Back to Journals » International Journal of Nanomedicine » Volume 11
Pluronic-based micelle encapsulation potentiates myricetin-induced cytotoxicity in human glioblastoma cells
Authors Tang XJ, Huang KM, Gui H, Wang JJ, Lu JT, Dai LJ , Zhang L, Wang G
Received 4 June 2016
Accepted for publication 7 September 2016
Published 3 October 2016 Volume 2016:11 Pages 4991—5002
DOI https://doi.org/10.2147/IJN.S114302
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Thomas Webster
Xiang-Jun Tang,1,* Kuan-Ming Huang,1,* Hui Gui,1,* Jun-Jie Wang,2 Jun-Ti Lu,1 Long-Jun Dai,1,3 Li Zhang,1 Gang Wang2
1Department of Neurosurgery, TaiHe Hospital, Hubei University of Medicine, Shiyan, 2Department of Pharmaceutics, Shanghai Eighth People’s Hospital, Jiangsu University, Shanghai, People’s Republic of China; 3Department of Surgery, University of British Columbia, Vancouver, BC, Canada
*These authors contributed equally to this work
Abstract: As one of the natural herbal flavonoids, myricetin has attracted much research interest, mainly owing to its remarkable anticancer properties and negligible side effects. It holds great potential to be developed as an ideal anticancer drug through improving its bioavailability. This study was performed to investigate the effects of Pluronic-based micelle encapsulation on myricetin-induced cytotoxicity and the mechanisms underlying its anticancer properties in human glioblastoma cells. Cell viability was assessed using a methylthiazol tetrazolium assay and a real-time cell analyzer. Immunoblotting and quantitative reverse transcriptase polymerase chain reaction techniques were used for determining the expression levels of related molecules in protein and mRNA. The results indicated that myricetin-induced cytotoxicity was highly potentiated by the encapsulation of myricetin. Mitochondrial apoptotic pathway was demonstrated to be involved in myricetin-induced glioblastoma cell death. The epidermal growth factor receptor (EGFR)/PI3K/Akt pathway located in the plasma membrane and cytosol and the RAS-ERK pathway located in mitochondria served as upstream and downstream targets, respectively, in myricetin-induced apoptosis. MiR-21 inhibitors interrupted the expression of EGFR, p-Akt, and K-Ras in the same fashion as myricetin-loaded mixed micelles (MYR-MCs) and miR-21 expression were dose-dependently inhibited by MYR-MCs, indicating the interaction of miR-21 with MYR-MCs. This study provided evidence supportive of further development of MYR-MC formulation for preferentially targeting mitochondria of glioblastoma cells.
Keywords: myricetin, glioblastoma, EGFR, miR-21, mitochondrial apoptosis, mixed micelles, anticancer, drug delivery
Introduction
Gliomas account for the majority (almost 80%) of primary malignant brain tumors.1 Glioblastoma multiforme (GBM) is the most common and aggressive type of glioma, which is also known as grade IV astrocytoma. GBM is characterized by high proliferative activity with very poor survival rates. The mainstay of GBM treatment has been surgical resection to the extent feasible, followed by chemotherapy and radiotherapy.2 Although enormous advances have occurred in treating other solid cancers, the median survival rates for GBM stayed nearly the same over the last half century, averaging around 1 year (15–18 months, from diagnosis to death).3,4 Because GBM infiltrates surrounding tissues and is sometimes located in privileged sites, its complete resection is almost impossible. In addition, the existence of the blood–brain barrier and local hypoxia microenvironment make tumor cells resistant to standard radiation and chemotherapy.5 In order to explore truly effective therapy for this most devastating and difficult-to-treat cancer, it is essential to search for an agent which specifically acts on tumor cells with high bioadaptability to the unique microenvironment. Recently, natural dietary compounds have been increasingly studied for their significant antitumoral properties and negligible side effects.6,7
Myricetin (MYR, 3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-4-chromenone) is an important and common type of herbal flavonoid that abundantly exists in various plant sources.8 A growing number of studies have demonstrated the antitumoral effects of MYR against different types of cancer by modifying varied cancer hallmarks, including aberrant cell proliferation, signaling pathways, apoptosis, angiogenesis, and tumor metastasis.9–11 MYR has proven to be a potent inhibitor of PI3K inhibiting Akt and activating apoptotic signaling pathways in several cancer cell lines, including glioblastoma cells.12–16 Despite its relative polarity, MYR is slightly soluble in water and its aqueous solubility is only 16.6 μg/mL.17 Therefore, MYR is hardly absorbed from the intestine. Its absolute bioavailability is <10% in rats.18 It is essential to develop an ideal form of MYR so as to fully explore its antitumoral properties. Our recent studies have suggested that the bioavailability of MYR is significantly enhanced by Pluronic-based micelle encapsulation, and its prioritized distribution in brain tumors was demonstrated with in vivo models.15,16 The present study aimed to investigate the molecular mechanisms underlying anticancer effects of MYR in human glioblastoma cells. Considering the similarity of miR-21 with MYR in respect to signal transduction pathway, the interactions of these two molecules were also studied.
