Back to Journals » Clinical Ophthalmology » Volume 14
Perceived Surgeon Stress During No-Sedation Topical Phacoemulsification
Authors Mansour A , Stewart MW, Charbaji AR , El Jawhari KM, El Zein L, Mansour MA, Saade JS
Received 7 June 2020
Accepted for publication 4 August 2020
Published 18 August 2020 Volume 2020:14 Pages 2373—2381
DOI https://doi.org/10.2147/OPTH.S266516
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Ahmad Mansour,1,2 Michael W Stewart,3 Abdul Razzak Charbaji,4,5 Khalil M El Jawhari,6 Lulwa El Zein,1 Mohamad A Mansour,1 Joanna S Saade1
1Department of Ophthalmology, American University of Beirut, Beirut, Lebanon; 2Department of Ophthalmology, Rafic Hariri University Hospital, Beirut, Lebanon; 3Department of Ophthalmology, Mayo Clinic, Jacksonville, FL, USA; 4Department of Statistics and Research Methodology, Lebanese American University, Beirut, Lebanon; 5Department of Statistics and Research Methodology, Lebanese University, Beirut, Lebanon; 6Faculty of Medicine, Medical University of Lodz, Lodz, Poland
Correspondence: Ahmad Mansour Email [email protected]
Purpose: To determine the patient-related factors that contribute to surgeon stress during phacoemulsification cataract extraction (PCE) performed under unassisted topical anesthesia.
Methods: This is a prospective study of perceived surgeon stress during phacoemulsification by a single surgeon of consecutive patients undergoing PCE. At the conclusion of each procedure, the surgeon recorded the perceived stress according to the following three indices: surgeon score, qualitative score (yes or no), and total score (sum of itemized causes of stress). Patient variables included in the analysis included gender, age, diabetes mellitus, morbid obesity, intake of oral alpha blockers, floppy iris, laterality, pseudo-exfoliation, intraocular lens power, and initial visual acuity.
Results: During the 10-year study, 1097 eyes underwent surgery. The following patient variables were seen frequently: floppy iris syndrome (92), pseudo-exfoliation (72), and morbid obesity (36). Surgeon identified stress was reported after 250 procedures. On multivariable analysis, the following patient characteristics were associated with surgeon stress: age > 80 years; morbid obesity; floppy iris syndrome; severe nuclear sclerosis; and poor baseline distance corrected visual acuity.
Conclusion: Several ocular and systemic patient-related characteristics contribute to surgeon stress during PCE.
Keywords: brunescent cataract, cataract, floppy iris, morbid obesity, phacoemulsification, pseudo-exfoliation, surgeon stress, surgery time, topical anesthesia
Plain Language Summary
Causes of cataract surgeon stress include patient factors such as advanced age, morbid obesity, floppy iris, dense cataract, and poor initial visual acuity.
Introduction
Modern cataract surgery is a highly technical specialty that requires fine motor skills, precise microscopic control, excellent judgement by the surgeon, and the availability of highly refined instruments. Because the ocular structures are delicate, minor trauma to the cornea or lens capsule can result in permanent vision loss due to corneal edema, cystoid macular edema, retained lens fragments, and retinal detachment. Surgical patients have high expectations and the margin for procedural error is low, which means that phacoemulsification surgery can be stressful for even skilled and experienced surgeons.1–6 Low levels of stress may improve surgical performance but higher levels can adversely affect cognitive function and systemic health.7–18 Unfortunately, few studies regarding stress among ophthalmic surgeons have been published in the literature.19–22
The present study aims to improve our understanding of surgeon-related stress that occurs during phacoemulsification. The primary goals of the study are as follows: (1) identify patient-related factors that cause surgeon stress during phacoemulsification; and (2) evaluate the relationship between these factors and the surgeon’s reported stress levels.
