Back to Journals » Clinical Interventions in Aging » Volume 14
Reticulocyte levels have an ambivalent association with hypertension and atherosclerosis in the elderly: a cross-sectional study
Authors Shimizu Y , Kawashiri SY, Yamanashi H, Koyamatsu J, Fukui S, Kondo H , Tamai M, Nakamichi S, Maeda T
Received 12 December 2018
Accepted for publication 15 March 2019
Published 8 May 2019 Volume 2019:14 Pages 849—857
DOI https://doi.org/10.2147/CIA.S197982
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Richard Walker
Yuji Shimizu,1,2 Shin-Ya Kawashiri,1 Hirotomo Yamanashi,3,4 Jun Koyamatsu,3 Shoichi Fukui,1 Hideaki Kondo,1,4 Mami Tamai,1 Seiko Nakamichi,4 Takahiro Maeda1,3–4
1Department of Community Medicine, Nagasaki University Graduate School of Biomedical Sciences, Nagasaki, Japan; 2Department of Cardiovascular Disease Prevention, Osaka Center for Cancer and Cardiovascular Disease Prevention, Osaka, Japan; 3Department of Island and Community Medicine, Nagasaki University Graduate School of Biomedical Sciences, Nagasaki, Japan; 4Department of General Medicine, Nagasaki University Graduate School of Biomedical Sciences, Nagasaki, Japan
Purpose: Age-related reduction in bone marrow activity has been shown to cause anemia, and hypertension and endothelial dysfunction (atherosclerosis) are age-related diseases. However, recent studies have revealed a close association between bone marrow activity and endothelial maintenance. This study aimed to determine the association between elevated reticulocyte levels in conjunction with vigorous bone marrow activity and hypertension and atherosclerosis among the elderly.
Study population and Methods: To determine the associations between reticulocyte levels and hypertension and atherosclerosis, we conducted a cross-sectional study of 2,098 elderly Japanese individuals, aged between 60 and 89 years, who had participated in an annual health check-up in 2014.
Results: Of the total study population, 1,348 individuals were diagnosed with hypertension (systolic blood pressure ≥140 mmHg and/or diastolic blood pressure ≥90 mmHg and/or having used antihypertensive medication), and 393 were diagnosed with atherosclerosis (carotid intima-media thickness ≥1.1 mm). Reticulocyte levels were found to be significantly positively associated with hypertension and inversely associated with atherosclerosis. Cardiovascular risk factor-adjusted odds ratios and 95% confidence intervals for hypertension and atherosclerosis, when raised incrementally by 1 standard deviation to determine reticulocyte levels (5.5×104 cells/μL for men and 5.0×104 cells/μL for women), were 1.12 (1.01, 1.25) and 0.83 (0.72, 0.94), respectively.
Conclusion: Along with established cardiovascular risk factors, reticulocyte levels in elderly Japanese individuals were found to be positively associated with hypertension and inversely associated with atherosclerosis. This finding may help clarify the background mechanisms concerning the association between bone marrow activity and vascular remodeling.
Keywords: atherosclerosis, hypertension, reticulocyte
Introduction
Recent studies have reported a close association between bone marrow activity and endothelial maintenance.1,2 The process of aging results in reduced bone marrow activity, which is associated with age-related anemia.3–5 Recent studies have indicated that erythropoietic activity, estimated according to reticulocyte levels, could be an indicator of endothelial maintenance capacity among elderly individuals.
Moreover, previous studies have reported a positive association between hemoglobin and hypertension,6–8 indicating that hypertension might be positively associated with hemoglobin production that could be determined using reticulocyte levels. Since hypertension is a recognized factor in endothelial injury and, given that hypertension may promote a maximum production capability of hematopoietic stem cells (CD34-positive cells) in the elderly,9 a positive association between hypertension and hemoglobin production suggests that hypertension stimulates endothelial repair activity.
Furthermore, aggressive endothelial repair is a recognized cause of atherosclerosis.10,11 Therefore, it is possible to establish a bidirectional association between hypertension and atherosclerosis-hypertension that induces endothelial dysfunction (atherosclerosis) and vice versa.12 We considered that a positive association between hypertension and atherosclerosis would likely be observed in our study population.