Materials and methods
Preparation of MYR-MCs
Myricetin-loaded mixed micelles (MYR-MCs) were prepared as previously described.16 Pluronic-based nano-carriers were prepared by a thin-film hydration method. Briefly, 200 mg of Pluronic P123 and 100 mg of Pluronic F68 (BASF Ltd, Shanghai, People’s Republic of China) were co-dissolved in 5.0 mL acetonitrile, and MYR (National Institute for the Control of Pharmaceutical and Biological Products, NICPBP, Beijing, People’s Republic of China) was added to the copolymers in a round-bottom flask. The solvent was subsequently stirred at 800 rpm for 60 min to obtain a thin film and kept in a vacuum at room temperature to remove the residual acetonitrile. Then, the thin film was diluted with de-ionized water and added into a Pluronic F68 solution at 800 rpm for 30 min. The resulting micelle solution was then filtered through a filter membrane to remove the non-incorporated drugs thereby generating Pluronic P123/F68 mixed micelles. As the nanoparticles’ aqueous dispersibility can be enhanced by chitosan, chitosan was incorporated into Pluronic P123/F68 mixed micelles to facilitate their cellular uptake.15 Chitosan-functionalized Pluronic P123/F68 nano-carriers were produced by adding de-ionized water and soluble chitosan into Pluronic P123/F68 mixed micelles. Briefly, chitosan (chitooligosaccharide, molecular weight =450.0 kDa, 95% deacetylated degree; Chemical Reagent, Shanghai, People’s Republic of China) was dissolved in pure water at 75°C, and Pluronic P123/F68 mixed micelles were added to the chitosan solution. The final preparation of chitosan-functionalized Pluronic P123/F68-based MYR micelles (MYR-MCs) was obtained through dialysis against de-ionized water using a dialysis membrane (molecular weight cutoff 100 kDa) for a period of 24 hours. The drug loading percentage and encapsulation efficiency were determined as previously described.15 Control micelles were prepared by the same method without adding MYR. Before being applied in cell experiments, the prepared MYR-MCs were distributed in Dulbecco’s Modified Eagle’s Medium (DMEM) containing 10% (v/v) fetal bovine serum (FBS) using ultrasound agitation to obtain nanosuspensions. Particle size and morphology of MYR-MCs were measured using a transmission electron microscope (JEM-2100; JEOL, Tokyo, Japan). Free MYR was used as a comparator of MYR-MCs in the present study. Because of its poor solubility, MYR was dissolved in dimethyl sulfoxide (DMSO) before applying to the cell culture. The final concentration of DMSO was limited to 0.01%.
Cell culture
The human glioblastoma cell line (DBTRG) was purchased from American Type Culture Collection (ATCC, Manassas, VA, USA) and cultured in DMEM with 10% FBS, 2 mM L-glutamine, and 1% penicillin–streptomycin solution (all from Life Technologies, Carlsbad, CA, USA) and incubated at 37°C in a humidified, 5% CO2 atmosphere. The DBTRG cells used in this study were limited to passages 5–7 and subculture was performed with 0.25% trypsin-EDTA solution.
Cell viability assay
Cell viability was evaluated using a methylthiazol tetrazolium (MTT) assay as previously described.19 DBTRG-05MG glioblastoma cells were seeded into a 96-well microplate at a density of 1×105 cells/well. DBTRG cells were respectively exposed to free MYR, MYR-MCs, or control micelles containing 0.01% DMSO and incubated for various periods before cytotoxicity determination. The kinetics of cell killing in DBTRG cells was detected at 12, 24, and 48 h after treatment with 50, 100, and 200 μM of free MYR or MYR-MCs. The MYR-induced cytotoxicity in DBTRG cells was assessed with MTT (Abcam, Cambridge, MA, USA). The optical density (OD) at the wavelength of 570 nm was used to calculate cell viability. The value of OD570 is proportional to the number of viable cells in each well. In this study, the cell viability was presented by the cell death rate, which was defined as: Cell death (%) = (1 − Sample A570/Control A570) ×100%.
