Back to Journals » International Journal of Nanomedicine » Volume 15
Recent Advances in Oral Nano-Antibiotics for Bacterial Infection Therapy
Received 1 September 2020
Accepted for publication 2 November 2020
Published 1 December 2020 Volume 2020:15 Pages 9587—9610
DOI https://doi.org/10.2147/IJN.S279652
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Yan Shen
Ze-Liang Wu, 1,* Jun Zhao, 2,* Rong Xu 1, 3
1Department of Pharmacology, School of Basic Medicine, Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430030, People’s Republic of China; 2Department of Anatomy, School of Basic Medicine, Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430030, People’s Republic of China; 3The Key Laboratory for Drug Target Researches and Pharmacodynamic Evaluation of Hubei Province, Wuhan 430030, People’s Republic of China
Correspondence: Rong Xu
Department of Pharmacology, School of Basic Medicine, Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430030, People’s Republic of China
Tel/Fax +86-27-83691785
Email [email protected]
*These authors contributed equally to this work
Abstract: Bacterial infections are the main infectious diseases and cause of death worldwide. Antibiotics are used to treat various infections ranging from minor to life-threatening ones. The dominant route to administer antibiotics is through oral delivery and subsequent gastrointestinal tract (GIT) absorption. However, the delivery efficiency is limited by many factors such as low drug solubility and/or permeability, gastrointestinal instability, and low antibacterial activity. Nanotechnology has emerged as a novel and efficient tool for targeting drug delivery, and a number of promising nanotherapeutic strategies have been widely explored to overcome these obstacles. In this review, we explore published studies to provide a comprehensive understanding of the recent progress in the area of orally deliverable nano-antibiotic formulations. The first part of this article discusses the functions and underlying mechanisms by which nanomedicines increase the oral absorption of antibiotics. The second part focuses on the classification of oral nano-antibiotics and summarizes the advantages, disadvantages and applications of nanoformulations including lipid, polymer, nanosuspension, carbon nanotubes and mesoporous silica nanoparticles in oral delivery of antibiotics. Lastly, the challenges and future perspective of oral nano-antibiotics for infection disease therapy are discussed. Overall, nanomedicines designed for oral drug delivery system have demonstrated the potential for the improvement and optimization of currently available antibiotic therapies.
Keywords: bacterial infection, nanomedicine, antibiotic, oral drug delivery system
Introduction
Infectious diseases refer to the illnesses that are caused by organisms such as bacteria, fungi, viruses, and parasites.1 With the resurgence of known ones and the emergence of new ones, infectious diseases continue to cause high morbidity and mortality in recent years all over the world, especially in developing countries.2 The overall morbidity of 39 notable infectious diseases in China increased from 3,906,566 cases (7248 deaths) in 2004 to 6,944,240 cases (18,237 deaths) in 2016.3 The global burden of disease study (GBD) 2017 report showed that four infectious diseases were listed in the top 10 causes of death globally, including lower respiratory infections (ranked fourth), diarrheal diseases (5th), AIDS (8th) and malaria (10th).4 Bacteria are the most common pathogenic organisms that can invade organs such as facial features, respiratory tract, gastrointestinal tract (GIT), and genitourinary system, whereas the symptoms range from minor illnesses such as strep throat or ear infections to life-threatening conditions.5–9 Statistics shows that the annual death toll caused by bacterial infections could reach 14 million,10 most of which are caused by Staphylococcus aureus, Pseudomonas aeruginosa, Clostridium labile and some drug-resistant bacteria including methicillin-resistant Staphylococcus aureus (MRSA), third-generation cephalosporin-resistant Escherichia coli and vancomycin-resistant Enterococci (VRE).11 Furthermore, bacterial infections pose significant economic burden on the healthcare system. In the United States, the medical costs and labor losses caused by antibiotic resistance alone amount to U$55 billion per year, while the cost for non-resistant bacteria further adds up to this stunning number.12
Most bacterial infections require treatment with antibiotics although some of them may resolve by themselves. Since the discovery of penicillin by Alexander Fleming in 1928,13 numerous antibiotic drugs have been isolated and identified that can kill bacteria (bactericidal) or prevent their reproduction or proliferation (bacteriostatic) through different mechanisms (Table 1).14 As a result, infections that are previously severe or fatal can now be managed effectively. However, the worldwide overuse and misuse of antibiotics led to the rapid emergence of drug-resistant bacteria, which are now reversing the therapeutic miracles and threatening the effective prevention and treatment of infectious diseases.15,16 Furthermore, the development of novel antibiotics has dropped by 30% over the last 30 years due to weakened economic incentives and more stringent regulations. Only six antibiotics were approved by the Food and Drug Administration of the United States between 2015 and 2018, and most of them only target Gram-positive bacteria and cause antimicrobial resistance (AMR) nonetheless.17–19 On the other hand, novel treatments including monoclonal antibodies, bacteriophages, stem cells, etc. have so far been unsuccessful in clinical trials or bedside applications due to various reasons.11 In summary, the aforementioned challenges in the treatment of bacterial infections still warrant further development of antibiotic formulations.
 |
Table 1 Main Types and Mechanisms of Drugs Used for Bacterial Infections |
Antibiotics can be administered via oral, intravenous, transpulmonary, and transdermal routes.20–22 Oral administration is the most popular route due to its safety, convenience, and excellent patient compliance, especially for long-term medications.23 However, its efficiency is complexed by many physical and chemical barriers in GIT including mucus layer, epithelial tight junction, drug metabolic enzymes, P-glycoprotein, etc.24–26 On the other hand, according to the biopharmaceutical classification system (BCS), many highly effective antibiotics, eg ciprofloxacin, vancomycin, cefpodoxime, rifampicin, and clarithromycin belong to BCS class II (low solubility) or IV (low solubility and low permeability) drugs. The poor solubility of most antibiotics also hinders their absorption through GIT and therefore significantly restricted their bioavailability.27–30 Although a variety of formulation strategies including micronization, salt formation, solid dispersion, and emulsion have been explored to improve the bioavailability of insoluble antibiotics, only limited success has been achieved due to difficulties in production, batch-to-batch consistency, and quality control.18
Nanotechnology offers new opportunities for drug dosing and delivery.31 With unique structural, chemical, mechanical, magnetic, electrical, and biological properties,32 nanomedicine demonstrates numerous advantages over ‘free’ therapeutic molecules. For example, the large specific surface area of nanomedicine increases drug solubility and gastrointestinal contact area.31,33 The encapsulated drug payloads are also protected from degradation in GIT by the nanoscale carrier. The nanocarriers can be further modified to improve the profiles of delivery, penetration, and controlled release of encapsulated drugs, and thereby achieve desirable characteristics including higher antibiotic uptake,34,35 less adverse reactions, alleviated drug resistance, shorter treatment duration, lower dose, and lower cost, especially for patients with severe infections.36,37 The palatability of antibiotics can be improved as well.38
Despite the great promise brought by orally deliverable nano-antibiotic formulations, the understanding of these formulations is still lacking compared to those of anti-neoplastic and biomolecules. In this review, we will discuss the present state-of-art and future prospects of the nanotechnology-based approaches to orally deliverable antibiotics.
Improved Oral Absorption of Poorly Soluble Antibiotics by Nano-Drug Delivery System
The orally administered antibiotics face a number of challenges before they can function properly. First, the absorption of antibiotics is physically barricaded by the tight connection of mucus layer lining the GIT. Drug capture by the mucus layer along with the fast turn-over of mucus layer cells also limits the efficiency of drug absorption.39 Some antibiotics are substrates of P-glycoprotein (P-gp) and therefore can be re-shuttled back into the GIT. In addition, pH values in the GIT, gastrointestinal motility, and intestinal contents also affect drug absorption.40 Second, the antibiotics need to retain their structural integrity before being absorbed. Some glycopeptide and lipopeptide antibiotics are especially vulnerable to the endo- or exo-peptidase secreted in the GIT.41 The extensive metabolism by cytochrome P450 enzymes expressed in small intestine or liver may also lead to premature destruction of the antibiotics.42 Researchers have designed different nano-drug delivery systems in order to overcome these challenges (Figure 1).
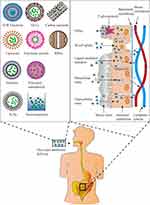 |
Figure 1 Types of oral antibiotic nanopreparations and mechanisms by which oral absorption improvement of antibiotics through nano-drug delivery system. |
Increase the Stability and Solubility in the GIT
Peptide-based antibiotics can be degraded by various enzymes in GIT, while acid- or base-labile antibiotics are prone to the gastrointestinal pH variations. Encapsulation of antibiotics in nanocarriers forms a protective shell for the active ingredient. For example, liposomes composed of bile salt-loaded cefotaxime effectively prevented the damage from bile acids toward the payload, and subsequently improved the drug stability.43 The large surface area of nanoformulations also improves drug solubility.
Promote the Mucus Adhesion
Positively charged polymers like chitosan-coated clarithromycin can interact with the negatively charged mucus layer to promote the adhesion and subsequent absorption of the polymer.44 Carriers containing thiol functionality can form disulfide bonds with the mucosa,45 which prolongs the exposure time of antibiotic payload in the small intestine, and thereby increase the probability of mucosa penetration.