Aging has been reported to be associated with low-grade inflammation,13 which can result in age-related diseases such as atherosclerosis.14 Since low-grade inflammation has been reported to be associated with anemia15 and low hemoglobin,16 aggressive endothelial repair that results in atherosclerosis could be associated with lower hemoglobin production.
In this study, we hypothesized that, for elderly Japanese individuals, reticulocyte levels would be positively associated with hypertension, that hypertension would be positively associated with atherosclerosis, and that reticulocyte levels would be inversely associated with atherosclerosis.
To confirm our hypothesis, we conducted a cross-sectional study of elderly Japanese individuals, between 60 and 89 years old, who had participated in an annual health check-up in 2014.
Materials and methods
Study population
The study population comprised 2,374 elderly residents aged 60–89 years from the city of Goto and the town of Saza in western Japan, who underwent an annual medical check-up in 2014, as recommended by the Japanese government.
Participants for whom data were not available for body mass index (BMI) (n=1), blood pressure (n=2), carotid intima-media thickness (CIMT) (n=3), or laboratory findings (n=270) were excluded from the study. The remaining 2,098 participants with a mean age of 70.1 years (standard deviation (SD), 6.3; range, 60–89 years) were enrolled.
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institution research committee and with the 1964 Helsinki Declaration, and its later amendments for comparable ethical standards. This study was approved by the Ethics Committee of Nagasaki University Graduate School of Biomedical Sciences (project registration number 14051404). Written consent forms were available in Japanese to ensure comprehensive understanding of the study objectives, and informed consent was provided by the participants.
Data collection and laboratory measurements
Body weight and height were measured with an automatic body composition analyzer (BF-220; Tanita, Tokyo, Japan), and BMI (kg/m2) was calculated. Trained examiners used a blood pressure measuring device (HEM-907; Omron, Kyoto, Japan) to measure and record systolic and diastolic blood pressure of the right arm after at least 5 mins of rest with the participant in a seated position. Hypertension was defined as systolic blood pressure ≥140 mmHg and/or diastolic blood pressure ≥90 mmHg and/or the use of antihypertensive medication.
Fasting blood samples were collected in a heparin sodium tube, an EDTA-2K tube and a siliconized tube. Concentrations of triglyceride (TG), HDL-cholesterol (HDL-C), γ-glutamyltranspeptidase (γ-GTP), hemoglobin A1c (HbA1c), and creatinine were measured with standard laboratory procedures. All measurements were performed at SRL, Inc. (Tokyo, Japan). The glomerular filtration rate (GFR) was estimated with an established method recently adapted and introduced by a working group of the Japanese Chronic Kidney Disease Initiative,17 which yielded an estimate of GFR (ml/min/1.73 m2)=194×(serum creatinine (enzyme method))−1.094×(age)−0.287×(0.739 for women).
Quantities of white blood cells (WBC), platelets, red blood cells (RBC), and reticulocytes in samples from the EDTA-2K tube were measured using an automated procedure at SRL, Inc. Reticulocyte levels were determined using the following formula: reticulocytes (×104 cells/μL)=(reticulocytes, ‰)×RBC (×104 cells/μL)/1,000. (for men: 5.45±1.77 [×104 cells/μL] and for women: 5.45±1.77 [×104 cells/μL]).
Ultrasonography was conducted to measure the carotid intima-media thickness (CIMT) of the left and right common carotid arteries by an experienced vascular technician using a LOGIQ Book XP with a 10-MHz transducer (GE Healthcare, Milwaukee, WI, USA). Maximum values for the left and right CIMT were calculated with automated digital edge-detection software (Intimascope; MediaCross, Tokyo, Japan) following a protocol that has been described in detail elsewhere.18 This software automatically identifies the edges of the internal and external membranes of the blood vessels, and can also determine automatically distance at a sub-pixel level (estimated to be 0.01 mm which measures CIMT at a 10×higher axial resolution) using a polynomial measurement formula.19 The right and left CIMT values, excluding plaque measurements, were then calculated and the maximum left or right CIMT value was used for analysis. Since a previous study reported a normal CIMT value as <1.1 mm, we defined atherosclerosis as a CIMT value of ≥1.1 mm.20
Statistical analysis
Characteristics of the study population in relation to reticulocyte levels and hypertension status were expressed as mean ± standard deviation for contentious variables, and as prevalence for medication status. A trend test was performed using a regression model for mean values.
