Back to Journals » Clinical Ophthalmology » Volume 14
Physiochemical Properties and Cytotoxicity of a Benzalkonium Chloride-Free, Micellar Emulsion Ophthalmic Formulation of Latanoprost
Authors Halder A, Khopade AJ
Received 25 June 2020
Accepted for publication 11 September 2020
Published 7 October 2020 Volume 2020:14 Pages 3057—3064
DOI https://doi.org/10.2147/OPTH.S266453
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Arindam Halder, Ajay J Khopade
Formulation Department, Sun Pharmaceutical Industries, Ltd, Tandalja, Vadodara, India
Correspondence: Ajay J Khopade
Sun Pharmaceutical Industries, Ltd., Tandalja, Vadodara 390020, Gujarat, India
Tel +91 22 6645 5645 Ext 1900
Email [email protected]
Purpose: Prostaglandin derivatives are used widely to reduce intraocular pressure associated with open-angle glaucoma. The most widely used prostaglandin derivative, latanoprost, is available in an ophthalmic solution that is solubilized and preserved with 0.02% benzalkonium chloride (BAK), which has been shown to be cytotoxic to corneal cells. Latanoprost ophthalmic solution with BAK requires specific storage temperatures, which can impact the supply cycle. Here, we describe the production, physicochemical characteristics, and cytotoxicity profile of a micelle formulation that solubilizes latanoprost without the need for BAK.
Methods: The optimum concentration of castor oil with the surfactant polyethylene glycol (15) hydroxystearate was determined, and the mixture stirred. Various surfactants were tested to determine the ideal mixture to form a micelle formulation. Viscosity, zeta potential, surface tension, droplet size, and osmolality of the batches were tested. The cytotoxicity of the micelle formulation was determined in a corneal cell viability assay that compared positive and negative controls, latanoprost without BAK, latanoprost with BAK, and placebo.
Results: A castor oil concentration of 0.15% produced a micelle formulation with a diameter of < 100 nm. This micelle formulation had unique characteristics that were not mimicked when either the surfactant or the oil was changed. The physicochemical characteristics in multiple batches of the micelle formulation did not vary significantly between batches. Long-term and accelerated stability studies showed latanoprost potency remained constant for 24 months at 25°C/75% relative humidity (RH) and at 40°C/25% RH for 6 months.
Conclusion: The micelle formulation technology system is capable of solubilizing latanoprost in an ophthalmic formulation without the need for BAK. The system is stable at room temperature.
Keywords: open angle glaucoma, micelle formulation, benzalkonium chloride, latanoprost
Introduction
Glaucoma is a leading cause of irreversible blindness in people aged 50 years and older.1,2 The primary risk factor for open-angle glaucoma (OAG) is increased intraocular pressure (IOP).2 Prostaglandin derivatives are the most effective class of medications for increasing the outflow of aqueous humor, thereby lowering IOP.3
Latanoprost 0.005% ophthalmic solution (Xalatan®, Pfizer & Upjohn Co, Puurs, Belgium), an F2α analogue, is indicated for the reduction of IOP in patients with OAG or ocular hypertension.4 It contains 0.02% benzalkonium chloride (BAK) as a preservative; BAK is also used to solubilize the latanoprost in the solution.4 Prostaglandin derivatives are poorly soluble in an aqueous environment due to their hydrophobic structures.2 BAK is known to be cytotoxic at concentrations as low as 0.00002%.5–7 Furthermore, OAG treatment requires chronic drug administration to maintain lowered IOP, and studies have demonstrated that chronic use of solutions with BAK can cause damage to the cornea.5,8
The poor water solubility seen with prostaglandin derivatives may lower the concentration of latanoprost delivered to the ocular tissue.9 Also, latanoprost is absorbed into the cornea and hydrolyzed into its active form, latanoprost acid;4 BAK then resorbs the latanoprost acid from the cornea into the tears, which further decreases the concentration of latanoprost delivered to the ocular tissues.
Latanoprost 0.005% ophthalmic solution must be stored under refrigeration at 2°C to 8°C (36°F–46°F).4 During shipment to the patient, the bottle may be maintained at temperatures up to 40°C (104°F) for up to 8 days. Once a bottle is open, it may be stored at room temperature up to 25°C (77°F) for 6 weeks.4 The issues with the use of BAK as a preservative and a solubilizing agent create an unmet need for a prostaglandin derivative that does not use BAK and has increased stability at room temperature.