Facilitate Translocation Across the Mucus Layers
In general, the rapid secretion and shedding of mucus cover the epithelium surface with a tenacious layer of viscoelastic hydrogel that lubricates and protects the exposed epithelium from external threats and enzymatic degradation. The mucus is composed of mucin glycoproteins with negatively charged sialic residues. The mucin monomers can also be crosslinked into a multimer network with hydrophobic pockets via disulfide bonds between the cysteine-rich domains of different monomers. This tenacious, negatively charged, hydrophobic viscoelastic hydrogel can quickly capture foreign substances via physical, electrostatic, or hydrophobic interactions, and subsequently remove them when the mucus layer is shed off.39 Polyethylene glycol (PEG) coating, on the other hand, imbues the nanoparticles with an electrically neutral and hydrophilic surface, and therefore can prevent the hydrophobic and electrostatic interactions between the nanoparticles and the mucus layer.46,47
Enhance Permeation Across the Enteric Epithelia
The permeation across the enteric epithelia can be increased by using absorption enhancer, eg chitosan and its derivatives.48 On the other hand, some natural and synthetic excipients, such as Cremophor EL, Tween-80, Tween-20,
Enhance Ligand-Mediated Endocytosis and Uptake by Microfold Cells (M Cells)
Nanocarriers can target specific receptors on the enteric epithelia through receptor-specific ligands, thereby enhancing the cellular uptake and transepithelial transport of payloads. The receptor-mediated endocytosis is generally mediated by reticulin and caveolin.53 For instance, folate-conjugated liposomes facilitate the endocytic delivery of vancomycin via the folate receptors that are ubiquitously expressed on intestinal epithelial cells.54 Microfold cells (M cells) presented in Peyer’s patches are epithelial cells with specialized antigen-sampling ability. With high endocytic rate and low degradation ability, M cell can take up foreign substances, and move them to the intraepithelial pocket beneath the M-cell basolateral membrane to be processed in the lymphoid tissues. Although fewer than enterocytes, the high transcytosis ability of M cells makes them an excellent target for antibiotic delivery, including the delivery of dapsone-loaded solid lipid nanoparticles.55–57
Classification of Oral Nano-Antibiotics
Lipid-Based Nano-Antibiotics
Lipid-based nanoparticles in antibiotic delivery can be categorized into self-nanoemulsifying drug delivery system (SNEDDS), liposomes, niosomes, solid lipid nanoparticles (SLNs), and nanostructured lipid carriers (NLCs) (Figure 1). Lipids are biocompatible and biodegradable. In addition to protecting drugs from gastrointestinal damage and controlling drug release, lipids also promote drug absorption through transcellular, paracellular and lymphatic transport, and eventually increase the bioavailability.58–61 The properties of lipid-based nano-antibiotics are summarized in Table 2.
    |
Table 2 Composition and Properties of Lipid-Based Nanocarriers for Oral Delivery of Antibiotics |
SNEDDS
Developed on the concept of nanoemulsion, SNEDDS is a stable complex composed of oil, water, surfactants, cosurfactants and fat-soluble drugs. While the nanoemulsions are sensitive and metastable, SNEDDS spontaneously form oil-in-water nanoemulsions after oral administration under the mechanical force in the GIT with a particle size between 20 and 200 nm.62,63 SNEDDS promotes drug transportation through the intestinal lymphatic system and prevent their degradation in the digestive tract.64 In SNEDDS, medium- or long-chain triglycerides with different degrees of saturation constitute the oil phase, while Tween-80 or Span-20 function as surfactants to reduce the surface tension and increase the degree of dispersion. Medium chain alcohols (C3-C8), including propylene glycol and 1, 2-octanediol, are used as cosurfactants.65 Appropriate choice of excipients and their ratios are critical to optimize particle size, drug-loading efficiency, encapsulation efficiency, and profile of drug release.66 The advantages of using SNEDDS for antibiotic delivery include solubilization of insoluble drugs, reduction of P-gp-mediated active efflux and first-pass metabolism, promotion of drug transcellular, paracellular or intestinal lymphatic transport, and reduction of drug resistance.67–69 To further improve the storage stability, solid self-nanoemulsifying drug delivery system (SSNEDDS) can be prepared by adding re-dispersants to the liquid preparations, and then curing the mixture by solid carrier adsorption, spray drying, etc.70
Cefpodoxime proxetil (CP) is a prodrug of cefpodoxime, a BCS IV class β-lactam antibiotic with low oral bioavailability. Bajaj et al52 encapsulated CP with different SNEDDS formulations using Tween-80, propylene glycol and tocopherol polyethylene glycol succinate (TPGS) as the surfactants/co-surfactants, and Capmul MCM as the oil compared with conventional formulations, CP-SNEDDS had significantly faster dissolution and higher permeability in vitro. The minimum inhibitory concentration (MIC) of CP-SNEDDS against Escherichia coli, Staphylococcus aureus and Bacillus subtilis were 1500 ng/mL, 100 ng/mL and 750 ng/mL, respectively; while the MIC of the conventional preparation were 2250 ng/mL, 250 ng/mL and 1000 ng/mL, respectively. The AUC0-∞ value of CP-SNEDDS was 5.36-fold higher than that of commercial CP (75.55 ± 2.8 µg·h/mL vs 14.07 ± 2.4 µg·h/mL), whereas its oral bioavailability was 4 times higher. The liquid CP-SNEDDS was further spray-dried to form solid CP-SSNEDDS to improve storage stability. The improved bioavailability of CP-SNEDDS can be attributed to the Tween 80 and TPSG that increased membrane permeability, as well as the SNEDDS formulation that minimized P-gp efflux and cytochrome enzyme pre-absorption metabolism, promoted lymphatic transport, and increased gastrointestinal membrane permeability. Rifampicin (RIF) is a first-line anti-Mycobacterium tuberculosis drug. Liquid RIF-SNEDDS was prepared by preconcentration method using Labrasol as the surfactant, Cremophor-EL as the cosurfactant and Capmul MCM C8 as the oil phase. RIF-SSNEDDS was obtained via adsorption technology where RIF-SNEDDS was adsorbed to the adsorbent Aerosil 200. These preparations dramatically promoted osmosis in vitro. Their relative bioavailability was significantly improved compared with RIF suspension, and the oral bioavailability of RIF-SSNEDDS reached as high as 96.83%.71,72
Liposome and Niosome
Liposomes have been explored for oral delivery antibiotics for more than 40 years.73 Conventional liposomes are composed of phospholipids and cholesterol, and exhibit a bilayer vesicle structure from 25 to 1000 nm in size. Drugs are distributed in the hydrophilic or hydrophobic compartments of liposomes according to their lipophilicity.74,75 The resemblance to cell membrane imbues liposomes with excellent biocompatibility. Liposomes have shown promising properties for drug delivery, such as high entrapment efficiency, controlled drug release, satisfactory safety profile, convenient drug loading and surface modification, and protection of drug payloads.76–78 On the other hand, they are also facing several challenges for antibiotic delivery. For example, gastric acid, bile salts, and digestive enzymes in GIT may compromise the liposome structure and cause premature drug release.79 The large size of liposomes may hinder their penetration through the gastrointestinal barriers.80 The industrial large-scale production of liposomal drug formulation may also present a technical difficulty.81 These challenges can be overcome by surface modification and composition adjustment.
Surface modification is an important method to overcome the drug destruction in the gastrointestinal tract. Anderson et al54 prepared a liposome formulation of vancomycin and conjugated folic acid as well as poly (ethylene glycol) (PEG) onto the liposome surface. The GIT specific and pH-dependent absorption of folic acid through receptor-mediated endocytosis increased drug bioavailability by 12.5-fold. As a type of surfactant secreted by hepatocytes, bile salts have been considered to be the main factor for the disruption of liposomes in GIT and lead to reduced concentrations of intact liposomes and release incorporated drugs. Paradoxically, studies also revealed that prior incorporation of bile salts into liposomal bilayers during liposome preparation stabilized the membrane to resist the destructive effects of physiological bile salts.79,82 Daniela et al modified Tween 80, linolenic acid, and dioleoylphosphatidylethanolamine (DOPE) to synthesize liposomes encapsulating amoxicillin for the treatment of Helicobacter pylori infection. Tween 80 can separate the outer membrane of Helicobacter pylori. As an unsaturated fatty acid, linolenic acid can play an anti-Helicobacter pylori effect by disrupting the integrity of the bacterial membrane. In addition, because of the presence of phosphatidylethanolamine receptors in these bacteria, DOPE was added as a targeting agent for Helicobacter pylori. The results showed that the nanoparticles (F4) modified by Tween 80, linolenic acid and DOPE are stable for at least 6 months at 4°C. At the same time, the nanoparticles have high tolerance to harsh conditions including acidic pH and physiological temperature. Nanoparticles have low cytotoxicity in fibroblasts and gastric cell lines, and increase the residence time of the infection site (gastric mucosa). Figure 2 shows the overall design of this liposome. Four formulations of LNPs have been studied and characterized, which are distinguished by the presence or absence of linolenic acid and DOPE.83 Arafat et al43 prepared sodium deoxycholate-containing liposomes to encapsulate cefotaxime, a BCS III drug. The resultant formulation maintained good stability in gastric juice or even more destructive intestinal fluid. In vivo pharmacokinetics experiments showed that, compared with free cefotaxime, the sodium deoxycholate-containing liposomal drug increased the maximum concentration (Cmax) by 3.39-fold (1.71 ± 0.3 vs 0.51 ± 0.2 µg/mL), and the area-under-the plasma concentration–time curve from 0 to infinity (AUC0-∞) by 5.12-fold (5.73 ± 0.7 vs 1.12 ± 0.5 µg·h/mL). Notably, the oral bioavailability of the sodium deoxycholate-modified liposomes was five fold and two fold higher than that of free drug and unmodified liposome, respectively.
