Back to Journals » Drug Design, Development and Therapy » Volume 14
Repurposing Anti-Cancer Drugs for COVID-19 Treatment
Authors Borcherding N, Jethava Y, Vikas P
Received 16 September 2020
Accepted for publication 29 October 2020
Published 18 November 2020 Volume 2020:14 Pages 5045—5058
DOI https://doi.org/10.2147/DDDT.S282252
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Tuo Deng
Nicholas Borcherding,1– 4 Yogesh Jethava,1,5 Praveen Vikas1,5
1Holden Comprehensive Cancer Center, University of Iowa, College of Medicine, Iowa City, IA, USA; 2Department of Pathology, University of Iowa, College of Medicine, Iowa City, IA, USA; 3Cancer Biology Graduate Program, University of Iowa, College of Medicine, Iowa City, IA, USA; 4Medical Scientist Training Program, University of Iowa, College of Medicine, Iowa City, IA, USA; 5Department of Internal Medicine, University of Iowa, College of Medicine, Iowa City, IA, USA
Correspondence: Praveen Vikas
Holden Comprehensive Cancer Center, 5983 JPP, 200 Hawkins Drive, Iowa City, Iowa 52242, USA
Tel + 1 319-356-2757
Email [email protected]
Abstract: The novel coronavirus disease 2019 (COVID-19) pandemic has caused catastrophic damage to human life across the globe along with social and financial hardships. According to the Johns Hopkins University Coronavirus Resource Center, more than 41.3 million people worldwide have been infected, and more than 1,133,000 people have died as of October 22, 2020. At present, there is no available vaccine and a scarcity of efficacious therapies. However, there is tremendous ongoing effort towards identifying effective drugs and developing novel vaccines. Early data from Adaptive COVID-19 Treatment Trials (ACTT) sponsored by the National Institute of Allergy and Infectious Diseases (NIAID) and compassionate use study have shown promise for remdesivir, leading to emergency authorization by the Food and Drug Administration (FDA) for treatment of hospitalized COVID-19 patients. However, several randomized studies have now shown no benefit or increased adverse events associated with remdesivir treatment. Drug development is a time-intensive process and requires extensive safety and efficacy evaluations. In contrast, drug repurposing is a time-saving and cost-effective drug discovery strategy geared towards using existing drugs instead of de novo drug discovery. Treatments for cancer and COVID-19 often have similar goals of controlling inflammation, inhibiting cell division, and modulating the host microenvironment to control the disease. In this review, we focus on anti-cancer drugs that can potentially be repurposed for COVID-19 and are currently being tested in clinical trials.
Keywords: COVID-19, drug-repurposing, anti-cancer drugs
Introduction
In December of 2019, a novel coronavirus structurally related to the virus that causes severe acute respiratory syndrome (SARS) was identified as the cause of respiratory disease in Wuhan, a city in the Hubei Province of China and reported to the World Health Organization (WHO).1 On January 30, 2020, the WHO declared this outbreak from novel coronavirus as a public health emergency of international concern and, in March 2020, began to characterize it as a pandemic. The virus that causes COVID-19 is designated severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2); previously, it was referred to as 2019-nCoV. In February 2020, the World Health Organization designated the disease COVID-19.
At present, there is a paucity of therapies or vaccines with demonstrated efficacy for the treatment or prevention of COVID-19. Clinical management is currently focused on supportive care and prevention and management of complications like ARDS. Several available agents are being tested, and few have been provided for expanded access use. Remdesivir, a broad-based antiviral drug, an inhibitor of RNA-dependent RNA polymerase, was developed by Gilead Sciences originally to target Ebola and is being repurposed for COVID-19 in clinical trials and for compassionate use. Remdesivir showed promise in a compassionate-use program case series for patients with severe COVID-19, and clinical improvement was noted in 68% of patients treated, but there was no control group.2 Final data on from Adaptive COVID-19 Treatment Trials (ACTT) sponsored by NIAID found that patients who received remdesivir had faster time to recovery when compared to placebo.3 Remdesivir has subsequently been approved by the Food and Drug Administration (FDA) for emergency authorization based on several trials that showed benefits outweighing risks.4,5 Enthusiasm for a possible treatment modality for COVID-19 is also tempered by two cohorts of remdesivir-treated patients which found no associated clinical benefit for remdesivir.6,7 One of these cohorts, consisting of 237 randomized COVID-19 patients, was discontinued early due to adverse events at 12% compared to 5% of the placebo-treated arm.6 Other drugs being used and tested include HIV protease inhibitors like darunavir8 and lopinavir-ritonavir, which showed no efficacy in clinical improvements in a recent randomized control trial.9 Anti-inflammatory agents used for rheumatoid arthritis and glucocorticoids are also being tested, with the latter the first agent to show a reduction in mortality for COVID-19.10 There has been both excitement and controversy around antimalarial drugs being tested for COVID-19. Some of the early results from these drugs have been inconclusive. The FDA issued an emergency use authorization to allow the use of chloroquine and hydroxychloroquine, which has since been revoked based on trial results published that showed no effect on clinical course and elevated side effects.11–14 The antiparasitic drug ivermectin has shown in vitro activity against SARS-CoV-2.15 Convalescent plasma from recovered patients has also been a focus in the treatment of severe COVID-19 infection and is being studied in clinical trials and also being used outside of clinical trials.16,17
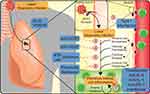
In order to address the clinical gap, the medical community is turning to repurposing of available antiviral, anti-inflammatory, and certain anti-cancer drugs in the context of individual patients, as well as initiating clinical trials exploring some of these available drugs. Given our expertise in anti-cancer treatments, we here focus on repurposing of anti-cancer drugs that have already been approved by regulatory agencies or are being fast-tracked and are supported by published data and being used in management of cancer patients. Drug repurposing is a valuable strategy in times of crisis like COVID-19 as it provides quick access to agents that not only have available safety data but also established production lines and supply chains.18 In addition, these therapies have early- and late-phase data on toxicity and managing complications, particularly advantageous in the pandemic setting versus novel therapies. Underscoring the potential of repurposing, estimates using the Covid19_db found 64.4% of trials for COVID-19 include repurposed drugs as of August 3, 2020.19 Several antineoplastic agents have the potential to improve outcomes of COVID-19 through some of the similar mechanisms utilized in management of cancer mostly pertaining to controlling inflammation, inhibiting cell division, and modulating the host–tumor microenvironment (Figure 1). In this review, we are discussing various anti-cancer agents currently being evaluated as a potential treatment option for COVID-19 (Table 1). As previously mentioned, a major antineoplastic therapy class, steroids, has recently shown efficacy in reducing the risk of death in ventilated patients (relative ratio 0.65) and patients receiving oxygen (relative ratio 0.8)10 compared to standard of care. As the first treatment to show reduction in mortality in COVID-19, steroids have been summarized previously20 and will not be the focus of this review.
