Back to Journals » Patient Preference and Adherence » Volume 13
Preferences for cervical cancer screening service attributes in rural China: a discrete choice experiment
Authors Li S , Liu S, Ratcliffe J , Gray A, Chen G
Received 17 January 2019
Accepted for publication 27 March 2019
Published 30 May 2019 Volume 2019:13 Pages 881—889
DOI https://doi.org/10.2147/PPA.S201913
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Johnny Chen
Shunping Li,1,2 Shimeng Liu,1,2 Julie Ratcliffe,3 Alastair Gray,4 Gang Chen5
1School of Health Care Management, Shandong University, Jinan 250012, People’s Republic of China; 2NHC Laboratory of Health Economics and Policy Research (Shandong University), Jinan, 250012, People’s Republic of China; 3Health Economics Group, College of Nursing and Health Sciences, Flinders University, Adelaide, South Australia 5042, Australia; 4Health Economics Research Centre, Nuffield Department of Population Health, University of Oxford, Oxford, UK; 5Flinders Centre for Innovation in Cancer, College of Medicine and Public Health, Flinders University, Adelaide, South Australia 5042, Australia
Objectives: Compared with other cancers, screening for cervical cancer is highly cost-effective. However, due to limited awareness about cervical cancer and many other factors, women’s attendance rate in rural China for cervical cancer screening remains low. This study aimed to determine women’s preferences for cervical cancer screening, to help enhance screening uptake.
Methods: A discrete choice experiment (DCE) was conducted among a population-based random sample of 420 women (30–65 years old) in August 2015. Attributes included the percentage of cervical cancer-related death reduction, screening interval, screening location, screening pain, waiting time for screening results and out-of-pocket costs. Mixed logit models were used to analyze the relative importance of each screening attribute.
Results: When considering a screening program, the screening cost, location and the percentage of cervical cancer-related death reduction were of most concern to women. Among the presented attributes, the pain associated with the process of screening was of the least concern.
Conclusions: All six attributes in our study were found to have a large influence on the preference for cervical cancer screening, and significant preference heterogeneity existed among participants. The findings indicate that the maintenance of a free screening program is essential to increasing screening uptake in this vulnerable population.
Keywords: discrete choice experiment, cervical cancer, screening, preference, China
Introduction
Cervical cancer is the fourth most common female cancer globally and the leading cause of cancer death among women, accounting for almost 12% of all female cancers.1 Around 87% of the cervical cancer deaths occur in developing countries, where the number of new cases ranks cervical cancer second among malignancies in female patients.2,3
In China, there were an estimated 100,700 new cases of cervical cancer in 2013, ranking as the sixth most common incident cancer among all newly diagnosed cancers in females.4 Although the incidence of cervical cancer in China is low in comparison to that of western countries, the mortality rate remains high, especially in rural areas.5 A total of 29,526 women died of the disease in 2012 in China, accounting for 11% all cervical cancer deaths worldwide.2 Furthermore, forward projections indicate that the number of new cases will rise as high as 186,600 by 2050.6
As a preventable and treatable disease, screening of precancerous lesions can reduce cervical cancer’s incidence and mortality. Compared to other cancers, screening for cervical cancer is very cost-effective, because no other cancer offers as good a means for primary and secondary prevention as cervical cancer.7,8 However, in many developing countries, cervical cancer screening programs are unavailable or are poorly accessible.9 Although China has provided free cervical cancer screening programs in some rural areas for women aged 35 to 59 years since 2009, a lack of awareness and knowledge and the absence of a nationwide organized cervical cancer screening program has resulted in many rural women remaining unaware of the benefits of screening and unwilling to undergo the procedure. Consequently, cervical cancer remains an important public health problem in mainland China.5
To improve patient-centered health care, it is important to investigate women’s preferences for cervical cancer screening. Obtaining insights into these preferences will provide valuable information to clinicians and policy decision makers for improving screening uptake.10 The decision to participate or not in a screening program is preferably based on an individual decision-making process of weighting (or trading off) the test burden against the potential benefits of screening.
Worldwide, discrete choice experiment (DCE) has been extensively applied in health care research, in a wide range of contexts and addressing different policy and research issues.11 To date, there have been very limited studies using DCE to explore the preferences for cervical cancer screening.12–15 These studies found that cost, chance of being recalled, waiting time for test results, sensitivity of test, pain and the provider’s gender were relatively more important when choosing a cervical cancer screening program by women. The objective of this study was to use a DCE, a quantitative methodology for investigating these trade-offs, to assess women’s preferences for cervical cancer screening in rural China.