Methods
The Institutional Review Board of Rafic Hariri University Hospital, Beirut, Lebanon, approved this study. Prior to enrollment in this study each patient provided informed written consent in accordance with the Declaration of Helsinki. Patients consented to having their images published. Data were anonymized to maintain patient confidentiality.
Patient Accrual and Pre-Operative Evaluation
This prospective study of phacoemulsification cataract extraction (PCE) surgeries analyzed self-reported surgeon stress with respect to various patient-related ocular and systemic variables. Consecutive patients undergoing PCE under topical anesthesia by a fellowship trained phacoemulsification surgeon (AMM) between January 2010 and August 2019 were eligible for enrollment. Inclusion criteria were as follows: visually significant cataracts in subjects with extreme anxiety, claustrophobia, hearing impairment, intellectual disability, dementia, Parkinson’s disease, monocular status, various degrees of nuclear sclerosis of the lens, flat anterior chamber, or a maximally dilated pupil diameter of less than 3.0 mm.
Baseline distance corrected Snellen visual acuity was measured by the surgeon. Each patient underwent slit lamp and dilated fundus examinations. The degree of lens nuclear sclerosis was graded by slit lamp (Haag-Streit® 900, Haag-Streit AG, Koeniz, Switzerland) examination according to the 5-level Emery-Little classification system (soft, semi-soft, medium hard, hard, and rock-hard). Patients were queried about the ingestion of oral alpha agonist drugs both during the pre-operative ophthalmic examination and just before surgery.
Anesthesia Technique
Topical xylocaine 2% or 3% gel23–26 and subconjunctival lidocaine – to allow for fixation of the globe in case of sudden eye movements during anterior capsulorhexis – were administered by the surgeon in every case. Continuous flow of oxygen was delivered via a nasal cannula. An anesthesia assistant monitored the patient’s vital signs (blood pressure, heart rate, and oxygen saturation), supplemental anesthesia was administered to patients with uncontrolled systemic arterial hypertension, and intravenous dexmedetomidine (Precedex®, Pfizer Inc., NY, NY) was given to anxious patients after telephone consultation with the supervising anesthesiologist.
Surgical Technique
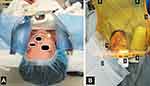
The pupil of the operative eye was dilated with phenylephrine 10%, cyclopentolate 1%, tropicamide 1%, and nonsteroidal anti-inflammatory drops. The surgical field was prepared with a standard betadine scrub and the patient was covered with a transparent sterile drape that facilitated CO2 washout and minimized claustrophobia by allowing the contralateral eye to fixate (Figure 1). An adjustable speculum was used to open the eyelids. Coaxial illumination with the operating microscope was kept to a minimum level.
Superior phacoemulsification incisions were performed in shallow orbits and temporal incisions in deep set eyes. Phacoemulsification was performed with moderate infusion pressure (low infusion pressure was used in high myopes), moderate aspiration pressure, and low phacoemulsification power. The majority of eyes with pseudo-exfoliation received intraocular capsular tension rings with circumferential removal of cortex. Standing phacoemulsification was performed in subjects with morbid obesity (body mass index ≥40).27 Mechanical devices to dilate small pupils were not available during the first 8 years of the study.
Measuring Stress
At the completion of each surgery the surgeon recorded the level of perceived stress on a scale from 0 to 2, the specific causes of stress, the duration of surgery, and any disruption of the procedure or surgical complication. The stress score was based on simplification of the Imperial Stress Assessment Tool.8 The score is the sum of 4 items with each item worth one half a point (“I feel situation out of control”; “I feel tense”; “I feel upset”; “I feel worried”). The investigators also used the following two stress tools: qualitative stress (yes or no); and total stress (sum of one of 13 listed stressors) (Table 1).
 |
Table 1 Itemized Stressors in Phacoemulsification |
Statistical Analysis
Statistical analyses were performed using SPSS version 22 (IBM, Chicago, IL). Paired samples statistics were performed with Pearson correlations. The Chi-square test was used to test the difference between independent binary variables and the t-test was used to test the difference between the independent continuous variables. Paired samples were used to test the difference between right and left eyes. Linear regression analyses were performed with two-tailed Pearson and Spearman Chi-square tests. Multivariate analysis (when the dependent categorical variable was defined as “yes” and “no”) was performed as a simple discriminant analysis using ANOVA, and confirmed by cross tabulation and Pearson Chi-square test. Spearman rank analysis was used to assess the correlation between continuous variables. Statistical significance was assumed when P values were less than 0.05.