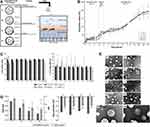 |
Figure 2 Delivering amoxicillin at the infection site – a rational design through lipid nanoparticles. (A) AMX-loaded LNPs, which were designed to release AMX near H. pylori. The double-emulsion LNPs are composed of cetyl palmitate, Tween 80, linolenic acid, and DOPE. Abbreviations: AMX, amoxicillin; DOPE, dioleoylphosphatidylethanolamine; H. pylori, Helicobacter pylori; LNPs, lipid nanoparticles. (B) In vitro AMX release profiles from the LNPs in three simulated conditions, namely 1) pH 1.6, 2) pH 5.0, and 3) pH 7.4. Notes: Vertical lines represent media changes. Values represent the mean ± SD of three independently produced formulations. (C) In vitro cell viability studies. L929 cell viability study and MKN-74 cell viability study. Different formulations in different solid lipid concentrations, from 0 (black) to 8 (light gray) mg/mL of solid lipid were evaluated. For free AMX, the same amount of AMX existent in those concentrations of LNPs was used, with the exception of 1 and 4 mg/mL. Notes: Values represent mean ± SD of three independently produced formulations. *P<0.05, **P<0.01, ***P<0.005, ****P<0.0001 relative to 0 mg/mL. Notes: Vertical lines represent media changes. Values represent the mean ± SD of three independently produced formulations. (D) Characterization of the AMX-loaded LNPs suspensions before (dark gray) and after (light gray) the incubation with mucins. 1) LNPs size and PDI. Bars represent the size (left y-axis) and dots represent the PDI (right y-axis). 2) LNPs zeta potential. Notes: Values represent the mean ± SD of three independently produced formulations. *P<0.05, **P<0.01, ****P<0.0001 relative to the LNPs without mucins. Abbreviations: PDI, polydispersity. (E) TEM images of the AMX-loaded LNPs and the corresponding unloaded LNPs. The difference among the four formulations (F1–F4) was their composition relative to the presence or absence of linolenic acid and DOPE. P1–P4 stands for AMX unloaded LNPs. P1, F1, P2, F2, F3, P4A, and F4A are at a magnification of 50,000×. P3 is at a magnification of 25,000×. P4B and F4B are at a magnification of 100,000×. Copyright © 2019. Dove Medical Press. Reproduced from Lopes-de-Campos D, Pinto RM, Lima SAC, et al. Delivering amoxicillin at the infection site - a rational design through lipid nanoparticles. Int J Nanomedicine. 2019;14:2781–2795.83 |
Solid proliposome is another tool to improve the stability of oral formulation. Daptomycin is a semisynthetic cyclic lipopeptide antibiotic with broad-spectrum activity against a variety of Gram-positive bacteria. It was incorporated into the proliposomes by hydrating a film of daptomycin, soybean phosphatidylcholine (SPC) and cholesterol. Stearylamine (SA) was also added provide a positive surface charge, which significantly increased the entrapment efficiency to 92% through interaction with daptomycin. Compared with daptomycin solution, the Cmax and AUC0-∞ of solid proliposomes were 8.35 ± 0.64 µg/mL and 46.39 ± 5.69 µg·h/mL, respectively, while DPT solution below lower limit of quantification (LLOQ), representing a significantly higher oral bioavailability.81
Phospholipids are prone to oxidative degradation in GIT in addition to its high cost of manufacture. As a result, efforts have been attempted to replace phospholipids with various nonionic surfactants, such as creatinine, creatinine derivatives, bergenin (3,4,8,10-tetrahydroxy-2-hydroxymethyl-9-meth-oxy-2,3,4,4a-tetrahydropyrano[3,2-c] isochromen-6-one), a-tocopherol, and other renewable resources. These novel formulations are collectively known as niosomes,84 and have shown promising properties including low toxicity, low cost, good biocompatibility and biodegradability, and stable physical/chemical properties.85–88
Cefixime is the third-generation cephalosporin against a variety of Gram-negative and Gram-positive bacteria, but its acidity results in poor solubility and low oral bioavailability. Imran et al87,89 synthesized two glycoside-based niosomal nanocarriers for cefixime delivery: LRC-BG (lauroyl chloride, bergenin) and BRM-BG (bromoundecane, bergenin). Cell viability and hemolysis assay showed that both had good biocompatibility. Compared with cefixime suspension, the Cmax and AUC0–24 of LRC-BG increased by 1.72 and 4.58 times, respectively; while those of BRM-BG increased by 1.9 and 4.97 times, respectively. In addition to niosomal cefixime, niosomes with other antibiotics, eg daptomycin, levofloxacin, clarithromycin and azithromycin, have all shown promising results after oral administration.85,86,88,90
Solid Matrix Mediated Lipid Nanoparticle Formulations (LNFs)
Solid lipid nanoparticles (SLNs) and nanostructured lipid carriers (NLCs) are two types of LNFs mainly used for antibiotic delivery. The unique shell-core structure of LNFs makes them suitable for delivering drugs of different lipophilicity or different composition, and subsequently increase the solubility, permeability, and bioavailability of encapsulated drugs.
Solid lipid nanoparticles (SLNs) were first designed in the 1990s by Schwarzetal et al.91 These are composed of medium- or long-chain lipids eg anisodylglycerol, tripalmitin, and stearic acid that have high melting points and are electrically neutral, SLNs are shell-core structured with a particle size between 10 and 1000 nm.92,93 SLNs are particularly effective in avoiding the first-pass metabolism and increasing the intestinal lymphatic transport via paracellular and transcellular pathways of enterocytes, and endocytosis of phagocytic cells.94,95 In addition to the solid lipids, surfactants such as Tween-80, PVA and Pluronic F-68 are also used in SLNs. The composition and properties of LNFs for oral delivery of antibiotics are listed in Table 2.
Sharma et al96 loaded the BCS II drug clarithromycin (CLA) in SLNs. The optimal feature of CLA-SLNs was achieved when Pluronic F-68 was used as the surfactant, and the ratio of stearic acid to trisearin was 1:1. These highly hydrophobic, negatively charged, sub-micron SLNs were conducive to M cell uptake and accumulation, and easily released from Peyer’s patches to the lymphatic system. Compared to free CLA, the Cmax of CLA-SLNs increased by 2.3-fold, and its relative bioavailability increased by 5-fold. Öztürk et al studied the effect of lipid skeletons, ie glyceryl behenate, tripalmitin, and stearic acid, on the properties and antibacterial activity of CLA-SLNs. Besides the improvement in bioavailability, drug loading and release efficiency are significantly related to carbon chain length.97 Rifabutin (RFB), with poor solubility, is a semisynthetic antibiotic to treat Mycobacterium tuberculosis and Mycobacterium avium. By loading RFB into glyceryl monostearate (GMS) via solvent diffusion evaporation, the oral bioavailability of resultant RFB-SLNs increased by 5-fold.98
Ion pairing of unsaturated fatty acids (FAs) with antibiotics is another method to improve the antibacterial activity and entrapment efficiency of SLN.99 Linoleic acid (LA), an unsaturated fatty acid, exhibits antibacterial activity.100 Kalhapure et al99 constituted VCM-LA2-SLNs by wrapping ion pair agent LA and vancomycin (VCM) in SLNs. The minimum inhibitory concentration (MICs) of vancomycin hydrochloric (VCM-HCL) SLNs to Staphylococcus aureus and MRSA were 250 g/mL and 500 g/mL, respectively, while those of VCM-LA2 SLNs were 31.25 g/mL and 15.62 g/mL, respectively. Unsaturated fatty acids exhibit antibacterial activity. The main target of unsaturated fatty acids action is the cell membrane, where it interferes with cell energy production by disrupting the electron transport chain and oxidative phosphorylation. Other mechanisms include a decrease in transfer frequency of conjugal DNA and an inhibition of bacterial enoyl-acyl carrier protein reductase FabI.100–102 Vieira et al56 prepared M cell-targeting SLNs by conjugating
 |
Figure 3 Solid lipid nanoparticles with enteric coating for improving stability, palatability, and oral bioavailability of enrofloxacin. (A) The production process of enrofloxacin enteric granules containing SLNs inner core. (B) Scanning electron microscopy photographs of enrofloxacin-loaded SLNs. (C) The accumulation release profiles of SLNs and granules in the simulated SIF (pH=8) (n=3). (D) The plasma enrofloxacin concentration profiles–time of the prepared granules and reference formulation (soluble powder) in pigs (n=6). (E) The influence of high temperature to release ability of enteric granules. (F) The influence of high humidity to release ability of enteric granules. (G) The influence of high light to release ability of enteric granules. SLNs, solid lipid nanoparticles; ENR, enrofloxacin; ENR-SLNs, enrofloxacin-loaded SLNs; RT, room temperature; PR, polyacrylic resin; SIF, simulated intestine fluid; Granules: 10% enrofloxacin enteric granules; Powder: 5% enrofloxacin-soluble powder; HT: high temperature (40°c); HI: high humidity (25°c, 90%±5%); HL: high light (4500±500 lx). Copyright© 2019. Li C, Zhou K, Chen D, et al. <p>Solid lipid nanoparticles with enteric coating for improving stability, palatability, and oral bioavailability of enrofloxacin. Int J Nanomedicine. 2019;14:1619–1631.38 |
NLCs are the second generation of LNFs. They are composed of both solid and liquid lipids at a defined ratio, although the carriers maintain solid features at room temperature or body temperature.103 By the adjustment in lipid composition, NLCs can encapsulate more antibiotics with higher entrapment efficiency and better biocompatibility compared to conventional SLNs.
First-line anti-tuberculosis drugs rifampicin and isoniazid are often used together in clinic, but rifampicin exhibits significant first-pass metabolism and gastric degradation. Rifampicin and isoniazid-loaded SLNs or NLCs were prepared using a modified multi-emulsification method. Although rifampicin and isoniazid showed similar compatibility in the matrices of both SLNs and NLCs, the NLCs system had higher drug loading and entrapment efficiency (Table 2), slower chemical degradation, and therefore exhibited great potential to increase the oral bioavailability of rifampicin.104
Polymer-Based Nanomedicines
Polymers used for oral drug delivery are expected to have good biocompatibility, biodegradability, high drug loading, gastrointestinal stability, safety and controlled release. The carrier composition and properties of polymer-based nanocarriers for oral delivery of antibiotics are listed in Table 3.
 |
Table 3 Composition and Properties of Polymer-Based Nanocarriers for Oral Delivery of Antibiotics |
Polymeric Micelle
Polymeric micelles are composed of amphiphilic copolymers that spontaneously assemble into 5 to 100 nm colloidal particles when their concentrations are above the critical micelle concentration (CMC) (Figure 1).105,106 Antibiotics are encapsulated in the hydrophobic core of micelles, which were stabilized by a hydrophilic corona.107 Polymeric micelles play an important role in the oral delivery of antibiotics not only because of its good solubilization, high drug-loading, and controlled drug release, but also due to its ability to reduce the non-specific uptake by the reticuloendothelial system (RES) and to remain stable in the GIT.28,108
Pyrinezolid, an oxazolidinone-based drug, is effective against many Gram-positive bacteria including methicillin-resistant Staphylococcus aureus (MRSA) and vancomycin-resistant Enterococcus albeit with poor solubility. Methoxy poly (ethylene glycol)-poly (lactide) (mPEG-PLLA) diblock copolymers were used to improve the oral bioavailability and lung targeting of pyrinezolid.28 While the hydrophilic mPEG block inhibited pyrinezolid elimination by RES, and increasing its stability and retention time in the body,109,110 the hydrophobic block PLLA had good biocompatibility and degradability.111 The pyrinezolid micelles were 57.8 ± 0.8 nm with a polydispersity index (PDI) of 0.68 ± 0.019 and an entrapment efficiency of 89.76 ± 3.19%. In vivo experiments showed that the oral bioavailability of pyrinezolid-micelles was 99.7%, significantly higher than that of free pyrinezolid (75.8%). Furthermore, higher lung tissue-targeted aggregation was beneficial to the treatment of MRSA-associated pneumonia.