Methods
Discrete choice experiment
The methodology of DCEs is grounded in random utility theory, where a discrete choice is offered and participants choose the option with the highest utility among candidate options.16 The technique has been increasingly used in health economics to consider attribute importance in delivering health care, with consideration both to the aspect of the patient experience and health outcomes, as well as to trade-offs between these and willingness to pay (WTP) for different attributes.11,17 In comparison with other quantitative methods (eg, a ranking task), a DCE task more closely resembles a real-world decision; in addition, the output from DCEs can facilitate future policy implementation (eg, the calculation of the uptake rate for hypothetical scenarios).18 In the context of this study, it is assumed that a cancer screening program can be described by a series of attributes and their corresponding levels.19 The DCE design and analysis was conducted following the International Society For Pharmacoeconomics and Outcomes Research (ISPOR) good practice principles outlined by Bridges et al.20
The salient features or characteristics (attributes) associated with the provision of cervical cancer screening and their associated levels were formulated from a literature review,12–15,21–24 coupled with a series of in-depth qualitative interviews with rural women (N=15) aged 30–65 years old, and consultation with clinical experts in the field of cervical cancer screening in China. During the interview, women were asked to comment on a candidate list of cervical cancer screening attributes which were extracted from the literature review and to indicate any additional candidate attributes that were omitted from the literature review. Following this process and after consulting with the clinical experts, a total of six final attributes most relevant to cervical cancer screening in rural China were selected: the percentage of cervical cancer-related death reduction (with levels been determined by consulting clinicians as well as the levels of attribute “chance of dying from cervical cancer” adopted by Wordsworth et al15), screening interval, screening pain, screening location, waiting time for screening results and the out-of-pocket (OOP) costs (with levels been determined by the relevant local government document) (Table 1).
 | Table 1 DCE attributes and levels |
The combination of six attributes with three levels associated with each attribute resulted in 729 (36) possible cervical cancer screening scenarios and a total of 265,356 possible pairwise choices ((729×728)/2). A sequential orthogonal factorial design was adopted to generate a more manageable 27 choice scenarios (which were further blocked into three versions containing 9 choice sets each) using the Ngene version 1.1.2 DCE design software (Choice-Metrics, Sydney, Australia). Within each version, a single choice set was duplicated to examine the internal consistency of participants.
Each discrete choice set consisted of two cervical cancer screening alternatives. Participants were first asked to consider two alternatives in a choice set and to choose the alternative that they preferred. Secondly, participants were asked a follow-up question as to whether in real life they would be willing to participate in their preferred scenario (see Table 2 for an example choice set, and another example in Chinese in
Participants
A stratified random sampling framework was utilized. In the first step, four counties (Rencheng County, Qufu County, Sishui County and Yutai County) out of a total of 11 counties in Jining Prefecture were randomly selected. Next, two townships were randomly selected within each county and then two villages were randomly selected from within each sampled township. Finally, a minimum of 100 women aged 30 to 65 years for each county were randomly selected from potential candidate participants identified by each village committee. Women who had previously had a hysterectomy or who indicated no prior sexual experience were excluded from participation.26
Women in rural China are often less well-educated relative to their urban counterparts. The DCE questionnaire was therefore administered as a one-to-one face-to-face interview to assist the participant in understanding and completing the DCE task, thereby ensuring the quality of the investigation. Participants received an insulated cup (35 Chinese Yuan; the average annual exchange rate between US$ and CNY in 2015 was US$1= CNY 6.227) as a gift following survey completion. Five researchers from Shandong University who were able to speak in the relevant local dialect were trained to administer the DCE questionnaire via a face-to-face interview. The survey contains four sections. Section A contained a series of questions regarding participants’ socio-demographic characteristics; Section B included cervical cancer knowledge; Section C assessed participants’ attitudes towards cervical cancer and its screening; Section D contained the DCE task. Results regarding participants’ cervical cancer knowledge and attitudes have been published in a separate paper.26 The full questionnaire was piloted among 10 rural women in Sishui County, aiming to examine the intelligibility, acceptability, and validity of the questionnaire. The survey data was collected in August 2015. The process of administering the questionnaire took about 30 to 40 mins on average and all completed questionnaires were returned directly to the investigators.