 |  |  |
Table 1 NCT Trials Using Anti-Cancer Agents Currently Underway in COVID-19 as of 07/06/2020 |
Materials and Methods
All registered interventional clinical trials (n=1358) were obtained from clinicaltrials.gov on July 6, 2020. No search terms were used, but rather the tab-delimited summary of the curated COVID-19 section of clinicaltrials.gov was recovered. Trials without phase designations were removed. R (v3.6.3) was used to parse through the listed interventions for anti-neoplastic therapies as identified by Y.J. and P.V. Trials suspended or terminated were removed. Further review of trials by hand eliminated trials with the majority of non-FDA-approved therapies, with the exception of drugs relevant to the review, and the use of steroids for the management of COVID-19. The code used to filter and summarize the data is available at https:www.github.com/ncborcherding/COVID19review.
Pathophysiology and Clinical Course of COVID-19
The pathophysiology of the SARS-CoV-2 infection is an emerging field, but broadly it can be separated into three major categories: 1) lung pathology, 2) inflammation, and 3) thromboembolic events. Although separated conceptually, the interaction of these three categories form a backbone of COVID-19. Based on the WHO–China Joint Report, signs and symptoms of infection occur 5–6 days after transmission (range 1–14 days).21 Symptoms vary, but over the course of COVID-19 include fever (87.9%), dry cough (67.7%), fatigue (38.1%), sputum production (33.4%), and shortness of breath (18.6%).21 Although roughly 85% of patients had mild to moderate disease, 14% developed severe (6% critical) disease defined by dyspnea, hypoxemia, and radiographic evidence of lung pathology. Lung pathology in COVID-19 patients has revealed increased alveolar exudate caused by aberrant host immune response and inflammatory cytokine storm that leads to acute respiratory distress syndrome (ARDS), with alveolar gas exchange abnormality leading to mortality of severe COVID-19 patients. In the pathogenesis of SARS-CoV-2 pneumonia, a study found that a cytokine release syndrome (CRS) involving a considerable release of proinflammatory cytokines occurred, including IL-6, IL-12, and tumor necrosis factor α (TNF-α).22 The serum levels of IL-2R and IL-6 in patients with COVID-19 are positively correlated with the severity of the disease.23 In addition to interleukins, patients in the ICU displayed increased serum levels of G-CSF, CXCL3, MCP-1, CCL-3, and TNF-α relative to non-ICU COVID patients.23 Modulation of the infection–immune response axis is a key strategy in limiting the morbidity and mortality from COVID-19. Thromboembolic complications have been noted in the treatment of COVID-19. Observational studies have found rates ranging from 20.6% to 42.7%.24–26 Increases in circulating D-dimer, Von Willebrand factor, and Factor VIII were also observed in up to 87% of patients,25 and elevation in these parameters upon admission are predictive of poor survival.27 The mechanistic basis of the high incidence of thromboembolic events in COVID-19 is currently unknown, with some proposing a link to overall inflammation.28 Supporting this are studies finding correlating cytokines, like IL-6, with pro-coagulant profiles.29 This pro-coagulation phenotype might also be connected to the underlying lung injury, with histological analysis showing obliterative lung patterning with extensive intra-alveolar lung fibrin deposition and intraluminal connective tissue deposition.30
Anti-Cancer Drugs for COVID-19 Treatment
Interleukin Inhibitors: Targeting IL-6 and Other Cytokines
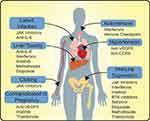
IL-6 or IL-6 receptor blocking antibodies like tocilizumab (Actemra), sarilumab (Kevzara), and siltuximab (Sylvant) are FDA-approved for various conditions including lymphoproliferative disorders, Castleman’s syndrome, and smoldering multiple myeloma. The IL-6 signaling pathway is being explored for targeted therapy for various malignancies.31 IL-6 signaling plays a role in the pathogenesis of multiple myeloma (MM), and disruption of IL-6 signaling is known to inhibit MM cell growth.32 IL-6 is among the key components of the inflammatory cascade responsible for host defense against several infections, but excessive IL-6 can lead to an acute severe systemic inflammatory response of CRS. IL-6 blockade has also been used in management of CRS,33 and tocilizumab is FDA-approved to manage CRS associated with the use of chimeric antigen receptor T cell therapy.34 The National Health Commission in China in March of 2020 included tocilizumab in their COVID-19 treatment guidelines for patients with severe lung damage and correspondingly high IL-6 levels (> 20 pg/mL).35 Monoclonal antibodies that target the IL-6 pathways can control the inflammatory storm (Figure 1), possibly by targeting highly inflammatory CD14+ CD16+ monocytes that have high expression of IL-6 in patients infected with COVID-19.36 Interestingly, increased levels of IL-6 have been associated with increased viral RNA load in the bloodstream,37 suggesting that the increased levels of IL-6 in COVID-19 patients may be preventing a proper immune response. Tocilizumab has shown promise in early results and can be an effective treatment in severelyill COVID-19 patients to mitigate the inflammatory storm; however, timing of anti-IL-6 induction may be important with reports of less effective results in criticallyill patients.38 The paper published by Fu et al reports preliminary data collected from 21 patients with COVID-19-induced ARDS treated with tocilizumab with persistence of fever associated with detectable levels of IL-6 after therapy.35 A secondary case report of a COVID-19 patient with multiple myeloma successfully treated with tocilizumab was recently reported.39 Interestingly, following administration of anti-IL-6 therapy, there was a transient decrease in CD8+ T cells and increase in CD4+ T cells, with decreased serum concentrations of IL-6.39 Another study found an overall increase in serum lymphocytes and decrease in C-reactive protein, a surrogate marker for overall inflammation, following tocilizumab induction.40 The reduction in C-reactive protein was also seen in a prospective cohort study from Italy, specifically in responders.41 This study also found the administration of tocilizumab led to stabilization or improvement in acute respiratory failure in 77% of patients administered but is limited by lack of control.41 Other direct inhibitors of cytokines are being evaluated in clinical trials for COVID-19, such as anakinra (NCT04341584, NCT04339712, NCT04357366, and NCT04324021), a peptide inhibitor of IL-1, and monoclonal antibodies targeting receptors or cytokines, such as IL-8 (NCT04347226). Anti-interleukin-based therapies for COVID-19 are one of the most common classes of drugs for interventional trials. However, these therapies likely have a narrow therapeutic window for effectiveness (ie, during the cytokine storm), should not be administered in patients with tuberculosis or other infections, and may lead to further complications in select patients. A retrospective cohort study found tocilizumab therapy to be associated with increased requirement for invasive ventilation, but shortened duration of vasopressor support,42 while a second study found no such link.43 Other limitations of monoclonal antibody therapy targeting interleukins for COVID-19 are the overall cost and availability of these agents and significant toxicity.