Micelles with more complicated structure, including mixed micelles and “flower-like” structures, are also used for antibiotic delivery. For example, micelles made of block copolymer with different structures or molecular weights showed better thermodynamic and kinetic stability.112,113 Ciprofloxacin, a P-gp substrate, was delivered by synthetic-mixed micelles made of poloxamer, phosphatidylcholine, and cholesterol. In addition to an outstanding drug-loading capability at 88%, the micelles also significantly increased the drug transport across Caco-2 cells.114 Arafat et al prepared micelles using phosphatidylcholine and sodium deoxycholate, and loaded them with a complex of cefotaxime and 3a,7a-dihydroxy-12-keto-5b-cholanate. The drug-loading capability was 10.5% to 18.9%, and the micelles increased the oral bioavailability of drugs by 4-fold.115
“Flower-like” micelles can be prepared from triblock copolymers.116,117 For example, poly(ε-caprolactone)-b-polyethylene glycol-b-poly(ε-caprolactone) (PCL-b-PEG-b-PCL) micelles were prepared using co-solvent/evaporation method for the delivery of rifampicin. The hydrophobic-hydrophilic-hydrophobic triblock copolymer spontaneously formed a “flower structure” micelle in the aqueous solution, which encapsulated rifampicin in a hydrophobic core, controlled its release, and protected rifampicin from isoniazid and the highly acidic environment in the stomach. Moreover, even in the presence of isoniazid, the oral bioavailability of rifampicin micelles was a 3.3-fold higher than that of free drugs.118
Polymeric Nanoparticles
Biodegradable polymers for antibiotics delivery include chitosan, alginate, gelatin of natural origin, and poly (lactic-co-glycolic acid) (PLGA), and polylactic acid (PLA) of synthetic origin can be used to encapsulate insoluble antibiotics (Figure 1).119,120 The gastrointestinal localization and paracellular or lymphoid transport can be improved by adjusting the molecular weight, hydrophobicity, and adhesion of polymers.121,122
As a natural cationic polysaccharide, chitosan (CS) has been used for the delivery of small molecules, proteins, polypeptides, polysaccharides, and genes.123–125 At the same time, many chitosan derivatives with better delivery properties have been developed such as trimethyl chitosan, carboxymethyl chitosan, and thiolated chitosan, all of which played an important role in the development of oral drug delivery systems.126–128 First, chitosan causes the relaxing of tight junctions between epithelial cells, and subsequently allows more drugs to pass through the epithelial barriers. Second, the positively charged chitosan interacts with the negatively charged mucus to improve the adhesion of the carrier on the mucus layer and promote the transcellular and paracellular transport of encapsulated antibiotics129,130 The reactive functional groups of chitosan and chitosan derivatives also provide a good opportunity for improving the stability in the stomach area and the permeability in the intestine. Gentamicin (GM) is a well-known aminoglycoside antibiotic, but its oral absorption is limited by enzymatic degradation and low bioavailability. Optimizing the oral controlled-release dosage form of GM by using poly(lactic-co-glycolic acid) (PLGA) nanoparticles (NPs) modified with chitosan. The mean residence time was increased to 11.22±0.42 h, indicating a sustained release from the nanoformulation. This observation was further supported by the higher elimination half-life value (6.23±0.53 h) of nanoformulation. Chitosan-modified GM-PLGA nanoparticles has the potential to improve the oral absorption of GM.131 PLGA nanoparticles coated with chitosan were used to load clarithromycin. Both PLGA and chitosan exhibit good biocompatibility and degradability, while the positively charged chitosan also had excellent stability and the ability to cross intestinal epithelial tight junction.48,132 Furthermore, studies showed that chitosan has antibacterial activity.133 In the nanoparticles containing chitosan and PLGA, the zeta potential increased significantly to a positive value, and the encapsulation efficiency was up to 85%. Antibacterial activity test results showed that mixing of natural and synthetic polymers significantly increases the antibacterial activity of clarithromycin against Staphylococcus aureus (ATCC 25,923), Listeria monocytogenes (ATCC 1911), and Klebsiella pneumoniae (ATCC 700,603).44 Similar improvement was also observed when clofazimine and clindamycin hydrochloride were loaded into PLGA polymeric nanoparticles.134 In another example, rifampicin-gantrez-ethylcellulose nanoparticles are prepared with particle sizes ranging from 400 to 500 nm. Compared with rifampicin-gantrez AN-119 nanoparticles, the incorporation of ethylcellulose increased the contact angle and decreased the adhesion, which are beneficial for Peyer’s patches uptake and lymphatic transport. As a result, the relative bioavailability of rifampicin–Gantrez–ethylcellulose nanoparticles nearly tripled, and more drugs accumulated in lung.122 Helicobacter pylori is a micro-aerobic gram-negative bacterium that colonizes the deep layer of human gastric mucus with acidic pH values.135,136 Therefore, therapeutic agents not only need to penetrate the gastric mucus layer but also need to be protected from the acidic environment. Since the solubility of chitosan is pH-dependent, Arif et al prepared pH-responsive sulfhydryl chitosan/poly (malic acid) nanoparticles to encapsulate amoxicillin for the treatment of Helicobacter pylori (H. pylori) infection. The sulfhydryl chitosan/poly (malic acid) was selected based on their mucoadhesive and anticoagulant properties, so that the formulated nanoparticles can adhere and then penetrate the mucous layer at the infection site. Amoxicillin-cysteine-CS/poly (malic acid) nanoparticles have good pH sensitivity and can delay the presence of amoxicillin in gastric acid. The release of the drug enables the drug to be effectively delivered and targeted to the survival area of H. pylori.137 Similarly, new pH-sensitive urea-based coupled chitosan/tripolyphosphate nanoparticles have also been designed for targeted therapy of H. pylori.138
Other Nanopreparations
Nanosuspension
Nanosuspension is a no-carrier two-phase dispersion system composed of surfactant, co-surfactant, and aqueous solution. Its size is generally smaller than 1000 nm (Figure 1).139,140 Sucrose, lactose, mannose and other re-dispersants can be added and then dried to form solid preparations.141,142
Cefdinir is the third-generation cephalosporin antibiotics with poorsolubility and permeability. Sawant et al prepared nanosuspensions with Poloxamer 407 as stabilizers and zirconia as the abrasive by a media milling method to improve cefdinir solubility. The saturated solubility of cefdinir nanosuspension in physiological saline was 1985.3 ± 10.2 mg/mL, while the pure drug was 352.2 ± 6.5 mg/mL. In the case of improved solubility, in vivo results showed that Cmax and Tmax of the marketed suspension were 6.37 mg/mL and 2.1 h, while nanosuspension were 28.45 mg/mL and 1.6 h, respectively. In addition, the oral bioavailability of nanosuspension was 3-fold higher than that of marketed suspension.143 Tetracycline, as a well-known, broad-spectrum, effective and cheap antibiotic for the treatment of diarrhea, is very popular in developing countries with limited medical budgets. However, the emergence of bacterial resistance is gradually eroding its market competitiveness. Mukherjee et al loaded tetracycline within calcium phosphate nanoparticle (Tet-CPNP) to develop a nano-drug that can cross the cellular membrane of resistant bacteria, therefore overcoming the efflux barrier. As the main component of human bones and teeth, calcium phosphate is biocompatible and biodegradable. At the same time, CPNP can be used as a potential carrier of DNA, RNA, protein and various therapeutic drugs to enter different cells, most importantly, tetracycline has a high chelating affinity with divalent metal ions. Tet-CPNP, at a concentration range of 20–40 µg/mL, could kill multi-antibiotics (including tetracycline) resistant diarrhea-causing bacteria like E. coli ETEC 4266, Salmonella Kentucky and Shigella flexneri 2a; whereas free tetracycline (up to 200 µg/mL) had no killing effect on any of the strains. Oral delivery of Tet-CPNP (4.5 µg/kg b.w.) showed better Shigella-infected mice treatment effect compared with the same dose of free tetracycline.144,145
Carbon Nanotubes
Carbon nanotubes are allotropes of carbon with an inner diameters as small as 1 nm (Figure 1).146 Multiwalled carbon nanotubes (MWCNTs) exhibited satisfactory properties such as small particle size, acid stability, high strength, high toughness, and non-swelling.147,148 MWCNTs, encapsulated by liquid crystalline molecularly imprinted polymer (LC-MIP), namely MWCNT@LCMIP, was prepared for oral drug delivery of levofloxacin (LVF).149,150 LC-MIP has the ability to float on an aqueous medium by its solvent-responsive deformation. This elastic structure had good gastric floating and controlled release characteristics, which prolonged the gastric residence time of drugs, and increased the half-life of levofloxacin. Since MWCNT@LC-MIP had a smaller size (average layer thickness was 60 nm), a higher pore property (171.10 × 10−3 cm3 g−1), as well as a longer sustained release time (15.6 h), compared with bared MWCNT, the relative bioavailability of MWCNT@LCMIP was increased by 578.9%, whereas only 11.7% of the former.
Mesoporous Silica Nanoparticles (MSNs)
Since 2001, mesoporous materials have been gradually used as drug delivery vehicles due to their smaller size (2–50 nm) and larger specific surface area (above 1000 m2/g) (Figure 1). In addition to low toxicity, large pore size and high drug loading, it also has good thermal stability, strong controlled release ability and acid stability.151–154 Synthetic lipid-coated mesoporous silica nanoparticles (L-MSNs) were used to improve the antibacterial activity of ciprofloxacin by Mudakavi et al.155 The submicron MSN has a surface area of more than 1000 m2 g−1, which is beneficial for loading more drugs and being absorbed by M cells and macrophages present in Peyer’s patches. Results showed that ciprofloxacin coated with L-MSN had the same or even higher antibacterial activity than free drugs at low doses, and the carrier-controlled drug release also prolongs antibiotic courses. In addition, rifampicin-loaded MSN also showed positive results.156
Conclusion
Numerous studies have shown that the advantages of nano-antibiotics in overcoming multiple gastrointestinal barriers and improving drug stability, solubility, permeability and oral bioavailability (Table 4). However, few review articles have been published to provide an overview on oral nano-antibiotics. To date, some new functional nanomaterials such as nano-selenium, nano-ZnO/TiO2/CuO/Cu2O, dendrimers, graphene, fullerenes, nano-carries for nitric oxide, nano-hydroxyapatite, and bioinspired nanomedicines have been explored in other routes of antibiotic administration, all of these provide scholars with more choices for in-depth exploration and design of convenient and effective oral nano-antibiotics. Although there are many studies on oral nano-antibiotics, few of them are translated to the clinic. Regardless of its effectiveness, there are many issues that need to be considered in the clinical translation of nanomedicine. The first is the synthesis, such as physical and chemical stability, biodegradability, and formulation design. We should work hard to solve the obstacles of large-scale production, such as repeatability and high cost, as well as the obstacles of quality control analysis for characterization, such as polydispersity, scalability, complexity, final product consistency and storage stability. The second issue is pre-clinical evaluation. We should conduct a systematic and effective nanomedicine evaluation to determine the pharmacodynamics, pharmacokinetics and toxicology. Finally, after entering the clinical evaluation stage, the design of nanomedicine, the safety of the human body, the toxicity, and the evaluation of the patient’s therapeutic effect should all minimize time and cost.
 |
Table 4 The Advantages and Disadvantages of Various Nanoparticle Delivery Systems |
Acknowledgments
This work was supported by grants from the National Natural Science Foundation of China [Grant number: 81672947, 81974450], Natural Science Foundation of Hubei Province for Distinguished Young Scholars [Grant number: 2018CFA032], the Fundamental Research Funds for the Central Universities [HUST: No. 2017KFKJXX004] and Wuhan Science and Technology Research Project [Grant number: 2017060201010149].