Data analysis
Taking into account potential preference heterogeneity, a mixed logit model was used to analyze the DCE data.27,28 The utility function can be specified as:
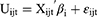
where Uijt is the utility individual i derives from choosing alternative j in choice scenario t, X is a vector of observed attributes (ie, the cervical cancer screening characteristics and corresponding levels), β is a vector of coefficients reflecting the desirability of the attributes, and εijt is an error term. Although most previous studies specify the coefficient for monetary attribute in choice models to be fixed, it is often unrealistic to assume that all participants have the same preferences regarding the costs of a cervical cancer screening program.29 In our study, all attributes were specified as having a random component. In addition, all attribute variables were coded as dummy variables except for cost which was specified as a continuous variable.
To reveal the trade-offs that participants were willing to make between screening attributes, the WTP for marginal improvements in the attributes (which were calculated by dividing the estimated coefficients for the remaining attributes by the estimated coefficient for the cost attribute) and associated confidence intervals were estimated.30 The WTP estimates will facilitate policy makers in understanding how much out-of-pocket cost an individual would be willing to incur for improvements in other attributes of a screening program. Finally, by using the estimated coefficients, the uptake rates of a cervical cancer screening program were calculated.31 Such information would be useful for future policy implementation in rural China. All statistical analyses were conducted in Stata version 12.1 (StataCorp LP, College Station, Texas, USA).
Results
Participants
A total of 420 women consented to participate in this study. Among these, 405 (96%) successfully completed the survey, 57 (14%) participants failed the consistency test in the duplicated DCE task, and there were no statistically significant differences in demographic characteristics between those who failed versus who passed the test (see Table 3). Women who passed the consistency test had a mean (standard deviation, SD) age of 49.1 (8.3) years, the vast majority (96.6%) were married, and around half (50.9%) had an annual family income lower than 20,000 CNY.
 | Table 3 Demographic characteristics of participants |
DCE results
The DCE results based on the analysis sample are reported in Table 4 and
 | Table 4 Main effects model for DCE (n=348) |
The WTP estimates for the analysis sample (n=348) are also presented in Table 4. Compared to the reference levels for each attribute, screening location and the percentage risk reduction in cervical cancer-related deaths had the largest impact on the preferences for screening. For example, compared to having screening in the county, participants were willing to pay 52 CNY and 110 CNY to participate in a cervical cancer screening program located in town and village, respectively. In comparison to a waiting time of between 3 and 6 months, participants were willing to pay 44 CNY and 61 CNY to reduce waiting times to between 2 weeks and 3 months, and to less than 2 weeks, respectively.
Selected subgroup analyses results are presented in
Finally, the calculated uptake rates based upon the full sample are shown in Figure 1. A reduction in the out-of-pocket costs associated with screening from 300 CNY to 50 and 0 CNY had the largest effect on preference of women to choose to participate in a cervical cancer screening program, increasing the probability of uptake by 93% and 96%, respectively. In contrast, compared with a moderate degree of pain associated with the screening process, the absence of pain enhances the probability of uptake by only 13% overall.
Discussion
To the best of our knowledge, this is the first study to utilize DCE methodology to elicit women’s preferences for cervical screening attributes in China. All six attributes considered in this study were found to be statistically significant in influencing individual preferences to participate (or not) in a cervical cancer screening program.
The percentage of cervical cancer-related death reduction has an important positive influence on preferences for cervical cancer screening. Our finding that women attach much importance to cervical cancer mortality reduction is consistent with the results of a previous study conducted in Scotland.15 Furthermore, the finding that participants were willing to pay more on average for screening programs associated with higher percentage of cervical cancer-related death reduction demonstrates that they trade benefits and costs of a screening test. When comparing the WTP estimates between different subgroups, women with higher education and income levels were prepared to pay more for the percentage of cervical cancer-related death reduction relative to those with lower education and income levels. This finding may be related to the ability to pay or the fact that women with higher education and income levels are more likely to pay more attention to their health and have more opportunities to obtain relevant information and thereby increase their cervical cancer screening knowledge.26
In relation to screening intervals, our findings reinforce those of previous studies which have identified that participants tend to prefer a shorter screening interval over a longer screening interval. There is evidence to suggest that women feel that longer waiting times expose them to a higher risk of contracting cervical cancer, and are therefore associated with increased worry and anxiety.15,32
With regards to OOP costs, compared with a screening program that require women to pay 300 CNY to access, providing a free cost screening program will increase the uptake rate by 96%. In comparison, a study conducted in the UK by Wordsworth et al15 found that a unit change in the cost of screening was the least important attribute. The difference may be due to relative affluence of the UK cohort relative to this study and therefore their increased capacity/ability to pay relative to rural Chinese women, or to difficulties in accepting the possibility of out-of-pocket costs in a predominantly free at point of use health system. In addition, several previous studies including those by Murray and McMillan33 in Northern Ireland (on breast cancer screening) and Doyle34 in the UK found the fear of the associated pain to be one of the most important reasons for declining screening. These findings contrast with the finding from our study that the pain associated with the screening process was considered to be relatively unimportant in influencing uptake.
