Back to Journals » Clinical Ophthalmology » Volume 10
Relationship between magnetic resonance imaging signal intensity and volume of extraocular muscles in thyroid-associated ophthalmopathy with methylprednisolone pulse therapy
Authors Higashiyama T, Nishida Y, Ohji M
Received 26 January 2016
Accepted for publication 3 March 2016
Published 20 April 2016 Volume 2016:10 Pages 721—729
DOI https://doi.org/10.2147/OPTH.S105096
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Tomoaki Higashiyama,1 Yasuhiro Nishida,2 Masahito Ohji1
1Department of Ophthalmology, 2Clinical Medical Education Center, Shiga University of Medical Science, Shiga, Japan
Purpose: To characterize the relationship between inflammation and swelling of extraocular muscles in thyroid-associated ophthalmopathy before and after methylprednisolone pulse therapy.
Methods: The signal intensities and volumes of the superior rectus (SR), inferior rectus (IR), lateral rectus (LR), medial rectus (MR), and superior oblique (SO) muscles were measured with magnetic resonance imaging in 25 eyes of 25 patients with thyroid-associated ophthalmopathy in the acute inflammatory phase before and after methylprednisolone pulse therapy. The signal intensity ratios (SIRs) of muscles and brain white matter were calculated.
Result: The mean SIRs before treatment were 2.28±0.74 in SR, 2.66±0.57 in IR, 2.03±0.42 in LR, 2.45±0.49 in MR, and 1.98±0.48 in SO muscles. The mean SIRs after treatment were 1.82±0.62, 1.84±0.52, 1.70±0.35, 1.95±0.46, and 1.60±0.36, respectively. The mean volumes (cm3) before treatment were 1.35±0.67 in SR, 1.21±0.39 in IR, 0.66±0.13 in LR, 0.94±0.31 in MR, and 0.58±0.14 in SO muscles. Those after treatment were 1.12±0.45, 0.91±0.31, 0.61±0.12, 0.85±0.28, and 0.49±0.11, respectively. The SIRs showed significantly positive correlations with volumes both before and after treatment in SR muscles (before, r=0.77; after, r=0.69), IR muscles (before, r=0.65; after, r=0.60), MR muscles (before, r=0.69; after, r=0.73), and SO muscles (before, r=0.52; after, r=0.50) (P<0.01 for all correlations).
Conclusion: Swelling reflected the inflammation in extraocular muscles of thyroid-associated ophthalmopathy both before and after treatment.
Keywords: thyroid-associated ophthalmopathy, methylprednisolone pulse therapy, extraocular muscle, magnetic resonance imaging, signal intensity, volume
Introduction
Thyroid-associated ophthalmopathy (TAO) is an inflammatory autoimmune disorder of the orbit. The symptoms of TAO caused by inflammatory swelling of the orbital tissues involve ocular motility dysfunction, proptosis, periorbital swelling, chemosis, lid retraction, and optic neuropathy.1–4 Ocular motility dysfunction, which affects quality of life, is caused by the inflammatory swelling of the extraocular muscles.
Previous studies reported immunosuppressive therapy, such as corticosteroids and orbital radiotherapy, for patients with TAO in the acute active inflammatory phase.2,5–9 The European Group on Graves’ Orbitopathy recommends methylprednisolone pulse therapy for moderate-to-severe active TAO.10 Previous studies reported the effectiveness of this treatment for inflammation and swelling of extraocular muscles in TAO.11–17
The short-tau inversion-recovery (STIR) technique of magnetic resonance imaging (MRI) is useful for evaluating inflammation in the extraocular muscles of TAO because the STIR images show high signal intensity in the edematous lesions caused by inflammation.15,16,18–23 We previously reported that signal intensities in the extraocular muscles of TAO after methylprednisolone pulse therapy were significantly lower than those before treatment,16 and the volumes in total extraocular muscles were improved after methylprednisolone pulse therapy.17
Although changes of inflammation and swelling in TAO have been reported, the relationships between inflammation and swelling of each extraocular muscle involved in TAO with methylprednisolone pulse therapy are unknown.15–17 To characterize these relationships, we measured and correlated the signal intensities and volumes of extraocular muscles before and after methylprednisolone pulse therapy.