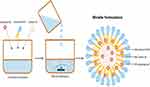
To help address this unmet need, we have synthesized a micelle formulation that stably dissolves the hydrophobic latanoprost and prevents a decrease in the administered dose of the drug. A microemulsion is a system of water, oil, and surfactants that is a thermodynamically stable liquid solution. However, when the hydrophobic solubilitate is oil and the oil/surfactant pair has hydrophobic domains of similar physicochemical properties, a relatively large quantity of oil can be solubilized. Such swollen micelles containing solubilized oil at the boundary of <100 nm diameter are termed nanomicellar emulsions. These micellar formulations form spontaneously without the addition of external energy, contain large liquid hydrophobic cores, and are surrounded by a surfactant monolayer that stabilizes the dispersion (Figure 1). The hydrophobic cores of the surfactant can solubilize the oil without transforming it to an emulsion system above its critical micelle concentration.
Here, we report the characteristics of a micellar formulation technology system with a nonionic surfactant whose hydrophobic properties can solubilize hydrophobic molecules. This system forms a micellar formulation without using BAK as a preservative. We also report on the cytotoxicity of the micellar formulation vs latanoprost 0.005% ophthalmic solution in Statens Serum Institut rabbit corneal cells.
Materials and Methods
Preparation of Micelle Formulation
Latanoprost (Chirogate International Inc., Taoyuan City, Taiwan) and castor oil 0.15% (J.T. Baker, USA) were continuously stirred with a glass rod in a beaker until the latanoprost was fully solubilized. Meanwhile, polyethylene glycol (15) hydroxystearate (hereafter referred to as Solutol HS 15), the nonionic surfactant (BASF, Ludwigshafen, Germany), was heated in a separated glass beaker at 65°−70°C until melted. After melting, the Solutol HS 15 was added to the latanoprost and castor oil phase and stirred with a dry glass rod at 65°−70°C until completely mixed, at which time it cooled to 60°C while undergoing gentle stirring. This solution was added dropwise with continuous stirring to water for injection heated to 55°−70°C. After the formation of the transparent microemulsion, propylene glycol (Dow Chemicals, Midland, MI, USA) was added while the temperature was maintained at 55°−60°C. After the addition of the propylene glycol, the solution was gently stirred while cooling to 25°−30°C; once this temperature range was achieved, gentle stirring continued for another 30−45 minutes. After this time, boric acid 0.30% (Merck Group, Darmstadt, Germany), sodium borate/Borax 0.11% (Merck Group), and potassium sorbate 0.47% (Merck Group) were dissolved in the solution. The pH was adjusted to 7.00 with 10% w/v sodium hydroxide solution/hydrochloric acid (Mallinckrodt, Staines-upon-Thames, UK). The solution was filtered through a 0.2µ polyethersulfone filter.
Nanomicellar Emulsion Characteristics
The optimum concentration of castor oil that can be solubilized in 0.25% Solutol HS 15 was determined. A range of castor oil concentrations (0.05%, 0.1%, 0.15%, 0.20%, 0.25%, and 0.30%) were tested with 0.25% Solutol HS 15, and the optical transmission was tested at 650 nm using a UV-1800 spectrophotometer (Shimadzu, Kyoto, Japan). The optical transmission was a surrogate marker for the critical micelle concentration and also an indication of solution clarity. The interbatch characteristics for the 6 solutions mixed in 1-liter and 20-liter quantities were also tested. Viscosity (an indicator of latanoprost retention), zeta potential (an indicator of the charge on the surface of the particle and a surrogate for potential corneal irritation), surface tension (which determines the potential for dripping and leaking), and osmolality of the batches were also tested. Viscosity testing was performed by a capillary-based method using Ubbelohde-type capillary viscometer (Lauda Viscometer, Delta Labo, France) at 25°C ± 0.1°C with capillary size Oa, zeta potential was measured by nano ZS/nano S Malvern Zeta Sizer (Malvern Panalytical Ltd., Malvern, UK), and surface tension was measured by k12 tensiometer (KRÜSS Scientific, Hamburg, Germany). The dynamic viscosity was calculated using the equation:
Dynamic viscosity = kinematic viscosity x density of sample (gm/mL)
Dynamic light scattering was used to determine the particle size using a nano ZS/nano S Malvern Zeta Sizer (Malvern Panalytical Ltd., Malvern, UK). For each batch, the individual containers were shaken and the contents of the 5 bottles were pooled into a clean, particle-free container using a cuvette to eliminate air bubble formation. The particle refractive index was set at 1.60 at a temperature of 25°C and an equilibrium time of 120 seconds and the particle size was measured. Droplet size was examined using varying surfactant-oil pairs. The process above was repeated with polysorbate 80 in place of Solutol HS15 and/or arachis oil or medium-chain triglyceride used in place of the castor oil.