All of the participants in our study came from rural areas, and for the majority, their annual family income was relatively low (less than 30,000 CNY). In addition, cervical cancer screening is currently provided free of charge in some rural areas. Hence, our finding that participants strongly preferred not to have to pay any out-of-pocket costs for screening is unsurprising. In a previous study conducted by our team we found that although Chinese governments currently provide free cervical cancer screening for target populations in pilot counties, if women were asked to contribute towards the cost of providing these screening services in the future, it will become an additional barrier preventing uptake for many rural Chinese women.26 Consequently maintaining a cervical cancer screening program that is freely accessible is crucial for increasing uptake amongst women in rural China.
A previous study conducted by Fort et al21 in rural locations in Malawi indicated that village participants were particularly frustrated with the waiting times to receive cervical cancer screening results from the District Hospital, and longer waiting times tended to negatively influence uptake rates. Our study found similar results, in that reducing the waiting time to receive results from 6 months to less than 2 weeks enhanced the probability of uptake by 38.1% overall. In relation to the subgroup analyses, we found that those with higher education and family income levels were willing to pay about 3 times more on average to receive their results within 2 weeks than those with lower education and family income levels. Women with higher education and income levels were more likely to be working full time and have higher capability to pay due to higher disposable income levels. In addition, waiting excessively long time periods for the results may be associated with a higher opportunity cost for them.
For screening location, the participants prefer the location “village” and there was no significant difference between the subgroups. Hence, this finding cannot be explained by knowledge levels, family income and previous cervical cancer screening history. The higher cost and inconvenience associated with attending cervical cancer screening far away from home for women could be a potential explanation for this finding. The majority (74%) of participants in our study were less than 55 years old and they were more likely to be working full time or taking care of younger children at home.26 A DCE study conducted in the Netherlands to investigate preferences for colorectal cancer screening also reported similar findings in that participants preferred the location “home” rather than hospital.35 Our findings indicate that the provision of cervical cancer screening programs in primary health care institutions located close to women’s homes has the potential to enhance the cervical cancer screening uptake in rural areas of China.
This study has some limitations. First, as with all DCE results it is based on ‘stated preferences’, rather than on ‘revealed preferences’. To verify the credibility of this study’s results, further research would ideally compare the stated preferences of women with their actual behavior in a cervical cancer screening program. Second, only rural women from four counties were included in this research and hence the findings are limited in their generalizability. Differences may exist between cervical cancer screening preferences and the likelihood of uptake for women living in rural China relative to those living in urban areas. Further research should be conducted to explore the potential for differences in women’s preferences according to geographical location. Thirdly, there may be some concerns about the appropriateness of the attribute “the percentage of cervical cancer-related death reduction” since the range of its level (20% to 80%) seems too large to be realistic. It should be noted that this does not refer to the reduction on the absolute mortality rate, but a percentage change. For example, in Wordsworth et al, the implied largest percentage change (reduction) on the “chance of dying from cervical cancer” was (0.4%-2%)/2%=−80%. We opted to present this attribute as percentage change rather than the absolute mortality since it is easier for our respondents to understand the magnitude of the vaccination benefit. Finally, since the participants in this study were from rural areas with relatively low education levels on average, there could be some concern about the extent to which the participants fully understood the DCE task. However, in the field, the DCE survey was conducted via a one-to-one face-to-face interview by trained researchers who spoke the local dialect to maximize the probability that participants would fully understand the DCE task. In addition, there was no significant association between education and passing the consistency check. Furthermore, following the strategy adopted by Milte et al,36 we have empirically investigated this potential issue by estimating a generalized multinomial logit model within which the lowest education level was specified to impact on scale heterogeneity. The statistically nonsignificant coefficient attached to the estimated dummy variable reflecting the lowest education level further indicates that those in the lowest education level did not reveal less consistent preferences relative to those with higher education levels.