Methods
Subjects
Twenty-five eyes of 25 Japanese patients (18 female and seven male) with TAO in the acute inflammatory phase were included in this study at the Department of Ophthalmology, Shiga University of Medical Science Hospital, Japan, from April 2008 to April 2015. This retrospective study was approved by the Institutional Review Board of Shiga University of Medical Science and was conducted in accordance with the tenets of the Declaration of Helsinki. Written informed consent was obtained from each patient in the study. All patients suffered from diplopia due to ocular motility dysfunction. The diagnosis of TAO in the acute inflammatory phase was based on a high signal intensity of the extraocular muscles on STIR images, high levels of serum thyroid autoantibodies, presence of typical clinical signs of TAO, and a history of hyperthyroidism or hypothyroidism. Patients were excluded if they had orbital diseases, such as trauma, an inflammatory disease of unknown origin in the orbit, a high risk for methylprednisolone pulse therapy, or had previously undergone orbital radiotherapy or surgery for TAO. Clinical activity of TAO was determined for each patient before treatment according to the clinical activity score (CAS), as recommended by the European Group on Graves’ Orbitopathy (a maximal score of 7 points).10 The eye in which the mean signal intensity of five measured extraocular muscles was highest was selected as representative of the patient.
All patients were admitted to the Department of Medicine in our hospital and received methylprednisolone pulse therapy, which was administered at a daily dose of 500 or 1,000 mg for three successive days. This treatment was repeated weekly three times for 3 weeks. In 19 cases (cases 6–25), the daily dose of methylprednisolone was decreased to 500 mg because the European Group on Graves’ Orbitopathy recommends that the total cumulative dose should be less than 8 g in one course of therapy to prevent acute liver damage.10 Instead, oral prednisolone was administered at a daily dose of 30 mg on four successive days as interpulse therapy. After three successive methylprednisolone pulse therapies, oral prednisolone therapy was administered at an initial daily dose of 30 or 40 mg/day and then gradually decreased while monitoring the activity of TAO.
Magnetic resonance image acquisition
The orbital MRI examinations were performed on a 3.0 Tesla MRI scanner (Achieva 3.0 T Quasar Dual; Royal Philips Electronics, Amsterdam, the Netherlands) with a standard head coil. The protocol of the examination consisted of the following sequence: STIR images (repetition time, 3,700 ms; echo time, 50 ms; inversion time, 200 ms; field of view, 120 mm; matrix, 256×320; number of excitations, two; and slice thickness, 2.5 mm), and T2-weighted spin echo (repetition time, 3,000 ms; echo time, 85 ms; field of view, 120 mm; matrix, 256×512; number of excitations, two; and slice thickness, 2.5 mm). The patients were instructed to gaze at the center during scanning. The MRI examinations were performed within 3 months before and after methylprednisolone pulse therapy.
Signal intensity measurements
The coronal STIR and T2-weighted images of MRI were used to measure the signal intensities in the superior rectus (SR), inferior rectus (IR), lateral rectus (LR), medial rectus (MR), and superior oblique (SO) muscles. The outlines of the extraocular muscles were traced in T2-weighted images because the contrast of STIR images was low. The outlines were superimposed in the STIR images on the same slice level of the T2-weighted images, and signal intensities of muscles in the STIR images were measured. The SR muscle and the levator palpebrae muscle were traced together because it was difficult to separate them on the MRI images. The signal intensity of the inferior oblique muscle was not measured in this study because this muscle included only a few slices of the coronal MRI image and it was difficult to calculate a representative signal intensity ratio (SIR) value. The signal intensities were measured by one of two ophthalmologists (TH or YN) who were experienced in TAO and orbital MRI.
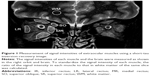
To standardize the signal intensities of the extraocular muscles, we measured the signal intensity of the brain white matter in the coronal STIR images as the standard tissue and calculated the SIR of the signal intensity in the extraocular muscle to that of the white matter from the same slice (Figure 1). The highest value among the mean SIRs of three consecutive slices in each subject was the representative value of the SIR in the muscle.
