Back to Journals » Clinical Ophthalmology » Volume 11
Refractive changes after lens-sparing vitrectomy for macular hole and epiretinal membrane
Authors Muto T , Nishimura T , Yamaguchi T, Chikuda M, Machida S
Received 14 May 2017
Accepted for publication 22 July 2017
Published 21 August 2017 Volume 2017:11 Pages 1527—1532
DOI https://doi.org/10.2147/OPTH.S141771
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Tetsuya Muto,1 Tomoharu Nishimura,1 Takefumi Yamaguchi,2 Makoto Chikuda,1 Shigeki Machida1
1Department of Ophthalmology, Dokkyo Medical University Koshigaya Hospital, Koshigaya, Japan; 2Department of Ophthalmology, Tokyo Dental College Ichikawa General Hospital, Ichikawa, Japan
Purpose: Cataract progression after lens-sparing vitrectomy might differ according to original posterior segment diseases. Our objective was to analyze the refractive values after lens-sparing vitrectomy for macular hole (MH) and epiretinal membrane (ERM).
Materials and methods: We reviewed the medical records of 25 MH patients (25 eyes) and 23 ERM patients (23 eyes) who underwent lens-sparing vitrectomy. Refractive changes in both groups were compared. All patients underwent 20-gauge three-port pars plana vitrectomy. Fluid–air exchange was performed during vitrectomy only in the MH group. The results were analyzed using the unpaired t-test, chi-square test, or Fisher exact probability test, and multivariate analysis.
Results: There were no significant differences in the patient’s age (P=0.45). The myopia progression rate (D/month) was higher in the MH group after surgery than that in the ERM group (P=0.035). MH group had more females (P=0.043), longer surgical time (P<0.001), and higher frequencies of surgical adjuvants use (triamcinolone acetonide, P=0.019; brilliant blue G, P<0.001). The myopia progression rate in the MH group (R=0.568, P<0.001) correlated with female gender. However, no correlation was observed between longer surgical time and the use of surgical adjuvants.
Conclusion: The rate of myopia progression was higher in the MH group. Fluid–air exchange and gender may affect the rate of myopia progression.
Keywords: cataract, vitrectomy, macular hole, epiretinal membrane
Introduction
With increasing age, the opacity and hardness of the nucleus of the crystalline lens gradually increases in all individuals.1 Nuclear sclerotic cataracts are characterized by visually significant nuclear opacity. Progressed nuclear sclerotic cataracts are more difficult to divide during surgery than cortical or anterior and posterior subcapsular cataracts. In most western populations, nuclear sclerotic cataracts are the most common reason for cataract surgery.2
Vitreous surgery is used to treat many types of vitreoretinal diseases, and it is often performed in phakic, as well as aphakic and pseudophakic eyes.3 Lens-sparing vitrectomy often results in postoperative formation of nuclear sclerotic cataracts.4–8 Because nuclear sclerotic cataracts frequently affect refraction of the eye, shifting it toward myopic in the majority of cases,9,10 symptoms of anisometropia should be carefully monitored after vitrectomy. This myopic shift can be useful for subjectively grading the rate of progression of nuclear sclerotic cataracts. There have been several reports on myopic shift after lens-sparing vitrectomy.9–13
Several reports6,12,14,15 have identified a connection between the presence of intraocular gas bubbles and the formation of nuclear sclerotic cataracts after lens-sparing vitrectomy. Because these reports are not recent or involved more than one surgeon, we decided to confirm this connection. Moreover, the surgical procedures for macular hole (MH) and epiretinal membrane (ERM) are almost similar, with the exception of fluid–air exchange in the former.
In the current report, the formation of nuclear sclerotic cataracts and myopia progression rate after lens-sparing vitrectomy performed by a single surgeon using the most recent vitrectomy system were compared between the MH and ERM groups.
Materials and methods
The medical records of 25 MH patients (25 eyes) and 23 ERM patients (23 eyes) who underwent lens-sparing vitrectomy between November 2011 and May 2015 were reviewed. All subjects analyzed in this study were followed-up for a period ranging from 4 to 34 months and had primary MH or ERM and phakic eyes. None of the patients had a visually significant cataract at the time of surgery. Exclusion criteria were a history of ocular surgery or the presence of ocular disease which could affect refraction and/or visual function. The protocol of this retrospective study was approved by the Institutional Review Board of Dokkyo Medical University Koshigaya Hospital and was conducted in accordance with the tenets of the Declaration of Helsinki. Written informed consent was obtained from all participants.