Conclusion
Cervical cancer is one of the most preventable cancers and is curable at early stages. Although a nationwide screening program, publicly funded and free at the point of access, has been launched for rural women in China, the universal screening rate has to date not attained high uptake with around one-third of candidate women never participating in screening. This study used DCE methodology to investigate six key factors influential in determining women’s preferences for participation in cervical cancer screening programs in rural China. All six attributes were found to be significantly influential in affecting women’s preferences for cervical cancer screening. In addition, there exists a certain degree of both observable and unobservable preference heterogeneity among participants. The findings from this study will help policy makers further improve cervical cancer screening uptake rates in rural China and have the potential to influence cervical cancer screening uptake rates in other developing countries with similar characteristics.
Details of ethics approval
Verbal informed consent was obtained from all participants prior to the survey. This study (including this consent process) was approved by the Medical Ethics Committee of Medical School, Shandong University (LL-201401048), and conforms to the ethics guidelines of the Declaration of Helsinki.
Acknowledgments
We thank Tongtong Liu, Luhong Sun for their assistance at the stage of data collection. We are grateful to all the participants for their time and effort. Responsibility for any remaining errors lies solely with the authors. This study was supported by China Medical Board Open Competition Grant (Grant number CMB14-195). An early version of this study has been presented at the 2016 International Society for Pharmacoeconomics and Outcomes Research (ISPOR) 7th Asia Pacific Conference and the abstract has been published online (
Author contributions
All authors contributed to data analysis, drafting and revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Ferlay J, Soerjomataram I, Dikshit R, et al. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. 2015;136(5):E359–386. doi:10.1002/ijc.29210
2.
3. Song B, Ding C, Chen W, Sun H, Zhang M, Chen W. Incidence and mortality of cervical cancer in China, 2013. Chin J Cancer Res. 2017;29(6):471–476. doi:10.21147/j.issn.1000-9604.2017.06.01
4. Chen WQ, Zheng RS, Zhang SW, et al. Cancer incidence and mortality in China, 2013. Cancer Lett. 2017;401:63–71. doi:10.1016/j.canlet.2017.04.024
5. Jing L, Leni K, Youlin Q. Review of the cervical cancer disease burden in mainland China. Asian Pac J Cancer Prev. 2011;12:1149–1153.
6. Shi JF, Canfell K, Lew JB, Qiao YL. The burden of cervical cancer in China: synthesis of the evidence. Int J Cancer. 2012;130(3):641–652. doi:10.1002/ijc.26042
7. Hakama M, Miller AB, Day NE. Screening for cancer of the uterine cervix: from the IARC Working Group on Cervical Cancer Screening and the UICC Project Group on the Evaluation of Screening Programmes for Cancer. Lyon: International Agency for Research on Cancer. 1986.
8. Petry KU. HPV and cervical cancer. Scand J Clin Lab Invest Suppl. 2014;244:59–62. doi:10.3109/00365513.2014.936683
9. Jia Y, Li S, Yang R, et al. Knowledge about cervical cancer and barriers of screening program among women in Wufeng County, a high-incidence region of cervical cancer in China. PLoS One. 2013;8(7):e67005. doi:10.1371/journal.pone.0067005
10. de Bekker-Grob EW, Rose JM, Donkers B, Essink-Bot ML, Bangma CH, Steyerberg EW. Men‘s preferences for prostate cancer screening: a discrete choice experiment. Br J Cancer. 2013;108(3):533–541. doi:10.1038/bjc.2013.5
11. de Bekker-Grob EW, Ryan M, Gerard K. Discrete choice experiments in health economics: a review of the literature. Health Econ. 2012;21(2):145–172. doi:10.1002/hec.1697
12. Basen-Engquist K, Fouladi RT, Cantor SB, et al. Patient assessment of tests to detect cervical cancer. Int J Technol Assess Health Care. 2007;23(2):240–247. doi:10.1017/S0266462307070171
13. Fiebig DG, Haas M, Hossain I, Street DJ, Viney R. Decisions about Pap tests: what influences women and providers? Soc Sci Med. 2009;68(10):1766–1774. doi:10.1016/j.socscimed.2009.03.002
14. Wittenberg E, Bharel M, Saada A, Santiago E, Bridges JF, Weinreb L. Measuring the preferences of homeless women for cervical cancer screening interventions: development of a best-worst scaling survey. Patient. 2015;8(5):455–467. doi:10.1007/s40271-014-0110-z
15. Wordsworth S, Ryan M, Skatun D, Waugh N. Women‘s preferences for cervical cancer screening: a study using a discrete choice experiment. Int J Technol Assess Health Care. 2006;22(3):344–350.