Results
Baseline Data
Seven hundred fifteen patients (mean and median age: 74 years; range: 20–99 years) were enrolled in this study and 387 of them underwent bilateral cataract removal. Surgeries were performed on 1097 eyes (599 right eyes and 498 left eyes) and were nearly equally distributed among men (509) and women (588).
Patients in the cohort had the following systemic and ocular co-morbidities: diabetes mellitus (257 eyes in subjects with mean diabetes duration of 15.3 years; range 1–51 years), morbid obesity (36 eyes), pseudo-exfoliation (79 eyes), oral alpha blockers for benign prostate hypertrophy (108 eyes – subjects continued receiving drug), and floppy iris syndrome (92 eyes) (Table 2).
 |
Table 2 Clinical Characteristics of Enrolled Patients and Eyes Undergoing Surgery |
Grading of the nuclear sclerosis was as follows: 0 (soft) in 112 eyes, 1 (soft) in 53 eyes, 2 (semi-soft) in 163 eyes, 3 (medium hard) in 425 eyes, 4 (hard) in 243 eyes, and 5 (rock-hard) in 101 eyes (including cataracta nigra in 4 eyes). Posterior subcapsular cataracts were present in 326 eyes.
Surgical Results
LogMAR distance corrected visual acuity (mean±standard deviation (SD)) improved significantly from the pre-operative visit (1.03±0.63; Snellen equivalent of 20/209) to the final follow-up (0.20±0.37; Snellen equivalent of 20/32; P<0.0001), which took place at a mean of 22.9±31.0 months (range 1–188 months) after surgery. The mean±SD power of the implanted intraocular lens was 19.8±1.6 D (range −3 to +31 D).
Stress Analyses
Stress was reported by the surgeon in 250 of the 1097 surgeries. The mean±SD total stress score among all surgeries was 0.36±0.75 and the frequencies of the scores were as follows: 846 surgeries were scored as 0; 141 surgeries were scored as 1; 80 surgeries were scored as 2; 23 surgeries were scored as 3; and 6 surgeries were scored as 4. The mean surgeon stress score was 0.25±0.48 with 817 surgeries scored as 0, 77 surgeries scored as 0.5, 169 surgeries scored as 1, 4 surgeries scored as 1.5, and 30 surgeries scored as 2. There was a strong correlation between the 3 stress score indices (qualitative, total, and surgeon; P<0.001).
Surgery on male patients was more stressful, particularly when those subjects had been taking oral alpha blockers (Table 3). Stress was reported in 21.8% (128) of surgeries on female patients, 27.7% (111) of surgeries on male patients who were not taking alpha blockers (P=0.02), and 36.1% (39) of male patients taking alpha blockers. Surgery on patients with diabetes was associated with a minor increase in surgeon stress.
 |
Table 3 Clinical Variables and Stress Indices Using Univariate Analysis in 1097 Eyes |
The power of the inserted intraocular lens did not correlate with stress indices despite analyses of several different groupings (hyperopia vs myopia, high ametropia (high hyperopia and high myopia grouped together or separately) vs the rest) (Table 3). Surgeries on eyes with pseudo-exfoliation produced stress scores similar to the rest of the cohort (Table 3; Student’s t-test) but a separate analysis found that 5.8% (49) of surgeries on eyes without recorded stress had pseudo-exfoliation while 11.6% (29) of eyes with a positive total stress score had pseudo-exfoliation (Pearson Chi-square 9.8; P=0.002).