In order to follow up MYR-induced cell viability changes continuously, an impedance-based real-time cell analyzer (RTCA) system (xCELLligence; Roche, Indianapolis, IN, USA) was also used for label-free and real-time monitoring of cell viability in DBTRG cells. The RTCA system uses specially designed microtiter plates containing interdigitated gold microelectrodes to noninvasively monitor the viability of cultured cells using electrical impedance as the readout.20,21 The interaction of cells with electrodes impedes the current and generates the impedance signal, which is displayed as an arbitrary unit referred to as cell index (CI). CI value is defined as relative change in measured impedance to background impedance and represents cell status, and is directly proportional to the quantity, size, and attachment forces of the cells. In the present study, DBTRG cells cultured in DMEM medium at 70%–80% confluency were trypsinized and counted and then were resuspended in culture medium. Background measurements were taken from the wells by adding 100 μL of the same medium to the E-Plate 16. A volume of 100 μL of cell suspension (1×105 cells) was then added to the wells to make a final volume of 200 mL. All cells were allowed to settle at the bottom of the wells at room temperature for 15 min and were then incubated at 37°C and 5% CO2. The impedance signals were recorded every 5 min for the first 25 sweeps (2 h) and every 10 min until the end of the experiment (up to 96 h). After 24 h as described above, 50 μL of medium was removed from each well and replaced with the same volume of medium containing an appropriate amount of MYR or MYR-MCs to make 100 and 200 μM final concentrations of these two forms of MYR. The impedance signals were continuously recorded using the same time interval until the end of the experiment (up to 96 h).
Transfection of miRNA mimics and inhibitors
The synthetic miR-21 mimics and miR-21 inhibitors were purchased from RiboBio (Guangzhou, People’s Republic of China). All mimics and inhibitors contain 2′-O-methyl modification at each base. Stealth™ RNAi (Life Technologies) was used as a negative control. Lipofectamine™ RNAiMAX reagent (Life Technologies) was used for the transfection of DBTRG cells. Briefly, DBTRG cells (0.5−1×106 cells/well in 6-well plates with 2 mL volume) were cultured in DMEM supplemented with 10% FBS for 24 h. MiR-21 mimics and inhibitors or negative controls (25 pmol) in 150 μL Opti-MEM medium (Life Technologies) were mixed with the same volume of Opti-MEM medium containing 9 μL of Lipofectamine RNAiMAX reagent and incubated at room temperature for 5 min to allow the formation of transfection complexes. The transfection complexes were added to the appropriate cell cultures by gently swirling the plates. The culture medium was replaced with fresh medium after 5–6 h and the cells were continuously incubated for 48 h.
Quantitative reverse transcriptase polymerase chain reaction (qRT-PCR) for miRNAs and mRNAs
Total RNA was extracted from DBTRG cells using TRIzol (Life Technologies). The miR-21 level was detected by qRT-PCR using Mir-X microRNAs (miRNA) first strand synthesis and SYBR® qRT-PCR kits (Clontech Laboratories, Mountain View, CA, USA) with siRNA negative controls as an internal control. The expression of PI3K mRNA in DBTRG cells was evaluated by qRT-PCR using a PrimeScript™ 1st strand cDNA synthesis kit (Takara Bio, Otsu, Japan) with GAPDH as an internal control. The PCR primers for miR-21 were 5′-CGGCGGTAGCTTATCAGACTGA-3′ (forward) and 5′-CTGGTGTCGTGGAGTCGGCAATTC-3′ (reverse), and the primers for PI3K were 5′-CTCCACGACCATCATCAG-3′ (forward) and 5′-TTCTTCACGGTTGCCTAC-3′ (reverse). All PCR reactions were performed in triplicate using a Step One Plus thermocycler (Applied Biosystems, Foster City, CA, USA). Fold-changes were used to present the relative quantitation of miR-21 and PI3K expression.
Immunoblotting analysis
DBTRG cells were harvested in lysis solution after treatment with 100–200 μM free MYR or MYR-MCs for 24 h. The total protein was isolated using radio-immunoprecipitation assay buffer containing protease inhibitors (Sigma-Aldrich, St Louis, MO, USA). Protein extracts (50 μg) were separated through 12% denaturing sodium dodecyl sulfate (SDS)-polyacrylamide gel electrophoresis and electrotransferred onto a nitrocellulose membrane. The membrane was incubated overnight with primary antibodies for KRAS and Raf1 (Abcam), epidermal growth factor receptor (EGFR), Akt, phosphorylated Akt, ERK, and phospho-ERK (Santa Cruz Biotechnology, Santa Cruz, CA, USA). Anti-β-actin antibody (Santa Cruz Biotechnology) was used as a loading control. The membranes were then incubated for 1 h with the appropriate horseradish peroxidase-conjugated secondary antibodies (Santa Cruz Biotechnology). The protein signal in the membrane was detected using an enhanced chemiluminescence (ECL) detection system (Pierce Biotechnology, Rockford, IL, USA).