Janus-Associated Kinase (JAK) Inhibitors
JAK inhibitors like ruxolitinib are approved for primary myelofibrosis and polycythemia vera and are now showing promise in the treatment of COVID-19. JAK functions as a relay in cytokine signaling, promoting immune cell activation and survival genetic programs (Figure 1) with implications in the hyperreactivity of immune response to infection.44 Inhibition of JAK-STAT pathway can limit systemic inflammatory response and cytokine production. A series of phase 1/2/3 trials are underway investigating the use of JAK2 inhibitor ruxolitinib, for patients with hyperinflammation, CRS, or severe lung pathology subsequent to COVID-19. Preliminary data for patients with severe lung inflammation treated with ruxolitinib showed sustained reduction in inflammation in 11 of 14 patients treated, leading to the initiation of the NCT04338958 trial.45 Recently, Incyte has made ruxolitinib available through an expanded access program for patients with COVID-19 (NCT04355793). The JAK1/2 kinase inhibitor baricitinib is also being extensively trialed for COVID-19 infections and may be working through inhibition of endocytosis.46 Further research into antiviral properties of JAK inhibition has found them to work by inhibiting human immunodeficiency virus (HIV) replication in primary peripheral blood mononuclear cells,47 although this may be offset by inhibition of type I interferons downstream of JAK activation.48 Results of the completed phase 2/3 trial of baricitinib with the antivirals lopinavir-ritonavir are still forthcoming (NCT04358614); however, preliminary results showed a 2-week mortality rate of 0% in the treatment arm compared to 6.4% in the control arm of the study.49 In addition, the JAK1/3 inhibitor, thought to be upstream of IL-6 signaling,50 tofacitinib is being trialed for interstitial pneumonia related to SARS-CoV-2 infection (NCT04332042). The modulation of cytokine signaling may also alter the immune response, with the JAK2 inhibitor, fedratinib, potentially useful to move inflammation away from a Th-17-type of response.51 Currently no trials are registered evaluating the efficacy of fedratinib for COVID-19. Like the anti-interleukin therapies, JAK inhibitors should not be used in patients with concomitant secondary infections, like tuberculosis, and can increase the risk of upper respiratory infections (Figure 2). More critically, JAK inhibitors, such as tofacitinib and baricitinib, have black box warnings for associations with increased risk of blood clots, pulmonary emboli, and death. Due to the high incidence of thrombotic issues in COVID-19, particular caution should be taken for these particular therapies.
Interferons
Interferons can be classified by the major downstream signaling pathways elicited, with type I interferons (IFN-ɑ/β) vital in antiviral responses, type II interferons (IFN-γ) important in adaptive immune responses, and type III playing a role in fungal and viral response.52 Both type I and type III interferons can reduce the ability of viruses to infect host cells, but importantly there is a temporal relationship, with early interferons associated with improved viral clearance and late interferon signaling possibly potentiating infections.53 As a cytokine mediator, type II interferons are indicated in treatment of chronic myelogenous leukemia, hairy cell leukemia, melanoma, renal cell cancer, and Kaposi sarcoma. The use of types I and III interferons is being extensively studied for use in COVID-19 (Figure 1). The majority of type I interferons are being studied accompanying anti-microbial or antiviral agents, such as hydroxychloroquine, lopinavir, and ritonavir. Preliminary results show that the addition of IFN-β1b to ritonavir and ribavirin increased viral clearance as measured by nasopharyngeal swab.54 However, several studies are also examining the efficacy of IFN-ɑ1 (NCT04293887 and NCT04349410) and IFN-β (NCT04449380 and NCT04385095) as single agents. Pegylated interferon-λ (type III interferon) infusions are also being studied in a number of phase 2 trials as single agents in the context of COVID-19. Another strategy for modulating the immune response to COVID-19 may be targeting type II interferons. The monoclonal antibody against IFN-γ, emapalumab, is approved for the treatment of hemophagocytic lymphohistiocytosis, a systemic form of immune activation. Emapalumab is currently being evaluated for possible use in limiting ventilator requirements for patients with COVID-19 (NCT04324021). Although potent antiviral agents, interferons are potent molecules with systemic effects, with most patients reporting flu-like symptoms, but can also include hematologic suppression, elevated liver enzymes, and psychiatric complications.55,56 In addition, unlike targeting interleukins to limit the inflammatory cascade of COVID-19, interferons should be targeted at limiting the extent of infection, requiring timely testing and diagnosis, which may limit the efficacy of these therapies. These agents have life-threatening side effect,s and so careful selection of patients would be necessary (Figure 2).