A multivariate analysis (Table 4) that included the following independent variables was performed: age, morbid obesity, baseline distance corrected visual acuity, floppy iris, severity of nuclear sclerosis, and duration of surgery. Floppy iris emerged up as a strong independent variable for stress while intake of alpha blockers did not (Tables 3 and 4). Fewer eyes (48 of 329; 14.6%) with nuclear sclerotic cataract grades of ≤2 were associated with total stress scores compared to eyes with nuclear sclerotic cataract grades >2 (202 of 768; 26.3%; Pearson Chi-square 17.8; P<0.001). More subjects over the age of 80 years (113 of 349, 32.4%) underwent surgeries with recordable total stress than did subjects under the age of 80 (137 of 749; 18.3%; Pearson Chi-square=26.6; P=0.001). Surgeries that lasted less than 30 minutes were less likely to have recordable total stress than surgeries lasting more than 30 minutes (15.5% vs 67.4%; Pearson Chi-square=181.6; P<0.001).
 |
Table 4 Stress Indices and Odds Ratios of the Surgical Variables That Correlated with Stress Using Multivariate Analysis in 1097 Eyes |
Surgeries with Reported Stress
The 250 surgeries with recorded stress underwent additional analyses. More stress was reported for surgeries on the right eye than the left eye (surgeon stress score 0.98±0.42 vs 0.46±0.50, paired t-test; P<0.001; total stress score 0.98±0.45 vs 0.48±0.50, P<0.001), perhaps related to the surgeon being right-handed. Surgeon stress scores were higher during surgeries performed in patients over the age of 80 years compared to those under the age of 80 (1.04±0.45 vs 0.92±0.40; P=0.01). Both surgeon stress scores and total stress scores were higher in patients with morbid obesity compared to those without (1.25±0.58 vs 0.96±0.41, P=0.008; 2.00±1.03 vs 1.55±0.73, P=0.02).
In the 383 subjects who underwent surgery in both eyes the chance of surgeon perceived stress in the second eye increased by 2.7-fold if stress was associated with surgery in the first eye (45.5% vs 16.8%; P<0.001) (Table 5). In 250 of these patients no surgeon reported stress was associated with surgery in either eye. If no stress was reported after surgery on the right eye, stress in the left eye occurred in 18.8% of patients; if no stress was reported after surgery in the left eye, stress in the right eye occurred in 14.7% of patients; if stress was associated with surgery in the right eye, stress in the left eye occurred in 42.7% of patients; and if stress was associated with surgery in the left eye, stress in the right eye occurred in 35.6% of patients.
Discussion
In the current study perceived surgeon stress was associated with advanced patient age, severity of nuclear sclerosis, profound visual loss, floppy iris syndrome, and morbid obesity. Anatomic characteristics – a right-handed surgeon has greater difficulties operating on left eyes, particularly when they are deep set with small palpebral fissures and a robust Bell’s phenomenon – also increase stress. Morbid obesity makes patient positioning difficult and increases the risk of posterior capsular rupture due to positive vitreous pressure.27 Floppy iris syndrome leads to intraoperative miosis though this can be countered with the use of phenylephrine and ketorolac intraocular solution 1%/0.3% (Omidria®) and dilating devices such as the Malyugin ring and iris hooks. Advanced patient age frequently stresses the surgeon because of associated mental deterioration (Alzheimer’s or Parkinson’s), sensorineural hearing loss, and poor patient cooperation. Advanced nuclear sclerosis requires the use of high ultrasound energy for extended periods and is frequently accompanied by zonular weakness, absent cortex, and increased risk of posterior capsular rupture. Pseudo-exfoliation increases the risk of vitreous loss because of poor pupillary dilation and weak zonules.