All lens-sparing vitrectomies were performed by an experienced surgeon at Dokkyo Medical University Koshigaya Hospital. All surgeries were three-port 20-gauge pars plana vitrectomies. Surgical procedures were performed under local anesthesia using the Constellation® Vision System (Alcon Laboratories, Fort Worth, TX, USA) along with handheld contact lenses (Nanoo Kougaku Inc., Tokyo, Japan) with perfusion. When required, triamcinolone acetonide (TA) was used to secure posterior vitreous detachment and prevent vitreous incarceration of the sclerotomy wound. Brilliant blue G (BBG) was used as needed to stain the inner limiting membrane. Air, 20% sulfur hexafluoride (SF6), or 12.5% octafluoropropane (C3F8) was used for total gas–fluid exchange, and all patients in the MH group were kept in the prone position for 1–2 weeks postoperatively. The gas (air or 20% SF6 or 12.5% C3F8) was selected according to the patient’s status. In patients who were considered to be unable to maintain prone position, 12.5% C3F8 was used. Air was used if the MH diameter was <30% of the optic nerve and in patients who were within two months of disease onset; 20% SF6 was used otherwise. None of the patients in the ERM group received total gas–fluid exchange during surgery. All sutures were removed at 3 months postoperatively.
Preoperative and postoperative objective refractive errors were measured in all eyes using an automatic refractometer (KR-7100; Topcon Corporation, Tokyo, Japan). The manifest spherical equivalent (SE) value was determined as the spherical power plus half the cylindrical power. Noncontact optical biometry (IOL Master; Carl Zeiss Meditec AG, Jena, Germany) was used to evaluate axial length before primary vitrectomy. Each clinical parameter was evaluated at baseline (preoperatively) and at the final medical check-up or just before the cataract surgery, to calculate the myopia progression rate (D/month). Myopia progression rate was defined as the mean refractive change per month between the baseline and the final medical check-up or just before the cataract surgery.
Differences in mean age, manifest SE value, surgical time, axial length, myopia progression rate, and follow-up period after lens-sparing vitrectomy were compared between the ERM and MH groups using the unpaired t-test. Discrete variables such as gender; ratio of left eyes to right eyes; use of TA and BBG; combinations of diabetes, hypertension, and hyperlipidemia; fluid–air exchange; receiving cataract surgery after vitrectomy; and the myopia progression rate of three factors (ratio of male to female, use of TA, and BBG) in the MH group were compared between the two groups using the chi-square test or Fisher exact probability test. Pearson coefficient of correlation was performed to examine the following factors: gender, surgical time, and surgical adjuvant use (TA and BBG), which were associated with myopia progression rate in MH group. Multiple linear regressions were used with a stepwise selection of variables with significance in the linear regression analysis. Unpaired t-test was performed to evaluate gender and TA and BBG use which were associated with myopia progression rate in MH group. Differences with a probability (P) value <0.05 were considered statistically significant.
Results
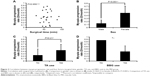
Preoperative, intraoperative, and postoperative factors of both MH and ERM groups are shown in Table 1. No statistically significant differences were observed between groups with respect to age; ocular; preoperative SE; axial length; follow-up period; or the combinations of diabetes, hypertension, and hyperlipidemia. The proportion of females and the mean surgical time were greater in the MH group than in the ERM group (P=0.043 and P<0.001, respectively). TA, BBG, and fluid–air exchange were used more frequently in the MH group than in the ERM group (P=0.019, P<0.001, and P<0.001, respectively). The myopia progression rate was lower in the ERM group than in the MH group (P=0.035, unpaired t-test) as shown in Figure 1.
Table 2 shows the rate of cataract surgery after lens-sparing vitrectomy, and the gas tamponade material used during lens-sparing vitrectomy. Cataract surgery was performed in 21 of 25 eyes (84.0%) in the MH group and 17 of 23 eyes (73.9%) in the ERM group roughly a year postoperatively. There were no statistically significant differences between the groups. The cataract type was nuclear sclerosis in every case, and there were no cases of cortical or posterior subcapsular cataract. All cataract surgeries were successful and uneventful. Neither intraoperative (eg, lens capsule rupture) nor postoperative (eg, zonule formation, persistent ocular hypertension) complications were observed during the follow-up period. SF6 was the most frequently used tamponade material during vitrectomy in the MH group.
Table 3 shows the correlation coefficients of four factors (gender, surgical time, TA, and BBG) related to myopia progression rate in the MH group. Myopia progression rate remained unaffected by surgical time, TA, and BBG; however, it demonstrated a correlation with the gender of the patient (R=0.568, P<0.001). Figure 2 shows the relative distribution of these four factors with the myopia progression rate. Myopia progression rate of females was significantly greater than that of males (P<0.001).
No significant relationships were found between the myopia progression rate and gender (R=0.022, P=0.92), surgical time (R=−0.275, P=0.21), TA (R=0.230, P=0.29), or BBG (R=0.158, P=0.47) in the ERM group.