Disclosure
The authors report no conflicts of interest in this work.
References
1. Drexler M. Institute of M. In: What You Need to Know About Infectious Disease. Washington (DC): National Academies Press (US) Copyright © National Academy of Sciences.; 2010.
2. Fauci AS, Morens DM. The perpetual challenge of infectious diseases. N Engl J Med. 2012;366(5):454–461. doi:10.1056/NEJMra1108296
3. Liu Q, Xu W, Lu S, et al. Landscape of emerging and re-emerging infectious diseases in China: impact of ecology, climate, and behavior. Front Med. 2018;12(1):3–22. doi:10.1007/s11684-017-0605-9
4. Collaborators G. Global, regional, and national age-sex-specific mortality for 282 causes of death in 195 countries and territories, 1980–2017: a systematic analysis for the global burden of disease study 2017. Lancet. 2018;392(10159):1736–1788.
5. McLellan LK, Hunstad DA. Urinary tract infection: pathogenesis and outlook. Trends Mol Med. 2016;22(11):946–957.
6. Cohen J. The immunopathogenesis of sepsis. Nature. 2002;420(6917):885–891.
7. Anjos LM, Marcondes MB, Lima MF, Mondelli AL, Okoshi MP. Streptococcal acute pharyngitis. Rev Soc Bras Med Trop. 2014;47(4):409–413.
8. Schilder AG, Chonmaitree T, Cripps AW, et al. Otitis media. Nat Rev Dis Primers. 2016;2:16063.
9. Kaur CP, Vadivelu J, Chandramathi S. Impact of Klebsiella pneumoniae in lower gastrointestinal tract diseases. J Dig Dis. 2018;19(5):262–271.
10. Nii-Trebi NI. Emerging and neglected infectious diseases: insights, advances, and challenges. Biomed Res Int. 2017;2017(6):1–15. doi:10.1155/2017/5245021
11. Rello J, Parisella FR, Perez A. Alternatives to antibiotics in an era of difficult-to-treat resistance: new insights. Expert Rev Clin Pharmacol. 2019;12(7):635–642. doi:10.1080/17512433.2019.1619454
12. Roberts RR, Hota B, Ahmad I, et al. Hospital and societal costs of antimicrobial-resistant infections in a Chicago teaching hospital: implications for antibiotic stewardship. Clin Infect Dis. 2009;49(8):1175–1184. doi:10.1086/605630
13. Bentley R. The development of penicillin: genesis of a famous antibiotic. Perspect Biol Med. 2005;48(3):444–452. doi:10.1353/pbm.2005.0068
14. Nemeth J, Oesch G, Kuster SP. Bacteriostatic versus bactericidal antibiotics for patients with serious bacterial infections: systematic review and meta-analysis. J Antimicrob Chemother. 2015;70(2):382–395. doi:10.1093/jac/dku379
15. Cars O, Hedin A, Heddini A. The global need for effective antibiotics-moving towards concerted action. Drug Resist Updat. 2011;14(2):68–69. doi:10.1016/j.drup.2011.02.006
16. Aslam B, Wang W, Arshad MI, et al. Antibiotic resistance: a rundown of a global crisis. Infect Drug Resist. 2018;11:1645–1658. doi:10.2147/IDR.S173867
17. Ruddaraju LK, Pammi SVN, Guntuku GS, Padavala VS, Kolapalli VRM. A review on anti-bacterials to combat resistance: from ancient era of plants and metals to present and future perspectives of green nano technological combinations. Asian J Pharm Sci. 2020;15(1):42–59. doi:10.1016/j.ajps.2019.03.002
18. Omolo CA, Kalhapure RS, Agrawal N, Rambharose S, Mocktar C, Govender T. Formulation and molecular dynamics simulations of a fusidic acid nanosuspension for simultaneously enhancing solubility and antibacterial activity. Mol Pharm. 2018;15(8):3512–3526. doi:10.1021/acs.molpharmaceut.8b00505
19. Ventola CL. The antibiotic resistance crisis: part 1: causes and threats. P T. 2015;40(4):277–283.
20. McMullan BJ, Andresen D, Blyth CC, et al. Antibiotic duration and timing of the switch from intravenous to oral route for bacterial infections in children: systematic review and guidelines. Lancet Infect Dis. 2016;16(8):e139–152. doi:10.1016/S1473-3099(16)30024-X
21. Esposito S, Rosazza C, Sciarrabba CS, Principi N. Inhaled antibiotic therapy for the treatment of upper respiratory tract infections. J Aerosol Med Pulm Drug Deliv. 2017;30(1):14–19. doi:10.1089/jamp.2016.1300
22. Williamson DA, Carter GP, Howden BP. Current and emerging topical antibacterials and antiseptics: agents, action, and resistance patterns. Clin Microbiol Rev. 2017;30(3):827–860.
23. MacGregor RR, Graziani AL. Oral administration of antibiotics: a rational alternative to the parenteral route. Clin Infect Dis. 1997;24(3):457–467. doi:10.1093/clinids/24.3.457
24. Moss DM, Curley P, Kinvig H, Hoskins C, Owen A. The biological challenges and pharmacological opportunities of orally administered nanomedicine delivery. Expert Rev Gastroenterol Hepatol. 2018;12(3):223–236. doi:10.1080/17474124.2018.1399794
25. Babadi D, Dadashzadeh S, Osouli M, Daryabari MS, Haeri A. Nanoformulation strategies for improving intestinal permeability of drugs: A more precise look at permeability assessment methods and pharmacokinetic properties changes. J Control Release. 2020;321:669–709. doi:10.1016/j.jconrel.2020.02.041
26. Drucker DJ. Advances in oral peptide therapeutics. Nat Rev Drug Discov. 2020;19(4):277–289. doi:10.1038/s41573-019-0053-0
27. Olivera ME, Manzo RH, Junginger HE, et al. Biowaiver monographs for immediate release solid oral dosage forms: ciprofloxacin hydrochloride. J Pharm Sci. 2011;100(1):22–33. doi:10.1002/jps.22259
28. Long H, Li X, Sang Z, et al. Improving the pharmacokinetics and tissue distribution of pyrinezolid by self-assembled polymeric micelles. Colloids Surf B Biointerfaces. 2017;156:149–156. doi:10.1016/j.colsurfb.2017.05.014
29. Khan F, Katara R, Ramteke S. Enhancement of bioavailability of cefpodoxime proxetil using different polymeric microparticles. AAPS PharmSciTech. 2010;11(3):1368–1375. doi:10.1208/s12249-010-9505-x
30. Rani S, Gothwal A, Pandey PK, et al. HPMA-PLGA based nanoparticles for effective in vitro delivery of rifampicin. Pharm Res. 2019;36(1):19. doi:10.1007/s11095-018-2543-x
31. Davis ME, Chen ZG, Shin DM. Nanoparticle therapeutics: an emerging treatment modality for cancer. Nat Rev Drug Discov. 2008;7(9):771–782. doi:10.1038/nrd2614
32. Patra JK, Das G, Fraceto LF, et al. Nano based drug delivery systems: recent developments and future prospects. J Nanobiotechnology. 2018;16(1):71.
33. Kesisoglou F, Panmai S, Wu Y. Nanosizing — oral formulation development and biopharmaceutical evaluation. Adv Drug Deliv Rev. 2007;59(7):631–644. doi:10.1016/j.addr.2007.05.003
34. Ghosh S, Ghosh S, Sil PC. Role of nanostructures in improvising oral medicine. Toxicol Rep. 2019;6:358–368. doi:10.1016/j.toxrep.2019.04.004
35. BS P, BP V. Understanding peroral absorption: regulatory aspects and contemporary approaches to tackling solubility and permeability hurdles. Acta Pharm Sin B. 2017;7(3):260–280. doi:10.1016/j.apsb.2016.09.005
36. Mabilat C, Gros MF, Nicolau D, et al. Diagnostic and medical needs for therapeutic drug monitoring of antibiotics. Eur J Clin Microbiol Infect Dis. 2020;39(5):791–797. doi:10.1007/s10096-019-03769-8
37. Cowling T, Farrah K. Fluoroquinolones for the Treatment of Other Respiratory Tract Infections: A Review of Clinical Effectiveness, Cost-Effectiveness, and Guidelines. Ottawa (ON): Canadian Agency for Drugs and Technologies in Health; 2019.