The potency (% of label claim) of the nanomicelle emulsion at 2 different temperatures and relative humidities were also examined using high-performance liquid chromatography with a C18 column. The test sample was injected with diluted isopropyl alcohol for solubilization. The latanoprost working sample was also injected into the system by diluting with isopropyl alcohol. The formulation-filled bottles were stored in a stability chamber with controlled temperature and humidity (25°C and 40% relative humidity [RH] vs 40°C and 25% RH). The bottles were withdrawn from the chamber at 1, 3, 6, 9, 12, 18, and 24 months and analyzed for the percent of latanoprost retained. The percent of latanoprost (% of label claim) was calculated by comparing the average area of the latanoprost peak response in test preparation vs in standard preparation.
An in-vitro release study was performed on 6 batches of formulation. A 200 mL bottle of simulated tear fluid with 20% alcohol was placed on a rotating bottle apparatus. The bottle rotated at 50 rotations per minute at a temperature of 37° C ± 0.5. Measurements were taken at 0.5 hours, 1, 2, 4, 6, 8, 10, and 12 hours and analyzed for dilution.
In vitro Cytotoxicity Study
Statens Seruminstitut rabbit cornea cells (Cell Cultivation Facility, SPARC, India) were seeded at 30,000 cells/well on a 96-well plate on day 1. After 24 hours, the cells were incubated with 50 ug/mL of neutral red dye for 3 hours. The cells were washed and incubated with positive and negative controls, latanoprost ophthalmic solution and latanoprost BAK-free, or placebo (latanoprost BAK-free composition without drug), for 30 minutes, 24 hours, or 72 hours at 37°C + 5% carbon dioxide. Cell viability was measured by fluorometric detection using a colorimeter using an excitation wavelength of 535 nm and an emission wavelength of 600 nm. Cultures around each sample were examined under a microscope and using a (3-(4,5-dimethylthiazol-2yl)-2,5-diphenyl tetrazolium bromide) as a stain. Reactivity was graded on a 0−4 scale, with 0 indicating no detectable cytotoxic zone around or under the specimen and 4 including the cytotoxic zone extended >1.0 cm.
Results
Nanomicellar Emulsion Characteristics
The optical transmission decreased as the concentration of castor oil increased. The solution without castor oil had an optical transmission of 100% vs 0% for the solution with 0.30% castor oil concentration. The zone of clarity, indicating the ideal solution clarity for the nanomicellar emulsion, was determined to be 80% to 100% optical transmission. The nanomicellar emulsion was defined when the castor oil concentration was 0.125% to 0.175%. The zone of clarity and the formation of the nanomicellar emulsion intersected when the observed concentration of castor oil was 0.15%. Increasing the lipid concentration decreased the clarity of the solution, while the particle size increased slightly (Figure 2). The use of increasing concentrations of lipids identified 0.8% as the lowest observed total lipid concentration that would maximize optical clarity and minimize particle size (Figure 2).
Replacing Solutol HS 15 with polysorbate 80 resulted in a self-emulsifying system with larger droplets and lower transmission than in the Solutol-castor oil system. Replacing castor oil with arachis oil resulted in a Solutol HS 15-arachis oil emulsion with a droplet size more than double that with castor oil (273 nm vs <100 nm, respectively) and an optical transmission <35% (Table 1). Overall, less than 0.01% of arachis oil dissolved in the Solutol HS 15, which is approximately 10 times less soluble than castor oil.