Vascular Endothelial Growth Factor (VEGF) Inhibitor: Bevacizumab
Bevacizumab is indicated in treatment of a variety of solid tumor cancers that include colorectal, lung, and renal cancer and is now being explored in treatment of COVID-19. Unlike the potential of other monoclonal antibodies to modulate the immune response in COVID-19, bevacizumab targets vascular endothelial growth factor (VEGF). Bevacizumab is used as antineoplastic through the inhibition of aberrant angiogenesis.57 In the context of COVID-19 and other respiratory infections, hypoxia and inflammation are thought to cause an upregulation of VEGF, acting in a positive feedback loop to promote vascular permeability, edema, and ultimately ARDS (Figure 1). A major side effect for bevacizumab is hypertension along with thromboembolic risk, which is particularly important as hypertension has been linked to increased complications and mortality in COVID-19 (Figure 2).58
Immune Checkpoint Inhibitors
Immune checkpoint blockers have revolutionized treatment of several solid tumors and hematological malignancies and are indicated in a wide variety of cancers; they are now being explored in treatment of COVID-19. The methods of altering immune response using effector molecules on the immune cell surface are a growing mainstay in a diverse range of tumors. The most prominent agents, anti-PD-1/PD-L1 and anti-CTLA-4, promote immune activity through the inhibition of negative feedback pathways.59 Unlike the use of anti-IL-6 therapies, immune checkpoint blockade promotes immune activity, running the risk of overstimulation, such as the autoimmune complications seen in the use of immune checkpoint blockade in oncology.59,60 Despite this risk, anti-PD-1 therapies have been used successfully in the setting of human polyomavirus type 2 central nervous system infections, leading to reduction in viral load and increased antiviral-specific activity in both CD4+ and CD8+ T cells.61 As of now, anti-PD-1 therapies are being evaluated for efficacy in overall clinical improvement (NCT04343144, NCT04413838, NCT04333914), viral clearance (NCT04356508), and modulation of lung injury (NCT04268537). A clinical trial with the combination of pembrolizumab (anti-PD-1) and tocilizumab is underway with a focus on improvement of ARDS (NCT04335305). This approach might stimulate and direct immune activation, providing improvement in viral clearance and limiting inflammation. Immune checkpoint inhibitors can cause several immune-mediated toxicities like pneumonitis, and careful selection of patients and close monitoring for toxicities would be essential (Figure 2).
CCR5 Inhibitor: Leronlimab
Leronlimab, or PRO 140, is a monoclonal antibody against the chemokine receptor, CCR5, currently being investigated as an anti-HIV therapy and inhibitor of metastasis in triple-negative breast cancer. Leronlimab received fast-track designation from the FDA for the use with carboplatin in treatments of metastatic triple-negative breast cancer. Leronlimab also has antiviral properties and has been granted fast-track designation as a combination therapy with HAART for HIV-infected patients. CCR5 represents a major method of viral entry for the HIV virus. In contrast to the antiviral effects, the monoclonal antibody CCR5 is expressed on a large number of nonimmune and immune cells, playing a role in the migration of immune cells into sites of inflammation62 and M1 polarization in myeloid cells,63 both of which may alter the natural immune response to COVID-19 (Figure 1). The two trials evaluating leronlimab in COVID-19 are examining symptom improvement in mild-to-moderate patients (NCT04343651) and reduction in mortality for severe patients (NCT04347239). The CCR5 receptor antagonist, maraviroc, used in HIV therapies, is also being tested (NCT04441385 and NCT04435522). Similar to VEGF inhibitors, phase 2a studies of leronlimab were associated with an increase in blood pressure,64 but overall have shown minor side effects in HIV trials (Figure 2).
Multiple Kinase Inhibitors
The small molecule multiple tyrosine kinase inhibitor imatinib which is approved for chronic myelogenous leukemia, gastrointestinal stromal tumor, acute lymphoblastic leukemia, myelodysplastic syndrome, systemic mastocytosis, and dermatofibrosarcoma protuberans is also being tested for COVID-19 treatment. Designed for the oncogenic product of the BCR-ABL gene fusion seen in chronic myelogenous leukemia, imatinib also inhibits other receptor kinases, like c-Kit and PDGFR.65 In vitro reports have demonstrated the ability of imatinib and other Abelson kinases to inhibit the viral replication cycle for SARS-CoV and MERS-CoV, two related coronaviruses.66 Thus, the use of imatinib in clinical trials for COVID-19, unlike other kinase inhibitors listed, may function more proximally on limiting the scale or scope of infection (Figure 1). In terms of safety, imatinib, as an anti-cancer drug, is relatively safe; however, it can increase susceptibility to infection, cause neutropenia, and is contraindicated in patients with congestive heart failure, liver impairment, and in pregnant women (Figure 2). Another nonspecific tyrosine kinase inhibitor, nintedanib, has approval for idiopathic pulmonary fibrosis and as a second-line treatment with docetaxel for non-small cell lung cancer.67 Like the primary indication, nintedanib is being tested to prevent pulmonary complications of COVID-19, namely pulmonary fibrosis as a consequence of infections (NCT04338802).
Thalidomide and Analogs
Thalidomide and the analog lenalidomide display immunomodulatory and anti-angiogenic properties and are being evaluated in the control of inflammatory complications related to COVID-19. Thalidomide and lenalidomide are both indicated in treatment of multiple myeloma, and lenalidomide is also indicated in myelodysplastic syndrome with 5q deletion. Although the mechanism is not fully elucidated, investigations have pointed to the inhibition of proliferation and angiogenesis through actions on ubiquitin-ligase complexes68 and the degradation of messenger RNA in blood cells reducing effector molecules, such as tumor necrosis factor-ɑ (TNF-ɑ).69 Thalidomide also appears to modulate the type of immune response, with reports in multiple myeloma of thalidomide-induced increases in NK cells, effectors in innate antiviral immune response, IL-2 induction, and secretion of interferon-γ.70,71 This may be due to a nonspecific costimulation of CD8+ T cells that has been reported using in vitro studies and tested as an adjuvant for chronic viral infections.72,73 Interestingly, an early case report in a patient with COVID-19 complicated by severe pneumonia observed that thalidomide led to an overall reduction in inflammatory cytokines, but an increase in T, B, and NK cells.74 This trend may be a reflection of feedback of the disease process itself or therapeutic effect. To differentiate this dichotomy, the authors of the case report have also begun a thalidomide clinical trial (NCT04273529) focusing on early PCR-confirmed COVID-19 with radiographic evidence of lung damage. The related immunomodulatory compound, lenalidomide, which is used in co-injection with steroids to treat multiple myeloma, is also being investigated using low doses in elderly patients with mild-to-moderate COVID-19 (NCT04361643). Both thalidomide and lenalidomide, in addition to increasing the risk for infections via myelosuppression, are teratogenic and should be avoided in pregnant women (Figure 2).