Techniques for cataract removal have evolved from large-incision intracapsular and extracapsular extractions to phacoemulsification through a small, suture-less corneal incision. Anesthesia techniques associated with cataract surgery have advanced from general anesthesia to regional blocks (retrobulbar, peribulbar, sub-Tenon’s, and subconjunctival) to the currently popular topical anesthesia with anesthetic drops or gel, together with intracameral anesthetic. Topical anesthesia requires surgery to be performed fast and efficiently to minimize stress on the patient. The presence of systemic conditions such as claustrophobia, repeated cough, chronic obstructive pulmonary disease, irritable bladder, heart failure, orthopnea, morbid obesity, and Alzheimer disease, or ocular conditions such as high myopia, miosis, pseudo-exfoliation syndrome, idiopathic floppy iris syndrome, corneal opacity, intumescent cataract, and cataracta nigra (brunescent cataract), coupled with loss of the anesthetic effect interfere with surgical efficiency and increase the duration of surgery. In a multivariate analysis our study found that length of time was an independent variable for surgeon stress.
Stress can be quantified using the validated State-Trait Anxiety Inventory, which is a 20-item self-reporting assessment device that has been validated in more than 30 languages.8 Other methods that are used to evaluate stress include the Imperial Stress Assessment Tool for measuring stress during surgery, heart rate variability, one-on-one interviews with subjects, and telemetry monitoring. At the molecular level, telomere shortening can be seen after a prolonged period of stress.2
Ophthalmology is a rapidly evolving, highly technical diagnostic and surgical specialty that combines intellectually challenging management of medical conditions with the performance of minor and major surgical procedures. Newly introduced equipment, drugs, and biologics create a paradigm shift every few years. This rapid explosion of knowledge and change in technology challenges physicians to meet constantly evolving standards of care, use digital medical records, face increased litigation, satisfy demanding patients who expect perfect outcomes, and receive decreasing financial reimbursement for services, all of which contribute to physician stress. Because ophthalmic surgery profoundly effects the lives of blind and visually impaired individuals, most ophthalmologists derive tremendous professional satisfaction from their work. Surgeons perform this challenging work to improve the lives of individuals confronted with serious vision-threatening problems, to experience the joy derived from healing, and to experience satisfaction from correcting complicated problems. The operating room is a stressful environment in which highly technical complex procedures must be performed in a timely manner. This requires an efficiently working team composed of an anesthesiologist, nurse, technician, pharmacist, and surgeon. Surgical procedures may be plagued by distractions (equipment-related disruptions and acoustic disruptions)9 and interruptions.29 Excessive stress from distractions and disruptions can compromise a surgeon’s psychomotor ability and, if sufficiently frequent, causes distress, exhaustion, or burnout.12 Burnout is a long-term sequela that has been defined as a triad of “psychological syndrome of emotional exhaustion, depersonalization, and reduced personal accomplishment”.12
The literature on perceived surgeon stress is limited to small studies, many of which were conducted in laboratory settings. One objective measure of stress, the heart rate, does not distinguish between the effects of physical activity and mental stress but a validated algorithm based on heart rate and pulse variability is commercially available as the smart patch (Health Patch™).7 Activation of the hypothalamic-pituitary-adrenocortical axis by acute stress is associated with release of salivary cortisol, which can be used as a hormonal biomarker for stress. One study with Internal Medicine residents-in-training found higher cortisol concentrations during an emergency room-duty day than during a regular working day.15
A more relevant way to measure perceived surgeon stress is through self-reporting with the widely accepted scale known as State-Trait Anxiety Inventory.15 This psychological inventory is based on a 4-point Likert scale and consists of 40 questions that measure anxiety regarding an event. The Imperial Stress Assessment Tool reliably assesses stress in the operating room and is a concise version of the State-Trait Anxiety Inventory.8 The current scoring scheme incorporates 3 of the 4 points on the State-Trait Anxiety Inventory (each item is graded from 1 to 4) but with yes or no scoring.