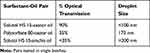 |
Table 1 Characteristics of Surfactant-Oil Pairs |
Interbatch consistency for the 6 batches demonstrated consistent particle sizes. Particle sizes were tested across 10 bottles per batch; the particles in 1-liter batch vs the 20-liter batches were distributed in similar manners across the size ranges (Table 2). Across the 6 batches, the viscosity was 0.993 to 0.997; the zeta potential was −3.14 to −5.94; surface tension was 38.94 to 39.62; and osmolality was 361 to 375 mOsm/kg. Optical transmission ranged from 88.37% to 91.61%, well within the zone of clarity as shown in Figure 2. All parameters showed a high degree of consistency across all batches (Table 3).
 |
Table 2 Interbatch Particle Size Distribution |
 |
Table 3 Physicochemical Characteristics of the Micellar Formulation Across the 6 Batches |
The micelle formulation showed a tight range of latanoprost potency (98.0%–102.4% of label claim) when tested for 24 months at 25°C and 40% RH. When the micelle formulation was kept at 40°C/25% RH for 6 months, the potency of latanoprost in the micellar formulation was maintained within a range of 97.2 to 102.4 nm (Table 4).
 |
Table 4 Average Potency of Latanoprost in Stability Study |
The dilution study showed consistent dilution across all batches and time points with 92%–106% of the nanomicelle formulation dissolved after 12 hours (Table 5).
 |
Table 5 In vitro Dilution Study |
In vitro Cytotoxicity
The micellar formulation showed slight cytotoxicity of 10% to 20% in the neutral red assay vs 30% to 50% for latanoprost 0.005% ophthalmic solution. The USP<87> Biological Reactivity test in the nanomicellar emulsion detected malformed cells around or under the specimen, while latanoprost 0.005% ophthalmic solution treatment showed malformed or degenerated cells (Table 6).
 |
Table 6 Cytotoxicity of Positive and Negative Controls and Micellar Formulation with Latanoprost 0.005% and Latanoprost 0.005% with BAK in Neutral Red Assay |
Discussion
The micellar formulation described here consisting of a nonionic surfactant (Solutol HS 15), castor oil, and water with the prostaglandin derivative latanoprost was stable at room temperature. The physiochemical properties of the micellar formulation of latanoprost enable its solubility within an aqueous environment, without the need for a preservative/solubilizing agent like BAK. This formulation of latanoprost BAK-free uses potassium sorbate as its preservative,4 which is a milder preservative system with lower toxicity to the ocular surface, as demonstrated by absence of cell death upon exposure of primary human corneal epithelial cells in vitro.10
Several studies demonstrate the efficacy of the latanoprost micellar formulation without BAK. A Phase 3 efficacy and safety study of latanoprost 0.005% without BAK showed a statistically significant mean change from baseline in both peak and trough IOP in patients with OAG and ocular hypertension (P <0.0001 for both).11 Patients receiving latanoprost 0.005% without BAK experienced comparable adverse events (AEs) to patients receiving latanoprost 0.005% with BAK; most common AEs were eye irritation (7.6% vs 0%) and eye pruritis (1.9% vs 5.9%).11 Additionally, in a prospective, open-label study, treatment with the micellar formulation of latanoprost without BAK resulted in significant improvements from baseline in mean tear break-up time and inferior corneal staining scores (P <0.0001 and P = 0.0033, respectively).12
When oil-in-water emulsions are close to their phase inversion temperatures, more complicated mechanisms of solubilization are observed; these include the formation of intermediate phases and dynamic Marangoni effects, which are caused by the partial surfactant solubility in the oil phase and the ultra-low interfacial tension. This was noted in the preparation of the micellar formulation when it was determined that the percent of optical transmission (solubility) was affected by the stirring speed and efficient solubilization was noted only at a high stirring speed. The additional shear stress at the interface between a droplet and an ambient phase generates complex convection patterns that temporarily increase the global drag coefficient of the droplet and decrease the droplet size.13 There is a transient stirring speed effect on the interfacial surfactant/triglyceride ratio and concomitant interfacial oil-in-water tension; therefore, a degree of polydispersity is obtained in the solubilized system diameter. The stability of the micellar formulation makes it ideal for the delivery of insoluble therapeutic agents such as latanoprost.14,15 Also, the ability of the micellar formulation to withstand high temperatures for extended time periods will allow the drug’s supply chain to be managed with relative ease.