Table 4 shows the mean age of male and female patients in the MH group. The mean age of male patients was significantly higher than female patients (P<0.01).
  | Table 4 The mean age of patients in the MH group |
Discussion
The findings of the present study indicated that myopia progression rate after lens-sparing vitrectomy was higher in the MH group than that in the ERM group. Kawakubo et al reported a higher myopia progression rate after lens-sparing vitrectomy in the MH group than in the ERM group 6 months postoperatively (−2.11±1.89 D vs −0.99±1.34 D, respectively, P=0.0002).12 The authors also speculated that peripheral vitreous body shaving and fluid–air exchange were responsible for the relatively high myopia progression rate.12 Thompson reported that the incidence of nuclear sclerosis was about 60% higher in the MH group than that in the ERM group, and vitreomacular traction syndrome occurred after lens-sparing vitrectomy.6,14 However, in these reports, nuclear sclerosis was analyzed using Chylack Lens Opacity Classification System (LOCS) II lens grading scale; not by refraction. The use of gas during vitrectomy might have caused the high nuclear sclerosis progression rate in the MH group.6,14
To the best of our knowledge, no other studies have reported the association between myopia progression rate, gender, and use of surgical adjuvant. Hsuan et al reported that the use of fluid–air exchange was the underlying cause of nuclear sclerosis progression after vitrectomy.15 Cheng et al reported that the duration of vitrectomy did not increase the risk of nuclear sclerosis progression in MH,16 while Smiddy and Feurer reported that it did not increase the risk of nuclear sclerosis progression in diabetic retinopathy, ERM, and MH groups.4 Okamoto et al reported that the duration of vitrectomy did not increase the risk of myopia progression in retinal detachment.9 According to our results, the use of fluid–air exchange and being female has a connection with myopia progression rate. However, further research is needed to confirm these suspicions.
Feng and Adelman reported that visual impairment due to cataract development after vitrectomy was likely influenced by a variety of factors including the level of oxygen in the vitreous cavity, the amount of vitreous removed, the amount of inflammation owing to ocular disease and surgery, and the length and difficulty of surgery.7 Moreover, biochemical effects such as the perfusion solution,17 the aqueous humor dynamics, and a change of constitution18 are also known to promote changes in lens metabolism. Holekamp et al reported that nuclear sclerosis after lens-sparing vitrectomy for diabetic retinopathy was less progressive than in other diseases due to the presence of ischemia and low oxygen supply.2 In fact, they measured the oxygen tension level of the vitreous cavity and found that it was significantly lower in patients with diabetes than in those without the condition.19 They concluded that oxidative damage to the lens nucleus and increased intraocular oxygen tension was associated with nuclear sclerotic cataracts. Also, Beebe et al and Shui et al reported that age-related degeneration of the vitreous body or vitrectomy exposes the posterior part of the lens to increased oxygen, resulting in the formation of nuclear sclerotic cataracts.20,21 The lens is exposed to oxygen during vitrectomy for MH because fluid–air exchange is needed. This procedure may cause severe progression of myopia. In future studies, we plan to investigate the association of nuclear cataract progression after vitrectomy with proliferative diabetic and non-diabetic retinopathy. Hamoudi et al reported that removal of vitreous gel itself played a role in the myopic change after vitrectomy without fluid gas exchange.22
Cheng et al reported that the incidence of nuclear sclerosis progression in eyes after vitrectomy was 98% at 1 year and 100% at 2 years postoperatively.16 Smiddy and Feuer reported that 16 of 22 patients (73%) required cataract surgery after lens-sparing vitrectomy for ERM at an average follow-up period of 38 months, and the same was true for 36 of 48 MH patients (75%) at an average follow-up period of 44 months.4 The mean age at surgery was 70 years in the ERM group and 67 years in the MH group. Six (27%) patients in the ERM group and 12 (25%) in the MH group did not undergo cataract surgery during relatively long follow-up periods. If we apply our results, myopic changes in the ERM group at 38 months and the MH group at 44 months after vitrectomy can be calculated as follows: 38×0.29=11.02 D and 44×0.42=18.48 D, respectively. These scores are unusual. A myopic change of 10 D causes severe nuclear sclerosis; thus, surgical intervention may be challenging. In a vitrectomized eye, there is a higher risk of posterior capsular rupture. In an eye where the anterior hyaloid is already disrupted, there is a higher risk of nucleus drop. Also, anisometropia can be very severe in some patients.
Our study has several limitations. First, it was a retrospective study; a prospective study with objective assessments of lens changes would have been preferable. Second, all procedures were performed by the same surgeon, which could have resulted in bias. Third, our sample size was small; hence, further studies with adequate sample size are needed to provide a better understanding of the pathophysiology of cataract after vitrectomy.
Conclusion
The findings of the present study indicated a higher myopia progression rate in the MH group than in the ERM group after lens-sparing vitrectomy. This difference may be attributable to the use of fluid–air exchange and a greater proportion of female complex in the MH group.