To identify MYR-induced changes of intrinsic apoptotic pathway in DBTRG cells, the mitochondrial lysates were used for the related immunoblotting analysis. The mitochondrial lysates were obtained using a mitochondrial isolation kit as per the manufacturer’s instruction. Briefly, the pelleted cells were permeabilized for 1 min in isotonic buffer on ice and then centrifuged at 15,000 rpm for 10 min. The pellet (mitochondrial fraction) and the supernatant (cytosolic fraction) were collected, and the pellet was further lysed to produce the final mitochondrial lysate for Western blotting. For protein analysis, the final mitochondrial lysates were sonicated for 30 seconds on ice and then lysed at 4°C for 60 min. The mitochondrial lysates were centrifuged at 12,000× g for 30 min at 4°C. The protein concentration of the mitochondrial lysate was determined using a BCA protein assay kit (Thermo Fisher Scientific Inc, Waltham, MA, USA). Total protein was separated by denaturing 12% SDS-polyacrylamide gel electrophoresis and the resolved bands were electro-transferred by a semi-dry blotting electroblotter (Bio-Rad Laboratories, Shanghai, People’s Republic of China) onto a nitrocellulose membrane. The membrane was incubated with the following antibodies at indicated dilutions: Anti-p53, phosphorylated p53, and MDM2 antibodies (1:1,000; Cell Signaling, Danvers, MA, USA); anti-caspase-3 and caspase-9 antibodies (1:800; Cell Signaling); and anti-Bax and anti-Bcl-2 antibodies (1:1,000; Santa Cruz Biotechnology). Anti-β-actin antibody (1:1,000) was used as a sample loading control. Protein signals were detected using an ECL detection system. Western blotting analysis of the corresponding strips’ gray value of the relative protein content was imaged using a Bio-Rad image analysis system with Image-Pro software analysis.
Statistical analysis
Data are presented as mean ± standard error of mean of three different experiments. Statistical analysis was assessed by one-way or repeated measures analysis of variance, with least significant difference or Dunnett’s T3 selected for post hoc evaluation (SPSS, Corp, Shanghai, People’s Republic of China). P<0.05 was used as the cutoff for statistically significant differences. The SPSS statistical software package (version 13.0) was used for single- or multi-factorial analyses and the charts were created using GraphPad Prism V6 for Windows.
Results
Characterization of MYR-MCs
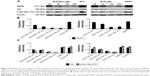
The size distribution of MYR-MCs was 20–300 nm (Figure 1A), which was observed using a laser particle analyzer, and the average diameter of the nanoparticles was found to be 101.46±27.32 nm. As examined with transmission electron microscopy, MYR-MCs were spherical particles with small diameters and a narrow size distribution (Figure 1B). The MYR-MCs exhibited 5.63% of drug loading percentage and 91.7% of encapsulation efficiency.
The effects of MYR on cell viability in human glioma cells
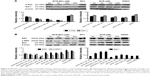
The cell viability was assessed by both RTCA and MTT assay. As shown in Figure 2, all three tested human glioma cell lines were very sensitive to both MYR and MYR-MCs. Under the same drug concentration, MYR-MCs exhibited more potency than MYR in terms of CI throughout the experimental period. However, the MYR-induced cell viability reduction appeared sizable at around 6 h after the drug treatment on DBTRG cells. This latent period was not observed on either U251 or U87 cells. MTT assay was used to examine the dose-responsive MYR-induced cytotoxicity in DBTRG cells. As shown in Figure 3, both MYR and MYR-MCs exhibited dose-dependent cytotoxic responses at all tested time points, 12, 24, and 48 h, compared to controls. Nevertheless, when comparing the responses of these two forms of the drug at the same dose and same time point, MYR-MCs showed significantly more potent cytotoxic effect than MYR (P<0.05). These results further confirmed the beneficial effect of micelle encapsulation on MYR.
MYR-MCs induce changes of upstream pathway of apoptosis in cell lysates
The EGFR is a member of receptor tyrosine kinases. The binding of ligands (EGF and TGF-α) induces a conformational change and subsequently leads to the activation of its intracellular tyrosine kinase domain. Figure 4 shows the MYR-MCs-induced changes of EGFR, Akt, and phosphorylated Akt expression. The expression of EGFR was restrained by both MYR-MCs and MYR in a dose-responsive manner, but DBTRG cells appeared more sensitive to MYR-MCs than MYR as demonstrated by the earlier response to the low dose of MYR-MCs (Figure 4). The expression levels of Akt and p-Akt (Ser 473) were significantly inhibited by MYR-MCs in DBTRG cells. Approximately 54% and 83% inhibition was observed at the concentration of 50 and 100 μM, respectively (Figure 4).