Bruton Tyrosine Kinase (BTK) Inhibitors
In addition to monoclonal therapies, there is interest in the ability of small molecule inhibitors to modulate the immune response to COVID-19. The inhibition of Bruton’s tyrosine kinase (BTK), which is FDA-approved for the treatment of chronic lymphocytic lymphoma and Waldenstrom’s macroglobulinemia, has shown anecdotal efficacy in controlling severe infection in COVID-19 patients. Recent clinical observations of the incidental use of ibrutinib in patients with Waldenstrom’s macroglobulinemia and COVID-19 showed marked improvement in symptoms for 5 of 6 patients taking 420 mg/day and required no hospitalization.75 The sixth patient was prescribed 140 mg/day of ibrutinib, had a more complicated course, with worsened hypoxia, and required intubation. The course improved with the increase of ibrutinib to 420 mg/day. Acalabrutinib is another BTK inhibitor indicated for CLL and mantle cell lymphoma with potential to treat COVID-19. Intriguingly, the inhibition of BTK or the off-target effects may prove to have multifactorial benefits as acalabrutinib treatment has shown reduction in platelet aggregation compared to healthy controls, a serious and common complication of COVID-19 infections.76 Currently, trials of ibrutinib and acalabrutinib, to assess the effect on the exaggerated immune response of patients who are severely ill with the COVID-19 infection, are underway. BTK inhibitors have side effect profiles that include increased bleeding and increased risk of infections (Figure 2).
BCG Vaccination
An early report found an epidemiological link between reduced morbidity and mortality of COVID-19 with countries that maintain a Bacillus Calmette–Guerin (BCG) vaccination program for tuberculosis.77 BCG is likely not an effective vaccine for COVID-19, but should be thought of as an adjuvant for an individual’s immune systems, priming the adaptive immune system (Figure 1). Vaccination with BCG works through the development of antigen-specific T cell responses and has been a mainstay of bladder cancer immunotherapy.78 A number of BCG-based preventative phase 3 and phase 4 trials have been registered to prevent SARS-CoV-2 infection. Several of these trials, such as the BRACE (NCT04327206), BCG-CORONA (NCT04328441), and BADAS (NCT04348370), focus on inoculating healthcare workers, with outcome measures of reduced infection rates or unplanned absenteeism. Other trials seek to immunize at-risk populations, for example ACTIVATE II (NCT04414267), and even the general population (NCT04369794). As of now, the WHO does not recommend BCG vaccinations as a preventative for COVID-19. BCG vaccinations can cause skin reactions and, in serious cases, abscess and bone inflammation.
XPO-1 Inhibitor (Selective Inhibitor of Nuclear Export) Selinexor
Selinexor, a selective nuclear export inhibitor, has been approved for relapsed/refractory multiple myeloma in combination with steroids.79 However, there is a growing literature on the direct antiviral effects of XPO-1 inhibitors in a variety of viruses, including HIV, influenza, respiratory syncytial virus, rabies, dengue, and cytomegalovirus by limiting nuclear transport.80 The side effect profile for selinexor includes moderate rates of bone marrow suppression (Figure 2). The two trials to evaluate selinexor will evaluate the improvement in clinical parameters for moderate to severe COVID-19 infections.
DNA-Damaging Agents: Etoposide and Methotrexate
A commonly used antineoplastic drug in therapeutic regimens for lung cancer, testicular cancer, and hematological malignancies, etoposide inhibits DNA replication and leads to preferential apoptosis in cancer cells. Etoposide has a narrow therapeutic window, and the immunosuppressive effect is being explored to treat cytokine storms in COVID-19 patients (Figure 1). A trial (NCT04356690) will evaluate whether pulse dose of etoposide, given on days one and four, will abate the pulmonary and inflammatory complications due to COVID-19. Likewise, the antifolate metabolite methotrexate, complexed with nanoparticles, is being evaluated for reduction of COVID-19-related lung injury (NCT04356690). Both of these agents target DNA replication, and as such are highly suppressive to bone marrow leading to increased risk of infection (Figure 2).
Conclusion and Future Perspective
The COVID-19 pandemic has resulted in insurmountable loss of life and challenged the medical and scientific community to develop effective treatment. Repurposing of available drugs has the potential to serve the unmet need but requires careful consideration, and clinical trials are underway. Despite the drive to improve clinical outcomes for patients with symptomatic COVID-19, caution should be exercised when using these potential agents and emphasis should be placed on enrollment into carefully constructed clinical trials. This is underscored by the hype surrounding and the subsequent negative randomized-controlled trial results of the use of hydroxychloroquine for COVID-19.7,13,14 Along with antiviral and anti-inflammatory drugs, several anti-cancer drugs can be potentially repurposed in the management of COVID-19. Challenges exist in repurposing anti-cancer drugs herein reviewed; like their initial development, these therapies are principally designed to alter proliferation. These agents are now being evaluated for a similar effect in blunting the hyperinflammatory response subsequent to COVID-19. Cytotoxic agents, like etoposide and methotrexate, or immune modulators, like BTK inhibitors or imatinib, run the risk of impairing humoral and cellular immune responses and subsequently leading to secondary infections and complications. This is underscored by the recent analysis comparing differential susceptibilities of patients with cancer, with 30.6% mortality across all cancer types and increased susceptibility of infection in patients with hematological malignancies compared to patients with solid malignancies.81 This susceptibility for COVID-19 infection in hematologic cancers was increased in patients who recently received chemotherapy, with drugs presumably directed against immune cells.81 As these anti-cancer drugs have general mechanisms of directing or impairing the immune system, particular attention should be placed on titrating the therapies to prevent over-suppression and further complications for patients hospitalized with COVID-19 (Figure 2). Future clinical trials, either single arm studies or randomized controlled trials, will be useful in determining the usefulness of anti-cancer drugs in COVID-19 patients.
Acknowledgments
The authors are supported by the National Cancer Institute of the National Institutes of Health under award number CA206255 (N.B.).
Disclosure
The authors report no conflicts of interest in this work.