Volume measurements
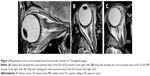
The cross-sectional areas of the five extraocular muscles (ie, SR, IR, LR, MR, and SO muscles) were measured by tracing outlines of each tissue on T2-weighted images with Image J software (National Institutes of Health, http://rsbweb.nih.gov/ij/index.html) (Figure 2).24 The cross-sectional areas for the SR and IR muscles were measured on sagittal images, and for MR, LR, and SO muscles on axial images. The SR and the levator palpebrae muscles were traced together, the same as the measurement of signal intensity. The volume of each tissue was calculated by multiplying the sum of the cross-sectional areas by the slice increment (slice thickness =2.5 mm) in the same manner as previous studies.17,25,26 The volumes were measured by one ophthalmologist (TH).
Assessment of CAS
The CAS was measured based on the following clinical findings: spontaneous retrobulbar pain, pain on attempted up or down gaze, redness of the eyelids, redness of the conjunctiva, swelling of the eyelids, and inflammation of the caruncle and/or plica and conjunctival edema.10 CAS was the sum of all items, and the maximal score was 7 points. The CAS values before and after treatment were determined by the same ophthalmologist (TH or YN). We analyzed the correlations of CAS values with the mean SIRs of five measured extraocular muscles or the total volume of five measured extraocular muscles to evaluate the correlation of the activity of the whole orbital inflammation and that of the muscle inflammation during TAO.
Statistical analysis
The Shapiro–Wilk test was used to test the normality of the numerical variables. A paired t-test was used to compare the SIRs, the volumes, and CAS values before and after treatment. An unpaired t-test was used to compare the SIRs and volumes between the two thyroid function groups. Pearson product-moment correlation coefficient or Spearman’s rank correlation coefficient was used to analyze the correlations between the SIRs and the volumes before and after treatment, those of CAS values with SIRs or volumes, and those of the duration before MRI with SIRs or volumes. PASW Statistics 18 software (IBM, Armonk, NY, USA) was used for all analyses. Data were expressed as the mean ± standard deviation. P<0.05 was considered statistically significant.
Results
Characteristics of patients
The characteristics of the patients before treatment are shown in Table 1. The mean ± standard deviation of patient age was 56.0±9.6 years (range, 35–74 years). The mean duration between the onset of diplopia and the start of treatment was 3.6±2.1 months (range, 1–8 months). Twenty-four patients had Graves’ disease and one patient had Hashimoto’s thyroiditis.
  | Table 1 Characteristics of patients with TAO |
SIRs
The mean SIRs before treatment were 2.28±0.74 in the SR, 2.66±0.57 in the IR, 2.03±0.42 in the LR, 2.45±0.49 in the MR, and 1.98±0.48 in the SO muscles. Those after the treatment were 1.82±0.62, 1.84±0.52, 1.70±0.35, 1.95±0.46, and 1.60±0.36, respectively. The mean SIRs after treatment were significantly decreased compared with those before treatment in all muscles (P<0.001) (Figure 3).
Volumes
The mean volumes before treatment were 1.35±0.67 cm3 in the SR, 1.21±0.39 cm3 in the IR, 0.66±0.13 cm3 in the LR, 0.94±0.31 cm3 in the MR, and 0.58±0.14 cm3 in the SO muscles. Those after treatment were 1.12±0.45, 0.91±0.31, 0.61±0.12, 0.85±0.28, and 0.49±0.11 cm3, respectively. The mean volumes after treatment were significantly decreased compared with those before treatment in all muscles (SR, MR, P<0.01; IR, SO, P<0.001; LR, P=0.02) (Figure 4).
Correlations of SIRs and volumes
The SIRs showed significant positive correlations with the volumes in the SR, IR, MR, and SO muscles both before and after treatment (before: SR, r=0.77, P<0.001; IR, r=0.65, P<0.001; MR, r=0.69, P<0.001; and SO, r=0.56, P=0.003; after: SR, r=0.69, P<0.001; IR, r=0.60, P=0.001; MR, r=0.73, P<0.001; SO, and r=0.51, P=0.01) (Figures 5 and 6). However, the volumes remained large, although signal intensities were low after treatment in some extraocular muscles.