38. Li C, Zhou K, Chen D, et al. <p>Solid lipid nanoparticles with enteric coating for improving stability, palatability, and oral bioavailability of enrofloxacin. Int J Nanomedicine. 2019;14:1619–1631. doi:10.2147/IJN.S183479
39. Murgia X, Loretz B, Hartwig O, Hittinger M, Lehr C-M. The role of mucus on drug transport and its potential to affect therapeutic outcomes. Adv Drug Deliv Rev. 2018;124:82–97. doi:10.1016/j.addr.2017.10.009
40. Agarwal SK, Tong B, Bueno OF, Menon RM, Salem AH. Effect of azithromycin on venetoclax pharmacokinetics in healthy volunteers: implications for dosing venetoclax with p-gp inhibitors. Adv Ther. 2018;35(11):2015–2023. doi:10.1007/s12325-018-0793-y
41. Zupancic O, Partenhauser A, Lam HT, Rohrer J, Bernkop-Schnurch A. Development and in vitro characterisation of an oral self-emulsifying delivery system for daptomycin. Eur J Pharm Sci. 2016;81:129–136. doi:10.1016/j.ejps.2015.10.005
42. Thummel K. Enzyme-catalyzed processes of first-pass hepatic and intestinal drug extraction. Adv Drug Deliv Rev. 1997;27(2–3):99–127. doi:10.1016/S0169-409X(97)00039-2
43. Arafat M, Kirchhoefer C, Mikov M, Sarfraz M, Lobenberg R. Nanosized liposomes containing bile salt: a vesicular nanocarrier for enhancing oral bioavailability of BCS class iii drug. J Pharm Pharm Sci. 2017;20:305–318. doi:10.18433/J3CK88
44. Ozturk AA, Yenilmez E, Ozarda MG. Clarithromycin-loaded poly (lactic-co-glycolic acid) (PLGA) nanoparticles for oral administration: effect of polymer molecular weight and surface modification with chitosan on formulation, nanoparticle characterization and antibacterial effects. Polymers. 2019;11(10):1632. doi:10.3390/polym11101632
45. Zhao R, Du S, Liu Y, et al. Mucoadhesive-to-penetrating controllable peptosomes-in-microspheres co-loaded with anti-miR-31 oligonucleotide and Curcumin for targeted colorectal cancer therapy. Theranostics. 2020;10(8):3594–3611. doi:10.7150/thno.40318
46. Maisel K, Reddy M, Xu Q, et al. Nanoparticles coated with high molecular weight PEG penetrate mucus and provide uniform vaginal and colorectal distribution in vivo. Nanomedicine. 2016;11(11):1337–1343. doi:10.2217/nnm-2016-0047
47. Umeyor C, Attama A, Uronnachi E, et al. Formulation design and in vitro physicochemical characterization of surface modified self-nanoemulsifying formulations (SNEFs) of gentamicin. Int J Pharm. 2016;497(1–2):161–198. doi:10.1016/j.ijpharm.2015.10.033
48. Huang W, Zhang C. Tuning the size of poly(lactic-co-glycolic acid) (PLGA) nanoparticles fabricated by nanoprecipitation. Biotechnol J. 2018;13(1):8. doi:10.1002/biot.201700203
49. Gurjar R, Chan CYS, Curley P, et al. Inhibitory effects of commonly used excipients on p-glycoprotein in vitro. Mol Pharm. 2018;15(11):4835–4842. doi:10.1021/acs.molpharmaceut.8b00482
50. Jiang T, Zhang C, Sun W, et al. Doxorubicin encapsulated in TPGS-modified 2d-nanodisks overcomes multidrug resistance. Chemistry. 2020;26(11):2470–2477. doi:10.1002/chem.201905097
51. Tuguntaev RG, Chen S, Eltahan AS, et al. P-gp inhibition and mitochondrial impairment by dual-functional nanostructure based on vitamin e derivatives to overcome multidrug resistance. ACS Appl Mater Interfaces. 2017;9(20):16900–16912. doi:10.1021/acsami.7b03877
52. Bajaj A, Rao MRP, Khole I, Munjapara G. Self-nanoemulsifying drug delivery system of cefpodoxime proxetil containing tocopherol polyethylene glycol succinate. Drug Dev Ind Pharm. 2013;39(5):635–645. doi:10.3109/03639045.2012.683440
53. Hillaireau H, Couvreur P. Nanocarriers’ entry into the cell: relevance to drug delivery. Cell Mol Life Sci. 2009;66(17):2873–2896.
54. Anderson KE, Eliot LA, Stevenson BR, Rogers JA. Formulation and evaluation of a folic acid receptor-targeted oral vancomycin liposomal dosage form. Pharm Res. 2001;18(3):316–322. doi:10.1023/A:1011002913601
55. Araújo F, Pereira C, Costa J, Barrias C, Granja PL, Sarmento B. In vitro M-like cells genesis through a tissue-engineered triple-culture intestinal model. J Biomed Mater Res B Appl Biomater. 2016;104(4):782–788.
56. Vieira AC, Chaves LL, Pinheiro M, Ferreira D, Sarmento B, Reis S. Design and statistical modeling of mannose-decorated dapsone-containing nanoparticles as a strategy of targeting intestinal M-cells. Int J Nanomedicine. 2016;11:2601–2617.
57. Des Rieux A, Fievez V, Garinot M, Schneider YJ, Préat V. Nanoparticles as potential oral delivery systems of proteins and vaccines: a mechanistic approach. J Control Release. 2006;116(1):1–27.
58. Porter CJ, Charman WN. In vitro assessment of oral lipid based formulations. Adv Drug Deliv Rev. 2001;50(Suppl 1):S127–147.
59. Charman WN, Porter CJ, Mithani S, Dressman JB. Physiochemical and physiological mechanisms for the effects of food on drug absorption: the role of lipids and pH. J Pharm Sci. 1997;86(3):269–282.
60. Charman WN. Lipids, lipophilic drugs, and oral drug delivery-some emerging concepts. J Pharm Sci. 2000;89(8):967–978.
61. Das S, Chaudhury A. Recent advances in lipid nanoparticle formulations with solid matrix for oral drug delivery. AAPS PharmSciTech. 2011;12(1):62–76.
62. Rai VK, Mishra N, Yadav KS, Yadav NP. Nanoemulsion as pharmaceutical carrier for dermal and transdermal drug delivery: formulation development, stability issues, basic considerations and applications. J Control Release. 2018;270:203–225.
63. Gursoy RN, Benita S. Self-emulsifying drug delivery systems (SEDDS) for improved oral delivery of lipophilic drugs. Biomed Pharmacother. 2004;58(3):173–182.
64. Kollipara S, Gandhi RK. Pharmacokinetic aspects and in vitro-in vivo correlation potential for lipid-based formulations. Acta Pharm Sin B. 2014;4(5):333–349.
65. Date AA, Desai N, Dixit R, Nagarsenker M. Self-nanoemulsifying drug delivery systems: formulation insights, applications and advances. Nanomedicine. 2010;5(10):1595–1616.
66. Wang L, Dong J, Chen J, Eastoe J, Li X. Design and optimization of a new self-nanoemulsifying drug delivery system. J Colloid Interface Sci. 2009;330(2):443–448.
67. Constantinides PP. Lipid microemulsions for improving drug dissolution and oral absorption: physical and biopharmaceutical aspects. Pharm Res. 1995;12(11):1561–1572.
68. AboulFotouh K, Allam AA, El-Badry M, El-Sayed AM. Self-emulsifying drug-delivery systems modulate P-glycoprotein activity: role of excipients and formulation aspects. Nanomedicine. 2018;13(14):1813–1834.
69. Elgart A, Cherniakov I, Aldouby Y, Domb AJ, Hoffman A. Improved oral bioavailability of BCS class 2 compounds by self nano-emulsifying drug delivery systems (SNEDDS): the underlying mechanisms for amiodarone and talinolol. Pharm Res. 2013;30(12):3029–3044.
70. Almeida SRD, Tippavajhala VK. A rundown through various methods used in the formulation of solid self-emulsifying drug delivery systems (S-SEDDS). AAPS PharmSciTech. 2019;20(8):323.
71. Hussain A, Kumar Singh S. et al. Experimental design-based optimization of lipid nanocarrier as delivery system against Mycobacterium species: in vitro and in vivo evaluation. Pharm Dev Technol. 2017;22(7):910–927.
72. Hussain A, Shakeel F, Singh SK, et al. Solidified SNEDDS for the oral delivery of rifampicin: evaluation, proof of concept, in vivo kinetics, and in silico GastroPlus(TM) simulation. Int J Pharm. 2019;566:203–217.
73. He H, Lu Y, Qi J, Zhao W, Dong X, Wu W. Biomimetic thiamine- and niacin-decorated liposomes for enhanced oral delivery of insulin. Acta Pharm Sin B. 2018;8(1):97–105.
74. Santo IE, Campardelli R, Albuquerque EC. Liposomes size engineering by combination of ethanol injection and supercritical processing. J Pharm Sci. 2015;104(11):3842–3850.
75. Lee WH, Loo CY, Young PM, Traini D, Mason RS, Rohanizadeh R. Recent advances in curcumin nanoformulation for cancer therapy. Expert Opin Drug Deliv. 2014;11(8):1183–1201.
76. Nguyen TX, Huang L, Gauthier M, Yang G, Wang Q. Recent advances in liposome surface modification for oral drug delivery. Nanomedicine. 2016;11(9):1169–1185.
77. Zylberberg C, Matosevic S. Pharmaceutical liposomal drug delivery: a review of new delivery systems and a look at the regulatory landscape. Drug Deliv. 2016;23(9):3319–3329.
78. Daeihamed M, Dadashzadeh S, Haeri A, Akhlaghi MF. Potential of Liposomes for Enhancement of Oral Drug Absorption. Curr Drug Deliv. 2017;14(2):289–303.
79. Hu S, Niu M, Hu F, et al. Integrity and stability of oral liposomes containing bile salts studied in simulated and ex vivo gastrointestinal media. Int J Pharm. 2013;441(1–2):693–700.
80. Ensign LM, Cone R, Hanes J. Oral drug delivery with polymeric nanoparticles: the gastrointestinal mucus barriers. Adv Drug Deliv Rev. 2012;64(6):557–570.
81. Arregui JR, Kovvasu SP, Betageri GV. Daptomycin proliposomes for oral delivery: formulation, characterization, and in vivo pharmacokinetics. AAPS PharmSciTech. 2018;19(4):1802–1809.
82. Senior K. Bilosomes: the answer to oral vaccine delivery? Drug Discov Today. 2001;6(20):1031–1032.
83. Lopes-de-Campos D, Pinto RM, Lima SAC, et al. Delivering amoxicillin at the infection site - a rational design through lipid nanoparticles. Int J Nanomedicine. 2019;14:2781–2795.
84. Mahale NB, Thakkar PD, Mali RG, Walunj DR, Chaudhari SR. Niosomes: novel sustained release nonionic stable vesicular systems–an overview. Adv Colloid Interface Sci. 2012;183–184:46–54.
85. Zhong M, Feng Y, Liao H, et al. Azithromycin cationic non-lecithoid nano/microparticles improve bioavailability and targeting efficiency. Pharm Res. 2014;31(10):2857–2867.
86. Imran M, Shah MR, Ullah F, et al. Sugar-based novel niosomal nanocarrier system for enhanced oral bioavailability of levofloxacin. Drug Deliv. 2016;23(9):3653–3664.
87. Imran M, Shah MR, Ullah F, et al. Glycoside-based niosomal nanocarrier for enhanced in-vivo performance of Cefixime. Int J Pharm. 2016;505(1–2):122–132.
88. Ullah S, Shah MR, Shoaib M, et al. Development of a biocompatible creatinine-based niosomal delivery system for enhanced oral bioavailability of clarithromycin. Drug Deliv. 2016;23(9):3480–3491.
89. Imran M, Shah MR, Ullah F, et al. Double-tailed acyl glycoside niosomal nanocarrier for enhanced oral bioavailability of Cefixime. Artif Cells Nanomed Biotechnol. 2017;45(7):1440–1451.
90. Ullah S, Shah MR, Shoaib M, et al. Hydrophilically modified self-assembling α-tocopherol derivative as niosomal nanocarrier for improving clarithromycin oral bioavailability. Artif Cells Nanomed Biotechnol. 2018;46(3):568–578.
91. Muller RH, Mader K, Gohla S. Solid lipid nanoparticles (SLN) for controlled drug delivery - a review of the state of the art. Eur J Pharm Biopharm. 2000;50(1):161–177.
92. Pandya NT, Jani P, Vanza J, Tandel H. Solid lipid nanoparticles as an efficient drug delivery system of olmesartan medoxomil for the treatment of hypertension. Colloids Surf B Biointerfaces. 2018;165:37–44.