Stress on the molecular level is seen as telomere shortening.2 Telomere length was used to quantify the effects of stress in 250 interns (first-year residents) who endured repeated stressful experiences during one year of medical training.2
Sources of intraoperative stress include fatigue (particularly when operating at night), equipment-related disruptions, missing or defective instruments, deficient supplies, inadequate time, a complex or high-risk surgery, surgical errors by the assistant, scrub nurse, or the surgeon himself.3,4,15 Weigl et al29 found that intra-operative interruptions occur frequently (once every 6 minutes on average), with people entering or exiting the operating room, telephone and pager calls, and communications not related to the case occurring most commonly.
In the only PubMed referenced study on surgeon stress during phacoemulsification that we could find, Pandey et al23 randomized 75 patients to phacoemulsification under no anesthesia, topical anesthesia, or topical plus intracameral anesthesia. Surgeon stress was significantly greater in the no-anesthesia group than in either the topical or the topical plus intracameral groups. The authors concluded that surgeons are more likely to experience stress when anesthesia use is minimal or absent. Similarly, we found a high incidence of surgeon stress during surgery under topical anesthesia without sedation, especially in patients who were uncooperative (those who frequently moved their body or their eyes, or squeezed their eyelids) or coughed frequently due to heavy smoking.
The high level of surgeon-related stress measured in this study may have resulted from the large number of high-risk patients (Alzheimer, severe pulmonary or cardiac disease, morbid obesity) that are seen at this tertiary care center. Most of the patients had marked vision loss due to advanced cataracts, which increased the difficulty of surgery. Many of the patients in this series had no medical insurance so surgeries were performed in facilities that lacked skilled dedicated ophthalmic anesthesia support. The assigned anesthesia service in such facilities were highly proficient in general anesthesia but lacked expertise in managing difficult patients receiving topical anesthesia, which resulted in their giving minimal analgesia because of a “I can’t give anything, doctor” approach or they over-sedated patients and caused frequent episodes of apnea and agitation. Our approach, therefore, was to forego intervention by the anesthesia team unless complications arose. This required the surgeon to manage anesthesia and mandated that surgery on challenging patients be performed efficiently. An accomplished skilful anesthesia support helps to decrease the degree of stress in difficult cases, especially when complications arise.
Outside the surgical theater little is known about practice-related stress among ophthalmologists. A recent report by Viviers et al20 found high levels of stress among Quebec ophthalmologists due to work overload, organizational deficiencies, and little professional recognition. Physicians subjected to high stress levels can develop psychiatric problems such as burnout and psychological distress. A Canada-wide survey that was commissioned by the Canadian Medical Association in 2007–2008 (https://www.cma.ca) reported that 45.7% of physicians suffered from advanced stages of burnout due to the following contributory elements: time constraints, bureaucratization of procedures, an increase in the number of high-needs cases, introduction of information technologies, risk of errors, increased responsibilities, substantial lack of resources, poor human resources management, lack of collegial solidarity, rapid pace of technological advances, pressure related to patients’ high expectations, inaccessible or outdated equipment, being overworked, fears about complications (especially in complex high-risk cases), errors, and risk of professional reprimand.20
Other specialties have measured surgeon stress during surgery. Two neuro-otology fellows were monitored with wireless continuous electrocardiograms while performing six mastoidectomies and facial nerve dissections both on patients and on cadavers.16 Decreased heart rate variability and increased sympathetic tone – indicating high levels of mental stress – were observed during surgeries on patients, yet similar changes did not occur during cadaveric dissections. Practice-related stress may lead to physician burnout, family problems, decreased quality of patient care, depression, and professional impairment because of illicit drug or alcohol abuse. Burnout has been reported in 25.2% of ophthalmologists,19 30–40% of general surgeons,12 and 50–60% of orthopedic surgeons.12 Career satisfaction among 16 surgical specialties included pediatrics (86% to 96%), endocrinology (96%), orthopedic surgery (80%), vascular surgery (64%) and plastic surgery (33%).14 The yearly cost attributed to physician turnover and reduced clinical hours from burnout in the United States has been estimated at $4.6 billion.13
Stress management techniques, such as mental rehearsal and resilience, can lessen surgeon stress and burnout.3 Low volume classical music may improve the performance of surgery and lower the surgeon’s stress.30 Surgeon preparedness and pre-operative planning can decrease the incidence of poor outcomes and surgical “surprises”.