CAS
The mean CAS was 2.5±1.5 before treatment and 1.4±1.4 after treatment. The mean CAS was significantly decreased after treatment (P<0.001). The CAS values showed significant positive correlations with the mean SIR of the five extraocular muscles both before and after treatment (before; r=0.40, P=0.046; after; r=0.41, P=0.04; Figure 7). The CAS values showed significant positive correlations with the total volume of the five measured extraocular muscles both before and after treatment (before; r=0.49, P=0.01; after; r=0.44, P=0.03; Figure 8).
Correlation of durations before MRI with SIRs or volumes
Regarding the duration between the diagnosis of Graves’ disease or Hashimoto’s thyroiditis and the MRI examination before treatment, the mean was 47.4±79.6 months (range, 0–317 months). The durations showed significant positive correlations with the SIRs and the volumes in the IR before treatment (SIRs, r=0.51, P=0.009; volumes, r=0.51, P=0.009) (Figure 9). In the mean duration between the diagnosis of TAO and the MRI examination before treatment, the mean was 1.9±3.8 months (range, 0–13 months). The durations were not significantly correlated with the SIRs and volumes of the extraocular muscles.
Comparison of thyroid function between the two groups
The thyroid function before methylprednisolone pulse therapy was hyperthyroid in six patients with Graves’ disease (hyperthyroid group) and euthyroid in the other 19 patients (euthyroid group). The mean duration between the diagnosis of Graves’ disease or Hashimoto’s thyroiditis and the MRI before treatment was 16.2±39.6 months (range, 0–97 months) in the hyperthyroid group and 57.2±87.1 months (range, 1–317 months) in the euthyroid group. Five patients in the hyperthyroid group were diagnosed with Graves’ disease and TAO at the same time. The thyroid function after treatment was euthyroid in all patients.
The mean SIRs in hyperthyroid group before treatment were 1.93 in the SR, 2.47 in the IR, 1.91 in the LR, 2.10 in the MR, and 1.74 in the SO muscles. Those in the euthyroid group before treatment were 2.39, 2.72, 2.07, 2.56, and 2.06, respectively. The mean SIR in the MR of the hyperthyroid group was significantly smaller than that of the euthyroid group (P=0.04). The mean changes of SIR before and after treatment in the hyperthyroid group were −0.28, −0.88, −0.32, −0.45, and −0.34 and those in the euthyroid group were −0.53, −0.80, −0.33, −0.51, and −0.39, respectively. There was no significant difference between the two groups in the mean changes of SIR.
The mean volumes in the hyperthyroid group before treatment were 1.06 cm3 in the SR, 0.93 cm3 in the IR, 0.60 cm3 in the LR, 0.73 cm3 in the MR, and 0.51 cm3 in the SO muscles. Those after treatment were 1.44, 1.30, 0.68, 1.01, and 0.61 cm3, respectively. The mean volumes in the IR and MR of the hyperthyroid group were significantly smaller than those of the euthyroid group (IR, P=0.006; MR, P=0.01). The mean changes of volume before and after treatment in the hyperthyroid group were −0.11, −0.20, −0.05, −0.06, and −0.05 cm3 and those in the euthyroid group were −0.27, −0.33, −0.05, −0.10, and −0.11 cm3, respectively. There was no significant difference between the two groups in the mean changes of volume.
Discussion
The present study demonstrated that the SIRs show significant positive correlations with the volumes in SR, IR, MR, and SO muscles, both before and after treatment. Inflammation causes the swelling in the extraocular muscles of TAO. A previous study reported that the SIRs of the most inflamed extraocular muscles in STIR images showed significant positive correlations with total muscle volumes before treatment of TAO.27 In the present study, we showed that the swelling reflected the inflammation in each extraocular muscle of TAO both before and after treatment. Therefore, a decrease in swelling requires adequate anti-inflammatory treatment of the extraocular muscles.