93. Chokshi NV, Khatri HN, Patel MM. Formulation, optimization, and characterization of rifampicin-loaded solid lipid nanoparticles for the treatment of tuberculosis. Drug Dev Ind Pharm. 2018;44(12):1975–1989.
94. Porter CJ, Charman WN. Intestinal lymphatic drug transport: an update. Adv Drug Deliv Rev. 2001;50(1–2):61–80.
95. Jain S, Valvi PU, Swarnakar NK, Thanki K. Gelatin coated hybrid lipid nanoparticles for oral delivery of amphotericin B. Mol Pharm. 2012;9(9):2542–2553.
96. Sharma M, Gupta N, Gupta S. Implications of designing clarithromycin loaded solid lipid nanoparticles on their pharmacokinetics, antibacterial activity and safety. RSC Adv. 2016;6(80):76621–76631.
97. Ozturk AA, Aygul A, Senel B. Influence of glyceryl behenate, tripalmitin and stearic acid on the properties of clarithromycin incorporated solid lipid nanoparticles (SLNs): formulation, characterization, antibacterial activity and cytotoxicity. J Drug Deliv Sci Technol. 2019;54:16.
98. Nirbhavane P, Vemuri N, Kumar N, Khuller GK. Lipid nanocarrier-mediated drug delivery system to enhance the oral bioavailability of rifabutin. AAPS PharmSciTech. 2017;18(3):829–837.
99. Kalhapure RS, Mocktar C, Sikwal DR, et al. Ion pairing with linoleic acid simultaneously enhances encapsulation efficiency and antibacterial activity of vancomycin in solid lipid nanoparticles. Colloids Surf B Biointerfaces. 2014;117:303–311.
100. Zheng CJ, Yoo JS, Lee TG, Cho HY, Kim YH, Kim WG. Fatty acid synthesis is a target for antibacterial activity of unsaturated fatty acids. FEBS Lett. 2005;579(23):5157–5162. doi:10.1016/j.febslet.2005.08.028
101. Desbois AP, Smith VJ. Antibacterial free fatty acids: activities, mechanisms of action and biotechnological potential. Appl Microbiol Biotechnol. 2010;85(6):1629–1642. doi:10.1007/s00253-009-2355-3
102. Smith PA, Romesberg FE. Combating bacteria and drug resistance by inhibiting mechanisms of persistence and adaptation. Nat Chem Biol. 2007;3(9):549–556. doi:10.1038/nchembio.2007.27
103. Fang CL, Al-Suwayeh SA, Fang JY. Nanostructured lipid carriers (NLCs) for drug delivery and targeting. Recent Pat Nanotechnol. 2013;7(1):41–55. doi:10.2174/187221013804484827
104. Banerjee S, Roy S, Nath Bhaumik K, Kshetrapal P, Pillai J. Comparative study of oral lipid nanoparticle formulations (LNFs) for chemical stabilization of antitubercular drugs: physicochemical and cellular evaluation. Artif Cells Nanomed Biotechnol. 2018;46(sup1):540–558. doi:10.1080/21691401.2018.1431648
105. Torchilin VP. Structure and design of polymeric surfactant-based drug delivery systems. J Control Release. 2001;73(2–3):137–172. doi:10.1016/S0168-3659(01)00299-1
106. Gong J, Chen M, Zheng Y, Wang S, Wang Y. Polymeric micelles drug delivery system in oncology. J Control Release. 2012;159(3):312–323. doi:10.1016/j.jconrel.2011.12.012
107. Xiong XB, Falamarzian A, Garg SM, Lavasanifar A. Engineering of amphiphilic block copolymers for polymeric micellar drug and gene delivery. J Control Release. 2011;155(2):248–261. doi:10.1016/j.jconrel.2011.04.028
108. Judy E, Pagariya D, Kishore N. Drug Partitioning in Micellar Media and Its Implications in Rational Drug Design: insights with Streptomycin. Langmuir. 2018;34(11):3467–3484. doi:10.1021/acs.langmuir.7b04346
109. Chu B, Qu Y, Huang Y, et al. PEG-derivatized octacosanol as micellar carrier for paclitaxel delivery. Int J Pharm. 2016;500(1–2):345–359. doi:10.1016/j.ijpharm.2016.01.030
110. Li W, Li X, Gao Y, et al. Inhibition mechanism of P-glycoprotein mediated efflux by mPEG-PLA and influence of PLA chain length on P-glycoprotein inhibition activity. Mol Pharm. 2014;11(1):71–80. doi:10.1021/mp4004223
111. Lu A, Petit E, Jelonek K, et al. Self-assembled micelles prepared from bio-based hydroxypropyl methyl cellulose and polylactide amphiphilic block copolymers for anti-tumor drug release. Int J Biol Macromol. 2020;154:39–47. doi:10.1016/j.ijbiomac.2020.03.094
112. Sun C, Li W, Ma P, et al. Development of TPGS/F127/F68 mixed polymeric micelles: enhanced oral bioavailability and hepatoprotection of syringic acid against carbon tetrachloride-induced hepatotoxicity. Food Chem Toxicol. 2020;137:111126. doi:10.1016/j.fct.2020.111126
113. Zheng B, Zhang X, Peng S, Julian McClements D. Impact of curcumin delivery system format on bioaccessibility: nanocrystals, nanoemulsion droplets, and natural oil bodies. Food Funct. 2019;10(7):4339–4349. doi:10.1039/C8FO02510J
114. Sharif Makhmal Zadeh B, Esfahani G, Salimi A. Permeability of ciprofloxacin-loaded polymeric micelles including ginsenoside as p-glycoprotein inhibitor through a caco-2 cells monolayer as an intestinal absorption model. Molecules. 2018;23(8):1904. doi:10.3390/molecules23081904
115. Arafat M, Kirchhoefer C, Mikov M. Mixed micelles loaded with bile salt: an approach to enhance intestinal transport of the BCS class iii drug cefotaxime in rats. Eur J Drug Metab Pharmacokinet. 2017;42(4):635–645. doi:10.1007/s13318-016-0375-9
116. Najafi M, Kordalivand N, Moradi MA, et al. Native chemical ligation for cross-linking of flower-like micelles. Biomacromolecules. 2018;19(9):3766–3775. doi:10.1021/acs.biomac.8b00908
117. Higashi N, Matsubara S, Nishimura SN, Koga T. Stepwise thermo-responsive amino acid-derived triblock vinyl polymers: ATRP synthesis of polymers, aggregation, and gelation properties via flower-like micelle formation. Materials. 2018;11(3):424.
118. Moretton MA, Hocht C, Taira C, Sosnik A. Rifampicin-loaded ‘flower-like’ polymeric micelles for enhanced oral bioavailability in an extemporaneous liquid fixed-dose combination with isoniazid. Nanomedicine. 2014;9(11):1635–1650. doi:10.2217/nnm.13.154
119. George A, Shah PA, Shrivastav PS. Natural biodegradable polymers based nano-formulations for drug delivery: A review. Int J Pharm. 2019;561:244–264. doi:10.1016/j.ijpharm.2019.03.011
120. Rauta PR, Das NM, Nayak D, Ashe S, Nayak B. Enhanced efficacy of clindamycin hydrochloride encapsulated in PLA/PLGA based nanoparticle system for oral delivery. IET Nanobiotechnol. 2016;10(4):254–261. doi:10.1049/iet-nbt.2015.0021
121. Taipaleenmäki E, Städler B. Recent advancements in using polymers for intestinal mucoadhesion and mucopenetration. Macromol Biosci. 2020;20(3):e1900342. doi:10.1002/mabi.201900342
122. Bachhav SS, Dighe VD, Devarajan PV. Exploring Peyer’s patch uptake as a strategy for targeted lung delivery of polymeric rifampicin nanoparticles. Mol Pharm. 2018;15(10):4434–4445. doi:10.1021/acs.molpharmaceut.8b00382
123. Lang X, Wang T, Sun M, Chen X, Liu Y. Advances and applications of chitosan-based nanomaterials as oral delivery carriers: A review. Int J Biol Macromol. 2020;154:433–445. doi:10.1016/j.ijbiomac.2020.03.148
124. Huang TW, Ho YC, Tsai TN, Tseng CL, Lin C, Mi FL. Enhancement of the permeability and activities of epigallocatechin gallate by quaternary ammonium chitosan/fucoidan nanoparticles. Carbohydr Polym. 2020;242:116312. doi:10.1016/j.carbpol.2020.116312
125. Wang J, Kong M, Zhou Z, et al. Mechanism of surface charge triggered intestinal epithelial tight junction opening upon chitosan nanoparticles for insulin oral delivery. Carbohydr Polym. 2017;157:596–602. doi:10.1016/j.carbpol.2016.10.021
126. Kulkarni AD, Patel HM, Surana SJ, Vanjari YH, Belgamwar VS, Pardeshi CV. N,N,N-Trimethyl chitosan: an advanced polymer with myriad of opportunities in nanomedicine. Carbohydr Polym. 2017;157:875–902. doi:10.1016/j.carbpol.2016.10.041
127. Teng Z, Luo Y, Wang Q. Carboxymethyl chitosan-soy protein complex nanoparticles for the encapsulation and controlled release of vitamin D(3). Food Chem. 2013;141(1):524–532. doi:10.1016/j.foodchem.2013.03.043
128. Bernkop-Schnurch A, Guggi D, Pinter Y. Thiolated chitosans: development and in vitro evaluation of a mucoadhesive, permeation enhancing oral drug delivery system. J Control Release. 2004;94(1):177–186. doi:10.1016/j.jconrel.2003.10.005
129. Cole H, Bryan D, Lancaster L, Mawas F, Vllasaliu D. Chitosan nanoparticle antigen uptake in epithelial monolayers can predict mucosal but not systemic in vivo immune response by oral delivery. Carbohydr Polym. 2018;190:248–254. doi:10.1016/j.carbpol.2018.02.084
130. Palazzo C, Trapani G, Ponchel G, Trapani A, Vauthier C. Mucoadhesive properties of low molecular weight chitosan- or glycol chitosan- and corresponding thiomer-coated poly(isobutylcyanoacrylate) core-shell nanoparticles. Eur J Pharm Biopharm. 2017;117:315–323. doi:10.1016/j.ejpb.2017.04.020
131. Akhtar B, Muhammad F, Aslam B, Saleemi MK, Sharif A. Pharmacokinetic profile of chitosan modified poly lactic co-glycolic acid biodegradable nanoparticles following oral delivery of gentamicin in rabbits. Int J Biol Macromol. 2020;164:1493–1500. doi:10.1016/j.ijbiomac.2020.07.206