16. Lancsar E, Louviere J. Conducting discrete choice experiments to inform healthcare decision making: a user‘s guide. Pharmacoeconomics. 2008;26(8):661–677. doi:10.2165/00019053-200826080-00004
17. Hemming K. Discrete choice experiments: helping to understand how patients make decisions and promoting non-paternalistic care. Bjog. 2015;122(6):881. doi:10.1111/1471-0528.12965
18. Ryan M, Gerard K, Amaya-Amaya M. Using Discrete Choice Experiments to Value Health and Health Care. Dordrecht: Springer; 2008.
19. Ryan M. Discrete choice experiments in health care. Bmj. 2004;328(7436):360–361. doi:10.1136/bmj.328.7436.360
20. Bridges JFP, Hauber AB, Marshall D, et al. Conjoint analysis applications in health-a checklist: a report of the ISPOR good research practices for conjoint analysis task force. Value Health. 2011;14(4):403–413. doi:10.1016/j.jval.2010.11.013
21. Fort VK, Makin MS, Siegler AJ, Ault K, Rochat R. Barriers to cervical cancer screening in Mulanje, Malawi: a qualitative study. Patient Prefer Adherence. 2011;5:125–131. doi:10.2147/PPA.S17317
22. Moyer VA. Screening for cervical cancer: U.S. preventive services task force recommendation statement. Ann of Intern Med. 2012;156(12):880–891. doi:10.7326/0003-4819-156-12-201206190-00424
23. Watts L, Joseph N, Velazquez A, et al. Understanding barriers to cervical cancer screening among Hispanic women. Am J Obstet Gynecol. 2009;201(2):199e191–198. doi:10.1016/j.ajog.2009.05.014
24. Abdullahi A, Copping J, Kessel A, Luck M, Bonell C. Cervical screening: perceptions and barriers to uptake among Somali women in Camden. Public Health. 2009;123(10):680–685. doi:10.1016/j.puhe.2009.09.011
25. Marshall HS, Chen G, Clarke M, Ratcliffe J. Adolescent, parent and societal preferences and willingness to pay for meningococcal B vaccine: a discrete choice experiment. Vaccine. 2016;34(5):671–677. doi:10.1016/j.vaccine.2015.11.075
26. Liu T, Li S, Ratcliffe J, Chen G. Assessing knowledge and attitudes towards cervical cancer screening among rural women in Eastern China. Int J Environ Res Public Health. 2017;14(9):967. doi:10.3390/ijerph14090967
27. Kruk ME, Johnson JC, Gyakobo M, et al. Rural practice preferences among medical students in Ghana: a discrete choice experiment. Bull World Health Organ. 2010;88(5):333–341. doi:10.2471/BLT.09.072892
28. McFadden D, Train K. Mixed MNL models for discrete response. J Appl Econ. 2000;15(5):447–470. doi:10.1002/(ISSN)1099-1255
29. Hole AR, Kolstad JR. Mixed logit estimation of willingness to pay distributions: a comparison of models in preference and WTP space using data from a health-related choice experiment. Empir Econ. 2012;42(2):445–469. doi:10.1007/s00181-011-0500-1
30. Hole AR. A comparison of approaches to estimating confidence intervals for willingness to pay measures. Health Econ. 2007;16(8):827–840. doi:10.1002/hec.1197
31.
32. Hawkins NA, Benard VB, Greek A, Roland KB, Manninen D, Saraiya M. Patient knowledge and beliefs as barriers to extending cervical cancer screening intervals in federally qualified health centers. Prev Med. 2013;57(5):641–645. doi:10.1016/j.ypmed.2013.08.021
33. Murray M, McMillan C. Social and behavioural predictors of women‘s cancer screening practices in Northern Ireland. J Public Health Med. 1993;15(2):147–153.
34. Doyle Y. A survey of the cervical screening service in a London district, including reasons for non-attendance, ethnic responses and views on the quality of the service. Soc Sci Med. 1991;32(8):953–957.
35. Dam L, Bekker-Grob D. What determines individuals‘ preferences for colorectal cancer screening programmes? A discrete choice experiment. Eur J Cancer. 2010;46(1):150–159. doi:10.1016/j.ejca.2009.07.014
36. Milte R, Ratcliffe J, Chen G, Lancsar E, Miller M, Crotty M. Cognitive overload? An exploration of the potential impact of cognitive functioning in discrete choice experiments with older people in health care. Value Health. 2014;17(5):655–659. doi:10.1016/j.jval.2014.05.005
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.