The CAS values showed significant positive correlations with not only the mean SIR but also with the total volume of the five extraocular muscles before and after treatment. A previous study reported that the SIRs of the extraocular muscles in TAO showed significant positive correlations with CAS values.23 Another study also reported that the SIRs of the extraocular muscles in TAO showed significant positive correlations with the Morits score, which is the basis for CAS.22 In the present report, we show that not only inflammation but also swelling of the extraocular muscles could be correlated with the CAS values in TAO.
The SIRs did not show statistically significant correlations with the volume in LR muscles both before and after treatment because the involvement of LR muscles was low. Wiersinga et al28 reported that orbital computed tomography in 80 patients with TAO showed enlargement of the IR muscle in 60%, MR muscle in 50%, SR muscle in 40%, and LR muscle in 22% of patients. Many previous studies have reported similar results.29,30
In the present study, the volumes of the extraocular muscles were not standardized as they were in previous studies.31–33 However, the results were not affected because differences according to sex or age were not found in the volumes of the extraocular muscles.34,35
The mean SIR in the MR and the mean volumes in the IR and MR of the hyperthyroid group were significantly smaller than those of the euthyroid group. Dysthyroidism had been associated with worsening of TAO in a previous report.36 However, our results showed that SIR and volume in the hyperthyroid group were worse than those in the euthyroid group. We assume the reason was that five patients in the hyperthyroid group were recent cases who were diagnosed with Graves’ disease and TAO at the same time.
There are some limitations associated with this study. First, the mean CAS of patients in this study was low. The CAS tends to be low in Japanese TAO patients who require treatment.37,38 The CAS of some patients was low in this study when the diagnosis of TAO in the acute inflammatory phase was confirmed, based on high signal intensity of the extraocular muscles in the STIR images, high levels of serum thyroid autoantibodies, and a history of hyperthyroidism or hypothyroidism. Second, the follow-up period to evaluate the changes of SIRs and volumes with treatment was within 3 months after methylprednisolone pulse therapy. An assessment of long-term changes is therefore needed for a better evaluation. However, TAO may naturally improve over time.39 Natural improvement had little effect on the results of this study, because the follow-up period after treatment was short. Third, the volumes remained large although signal intensities were low after treatment in some extraocular muscles. Fibrosis of extraocular muscles might therefore appear. Fourth, the patients who underwent orbital radiotherapy or were treated with other drugs were not enrolled in the current study. Many previous studies have reported the efficacies of treatments for TAO.40,41
In conclusion, SIRs statistically showed significant positive correlations with volumes both before and after treatment. The swelling reflected the inflammation in each extraocular muscle of TAO both before and after treatment, and a decrease in swelling required adequate anti-inflammatory treatment of the extraocular muscles.
Disclosure
The authors report no conflict of interest in this work.
References
Prummel MF, Bakker A, Wiersinga WM, et al. Multi-center study on the characteristics and treatment strategies of patients with Graves’ orbitopathy: the first European Group on Graves’ Orbitopathy experience. Eur J Endocrinol. 2003;148(5):491–495. | ||