MYR-MCs trigger changes of downstream pathway of apoptosis in mitochondrial lysate
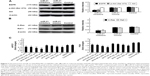
MYR-MC-induced changes of downstream pathway of apoptosis were examined in mitochondrial lysates of DBTRG cells. As shown in Figure 5A, both MYR and MYR-MCs did not induce considerable changes of p53 but significantly inhibited the expression of phosphorylated p53 in tested concentrations (100 and 200 μM) while the MDM2 level remained unchanged. The treatment of DBTRG cells with MYR and MYR-MCs influenced the expression of mitochondrial Bcl-2 and Bax differently; down-regulated for Bcl-2 and up-regulated for Bax (Figure 5B). Following exposure to MYR-MCs (100 and 200 μM), the cytochrome c protein level was decreased while the expression of caspase-9 was increased (Figure 5C). The cleaved caspase-3 level was significantly enhanced by MYR and MYR-MCs at tested concentrations (100 and 200 μM) although the total caspase-3 level remained relatively stable (Figure 5C). The results in Figure 6 showed the protein expression changes of K-Ras, Raf-1, ERK, p-ERK, and Bad with both MYR and MYR-MCs in DBTRG cells. A typical dose-dependent inhibition of K-Ras, Raf-1, and ERKs (ERK and p-ERK) was observed in MYR-MC-treated cells rather than MYR-treated cells. However, the expression of Bad was significantly up-regulated by MYR-MCs also in a dose-responsive manner.
The interaction of MYR-MCs and miR-21 in DBTRG cells
As an important oncogene, miR-21 is up-regulated in a wide range of cancers. The down-regulation of miR-21 in glioblastoma cells causes repression of growth, increased apoptosis, and cell cycle arrest.22 In order to verify the interaction of MYR-MCs with miR-21 in DBTRG cells, the inhibitors and mimics of miR-21 were utilized to reveal the changes of related signals. The direct interaction of MYR-MCs and miR-21 was demonstrated through assessing the expression level of miR-21 following the treatment with MYR-MCs. The cell lysate results of immunoblot analysis indicated that the expression of EGFR and phosphorylated Akt was significantly inhibited by miR-21 inhibitors, while Akt remained unchanged (Figure 7A). The expression level of K-Ras and Raf-1 in the mitochondrial lysates was also inhibited by miR-21 inhibitors (Figure 7B). There was no significant effect of miR-mimics on all tested signals in these two portions of lysates. Figure 7C shows the direct interaction of MYR-MCs and miR-21 using qRT-PCR. The expression level of miR-21 was repressed by the treatment of MYR-MCs in a dose-dependent manner, which was different from the treatment of MYR. A similar pattern of MYR-MC-induced inhibition of PI3K mRNA was observed using qRT-PCR (Figure 7C).
Discussion
As an important and common type of herbal flavonoid, MYR acts as a therapeutic bioactive compound against several human diseases, such as inflammation, cardiovascular disorders, and cancer. The beneficial effects of MYR have been acknowledged by dietary guidelines published in the past two decades.11 A growing number of studies have evidenced its antitumoral properties against various types of cancer through influencing cell proliferation, signal transduction, apoptosis, angiogenesis, and tumor metastasis with negligible side effects. Owing to its low polarity, MYR is sparingly soluble in water and therefore hardly absorbed from the intestine, thereby limiting its application for the treatment of brain tumor. In the last few years, several approaches have been investigated to enhance its bioavailability, including nanosuspension and use of cyclodextrin inclusion compounds.23,24 Most recently, we investigated the biomaterial properties of MYR encapsulated with F68/SDS- and Labrasol-modified lipid-based micelles. The improved cellular uptake of modified MYR was verified by in vitro studies and preferential distribution for the brain was demonstrated by in vivo studies.15,16 The main focus of the present study was to demonstrate the effects of Pluronic-based micelle encapsulation on MYR-induced cytotoxicity and explore the related mechanisms in human glioblastoma cells. MYR encapsulated with Pluronic P123/F68 micelles (MYR-MCs) were sized in a nanoscale range (101.46±27.32 nm, Figure 1) with a very high encapsulation efficiency (91.7%). The results of cell viability assessment by RTCA demonstrated the cytotoxic effects of MYR on all tested human glioma cells (Figure 2). Dose-dependent MYR-induced cytotoxicity was demonstrated by MTT assay in DBTRG cells (Figure 3). Remarkable potentiation of MYR-induced cytotoxicity by encapsulation was clearly revealed through comparing MYR-MC- and MYR-induced cytotoxic effects.