When the premixed Solutol/castor oil combination was dispersed in water, the interfacial surface tension was reduced to the point that immediate solubilization occurred. It is worth noting that the hydrophobic fatty portion of Solutol is close to the structural analogue of the castor oil.16 Solutol has 12-hydroxystearic acid (70%) as the hydrophobic component and polyethylene glycol (30%) as the hydrophilic component, and the main fatty acid component is 12–hydroxystearic acid (fatty acid with 18 carbon chain).17 Castor oil is a triglyceride in which approximately 90% of fatty acid chains are ricinoleic acid.18 Less than 0.01% of arachis oil could be dissolved in the Solutol, which is approximately 10 to 15 times less than the amount of castor oil that could be dissolved. This is because the hydrophobic portion of the arachis oil differs from the hydrophobic component of the castor oil. It was thought that the glass transition of the oil was equally important. When a medium-chain fatty triglyceride was used, the solution was clear, and more triglyceride could be dissolved. This indicates the polarity of the oil plays more of a role vs other structural characteristics.
The particle sizes (z-average) in the nanomicellar emulsion ranged from 54 to 63 nm, confirming the particle size falls in the nanomicellar range.19 The particle size falls within the approximate ranges of D10 31–36 nm, D50 51–59 nm, and D90 80–99 nm. The span value of the particles falls in the narrow range (close to 1.0), indicating a uniform droplet distribution. The particle size data indicate the manufacturing process is robust and produces highly uniform particle size distribution across batches, scale, and stability stations.
The dilution study indicates the encapsulation of the latanoprost molecules in the micelles helps ensure bioavailability and dilution across all time points that the medication is in contact with the ocular surface.
Cytotoxicity is a key indicator to assess for biological responses to chemical formulations. Chemicals have varying cytotoxicity mechanisms, including destruction of cell membranes, prevention of protein synthesis and irreversible binding to receptors. To determine cellular death caused by these cytotoxic mechanisms, initial screening is done before proceeding to in-vivo toxicity studies. The micellar formulation was tested on rabbit corneal cell lines.
The micellar formulation demonstrated less toxicity than latanoprost 0.005% ophthalmic solution in the rabbit corneal cell cytotoxicity assay. In addition to minimizing damage to the cornea, the micellar formulation may also increase patient compliance due to decreased eye irritation vs formulations containing BAK.
Conclusions
Overall, it is possible to generate a stabilized version of latanoprost without BAK using a simple but robust manufacturing process. This micellar formulation could provide ocular surface benefits to patients while delivering latanoprost at therapeutic levels to the eye.
Abbreviations
AE, adverse events; BAK, benzalkonium chloride; IOP, intraocular pressure; OAG, open-angle glaucoma; RH, relative humidity.
Acknowledgments
Writing and editorial assistance was provided by Jennifer Meyering, RN, MS, CMPP, of AlphaBioCom (King of Prussia, PA) with funding provided by Sun Pharmaceutical Industries, Inc (Princeton, NJ).
Author Contributions
All authors contributed to data analysis, participated in drafting or revising the article, have agreed on the journal to which the article will be submitted, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Funding
This study was sponsored and funded by Sun Pharma Advanced Research Company, Ltd.
Disclosure
AH and AJ are both employees of Sun Pharmaceutical Industries, Ltd. The authors report no other conflicts of interest in this work.
References
1. Flaxman SR, Bourne RRA, Resnikoff S, et al. Global causes of blindness and distance vision impairment 1990–2020: a systematic review and meta-analysis. Lancet Glob Health. 2017;5(12):e1221–e1234. doi:10.1016/S2214-109X(17)30393-5
2. Winkler NS, Fautsch MP. Effects of prostaglandin analogues on aqueous humor outflow pathways. J Ocul Pharmacol Ther. 2014;30(2–3):102–109. doi:10.1089/jop.2013.0179
3. Lim KS, Nau CB, O’Byrne MM, et al. Mechanism of action of bimatoprost, latanoprost, and travoprost in healthy subjects. A crossover study. Ophthalmology. 2008;115(5):790–795 e794. doi:10.1016/j.ophtha.2007.07.002
4. Xalatan(R) (Latanoprost Ophthalmic Solution) 0.005%. Full Prescribing Information. New York, NY:Pharmacia & Upjohn Co. 2012.