Kuriyan AE, Phipps RP, Feldon SE. The eye and thyroid disease. Curr Opin Ophthalmol. 2008;19(6):499–506. | ||
Bahn RS. Graves’ ophthalmopathy. N Engl J Med. 2010;362(8):726–738. | ||
Kuriyan AE, Woeller CF, O’Loughlin CW, Phipps RP, Feldon SE. Orbital fibroblasts from thyroid eye disease patients differ in proliferative and adipogenic responses depending on disease subtype. Invest Ophthalmol Vis Sci. 2013;54(12):7370–7377. | ||
Burch HB, Wartofsky L. Graves’ ophthalmopathy: current concepts regarding pathogenesis and management. Endocr Rev. 1993;14(6):747–793. | ||
Bartalena L, Pinchera A, Marcocci C. Management of Graves’ ophthalmopathy: reality and perspectives. Endocr Rev. 2000;21(2):168–199. | ||
Bartalena L, Marcocci C, Tanda ML, et al. Orbital radiotherapy for Graves’ ophthalmopathy. Thyroid. 2002;12(3):245–250 | ||
Prummel MF, Terwee CB, Gerding MN, et al. A randomized controlled trial of orbital radiotherapy versus sham irradiation in patients with mild Graves’ ophthalmopathy. J Clin Endocrinol Metab. 2004;89(1):15–20. | ||
Zoumalan CI, Cockerham KP, Turbin RE, et al. Efficacy of corticosteroids and external beam radiation in the management of moderate to severe thyroid eye disease. J Neuroophthalmol. 2007;27(3):205–214. | ||
Bartalena L, Baldeschi L, Dickinson A, et al. Consensus statement of the European Group on Graves’ Orbitopathy (EUGOGO) on management of GO. Eur J Endocrinol. 2008;158(3):273–285. | ||
Hiromatsu Y, Tanaka K, Sato M, et al. Intravenous methylprednisolone pulse therapy for Graves’ ophthalmopathy. Endocr J. 1993;40(1):63–72. | ||
Tagami T, Tanaka K, Sugawa H, et al. High-dose intravenous steroid pulse therapy in thyroid-associated ophthalmopathy. Endocr J. 1996;43(6):689–699. | ||
Ohtsuka K, Sato A, Kawaguchi S, Hashimoto M, Suzuki Y. Effect of high-dose intravenous steroid pulse therapy followed by 3-month oral steroid therapy for Graves’ ophthalmopathy. Jpn J Ophthalmol. 2002:46(5):563–567. | ||
Aktaran S, Akarsu E, Erbağci I, Araz M, Okumuş S, Kartal M. Comparison of intravenous methylprednisolone therapy vs oral methylprednisolone therapy in patients with Graves’ ophthalmopathy. Int J Clin Pract. 2007;61(1):45–51. | ||
Tortora F, Prudente M, Cirillo M, et al. Diagnostic accuracy of short-time inversion recovery sequence in Graves’ ophthalmopathy before and after prednisone treatment. Neuroradiology. 2014;56(5):353–361. | ||
Higashiyama T, Nishida Y, Morino K, et al. Use of MRI signal intensity of extraocular muscles to evaluate methylprednisolone pulse therapy in thyroid-associated ophthalmopathy. Jpn J Ophthalmol. 2015;59(2):124–130. | ||
Higashiyama T, Nishida Y, Ohji M. Changes of orbital tissue volumes and proptosis in patients with thyroid extraocular muscle swelling after methylprednisolone pulse therapy. Jpn J Ophthalmol. 2015;59(6):430–435. | ||
Hiromatsu Y, Kojima K, Ishisaka N, et al. Role of magnetic resonance imaging in thyroid-associated ophthalmopathy: its predictive value for therapeutic outcome of immunosuppressive therapy. Thyroid. 1992;2(4):299–305. | ||
Hoh HB, Laitt RD, Wakeley C, et al. The STIR sequence MRI in the assessment of extraocular muscles in thyroid eye disease. Eye (Lond). 1994;8(Pt 5):506–510. | ||
Laitt RD, Hoh B, Wakeley C, et al. The value of the short tau inversion recovery sequence in magnetic resonance imaging of thyroid eye disease. Br J Radiol. 1994;67(795):244–247. | ||
Bailey CC, Kabala J, Laitt R, et al. Magnetic resonance imaging in thyroid eye disease. Eye (Lond). 1996;10(Pt 5):617–619. | ||
Mayer E, Herdman G, Burnett C, Kabala J, Goddard P, Potts MJ. Serial STIR magnetic resonance imaging correlates with clinical score of activity in thyroid eye disease. Eye (Lond). 2001;15(Pt 3):313–318. | ||