Weaknesses of the present study include the reliance on only one surgeon’s experience with stress – the surgeon’s a type A personality may have experienced more stress than a type B personality – lack of objective signs of stress (such as heart rate variation), and absence of an independent observer to assess the stressors in the operating room. Comparison of surgeon stress between topical and peribulbar anesthesia can help delineate the factors inherent in surgery under topical anesthesia. Additional studies are needed to better define the stressors, and the short- and long-term consequences of intraoperative stress during cataract surgery. Moreover, strategies to prevent and treat surgeon stress need to be developed.
Conclusion
Operating room stress is a real entity that can be perceived subjectively and quantified objectively. Several factors within the operating theatre – disruptions, time pressure, complex or high-risk cases, and surgical errors – can compromise patient safety and cause stress to the surgeon. In the current study, patient characteristics such as advanced age or morbid obesity and ocular characteristics such as severe cataract, pseudo-exfoliation and floppy iris were the most stressful variables for the cataract surgeon. Successful management of these stressors includes pre-operative planning, precise and efficient surgical technique, presence of dedicated ophthalmic anesthesia support, and management of intraoperative distractions.
Disclosure
Michael W. Stewart, MD: Consultant – Alkahest, Bayer; Institutional Research Support – Allergan, Regeneron. The authors report no other conflicts of interest in this work.
References
1. Clare G. Cochrane corner: patient safety in cataract surgery. Eye (London). 2019;33(10):1517–1518. doi:10.1038/s41433-019-0526-8
2. Ridout KK, Ridout SJ, Guille C, Mata DA, Akil H, Sen S. Physician-training stress and accelerated cellular aging. Biol Psychiatry. 2019;
3. Ng R, Chahine S, Lanting B, Howard J. Unpacking the literature on stress and resiliency: a narrative review focused on learners in the operating room. J Surg Educ. 2019;76(2):343–353. doi:10.1016/j.jsurg.2018.07.025
4. Sami A, Waseem H, Nourah A, et al. Real-time observations of stressful events in the operating room. Saudi J Anaesth. 2012;6(2):136–139. doi:10.4103/1658-354X.97026
5. González-Cabrera JM, Fernández-Prada M, Iribar C, Molina-Ruano R, Salinero-Bachiller M, Peinado JM. Acute stress and anxiety in medical residents on the emergency department duty. Int J Environ Res Public Health. 2018;15(3):E506. doi:10.3390/ijerph15030506
6. Georgiou K, Larentzakis A, Papavassiliou AG. Surgeons‘ and surgical trainees‘ acute stress in real operations or simulation: a systematic review. Surgeon. 2017;15(6):355–365. doi:10.1016/j.surge.2017.06.003
7. Weenk M, Alken APB, Engelen LJLPG, Bredie SJH, van de Belt TH, van Goor H. Stress measurement in surgeons and residents using a smart patch. Am J Surg. 2018;216(2):361–368. doi:10.1016/j.amjsurg.2017.05.015
8. Arora S, Tierney T, Sevdalis N, et al. The Imperial Stress Assessment Tool (ISAT): a feasible, reliable and valid approach to measuring stress in the operating room. World J Surg. 2010;34(8):1756–1763. doi:10.1007/s00268-010-0559-4