Logistic regression models were used to calculate odds ratios (ORs) and 95% confidence intervals (CIs) to determine associations between hypertension and atherosclerosis. Logistic regression models were also used to determine the association of reticulocytes with hypertension and atherosclerosis.
The present study focused on reticulocyte levels, hypertension, and atherosclerosis. Since recent studies have identified a close association between vascular remodeling and bone marrow activity,1,2 and age-related anemia is due to a reduction in bone marrow activity,3–5 these factors influencing both bone marrow activity and vascular remodeling (atherosclerosis) were assumed to be confounding factors for the present analysis.
BMI status is a determinant factor for the association between hemoglobin and hypertension,7 as well as for an increase in arterial stiffness.21 Furthermore, BMI status has been shown to influence the association between HbA1c and atherosclerosis,22 while glycated hemoglobin may be associated with erythrocyte turnover.23 BMI and HbA1c were considered confounding factors in the present analysis. Moreover, renal function is strongly associated with anemia24 and atherosclerosis,10 while TG can stimulate vascular repair.25 Furthermore, the activity of vascular remodeling (including atherosclerosis progression) can influence the association between blood pressure levels and TG26 and HDL-C.27 Therefore, GFR, TG, and HDL-C were considered important confounding factors in our study.
Factors such as alcohol consumption and smoking status influence vascular remodeling. Alcohol consumption influences γ-GTP and smoking status influences the WBC;28 therefore, we included γ-GTP and WBC as confounding factors rather than considering these factors directly as in a previous study.29
In addition, since a previous study reported that systolic but not diastolic hypertension is associated with an increase in atherosclerosis,30 we used systolic blood pressure rather than diastolic blood pressure as a confounding factor for the analysis to determine the association between reticulocyte levels and atherosclerosis.
Therefore, three different approaches were used to make adjustments for confounding factors. First, adjustment was made only for sex and age (Model 1). For Model 2, we included other potential confounding factors, namely TG (mg/dL), HDL-C (mg/dL), HbA1c (%), γ-GTP (U/L), WBC (cells/μL), and GFR (ml/min/1.73 m2), as well as systolic blood pressure (mmHg) for analysis of the relationship between reticulocyte levels and atherosclerosis. Finally, Model 3 was further adjusted for BMI (kg/m2). Continuous variables were used for all confounding factors.
All statistical analyses were performed using the SAS system for Windows (version 9.4; SAS Inc., Cary, NC). p-values of <0.05 were regarded as statistically significant.
Results
Characteristics of the study population
Characteristics of the study population in relation to reticulocyte levels are shown in Table 1. Systolic blood pressure, diastolic blood pressure, antihypertensive medication use, BMI, TG, γ-GTP, HbA1c, GFR, WBC, platelets, and RBC were positively associated with reticulocyte levels, while age and HDL-C showed an inverse association.
 | Table 1 Characteristics of the study population based on absolute values of reticulocyte levels |
Characteristics of the study population in relation to hypertension status are shown in Table 2. Compared to the non-hypertension group, the hypertension group showed significantly higher number of men; significantly higher values for age, systolic blood pressure, diastolic blood pressure, BMI, TG, γ-GTP, HbA1c, WBC, platelet, RBC, and reticulocyte levels; and significantly lower HDL-C and GFR levels.
 | Table 2 Characteristics of the study population based on hypertension |
Association between hypertension and reticulocytes
A positive association was found between hypertension and reticulocyte levels, as shown in Table 3, with the association remaining unchanged even after further adjustment for cardiovascular risk factors (Model 2). However, even though the statistical power remained significant, the association became weaker after further adjustment for BMI (Model 3).
 | Table 3 Odds ratios (ORs) and 95% confidence intervals (CIs) for hypertension in relation to reticulocyte levels |
Association between hypertension and atherosclerosis
As shown in Table 4, hypertension was significantly positively associated with atherosclerosis. This association and its degree remained unchanged, even after further adjustments for cardiovascular risk factors including BMI (Model 2, Model 3).