Mayer EJ, Fox DL, Herdman G, et al. Signal intensity, clinical activity and cross-sectional areas on MRI scans in thyroid eye disease. Eur J Radiol. 2005;56(1):20–24. | ||
Schneider CA, Rasband WS, Eliceiri KW. NIH Image to ImageJ: 25 years of image analysis. Nat Methods. 2012;9(7):671–675. | ||
Nishida Y, Tian S, Isberg B, Tallstedt L, Lennerstrand G. MRI measurements of orbital tissues in dysthyroid ophthalmopathy. Graefes Arch Clin Exp Ophthalmol. 2001;239(11):824–831. | ||
Nishida Y, Tian S, Isberg B, Hayashi O, Tallstedt L, Lennerstrand G. Significance of orbital fatty tissue for exophthalmos in thyroid-associated ophthalmopathy. Graefes Arch Clin Exp Ophthalmol. 2002;240(7):515–520. | ||
Rodríguez-González N, Pérez-Rico C, López-Para Giménez R, et al. Short-tau inversion-recovery (STIR) sequence magnetic resonance imaging evaluation of orbital structures in Graves’ orbitopathy. Arch Soc Esp Oftalmol. 2011;86(11):351–357. | ||
Wiersinga WM, Smit T, van der Gaag R, Mourits M, Koornneef L. Clinical presentation of Graves’ ophthalmopathy. Ophthalmic Res. 1989;21(2):73–82. | ||
Nugent RA, Belkin RI, Neigel JM, et al. Graves orbitopathy: correlation of CT and clinical findings. Radiology. 1990;177(3):675–682. | ||
Enzmann DR, Donaldson SS, Kriss JP. Appearance of Graves’ disease on orbital computed tomography. J Comput Assist Tomogr. 1979;3(6):815–819. | ||
Kvetny J, Puhakka KB, Røhl L. Magnetic resonance imaging determination of extraocular eye muscle volume in patients with thyroid-associated ophthalmopathy and proptosis. Acta Ophthalmol Scand. 2006;84(3):419–423. | ||
Majos A, Pajak M, Grzelak P, Stefańczyk L. Magnetic resonance evaluation of disease activity in Graves’ ophthalmopathy: T2-time and signal intensity of extraocular muscles. Med Sci Monit. 2007;13 Suppl 1:44–48. | ||
Lennerstrand G, Tian S, Isberg B, et al. Magnetic resonance imaging and ultrasound measurements of extraocular muscles in thyroid-associated ophthalmopathy at different stages of the disease. Acta Ophthalmol Scand. 2007;85(2):192–201. | ||
Bijlsma WR, Mourits MP. Radiologic measurement of extraocular muscle volumes in patients with Graves’ orbitopathy: a review and guideline. Orbit. 2006;25(2):83–91. | ||
Tian S, Nishida Y, Isberg B, Lennerstrand G. MRI measurements of normal extraocular muscles and other orbital structures. Graefes Arch Clin Exp Ophthalmol. 2000;238(5):393–404. | ||
Prummel MF, Wiersinga WM, Mourits MP, Koornneef L, Berghout A, van der Gaag R. Effect of abnormal thyroid function on the severity of Graves’ ophthalmopathy. Arch Intern Med. 1990;150(5):1098–1101. | ||
Tachibana S, Murakami T, Noguchi H, et al. Orbital magnetic resonance imaging combined with clinical activity score can improve the sensitivity of detection of disease activity and prediction of response to immunosuppressive therapy for Graves’ ophthalmopathy. Endocr J. 2010;57(10):853–861. | ||
Hiromatsu Y, Eguchi H, Tani J, Kasaoka M, Teshima Y. Graves’ ophthalmopathy: Epidemiology and natural history. Intern Med. 2014;53(5):353–360. | ||
Perros P, Crombie AL, Kendall-Taylor P. Natural history of thyroid associated ophthalmopathy. Clin Endocrinol (Oxf). 1995;42(1):45–50. | ||
Stein JD, Childers D, Gupta S, et al. Risk factors for developing thyroid-associated ophthalmopathy among individuals with Graves disease. JAMA Ophthalmol. 2015;133(3):290–296. | ||
Kim JW, Han SH, Son BJ, Rim TH, Keum KC, Yoon JS. Efficacy of combined orbital radiation and systemic steroids in the management of Graves’ orbitopathy. Graefes Arch Clin Exp Ophthalmol. Epub 2016 Feb 15. |
 © 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.