References
1. World Health Organization. Pneumonia of Unknown Cause - China; 2020.
2. Grein J, Ohmagari N, Shin D, et al. Compassionate use of remdesivir for patients with severe Covid-19. N Engl J Med. 2020;382(24):2327–2336. doi:10.1056/NEJMoa2007016
3. Beigel JH, Tomashek KM, Dodd LE, et al. Remdesivir for the treatment of Covid-19 — final report. N Engl J Med. 2020. doi:doi:10.1056/NEJMoa2007764
4. Goldman JD, Lye DCB, Hui DS, et al. Remdesivir for 5 or 10 days in patients with severe Covid-19. N Engl J Med. 2020. doi:doi:10.1056/nejmoa2015301
5. Spinner CD, Gottlieb RL, Criner GJ, et al. Effect of remdesivir vs standard care on clinical status at 11 days in patients with moderate COVID-19: a randomized clinical trial. JAMA. 2020;324(11):1048–1057. doi:doi:10.1001/jama.2020.16349
6. Wang Y, Zhang D, Du G, et al. Remdesivir in adults with severe COVID-19: a randomised, double-blind, placebo-controlled, multicentre trial. Lancet. 2020.
7. Pan H, Peto R, Karim QA, et al. Repurposed antiviral drugs for COVID-19; interim WHO SOLIDARITY trial results. medRxiv. 2020.
8. Kalil AC. Treating COVID-19 - off-label drug use, compassionate use, and randomized clinical trials during pandemics. JAMA. 2020. doi:10.1001/jama.2020.4742
9. Cao B, Wang Y, Wen D, et al. A trial of lopinavir–ritonavir in adults hospitalized with severe Covid-19. N Engl J Med. 2020;382(19):1787–1799. doi:10.1056/NEJMoa2001282
10. Horby P, Lim WS, Emberson J, et al. Effect of dexamethasone in hospitalized patients with COVID-19: preliminary report. medRxiv. 2020. doi:10.1101/2020.06.22.20137273
11. Chen J, Liu D, Liu L, et al. A pilot study of hydroxychloroquine in treatment of patients with common coronavirus disease-19 (COVID-19). J Zhejiang Univ (Medical Sci). 2020;49(1).
12. Geleris J, Sun Y, Platt J, et al. Observational study of hydroxychloroquine in hospitalized patients with Covid-19. N Engl J Med. 2020;382(25):2411–2418. doi:doi:10.1056/NEJMoa2012410
13. Boulware DR, Pullen MF, Bangdiwala AS, et al. A randomized trial of hydroxychloroquine as postexposure prophylaxis for Covid-19. N Engl J Med. 2020;383(6):517–525. doi:doi:10.1056/nejmoa2016638
14. Tang W, Cao Z, Han M, et al. Hydroxychloroquine in patients with mainly mild to moderate coronavirus disease 2019: open label, randomised controlled trial. BMJ. 2020:m1849. doi:doi:10.1136/bmj.m1849.
15. Caly L, Druce JD, Catton MG, Jans DA, Wagstaff KM, The FD. A-approved drug ivermectin inhibits the replication of SARS-CoV-2 in vitro. Antiviral Res. 2020;178:104787. doi:doi:10.1016/j.antiviral.2020.104787
16. Duan K, Liu B, Li C, et al. Effectiveness of convalescent plasma therapy in severe COVID-19 patients. Proc Natl Acad Sci. 2020;117(17):9490–9496. doi:doi:10.1073/pnas.2004168117
17. Chen L, Xiong J, Bao L, Shi Y. Convalescent plasma as a potential therapy for COVID-19. Lancet Infect Dis. 2020. doi:doi:10.1016/S1473-3099(20)30141-9
18. Bertolini F, Sukhatme VP, Bouche G. Drug repurposing in oncology—patient and health systems opportunities. Nat Rev Clin Oncol. 2015;12(12):732–742. doi:doi:10.1038/nrclinonc.2015.169
19. Pantziarka P, Vandeborne L, Meheus L, Bouche G. Covid19db – an online database of trials of medicinal products to prevent or treat COVID-19, with a specific focus on drug repurposing. medRxiv. 2020. doi:doi:10.1101/2020.05.27.20114371
20. Yang Z, Liu J, Zhou Y, Zhao X, Zhao Q, Liu J. The effect of corticosteroid treatment on patients with coronavirus infection: a systematic review and meta-analysis. J Infect. 2020;81(1):e13–e20. doi:doi:10.1016/j.jinf.2020.03.062
21. World Health Organization. Report of the WHO-China Joint Mission on Coronavirus Disease 2019 (COVID-19). Geneva; 2020.
22. Chen T, Wu D, Chen H, et al. Clinical characteristics of 113 deceased patients with coronavirus disease 2019: retrospective study. BMJ. 2020;368.
23. Huang C, Wang Y, Li X, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395(10223):497–506. doi:10.1016/S0140-6736(20)30183-5
24. Poissy J, Goutay J, Caplan M, et al. Pulmonary embolism in COVID-19 patients: awareness of an increased prevalence. Circulation. 2020;142(2):184–186. doi:10.1161/circulationaha.120.047430
25. Helms J, Tacquard C, Severac F, et al. High risk of thrombosis in patients with severe SARS-CoV-2 infection: a multicenter prospective cohort study. Intensive Care Med. 2020;46(6):1089–1098. doi:10.1007/s00134-020-06062-x
26. Klok FA, Kruip MJHA, van der Meer NJM, et al. Incidence of thrombotic complications in critically ill ICU patients with COVID-19. Thromb Res. 2020. doi:10.1016/j.thromres.2020.04.013
27. Tang N, Li D, Wang X, Sun Z. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J Thromb Haemost. 2020;18(4):844–847. doi:10.1111/jth.14768
28. Connors JM, Levy JH. COVID-19 and its implications for thrombosis and anticoagulation. Blood. 2020;135(23):2033–2040. doi:10.1182/blood.2020006000
29. Ranucci M, Ballotta A, Di Dedda U, et al. The procoagulant pattern of patients with COVID-19 acute respiratory distress syndrome. J Thromb Haemost. 2020;18(7):1747–1751. doi:10.1111/jth.14854
30. Copin MC, Parmentier E, Duburcq T, et al. Time to consider histologic pattern of lung injury to treat critically ill patients with COVID-19 infection. Intensive Care Med. 2020;46(6):1124–1126. doi:10.1007/s00134-020-06057-8
31. Guo Y, Xu F, Lu T, Duan Z, Zhang Z. Interleukin-6 signaling pathway in targeted therapy for cancer. Cancer Treat Rev. 2012;38(7):904–910. doi:10.1016/j.ctrv.2012.04.007
32. Mishra AK, Dingli D. Metformin inhibits IL-6 signaling by decreasing IL-6R expression on multiple myeloma cells. Leukemia. 2019;33(11):2695–2709. doi:10.1038/s41375-019-0470-4
33. Tanaka T, Narazaki M, Kishimoto T. Immunotherapeutic implications of IL-6 blockade for cytokine storm. Immunotherapy. 2016;8(8):959–970. doi:10.2217/imt-2016-0020
34. Le RQ, Li L, Yuan W, et al. FDA approval summary: tocilizumab for treatment of chimeric antigen receptor T cell‐induced severe or life‐threatening cytokine release syndrome. Oncologist. 2018;23(8):943. doi:10.1634/theoncologist.2018-0028
35. Fu B, Xu X, Wei H. Why tocilizumab could be an effective treatment for severe COVID-19? J Transl Med. 2020;18(1):1–5. doi:10.1186/s12967-020-02339-3
36. Zhou Y, Fu B, Zheng X, Wang D, Zhao C. Pathogenic T cells and inflammatory monocytes incite inflammatory storm in severe COVID-19 patients. Natl Sci Rev. 2020.