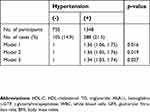 | Table 4 Odds ratios (ORs) and 95% confidence intervals (CIs) for atherosclerosis in relation to hypertension |
Association between atherosclerosis and reticulocyte count
As shown in Table 5, atherosclerosis was found to be inversely associated with reticulocyte levels. This association remained unchanged even after further adjustments for known cardiovascular risk factors including BMI (Model 2, Model 3).
 | Table 5 Odds ratios (ORs) and 95% confidence intervals (CIs) for atherosclerosis in relation to reticulocyte levels |
Sensitivity analysis
We evaluated sex-specific associations with hypertension and found these to be similar to our overall results. The age-adjusted ORs and 95% CIs for hypertension and atherosclerosis, when raised incrementally by 1 SD to determine reticulocyte levels (5.5×104 cells/μL for men and 5.0×104 cells/μL for women) were 1.34 (1.15, 1.59) and 0.83 (0.70, 0.98) for men, respectively, and 1.33 (1.18, 1.50) and 0.96 (0.81, 1.14) for women, respectively.
Discussion
In addition to established cardiovascular risk factors, reticulocyte levels were shown to be significantly positively associated with hypertension and were significantly inversely associated with atherosclerosis.
Bone marrow activity has recently been shown to be closely associated with vascular maintenance, since hematopoietic stem cells derived from bone marrow reportedly play a major role in vascular homeostasis.1,2 Hematopoietic bone marrow activity declines with age,3–5 resulting in anemia in elderly individuals. Therefore, erythropoietic activity determined according to reticulocyte levels may indicate the endothelial maintenance capacity of elderly individuals.
Hypertension has been reported to injure the endothelium, and hypertension may also influence the maximum production capability of hematopoietic stem cells;9 therefore, hypertension could be associated with erythropoietic activity as indicated through the level of endothelial repair activity occurring among elderly individuals. Hemoglobin has been found to be positively associated with hypertension,7 which may be due to a mechanism leading to disruption of the microcirculation resulting from insufficient endothelial repair.8 We found a significant positive association between reticulocyte levels and hypertension.
The platelet count can act as an indicator of vascular maintenance capacity.31 Therefore, our additional analysis that showed the platelet count was significantly positively associated with reticulocyte levels (sex and age-adjusted partial correlation coefficient (r)=0.10, p<0.001); this observation supports the aforementioned mechanism.
Aging is a process that has been reported to be associated with oxidative stress.32 Recently, it has been hypothesized that oxidative stress may function as a key factor in the pathogenesis of hypertension.33 Since oxidative stress also induces hematopoiesis, including that of the erythroid lineage,34 reticulocyte levels could be positively associated with hypertension through indicating oxidative stress activity.
Obesity is known to be closely associated with states of oxidative stress.35 Our analysis showed that the statistical power of this association remained significant, even when the degree of association between reticulocyte levels and hypertension became weaker after adjustment for BMI.
Hypertension has been reported to induce the progression of atherosclerosis and vice-versa.12 In our study, we found a significant positive association between hypertension and atherosclerosis. We also found a significant inverse association between reticulocyte and atherosclerosis.
Aging is recognized as a factor associated with oxidative stress32,36 and is also associated with chronic low-grade inflammation.13,15 Furthermore, aging is also known to be one of the main risk factors for the development of atherosclerosis.37,38
Since low-grade inflammation is reported to be associated with anemia,15 low hemoglobin levels16 and atherosclerosis,14 aggressive endothelial repair that causes atherosclerosis could be associated with lower hemoglobin production, as indicated by low-grade inflammation.
Furthermore, hemodynamic disturbances due to increased oxidative stress, premature cellular senescence, and impairment of synthesis and/or secretion of endothelium-derived vasoactive molecules, cause age-related structural and functional changes to the vasculature;38 therefore, age-related oxidative stress and inflammation can be expected to lead to the development of atherosclerosis.
Erythrocytes are an important component of the antioxidant capacity of blood,39 and activation of erythropoiesis can cause reduction in oxidative stress.40 Since reticulocytes are immature erythrocytes, an increase in the level of reticulocytes may indicate an increase in antioxidant activity that prevents atherosclerotic progression while, at the same time, an increase in antioxidant activity may be induced through oxidative stress and inflammation, which are related to hypertension. This process could explain our findings, in which reticulocyte levels showed a significantly positive association with hypertension, as well as showing an inverse association with atherosclerosis.