EGFR pathway is one of the most dysregulated molecular pathways in human cancers.25 EGFR is best known for its classical function as a receptor tyrosine kinase localized on the plasma membrane and activated upon ligand binding. Activated EGFR stimulates a number of downstream signaling molecules, leading to the activation of various major pathways which are essential to tumor growth, progression, prevention of apoptosis, drug resistance, and metastasis.26–28 According to their functions, the main downstream pathway molecules of activated EGFR can be activated through kinase-dependent reaction, such as PI3K, K-Ras, and Raf-1, or through its ability to physically interact with other proteins (kinase-independent), such as Akt and MAPK.25 As top-line molecules of EGFR/PI3K/Akt/mTOR pathway, the expressions of EGFR, PI3K, and Akt detected in the cell lysates were repressed by the treatment of MYR (Figures 4 and 7). MYR-MC showed more potent inhibition than MYR at all tested concentrations, which is believed to be attributed to the difference of cellular uptake for these two forms of MYR.15
The downstream molecules of EGFR pathway play an important role for the control of mitochondrial apoptotic pathway, which is regulated by caspase-9 and the balance between proapoptotic and antiapoptotic Bcl-2 protein family members.29–31 As discussed above, MYR-induced EGFR deprivation in DBTRG cells initiates the mitochondrial apoptotic pathway. The activation of this pathway leads to increased mitochondrial permeability, thereby promoting the release of proapoptotic proteins (eg, cytochrome c) from the intermitochondrial membrane space into the cytosol,32 ultimately cleaving numerous proteins and activating DNases. Immunoblotting assessments of apoptosis-related molecules in the mitochondrial lysates (Figures 5 and 6) were in favor of the hypothesis that MYR caused cell death through mitochondria-mediated apoptotic pathway in DBTRG cells. In a previous study, we demonstrated that p53 acts as an anti-oncogene and mediates apoptosis in C6 glioma cells through a mitochondrial signaling pathway.33 Following the treatment with MYR in the present study, slightly increased p53 levels were detected in DBTRG cells while MDM2 remained relatively stable. However, phosphorylated p53 levels were significantly reduced in a dose-dependent manner. The reduction of phosphorylated p53 might be involved in MYR-induced cytotoxicity, as phosphorylated p53 inhibits the interaction of p53 with MDM2, a negative regulator of p53, thereby preventing degradation of p53 protein.34 Deacetylated p53 binds to the mitochondrial outer membrane and out-competes Bax that binds with Bcl-2 protein. Moreover, the enhanced level of Bax might be generated as a more potent inducer of apoptotic cell death through Bax cleavage.35,36 The subsequent activation of Bax could lead to the release of cytochrome c from the mitochondria and the induction of apoptosis.37 In addition, MYR-induced decrease of Bcl-2 and increase of caspase-9 and 3 also played an important part in the activation of mitochondrial apoptotic pathway. MYR-MCs have been found to regulate p53 and Bad activation, constitutively inducing apoptosis in cancer cells through the PI3K/Akt and RAS-ERK pathways. A phosphorylated Bad-mediated ERK pathway was also identified by Kern et al in their recent study,38 which plays a key role in mitochondria-mediated apoptosis. In this study, MYR-MC-induced apoptosis through mitochondrial pathway was further supported by the assessments of RAS-ERK pathway-related proteins in the mitochondrial lysates, ie, dose-dependent stimulation of Bad and dose-dependent inhibition of K-Ras, Raf-1, and ERK.
miRNAs are small noncoding RNA molecules that regulate protein expression by targeting the mRNA of protein-coding genes. MiR-21 is a key oncogene which is highly expressed in most cancers.39,40 It is markedly up-regulated in human glioblastoma tissues and several established glioblastoma cell lines.41 Critical targets of miR-21 are mRNAs of tumor suppressor proteins, checkpoint regulators of cell cycle control, and intrinsic and extrinsic pathways of cellular apoptosis.42 Knockdown of miR-21 in cultured glioblastoma cells triggers activation of caspases and leads to increased apoptotic cell death.41 MiR-21 is a negative regulator of p53 signaling and stimulates the expression of the cell cycle promoter cyclin D1.22,43 It also induces tumor angiogenesis through targeting PTEN, leading to activated AKT and ERK1/2 signaling.44 In the present study, DBTRG cells treated with miR-21 inhibitors showed significant reduction of EGFR, phosphorylated Akt (Ser 473), K-Ras, and Raf-1 (Figure 7A and B). The similarity between miR-21 inhibitors and MYR-MCs in terms of related protein expression led us to speculate whether miR-21 could be the target of MYR-MCs. MiR-21 expression was significantly inhibited by MYR-MCs in a dose-dependent fashion (Figure 7C), which could be direct evidence showing the interaction between MYR-MCs and the expression of miR-21. However, further study is required to identify related molecules and verify the mechanisms underlying anticancer effects of MYR.
Conclusion
In conclusion, the encapsulation of MYR with nanosized Pluronic P123/F68 mixed micelles potentiated MYR-induced cytotoxicity in DBTRG cells. Mitochondrial apoptotic pathway was demonstrated to be involved in MYR-induced glioblastoma cell death. The EGFR/PI3K/Akt pathway located in the plasma membrane and cytosol and the RAS-ERK pathway located in mitochondria served as upstream and downstream targets, respectively, in MYR-induced apoptosis. The close relationship between miR-21 and MYR-MCs was sustained by the fact that miR-21 inhibitors interrupted the expression of EGFR, p-Akt, and K-Ras in the same fashion as MYR-MCs. Furthermore, the inhibition of miR-21 expression was dose-responsibly correlated with the application of MYR-MCs. Considering its remarkable antitumoral properties with negligible side effects, MYR could be developed as an ideal drug for varied clinical applications especially for the treatment of brain tumor. Targeted delivery of MYR-MCs will likely be achieved through innovations in the areas of nanotechnology and intracellular trafficking. Our study provided evidence for further development of MYR-MC formulation preferentially targeting mitochondria of glioblastoma cells.