132. Ahmed TA, Aljaeid BM. Preparation, characterization, and potential application of chitosan, chitosan derivatives, and chitosan metal nanoparticles in pharmaceutical drug delivery. Drug Des Devel Ther. 2016;10:483–507. doi:10.2147/DDDT.S99651
133. Varlamov VP, Il’ina AV, Shagdarova BT, Lunkov AP, Mysyakina IS. Chitin/Chitosan and Its Derivatives: fundamental Problems and Practical Approaches. Biochemistry (Mosc). 2020;85(Suppl 1):S154–S176. doi:10.1134/S0006297920140084
134. Chaves LL, Costa Lima SA, Vieira ACC, et al. Development of PLGA nanoparticles loaded with clofazimine for oral delivery: assessment of formulation variables and intestinal permeability. Eur J Pharm Sci. 2018;112:28–37. doi:10.1016/j.ejps.2017.11.004
135. Hejazi R, Amiji M. Stomach-specific anti-H. pylori therapy. I: preparation and characterization of tetracyline-loaded chitosan microspheres. Int J Pharm. 2002;235(1–2):87–94. doi:10.1016/S0378-5173(01)00985-1
136. Chang CH, Lin YH, Yeh CL, et al. Nanoparticles incorporated in pH-sensitive hydrogels as amoxicillin delivery for eradication of Helicobacter pylori. Biomacromolecules. 2010;11(1):133–142. doi:10.1021/bm900985h
137. Arif M, Dong QJ, Raja MA, Zeenat S, Chi Z, Liu CG. Development of novel pH-sensitive thiolated chitosan/PMLA nanoparticles for amoxicillin delivery to treat Helicobacter pylori. Mater Sci Eng C Mater Biol Appl. 2018;83:17–24. doi:10.1016/j.msec.2017.08.038
138. Jing ZW, Jia YY, Wan N, et al. Design and evaluation of novel pH-sensitive ureido-conjugated chitosan/TPP nanoparticles targeted to Helicobacter pylori. Biomaterials. 2016;84:276–285. doi:10.1016/j.biomaterials.2016.01.045
139. Patravale VB, Date AA, Kulkarni RM. Nanosuspensions: a promising drug delivery strategy. J Pharm Pharmacol. 2004;56(7):827–840. doi:10.1211/0022357023691
140. Patel VR, Agrawal YK. Nanosuspension: an approach to enhance solubility of drugs. J Adv Pharm Technol Res. 2011;2(2):81–87. doi:10.4103/2231-4040.82950
141. Fülöp V, Jakab G, Bozó T, et al. Study on the dissolution improvement of albendazole using reconstitutable dry nanosuspension formulation. Eur J Pharm Sci. 2018;123:70–78. doi:10.1016/j.ejps.2018.07.027
142. Medarevic D, Djuris J, Ibric S, Mitric M, Kachrimanis K. Optimization of formulation and process parameters for the production of carvedilol nanosuspension by wet media milling. Int J Pharm. 2018;540(1–2):150–161. doi:10.1016/j.ijpharm.2018.02.011
143. Sawant KK, Patel MH, Patel K. Cefdinir nanosuspension for improved oral bioavailability by media milling technique: formulation, characterization and in vitro-in vivo evaluations. Drug Dev Ind Pharm. 2016;42(5):758–768. doi:10.3109/03639045.2015.1104344
144. Mukherjee R, Patra M, Dutta D, Banik M, Basu T. Tetracycline-loaded calcium phosphate nanoparticle (Tet-CPNP): rejuvenation of an obsolete antibiotic to further action. Biochim Biophys Acta. 2016;1860(9):1929–1941. doi:10.1016/j.bbagen.2016.06.006
145. Mukherjee R, Dutta D, Patra M, Chatterjee B, Basu T. Nanonized tetracycline cures deadly diarrheal disease ‘shigellosis’ in mice, caused by multidrug-resistant Shigella flexneri 2a bacterial infection. Nanomedicine. 2019;18:402–413. doi:10.1016/j.nano.2018.11.004
146. Rode A, Sharma S, Mishra DK. Carbon nanotubes: classification, method of preparation and pharmaceutical application. Curr Drug Deliv. 2018;15(5):620–629. doi:10.2174/1567201815666171221124711
147. Wong BS, Yoong SL, Jagusiak A, et al. Carbon nanotubes for delivery of small molecule drugs. Adv Drug Deliv Rev. 2013;65(15):1964–2015. doi:10.1016/j.addr.2013.08.005
148. Karimi M, Solati N, Ghasemi A, et al. Carbon nanotubes part II: a remarkable carrier for drug and gene delivery. Expert Opin Drug Deliv. 2015;12(7):1089–1105. doi:10.1517/17425247.2015.1004309
149. Zhang LP, Tan XX, Huang YP, Liu ZS. Floating liquid crystalline molecularly imprinted polymer coated carbon nanotubes for levofloxacin delivery. Eur J Pharm Biopharm. 2018;127:150–158. doi:10.1016/j.ejpb.2018.02.012
150. Zhang LP, Wang XL, Pang QQ, et al. Solvent-responsive floating liquid crystalline-molecularly imprinted polymers for gastroretentive controlled drug release system. Int J Pharm. 2017;532(1):365–373. doi:10.1016/j.ijpharm.2017.09.008
151. Vadia N, Rajput S. Study on formulation variables of methotrexate loaded mesoporous MCM-41 nanoparticles for dissolution enhancement. Eur J Pharm Sci. 2012;45(1–2):8–18. doi:10.1016/j.ejps.2011.10.016
152. Doadrio AL, Sánchez-Montero JM, Doadrio JC, Salinas AJ, Vallet-Regí M. Mesoporous silica nanoparticles as a new carrier methodology in the controlled release of the active components in a polypill. Eur J Pharm Sci. 2017;97:1–8. doi:10.1016/j.ejps.2016.11.002
153. Florek J, Caillard R, Kleitz F. Evaluation of mesoporous silica nanoparticles for oral drug delivery - current status and perspective of MSNs drug carriers. Nanoscale. 2017;9(40):15252–15277. doi:10.1039/C7NR05762H
154. Tan X, Liu X, Zhang Y, et al. Silica nanoparticles on the oral delivery of insulin. Expert Opin Drug Deliv. 2018;15(8):805–820. doi:10.1080/17425247.2018.1503250
155. Mudakavi RJ, Raichur AM, Chakravortty D. Lipid coated mesoporous silica nanoparticles as an oral delivery system for targeting and treatment of intravacuolar Salmonella infections. RSC Adv. 2014;4(105):61160–61166. doi:10.1039/C4RA12973C
156. Xia X, Pethe K, Kim R, et al. Encapsulation of anti-tuberculosis drugs within mesoporous silica and intracellular antibacterial activities. Nanomaterials. 2014;4(3):813–826. doi:10.3390/nano4030813
157. Uhl P, Pantze S, Storck P, et al. Oral delivery of vancomycin by tetraether lipid liposomes. Eur J Pharm Sci. 2017;108:111–118. doi:10.1016/j.ejps.2017.07.013
158. Singh H, Jindal S, Singh M, Sharma G, Kaur IP. Nano-formulation of rifampicin with enhanced bioavailability: development, characterization and in-vivo safety. Int J Pharm. 2015;485(1–2):138–151. doi:10.1016/j.ijpharm.2015.02.050
159. Chaves LL, Lima S, Vieira ACC, Ferreira D, Sarmento B, Reis S. Overcoming clofazimine intrinsic toxicity: statistical modelling and characterization of solid lipid nanoparticles. J R Soc Interface. 2018;15:139. doi:10.1098/rsif.2017.0932
160. Dong Z, Xie S, Zhu L, Wang Y, Wang X, Zhou W. Preparation and in vitro, in vivo evaluations of norfloxacin-loaded solid lipid nanopartices for oral delivery. Drug Deliv. 2011;18(6):441–450. doi:10.3109/10717544.2011.577109
161. Xie S, Zhu L, Dong Z, Wang Y, Wang X, Zhou W. Preparation and evaluation of ofloxacin-loaded palmitic acid solid lipid nanoparticles. Int J Nanomedicine. 2011;6:547–555.
162. Bhandari R, Kaur IP. Pharmacokinetics, tissue distribution and relative bioavailability of isoniazid-solid lipid nanoparticles. Int J Pharm. 2013;441(1–2):202–212. doi:10.1016/j.ijpharm.2012.11.042
163. Patel MD, Date PV, Gaikwad RV, Samad A, Malshe VC, Devarajan PV. Comparative evaluation of polymeric nanoparticles of rifampicin comprising Gantrez and poly(ethylene sebacate) on pharmacokinetics, biodistribution and lung uptake following oral administration. J Biomed Nanotechnol. 2014;10(4):687–694. doi:10.1166/jbn.2014.1739
164. Rani S, Rana R, Saraogi GK, Kumar V, Gupta U. Self-emulsifying oral lipid drug delivery systems: advances and challenges. AAPS Pharm Sci Tech. 2019;20(3):129. doi:10.1208/s12249-019-1335-x
165. He H, Lu Y, Qi J, Zhu Q, Chen Z, Wu W. Adapting liposomes for oral drug delivery. Acta Pharm Sin B. 2019;9(1):36–48. doi:10.1016/j.apsb.2018.06.005
166. Raza A, Sime FB, Cabot PJ, Maqbool F, Roberts JA, Falconer JR. Solid nanoparticles for oral antimicrobial drug delivery: a review. Drug Discov Today. 2019;24(3):858–866. doi:10.1016/j.drudis.2019.01.004
167. Simoes SM, Figueiras AR, Veiga F, Concheiro A, Alvarez-Lorenzo C. Polymeric micelles for oral drug administration enabling locoregional and systemic treatments. Expert Opin Drug Deliv. 2015;12(2):297–318. doi:10.1517/17425247.2015.960841
168. Chen D, Ding PT, Deng YH, Wang SL. [Advances in the study of polymeric micelles used in oral administration]. Yao Xue Xue Bao. 2010;45(5):560–564. Chinese.
169. Singh SK, Vaidya Y, Gulati M, Bhattacharya S, Garg V, Pandey NK. Nanosuspension: principles, perspectives and practices. Curr Drug Deliv. 2016;13(8):1222–1246. doi:10.2174/1567201813666160101120452
170. Chen A, Shi Y, Yan Z, et al. Dosage form developments of nanosuspension drug delivery system for oral administration route. Curr Pharm Des. 2015;21(29):4355–4365.
171. Bharti C, Gulati N, Nagaich U. Mesoporous silica nanoparticles in target drug delivery system: A review. Int J Pharm Investig. 2015;5(3):124–133. doi:10.4103/2230-973X.160844
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