Furthermore, endothelial dysfunction has been recognized as one of the initial mechanisms leading to glomerular injury (reduced GFR) and atherosclerosis.41 Reduced renal function, a risk factor associated with anemia, is known as renal anemia. In our study, renal function (GFR) was considered a confounding factor. However, GFR showed a positive association with reticulocyte levels (Table 1) and, when compared to participants without hypertension, those with hypertension showed significant lower GFR values (Table 2). These associations also supported our results in relation to similar associations between reticulocyte levels and hypertension, and between reticulocyte levels and atherosclerosis.
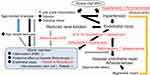
Figure 1 shows a summary of the possible mechanisms accounting for our results. Given age-related reduction in bone marrow activity is a well-recognized cause of anemia in elderly individuals,3 reticulocyte levels might also be influenced through age-related reduction in bone marrow. Low-grade inflammation, positively associated with aging,13 has also been reported to be positively associated with anemia15 and low hemoglobin levels.16 Therefore, an increase in reticulocyte levels may indicate that the influence of age-related reduction in bone marrow activity and chronic low-grade inflammation on bone marrow activity is not severe. Based on this phenomenon, hypertension might promote reticulocyte production through stimulating endothelial repair activity, while elevated reticulocyte levels might have a beneficial influence on endothelium maintenance. Therefore, an ambivalent association between reticulocyte levels and on age-related diseases, such as hypertension and atherosclerosis, was shown.
This study has some limitations that warrant consideration. First, while erythrocytes reportedly play an important role in anti-oxidative stress,39,40 no data concerning oxidative stress were available. Further investigations using data such as those for reactive oxygen species, superoxide dismutase, and catalase are necessary. Chronic low-grade inflammation might also play an important role in associations identified;13,15,16 however, no data on chronic low grade inflammation were available. Further investigations with high-sensitivity C-reactive protein and soluble tumor necrosis factor-αreceptor 2 are necessary. Our results could also have been influenced due to hemorrhagic anemia because hemorrhagic anemia stimulates reticulocyte production. However, this study was a general population study. Therefore, we consider that the influence of hemorrhagic anemia was likely to have been limited. Furthermore, because this was a cross-sectional study, causal relationships could not be established.
Conclusion
In conclusion, our study revealed that, in addition to well established cardiovascular risk factors, reticulocyte levels were significantly positively associated with hypertension and significantly inversely associated with atherosclerosis in the elderly. These results may help clarify the background mechanisms concerning age-related endothelial activity.
Acknowledgments
We are grateful to city council staff from Goto and from town council staff from Saza for their outstanding support. This study was supported by Grants-in-Aids for Scientific Research from the Japan Society for the Promotion of Sciences (No. 15K07243, No. 17H03740, and No. 18K06448).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Takahashi T, Kalka C, Masuda H, et al. Ischemia- and cytokine-induced mobilization of bone marrow-derived endothelial progenitor cells for neovascularization. Nat Med. 1999;5:434–438. doi:10.1038/7434
2. Shi Q, Rafii S, Wu MH, et al. Evidence for circulating bone marrow-derived endothelial cells. Blood. 1998;92:362–367.
3. Guralnik JM, Ershler WB, Schrier SL, et al. Anemia in the elderly: a public health crisis in hematology. Hematology Am Soc Hematol Educ Program. 2005;2005:528–532. doi:10.1182/asheducation-2005.1.528
4. Brusnahan SK, McGuire TR, Jackson JD, et al. Human blood and marrow side population stem cell and Stro-1 positive bone marrow stromal cell numbers decline with age, with an increase in quality of surviving stem cells: correlation with cytokines. Mech Ageing Dev. 2010;131:718–722. doi:10.1016/j.mad.2010.10.002
5. Garvin K, Feschuk C, Sharp JG, et al. Does the number or quality of pluripotent bone marrow stem cells decrease with age? Clin Orthop Relat Res. 2007;465:202–207. doi:10.1097/BLO.0b013e318159a9b8
6. Atsma F, Veldhuizen I, de Kort W, et al. Hemoglobin level is positively associated with blood pressure in a large cohort of healthy individuals. Hypertension. 2012;60:936–941. doi:10.1161/HYPERTENSIONAHA.112.193565
7. Shimizu Y, Nakazato M, Sekita T, et al. Association between the hemoglobin levels and hypertension in relation to the BMI status in a rural Japanese population: the Nagasaki Islands Study. Intern Med. 2014;53:435–440.