Acknowledgments
This work was supported by Taihe Hospital Foundation and Young Talent Project of the Education Department of Hubei Province (Q20162104). We thank Dr Shan Qing Jiang for his technical assistance, Dr Feng-Qian Li for revising this paper, and Dr Jia-Lan Guo for analyzing the results. We also thank James Dai for his assistance in preparing the manuscript and English proofreading.
Disclosure
The authors report no conflicts of interest in this work.
References
Schwartzbaum JA, Fisher JL, Aldape KD, Wrensch M. Epidemiology and molecular pathology of glioma. Nat Clin Pract Neurol. 2006;2(9):494–503. | ||
Urbanska K, Sokolowska J, Szmidt M, Sysa P. Glioblastoma multiforme – an overview. Contemp Oncol. 2014;18(5):307–312. | ||
Fomchenko EI, Holland EC. Mouse models of brain tumors and their applications in preclinical trials. Clin Cancer Res. 2006;12(18):5288–5297. | ||
Stupp R, Mason WP, van den Bent MJ, et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med. 2005;352(10):987–996. | ||
Chang JE, Khuntia D, Robins HI, Mehta MP. Radiotherapy and radiosensitizers in the treatment of glioblastoma multiforme. Clin Adv Hematol Oncol. 2007;5(11):894–902. | ||
Fulda S. Modulation of apoptosis by natural products for cancer therapy. Planta Med. 2010;76(11):1075–1079. | ||
Demain AL, Vaishnav P. Natural products for cancer chemotherapy. Microb Biotechnol. 2011;4(6):687–699. | ||
Harnly JM, Doherty RF, Beecher GR, et al. Flavonoid content of U.S. fruits, vegetables, and nuts. J Agric Food Chem. 2006;54(26):9966–9977. | ||
Weng CJ, Yen GC. Flavonoids, a ubiquitous dietary phenolic subclass, exert extensive in vitro anti-invasive and in vivo anti-metastatic activities. Cancer Metastasis Rev. 2012;31(1–2):323–351. | ||
Xu Y, Xie Q, Wu S, et al. Myricetin induces apoptosis via endoplasmic reticulum stress and DNA double-strand breaks in human ovarian cancer cells. Mol Med Rep. 2016;13(3):2094–2100. | ||
Devi KP, Rajavel T, Habtemariam S, Nabavi SF, Nabavi SM. Molecular mechanisms underlying anticancer effects of myricetin. Life Sci. 2015;142:19–25. | ||
Agullo G, Gamet-Payrastre L, Manenti S, et al. Relationship between flavonoid structure and inhibition of phosphatidylinositol 3-kinase: a comparison with tyrosine kinase and protein kinase C inhibition. Biochem Pharmacol. 1997;53(11):1649–1657. | ||
Williams RJ, Spencer JP, Rice-Evans C. Flavonoids: antioxidants or signalling molecules? Free Radic Biol Med. 2004;36(7):838–849. | ||
Kandaswami C, Lee LT, Lee PP, et al. The antitumor activities of flavonoids. In Vivo. 2005;19(5):895–909. | ||
Wang G, Wang JJ, Tang XJ, Du L, Li F. In vitro and in vivo evaluation of functionalized chitosan-Pluronic micelles loaded with myricetin on glioblastoma cancer. Nanomedicin. 2016;12(5):1263–1278. | ||
Wang G, Wang JJ, Li F, To ST. Development and evaluation of a novel drug delivery: pluronics/SDS mixed micelle loaded with myricetin in vitro and in vivo. J Pharm Sci. 2016;105(4):1535–1543. | ||
Yao Y, Lin G, Xie Y, et al. Preformulation studies of myricetin: a natural antioxidant flavonoid. Pharmazie. 2014;69(1):19–26. | ||
Dang Y, Lin G, Xie Y, et al. Quantitative determination of myricetin in rat plasma by ultra performance liquid chromatography tandem mass spectrometry and its absolute bioavailability. Drug Res. 2014;64(10):516–522. | ||
Tang XJ, Lu JT, Tu HJ, et al. TRAIL-engineered bone marrow-derived mesenchymal stem cells: TRAIL expression and cytotoxic effects on C6 glioma cells. Anticancer Res. 2014;34(2):729–734. | ||
Moniri MR, Sun XY, Rayat J, et al. TRAIL-engineered pancreas-derived mesenchymal stem cells: characterization and cytotoxic effects on pancreatic cancer cells. Cancer Gene Ther. 2012;19(9):652–658. | ||