37. Chen X, Zhao B, Qu Y, et al. Detectable serum SARS-CoV-2 viral load (RNAaemia) is closely correlated with drastically elevated interleukin 6 (IL-6) level in critically ill COVID-19 patients. Clin Infect Dis. 2020. doi:10.1093/cid/ciaa449
38. Luo P, Liu Y, Qiu L, Liu X, Liu D, Li J. Tocilizumab treatment in COVID-19: a single center experience. J Med Virol. 2020;92(7):814–818. doi:10.1002/jmv.25801
39. Zhang X, Song K, Tong F, et al. First case of COVID-19 in a patient with multiple myeloma successfully treated with tocilizumab. Blood Adv. 2020;4(7):1307. doi:10.1182/bloodadvances.2020001907
40. Xu X, Han M, Li T, et al. Effective treatment of severe COVID-19 patients with tocilizumab. Proc Natl Acad Sci U S A. 2020;117(20):10970–10975. doi:10.1073/pnas.2005615117
41. Toniati P, Piva S, Cattalini M, et al. Tocilizumab for the treatment of severe COVID-19 pneumonia with hyperinflammatory syndrome and acute respiratory failure: a single center study of 100 patients in Brescia, Italy. Autoimmun Rev. 2020;19(7):102568. doi:10.1016/j.autrev.2020.102568
42. Kewan T, Covut F, Al–Jaghbeer MJ, Rose L, Gopalakrishna KV, Akbik B. Tocilizumab for treatment of patients with severe COVID–19: a retrospective cohort study. EClinicalMedicine. 2020;24:100418. doi:10.1016/j.eclinm.2020.100418
43. Guaraldi G, Meschiari M, Cozzi-Lepri A, et al. Tocilizumab in patients with severe COVID-19: a retrospective cohort study. Lancet Rheumatol. 2020;2(8):e474–e484. doi:10.1016/S2665-9913(20)30173-9
44. Carey AJ, Tan CK, Ulett GC. Infection-induced IL-10 and JAK-STAT: A review of the molecular circuitry controlling immune hyperactivity in response to pathogenic microbes. Jak-Stat. 2012;1(3):159–167. doi:10.4161/jkst.19918
45. La Rosée F, Bremer HC, Gehrke I, et al. The Janus kinase 1/2 inhibitor ruxolitinib in COVID-19 with severe systemic hyperinflammation. Leukemia. 2020;34:1805–1815.
46. Stebbing J, Phelan A, Griffin I, et al. COVID-19: combining antiviral and anti-inflammatory treatments. Lancet Infect Dis. 2020;20(4):400–402. doi:10.1016/S1473-3099(20)30132-8
47. Gavegnano C, Detorio M, Montero C, Bosque A, Planelles V, Schinazi RF. Ruxolitinib and tofacitinib are potent and selective inhibitors of HIV-1 replication and virus reactivation in vitro. Antimicrob Agents Chemother. 2014;58(4):1977–1986. doi:10.1128/AAC.02496-13
48. Boor PPC, de Ruiter PE, Asmawidjaja PS, Lubberts E, van der Laan LJW, Kwekkeboom J. JAK-inhibitor tofacitinib suppresses interferon alfa production by plasmacytoid dendritic cells and inhibits arthrogenic and antiviral effects of interferon alfa. Transl Res. 2017;188(188):67–79. doi:10.1016/j.trsl.2016.11.006
49. Cantini F, Niccoli L, Nannini C, et al. Beneficial impact of Baricitinib in COVID-19 moderate pneumonia; multicentre study. J Infect. 2020;S0163–4453(20):30433. doi:10.1016/j.jinf.2020.06.052
50. Migita K, Izumi Y, Jiuchi Y, et al. Effects of J anus kinase inhibitor tofacitinib on circulating serum amyloid A and interleukin‐6 during treatment for rheumatoid arthritis. Clin Exp Immunol. 2014;175(2):208–214. doi:10.1111/cei.12234
51. Wu D, Yang XO. TH17 responses in cytokine storm of COVID-19: an emerging target of JAK2 inhibitor Fedratinib. J Microbiol Immunol Infect. 2020;53(3):368–370. doi:10.1016/j.jmii.2020.03.005
52. Sadler AJ, Williams BRG. Interferon-inducible antiviral effectors. Nat Rev Immunol. 2008;8(7):559–568. doi:10.1038/nri2314
53. Welsh RM, Bahl K, Marshall HD, Urban SL. Type 1 interferons and antiviral CD8 T-cell responses. PLoS Pathog. 2012;8:1.