8. Shimizu Y, Sato S, Koyamatsu J, et al. Possible mechanism underlying the association between higher hemoglobin level and hypertension in older Japanese men. Geriatr Gerontol Int. 2017;17:2586–2592. doi:10.1111/ggi.13068
9. Shimizu Y, Sato S, Koyamatsu J, et al. Height is an indicator of vascular maintenance capacity in older men. Geriatr Gerontol Int. 2017;17:1729–1736. doi:10.1111/ggi.12876
10. Shimizu Y, Yamanashi H, Noguchi Y, et al. Association between chronic kidney disease and carotid intima-media thickness in relation to circulating CD34-positive cell count among community-dwelling elderly Japanese men. Atherosclerosis. 2019;283:85–91. doi:10.1016/j.atherosclerosis.2019.02.004
11. Shimizu Y, Yamanashi H, Noguchi Y, et al. Cardio-ankle vascular index and circulating CD34-positive cell levels as indicator of endothelial repair activity in older Japanese men. Geriatr Gerontol Int. Epub 2019 Mar 28. doi 10.1111/ggi.13657
12. Shimizu Y, Sato S, Koyamatsu J, et al. Platelets and circulating CD34-positive cells as an indicator of the activity of the vicious cycle between hypertension and endothelial dysfunction in elderly Japanese men. Atherosclerosis. 2017;259:26–31. doi:10.1016/j.atherosclerosis.2017.02.016
13. Payne GW. Effect of inflammation on the aging microcirculation: impact on skeletal muscle blood flow control. Microcirculation. 2006;13:343–352. doi:10.1080/10739680600618918
14. Jain S, Khera R, Corrales-Medina VF, Townsend RR, Chirinos JA. Inflammation and arterial stiffness in humans. Atherosclerosis. 2014;237:381–390. doi:10.1016/j.atherosclerosis.2014.09.011
15. Artz AS, Xue QL, Wickrema A, et al. Unexplained anaemia in the elderly is characterized by features of low grade inflammation. Br J Haematol. 2014;16:286–289. doi:10.1111/bjh.12984
16. Kotzé SR, Pederson OB, Petersen MS, et al. Low-grade inflammation is associated with lower haemoglobin levels in healthy individuals: results from the Danish blood donor study. Vox Sang. 2016;111:144–150. doi:10.1111/vox.12396
17. Imai E, Horio M, Watanabe T, et al. Prevalence of chronic kidney disease in the Japanese general population. Clin Exp Nephrol. 2009;13:621–630. doi:10.1007/s10157-009-0199-x
18. Hara T, Takamura N, Akashi S, et al. Evaluation of clinical markers of atherosclerosis in young and elderly Japanese adults. Clin Chem Lab Med. 2006;44:824–829. doi:10.1515/CCLM.2006.149
19. Yanase T, Nasu S, Mukuta Y, et al. Evaluation of a new carotid intima-media thickness measurement by B-mode ultrasonography using an innovative measurement software, intimascope. Am J Hypertens. 2006;19:1206–1212. doi:10.1016/j.amjhyper.2006.05.010
20. Kawamori R, Yamasaki Y, Matsushima H, et al. Prevalence of carotid atherosclerosis in diabetic patients. Ultrasound high-resolution B-mode imaging on carotid arteries. Diabetes Care.. 1992;15:1290–1294.