Roshan Moniri M, Young A, Reinheimer K, Rayat J, Dai LJ, Warnock GL. Dynamic assessment of cell viability, proliferation and migration using real time cell analyzer system (RTCA). Cytotechnology. 2015;67(2):379–386. | ||
Papagiannakopoulos T, Shapiro A, Kosik KS. MicroRNA-21 targets a network of key tumor-suppressive pathways in glioblastoma cells. Cancer Res. 2008;68(19):8164–8172. | ||
Hong C, Dang Y, Lin G, et al. Effects of stabilizing agents on the development of myricetin nanosuspension and its characterization: an in vitro and in vivo evaluation. Int J Pharm. 2014;477(1–2):251–260. | ||
Lucas-Abellan C, Fortea I, Gabaldon JA, Nunez-Delicado E. Encapsulation of quercetin and myricetin in cyclodextrins at acidic pH. J Agric Food Chem. 2008;56(1):255–259. | ||
Han W, Lo HW. Landscape of EGFR signaling network in human cancers: biology and therapeutic response in relation to receptor subcellular locations. Cancer Lett. 2012;318(2):124–134. | ||
Yarden Y, Shilo BZ. SnapShot: EGFR signaling pathway. Cell. 2007;131(5):1018. | ||
Lo HW. EGFR-targeted therapy in malignant glioma: novel aspects and mechanisms of drug resistance. Curr Mol Pharmacol. 2010;3(1):37–52. | ||
Davis NM, Sokolosky M, Stadelman K, et al. Deregulation of the EGFR/PI3K/PTEN/Akt/mTORC1 pathway in breast cancer: possibilities for therapeutic intervention. Oncotarget. 2014;5(13):4603–4650. | ||
Youle RJ, Strasser A. The BCL-2 protein family: opposing activities that mediate cell death. Nat Rev Mol Cell Biol. 2008;9(1):47–59. | ||
Adams JM, Cory S. Bcl-2-regulated apoptosis: mechanism and therapeutic potential. Curr Opin Immunol. 2007;19(5):488–496. | ||
Hotchkiss RS, Strasser A, McDunn JE, Swanson PE. Cell death. N Engl J Med. 2009;361(16):1570–1583. | ||
Goldstein JC, Munoz-Pinedo C, Ricci JE, et al. Cytochrome c is released in a single step during apoptosis. Cell Death Differ. 2005;12(5):453–462. | ||
Wang G, Wang JJ, Chen XL, et al. The JAK2/STAT3 and mitochondrial pathways are essential for quercetin nanoliposome-induced C6 glioma cell death. Cell Death Dis. 2013;4:e746. | ||
Ryan KM, Phillips AC, Vousden KH. Regulation and function of the p53 tumor suppressor protein. Curr Opin Cell Biol. 2001;13(3):332–337. | ||
Wood DE, Thomas A, Devi LA, et al. Bax cleavage is mediated by calpain during drug-induced apoptosis. Oncogene. 1998;17(9):1069–1078. | ||
Toyota H, Yanase N, Yoshimoto T, Moriyama M, Sudo T, Mizuguchi J. Calpain-induced Bax-cleavage product is a more potent inducer of apoptotic cell death than wild-type Bax. Cancer Lett. 2003;189(2):221–230. | ||
Park CM, Park MJ, Kwak HJ, et al. Induction of p53-mediated apoptosis and recovery of chemosensitivity through p53 transduction in human glioblastoma cells by cisplatin. Int J Oncol. 2006;28(1):119–125. | ||
Kern HB, Niemeyer BF, Parrish JK, et al. Control of MicroRNA-21 expression in colorectal cancer cells by oncogenic epidermal growth factor/Ras signaling and Ets transcription factors. DNA Cell Biol. 2012;31(8):1403–1411. | ||
Medina PP, Nolde M, Slack FJ. OncomiR addiction in an in vivo model of microRNA-21-induced pre-B-cell lymphoma. Nature. 2010;467(7311):86–90. | ||
Krichevsky AM, Gabriely G. miR-21: a small multi-faceted RNA. J Cell Mol Med. 2009;13(1):39–53. | ||
Chan JA, Krichevsky AM, Kosik KS. MicroRNA-21 is an antiapoptotic factor in human glioblastoma cells. Cancer Res. 2005;65(14):6029–6033. | ||
Buscaglia LE, Li Y. Apoptosis and the target genes of microRNA-21. Chin J Cancer. 2011;30(6):371–380. | ||
Ng R, Song G, Roll GR, Frandsen NM, Willenbring H. A microRNA-21 surge facilitates rapid cyclin D1 translation and cell cycle progression in mouse liver regeneration. J Clin Invest. 2012;122(3):1097–1108. | ||
Liu LZ, Li C, Chen Q, et al. MiR-21 induced angiogenesis through AKT and ERK activation and HIF-1alpha expression. PLoS One. 2011;6(4):e19139. |
 © 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.