54. Hung IFN, Lung KC, Tso EYK, et al. Triple combination of interferon beta-1b, lopinavir–ritonavir, and ribavirin in the treatment of patients admitted to hospital with COVID-19: an open-label, randomised, phase 2 trial. Lancet. 2020;395(10238):1695–1704. doi:10.1016/S0140-6736(20)31042-4
55. Sleijfer S, Bannink M, Van Gool AR, Kruit WHJ, Stoter G. Side effects of interferon-α therapy. Pharm World Sci. 2005;27(6):423–431. doi:10.1007/s11096-005-1319-7
56. Raison CL, Demetrashvili M, Capuron L, Miller AH. Neuropsychiatric adverse effects of interferon-α: recognition and management. CNS Drugs. 2005. doi:10.2165/00023210-200519020-00002
57. Keating GM. Bevacizumab: a review of its use in advanced cancer. Drugs. 2014;74(16):1891–1925.
58. Singh AK, Gupta R, Misra A. Comorbidities in COVID-19: outcomes in hypertensive cohort and controversies with renin angiotensin system blockers. Diabetes Metab Syndr Clin Res Rev. 2020;14(5):729. doi:10.1016/j.dsx.2020.03.016
59. Borcherding N, Kolb R, Gullicksrud J, Vikas P, Zhu Y, Zhang W. Keeping tumors in check: a mechanistic review of clinical response and resistance to immune checkpoint blockade in cancer. J Mol Biol. 2018;430(14):2014–2029. doi:10.1016/j.jmb.2018.05.030
60. Bersanelli M. Controversies about COVID-19 and anticancer treatment with immune checkpoint inhibitors. Futur Med. 2020;12(5):269–273.
61. Cortese I, Muranski P, Enose-Akahata Y, et al. Pembrolizumab treatment for progressive multifocal leukoencephalopathy. N Engl J Med. 2019;380(17):1597–1608. doi:10.1056/NEJMoa1815039
62. Qin S, Rottman JB, Myers P, et al. The chemokine receptors CXCR3 and CCR5 mark subsets of T cells associated with certain inflammatory reactions. J Clin Invest. 1998;101(4):746–754. doi:10.1172/JCI1422
63. Halama N, Zoernig I, Berthel A, et al. Tumoral immune cell exploitation in colorectal cancer metastases can be targeted effectively by anti-CCR5 therapy in cancer patients. Cancer Cell. 2016;29(4):587–601.
64. Jacobson JM, Thompson MA, Lalezari JP, et al. Anti–HIV‐1 activity of weekly or biweekly treatment with subcutaneous PRO 140, a CCR5 monoclonal antibody. J Infect Dis. 2010;201(10):1481–1487. doi:10.1086/652190
65. Savage DG, Antman KH. Imatinib mesylate—a new oral targeted therapy. N Engl J Med. 2002;346(9):683–693. doi:10.1056/NEJMra013339
66. Coleman CM, Sisk JM, Mingo RM, Nelson EA, White JM, Frieman MB. Abelson kinase inhibitors are potent inhibitors of severe acute respiratory syndrome coronavirus and middle east respiratory syndrome coronavirus fusion. J Virol. 2016;90(19):8924–8933.
67. Popat S, Mellemgaard A, Fahrbach K, et al. Nintedanib plus docetaxel as second-line therapy in patients with non-small-cell lung cancer: a network meta-analysis. Futur Oncol. 2015;11(3):409–420. doi:10.2217/fon.14.290
68. Eichner R, Heider M, Fernández-Sáiz V, et al. Immunomodulatory drugs disrupt the cereblon–CD147–MCT1 axis to exert antitumor activity and teratogenicity. Nat Med. 2016;22(7):735. doi:10.1038/nm.4128
69. Moreira AL, Sampaio EP, Zmuidzinas A, Frindt P, Smith KA, Kaplan G. Thalidomide exerts its inhibitory action on tumor necrosis factor alpha by enhancing mRNA degradation. J Exp Med. 1993;177(6):1675–1680. doi:10.1084/jem.177.6.1675
70. Davies FE, Raje N, Hideshima T, et al. Thalidomide and immunomodulatory derivatives augment natural killer cell cytotoxicity in multiple myeloma. Blood, J Am Soc Hematol. 2001;98(1):210–216.
71. Hayashi T, Hideshima T, Akiyama M, et al. Molecular mechanisms whereby immunomodulatory drugs activate natural killer cells: clinical application. Br J Haematol. 2005;128(2):192–203. doi:10.1111/j.1365-2141.2004.05286.x
72. Haslett PAJ, Corral LG, Albert M, Kaplan G. Thalidomide costimulates primary human t lymphocytes, preferentially inducing proliferation, cytokine production, and cytotoxic responses in the CD8+ subset. J Exp Med. 1998;187(11):1885–1892. doi:10.1084/jem.187.11.1885
73. Haslett PAJ, Hanekom WA, Muller G, Kaplan G. Thalidomide and a thalidomide analogue drug costimulate virus-specific CD8+ T cells in vitro. J Infect Dis. 2003;187(6):946–955. doi:10.1086/368126
74. Chen C, Qi F, Shi K, et al. Thalidomide combined with low-dose glucocorticoid in the treatment of COVID-19. pneumonia. 2020.
75. Treon SP, Castillo J, Skarbnik AP, et al. The BTK-inhibitor ibrutinib may protect against pulmonary injury in COVID-19 infected patients. Blood. 2020;135(21):1912–1915. doi:10.1182/blood.2020006288
76. Byrd JC, Harrington B, O’Brien S, et al. Acalabrutinib (ACP-196) in relapsed chronic lymphocytic leukemia. N Engl J Med. 2016;374(4):323–332. doi:10.1056/NEJMoa1509981
77. Miller A, Reandelar MJ, Fasciglione K, Roumenova V, Li Y, Otazu GH. Correlation between universal BCG vaccination policy and reduced morbidity and mortality for COVID-19: an epidemiological study. medRxiv. 2020.
78. Alexandroff AB, Jackson AM, O’Donnell MA, James K. BCG immunotherapy of bladder cancer: 20 years on. Lancet. 1999;353(9165):1689–1694. doi:10.1016/S0140-6736(98)07422-4
79. Chari A, Vogl DT, Gavriatopoulou M, et al. Oral selinexor–dexamethasone for triple-class refractory multiple myeloma. N Engl J Med. 2019;381(8):727–738. doi:10.1056/NEJMoa1903455
80. Mathew C, Ghildyal R. CRM1 inhibitors for antiviral therapy. Front Microbiol. 2017;8(8):1171. doi:10.3389/fmicb.2017.01171
81. Lee LYW, Cazier JB, Starkey T, et al. COVID-19 prevalence and mortality in patients with cancer and the effect of primary tumour subtype and patient demographics: a prospective cohort study. Lancet Oncol. 2020;21(10):1309–1316. doi:10.1016/S1470-2045(20)30442-3
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.