Disclosure
None of the authors have a financial or proprietary interest in any materials or methods mentioned in this report. The authors report no conflicts of interest in this work.
References
Heys KR, Cram SL, Truscott RJ. Massive increase in the stiffness of the human lens nucleus with age: the basis for presbyopia? Mol Vis. 2004;10:956–963. | ||
Holekamp NM, Bai F, Shui YB, Almony A, Beebe DC. Ischemic diabetic retinopathy may protect against nuclear sclerotic cataract. Am J Ophthalmol. 2010;150(4):543–550. | ||
Ikeda T, Minami M, Nakamura K, et al. Progression of nuclear sclerosis based on changes in refractive values after lens-sparing vitrectomy in proliferative diabetic retinopathy. Clin Ophthalmol. 2014;8:959–963. | ||
Smiddy WE, Feuer W. Incidence of cataract extraction after diabetic vitrectomy. Retina. 2004;24(4):574–581. | ||
Almony A, Holekamp NM, Bai F, Shui YB, Beebe DC. Small-gauge vitrectomy does not protect against nuclear sclerotic cataract. Retina. 2012;32(3):499–505. | ||
Thompson JT. The role of patient age and intraocular gases in cataract progression following vitrectomy for macular holes and epiretinal membranes. Trans Am Ophthalmol Soc. 2003;101:485–498. | ||
Feng H, Adelman RA. Cataract formation following vitreoretinal procedures. Clin Ophthalmol. 2014;8:1957–1965. | ||
Wong SC, Clare G, Bunce C, Sullivan PM, Gregor ZJ, Ezra E. Cataract progression in macular hole cases: results with vitrectomy or with observation. J Cataract Refract Surg. 2012;38(7):1176–1180. | ||
Okamoto Y, Okamoto F, Hiraoka T, Oshika T. Refractive changes after lens-sparing vitrectomy for rhegmatogenous retinal detachment. Am J Ophthalmol. 2014;158(3):544–549. | ||
Tseng PC, Woung LC, Tseng GL, et al. Refractive change after pars plana vitrectomy. Taiwan J Ophthalmol. 2012;2(1):18–21. | ||
Sawa M, Saito Y, Hayashi A, Kusaka S, Ohji M, Tano Y. Assessment of nuclear sclerosis after nonvitrectomizing vitreous surgery. Am J Ophthalmol. 2001;132(3):356–362. | ||
Kawakubo H, Sato Y, Shimada H, Amano K, Kuwajima A, Matsui M. Myopic change in refraction due to nuclear sclerotic changes after vitreous surgery (comparison of epimacular membrane and macular hole). Folia Ophthalmologica Japonica. 1996;47:396–400. | ||
Saito Y, Lewis JM, Park I, et al. Nonvitrectomizing vitreous surgery: a strategy to prevent postoperative nuclear sclerosis. Ophthalmology. 1999;106(8):1541–1545. | ||
Thompson JT. The role of patient age and intraocular gas use in cataract progression after vitrectomy for macular holes and epiretinal membranes. Am J Ophthalmol. 2004;137(2):250–257. | ||
Hsuan JD, Brown NA, Bron AJ, Patel CK, Rosen PH. Posterior subcapsular and nuclear cataract after vitrectomy. J Cataract Refract Surg. 2001;27(3):437–444. | ||
Cheng L, Azen SP, El-Bradey MH, et al. Duration of vitrectomy and postoperative cataract in the vitrectomy for macular hole study. Am J Ophthalmol. 2001;132(6):881–887. | ||
Sakaue H, Negi A, Honda Y. Comparative study of vitreous oxygen tension in human and rabbit eyes. Invest Ophthalmol Vis Sci. 1989;30(9):1933–1937. | ||
Fecondo JV, Augusteyn RC. Superoxide dismutase, catalase and glutathione peroxidase in the human cataractous lens. Exp Eye Res. 1983;36(1):15–23. | ||
Holekamp NM, Shui YB, Beebe DC. Lower intraocular oxygen tension in diabetic patients: possible contribution to decreased incidence of nuclear sclerotic cataract. Am J Ophthalmol. 2006;141(6):1027–1032. | ||
Beebe DC, Shui YB, Siegfried CJ, Holekamp NM, Bai F. Preserve the (intraocular) environment: the importance of maintaining normal oxygen gradients in the eye. Jpn J Ophthalmol. 2014;58(3):225–231. | ||
Shui YB, Holekamp NM, Kramer BC, et al. The gel state of the vitreous and ascorbate-dependent oxygen consumption: relationship to the etiology of nuclear cataracts. Arch Ophthalmol. 2009;127(4):475–482. | ||
Hamoudi H, Kofod M, La Cour M. Refractive change after vitrectomy for epiretinal membrane in pseudophakic eyes. Acta Ophthalmol. 2013;91(5):434–436. |
 © 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.