treatment of infantile hemangioma: a systematic review. Lasers Surg Med. 2016;48(3):221–233. doi:10.1002/lsm.22455
20. Anderson R, Parrish JJS. Selective photothermolysis: precise microsurgery by selective absorption of pulsed radiation. Science. 1983;220(4596):524–527. doi:10.1126/science.6836297
21. Chen W, Liu S, Yang C, et al. Clinical efficacy of the 595 nm pulsed dye laser in the treatment of childhood superficial hemangioma - analysis of 10-year application in Chinese patients. J Dermatol Treat. 2015;26(1):54–58. doi:10.3109/09546634.2013.806979
22. Ying H, Zou Y, Yu W, et al. Prospective, open-label, rater-blinded and self-controlled pilot study of the treatment of proliferating superficial infantile hemangiomas with 0.5% topical timolol cream versus 595-nm pulsed dye laser. J Dermatol. 2017;44(6):660–665. doi:10.1111/1346-8138.13747
23. Poetke M, Philipp C, Berlien H. Flashlamp-pumped pulsed dye laser for hemangiomas in infancy: treatment of superficial vs mixed hemangiomas. Arch Dermatol. 2000;136(5):628–632. doi:10.1001/archderm.136.5.628
24. Zide B, Levine SM. Hemangioma update: pearls from 30 years of treatment. Ann Plast Surg. 2012;69(1):99–103. doi:10.1097/SAP.0b013e3182212881
25. Su W, Ke Y, Xue J, et al. Beneficial effects of early treatment of infantile hemangiomas with a long-pulse Alexandrite laser. Lasers Surg Med. 2014;46(3):173–179. doi:10.1002/lsm.22221
26. Li D, Zhang H, Chen B, et al. Experimental investigations on thermal effects of a long-pulse alexandrite laser on blood vessels and its comparison with pulsed dye and Nd: YAG lasers. Lasers Med Sci. 2020;35(7):1555–1566. doi:10.1007/s10103-020-02981-9
27. Vlachakis I, Gardikis S, Michailoudi E, et al. Treatment of hemangiomas in children using a Nd: YAG laser in conjunction with ice cooling of the epidermis: techniques and results. BMC Pediatr. 2003;3(1):2. doi:10.1186/1471-2431-3-2
28. Feng H, Kauvar A. Successful treatment of a residual, thick, infantile hemangioma in a darker phototype pediatric patient using the 755-nm long-pulsed alexandrite laser. Dermatol Surg. 2017;43(12):1514–1516. doi:10.1097/dss.0000000000001144
29. Rizzo C, Brightman L, Chapas A, et al. Outcomes of childhood hemangiomas treated with the pulsed-dye laser with dynamic cooling: a retrospective chart analysis. Dermatol Surg. 2009;35(12):1947–1954. doi:10.1111/j.1524-4725.2009.01356.x
30. Park J, Kang S, Choi T. Statistical analysis of influences on the psychosocial status of children with hemangiomas and their families. Arch Craniofac Surg. 2017;28(8):2056–2059. doi:10.1097/scs.0000000000003982
31. Ross E, Meehan K, Domankevitz Y, et al. Use of a variable long-pulse alexandrite laser in the treatment of facial telangiectasia. Dermatol Surg. 2010;36(4):470–474. doi:10.1111/j.1524-4725.2010.01475.x
32. Rosenthal A, Kolli H, Israilevich R, et al. Lasers for the prevention and treatment of hypertrophic scars: a review of the literature. J Cosmet Laser Ther. 2020;22(3):115–125. doi:10.1080/14764172.2020.1783451
33. Ouyang H, Li G, Lei Y, et al. Comparison of the effectiveness of pulsed dye laser vs pulsed dye laser combined with ultrapulse fractional CO2 laser in the treatment of immature red hypertrophic scars. J Cosmet Dermatol. 2018;17(1):54–60. doi:10.1111/jocd.12487
34. Kuo Y, Jeng S, Wang F, et al. Flashlamp pulsed dye laser (PDL) suppression of keloid proliferation through down-regulation of TGF-beta1 expression and extracellular matrix expression. Lasers Surg Med. 2004;34(2):104–108. doi:10.1002/lsm.10206
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