5. Pisella PJ, Pouliquen P, Baudouin C. Prevalence of ocular symptoms and signs with preserved and preservative free glaucoma medication. Br J Ophthalmol. 2002;86(4):418–423.
6. Kawa JE, Higginbotham EJ, Chang IL, Yue BY. Effects of antiglaucoma medications on bovine trabecular meshwork cells in vitro. Exp Eye Res. 1993;57(5):557–565. doi:10.1006/exer.1993.1160
7. De Saint Jean M, Brignole F, Bringuier AF, Bauchet A, Feldmann G, Baudouin C. Effects of benzalkonium chloride on growth and survival of Chang conjunctival cells. Invest Ophthalmol Vis Sci. 1999;40(3):619–630.
8. Hedengran A, Steensberg AT, Virgili G, Azuara-Blanco A, Kolko M. Efficacy and safety evaluation of benzalkonium chloride preserved eye-drops compared with alternatively preserved and preservative-free eye-drops in the treatment of glaucoma: a systematic review and meta-analysis. Br J Ophthalmol. 2020:
9. Rodriguez-Aller M, Guinchard S, Guillarme D, et al. New prostaglandin analog formulation for glaucoma treatment containing cyclodextrins for improved stability, solubility and ocular tolerance. Eur J Pharm Biopharm. 2015;95(Pt B):203–214. doi:10.1016/j.ejpb.2015.04.032
10. Tripathi BJ, Tripathi RC, Kolli SP. Cytotoxicity of ophthalmic preservatives on human corneal epithelium. Lens Eye Toxic Res. 1992;9(3–4):361–375.
11. Kabat AG Efficacy and safety of latanoprost 0.005% without benzalkonium chloride vs latanoprost 0.005% with benzalkonium chloride administered daily in patients with open angle glaucoma or ocular hypertension; 2019. Available from: https://www.aaopt.org/detail/knowledge-base-article/efficacy-and-safety-of-latanoprost-0-005-without-benzalkonium-chloride-vs-latanoprost-0-005-with-benzalkonium-chloride-administered-daily-in-patients-with-open-angle-glaucoma-or-ocular-hypertension-339195-3231591.
12. Walimbe T, Chelerkar V, Bhagat P, Joshi A, Raut A. Effect of benzalkonium chloride-free latanoprost ophthalmic solution on ocular surface in patients with glaucoma. Clin Ophthalmol. 2016;10:821–827.
13. Pan J, Wang W, Wei Y, Chen J, Wang L. A calculation model of critical liquid-carrying velocity of gas wells considering the influence of droplet shapes. Nat Gas Ind. 2018;5(4):337–343. doi:10.1016/j.ngib.2018.01.007
14. Natarajan JV, Ang M, Darwitan A, Chattopadhyay S, Wong TT, Venkatraman SS. Nanomedicine for glaucoma: liposomes provide sustained release of latanoprost in the eye. Int J Nanomedicine. 2012;7:123–131.
15. Natarajan JV, Darwitan A, Barathi VA, et al. Sustained drug release in nanomedicine: a long-acting nanocarrier-based formulation for glaucoma. ACS Nano. 2014;8(1):419–429. doi:10.1021/nn4046024
16. Williams HD, Trevaskis NL, Charman SA, et al. Strategies to address low drug solubility in discovery and development. Pharmacol Rev. 2013;65(1):315–499.
17. Ku S, Velagaleti R. Solutol HS15 as a novel excipient. Pharm Technol. 2010;34(11).
18. Patel VR, Dumancas GG, Kasi Viswanath LC, Maples R, Subong BJ. Castor oil: properties, uses, and optimization of processing parameters in commercial production. Lipid Insights. 2016;9:1–12. doi:10.4137/LPI.S40233
19. Cholkar K, Patel A, Vadlapudi AD, Mitra AK. Novel nanomicellar formulation approaches for anterior and posterior segment ocular drug delivery. Recent Pat Nanomed. 2012;2(2):82–95. doi:10.2174/1877912311202020082
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.