21. Shimizu Y, Nakazato M, Sekita T, et al. Association between hemoglobin levels and arterial stiffness for general Japanese population in relation to body mass index status: the Nagasaki Islands study. Geriatr Gerontol Int. 2014;14:811–818. doi:10.1111/ggi.12171
22. Shimizu Y, Nakazato M, Sato S, et al. Association between hemoglobin A1c and carotid atherosclerosis in rural community-dwelling elderly Japanese men. J Physiol Anthropol. 2015;34:16. doi:10.1186/s40101-015-0054-6
23. Beltran Del Rio M, Tiwari M, Amodu LI, Cagliani J, Rodriguez Rilo HL. Glycated hemoglobin, plasma glucose, and erythrocyte aging. J Diabetes Sci Technol. 2016;10:1303–1307. doi:10.1177/1932296816659885
24. Shimizu Y, Sato S, Koyamatsu J, et al. Associations between renal impairment and anemia in older, rural Japanese men: the Nagasaki Islands study. J Physiol Anthropol. 2014;33:7. doi:10.1186/1880-6805-33-7
25. Shimizu Y, Sato S, Koyamatsu J, et al. Circulating CD34-positive cells, glomerular filtration rate and triglycerides in relation to hypertension. Atherosclerosis. 2015;243:71–76. doi:10.1016/j.atherosclerosis.2015.08.035
26. Shimizu Y, Sato S, Noguchi Y, et al. Triglycerides and blood pressure in relation to circulating CD34-positive cell levels among community-dwelling elderly Japanese men: a cross-sectional study. Environ Health Prev Med. 2017;22:77. doi:10.1186/s12199-017-0684-x
27. Shimizu Y, Sato S, Koyamatsu J, et al. Association between high-density lipoprotein-cholesterol and hypertension in relation to circulating CD34-positive cell levels. J Physiol Anthropol. 2017;36:26. doi:10.1186/s40101-017-0143-9
28. Kawada T. Relationships between the smoking status and plasma fibrinogen, white blood cell count and serum C-reactive protein in Japanese workers. Diabetes Metab Syndr. 2015;9:180–182. doi:10.1016/j.dsx.2015.02.010
29. Shimizu Y, Sato S, Koyamatsu J, et al. Possible mechanism underlying the association between height and vascular remodeling in elderly Japanese men. Oncotarget. 2018;9:7749–7757. doi:10.18632/oncotarget.23660
30. Salonen R, Salonen JT. Carotid atherosclerosis in relation to systolic and diastolic blood pressure: Kuopio Ischaemic Heart Disease Risk Factor Study. Ann Med. 1991;23:23–27.
31. Shimizu Y, Sato S, Koyamatsu J, et al. Platelets as an indicator of vascular repair in elderly Japanese men. Oncotarget. 2016;7:44919–44926. doi:10.18632/oncotarget.10229
32. Liochev S. Reflections on the theories of aging, of oxidative stress, and of science in general. Is it time to abandon the free radical (oxidative stress) theory of aging? Antioxid Redox Signal. 2015;23:187–207. doi:10.1089/ars.2014.5928
33. Baradaran A, Nasri H, Rafieian-Kopaei M. Oxidative stress and hypertension: possibility of hypertension therapy with antioxidants. J Res Med Sci. 2014;19:358–367.
34. Zhao JL, Baltimore D. Regulation of stress-induced hematopoiesis. Curr Opin Hematol. 2015;22:286–292. doi:10.1097/MOH.0000000000000149
35. Suzuki K, Ito Y, Ochiai J, et al.;
36. Satoh T, Yamakage M, Satoh J, Namiki A. [Effect of aging on preoperative oxidative stress] (Article in Japanese) Masui. Masui. 2007;56:1422–1424.
37. Yildiz O. Vascular smooth muscle and endothelial functions in aging. Ann N Y Acad Sci. 2007;1100:353–360. doi:10.1196/annals.1395.038
38. Ghebre YT, Yakubov E, Wong WT, Krishnamurthy P. Vascular aging: implications for cardiovascular disease and therapy. Transl Med (Sunnyvale). 2016;6:
39. Grune T, Sommerburg O, Siems WG. Oxidative stress in anemia. Clin Nephrol. 2000;53:S18–S22.
40. Noguchi-Sasaki M, Sasaki Y, Matsuo-Tezuka Y, et al. Reduction of a marker of oxidative stress with enhancement of iron utilization by erythropoiesis activation following epoetin beta pegol administration in iron-loaded db/db mice. Int J Hematol. 2016;103:262–273. doi:10.1007/s12185-015-1929-3
41. Endemann DH, Schiffrin EL. Endothelial dysfunction. J Am Soc Nephrol. 2004;15:1983–1992. doi:10.1097/01.ASN.0000132474.50966.DA
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.