9. Wheelock A, Suliman A, Wharton R, et al. The impact of operating room distractions on stress, workload, and teamwork. Ann Surg. 2015;261(6):1079–1084. doi:10.1097/SLA.0000000000001051
10. Jung JJ, Elfassy J, Grantcharov T. Factors associated with surgeon’s perception of distraction in the operating room. Surg Endosc. 2020;34(7):3169–3175. doi:10.1007/s00464-019-07088-z
11. Klein J, Grosse Frie K, Blum K, von Dem Knesebeck O. Psychosocial stress at work and perceived quality of care among clinicians in surgery. BMC Health Serv Res. 2011;11:109. doi:10.1186/1472-6963-11-109
12. Arora M, Diwan AD, Harris IA. Burnout in orthopaedic surgeons: a review. ANZ J Surg. 2013;83(7–8):512–515. doi:10.1111/ans.12292
13. Han S, Shanafelt TD, Sinsky CA, et al. Estimating the attributable cost of physician burnout in the United States. Ann Intern Med. 2019;170(11):784–790. doi:10.7326/M18-1422
14. Pulcrano M, Evans SR, Sosin M. Quality of life and burnout rates across surgical specialties: a systematic review. JAMA Surg. 2016;151(10):970–978. doi:10.1001/jamasurg.2016.1647
15. Arora S, Hull L, Sevdalis N, et al. Factors compromising safety in surgery: stressful events in the operating room. Am J Surg. 2010;199(1):60–65. doi:10.1016/j.amjsurg.2009.07.036
16. Dedmon MM, O’Connell BP, Yawn RJ, et al. Measuring mental stress during otologic surgery using heart rate variability analysis. Otol Neurotol. 2019;40(4):529–534. doi:10.1097/MAO.0000000000002187
17. Yamanouchi K, Hayashida N, Kuba S, et al. Increase in operator’s sympathetic nerve activity during complicated hepatobiliary surgery: evidence for surgeons’ mental stress. Tohoku J Exp Med. 2015;237(3):157–162. doi:10.1620/tjem.237.157
18. Babyar JC. They did not start the fire: reviewing and resolving the issue of physician stress and burnout. J Health Organ Manag. 2017;31(4):410–417. doi:10.1108/JHOM-11-2016-0212
19. Nair AG, Jain P, Agarwal A, Jain V. Work satisfaction, burnout and gender-based inequalities among ophthalmologists in India: a survey. Work. 2017;56(2):221–228. doi:10.3233/WOR-172488
20. Viviers S, Lachance L, Maranda MF, Ménard C. Burnout, psychological distress, and overwork: the case of quebec’s ophthalmologists. Can J Ophthalmol. 2008;43(5):535–546. doi:10.3129/i08-132
21. Stewart WC, Adams MP, Stewart JA, Nelson LA. Survey of practice-related stress among United States and European ophthalmologists. Graefes Arch Clin Exp Ophthalmol. 2011;249(9):1277–1280. doi:10.1007/s00417-011-1686-9
22. Mansour AM, Hamam R. Operating room central serous chorioretinopathy. SAGE Open Med Case Rep. 2017;5:2050313X17740052. doi:10.1177/2050313X17740052
23. Pandey SK, Werner L, Apple DJ, Agarwal A, Agarwal A, Agarwal S. No-anesthesia clear corneal phacoemulsification versus topical plus intracameral anesthesia. Randomized clinical trial. J Cataract Refract Surg. 2001;27(10):1643–1650. doi:10.1016/S0886-3350(01)00793-3
24. Koch PS. Efficacy of lidocaine 2% jelly as a topical agent in cataract surgery. J Cataract Refract Surg. 1999;25(5):632–634. doi:10.1016/S0886-3350(99)00007-3
25. Assia EI, Pras E, Yehezkel M, Rotenstreich Y, Jager-Roshu S. Topical anesthesia using lidocaine gel for cataract surgery. J Cataract Refract Surg. 1999;25(5):635–639. doi:10.1016/S0886-3350(99)00026-7
26. Patel BCK, Burns TA, Crandall A, et al. A comparison of topical and retrobulbar anesthesia for cataract surgery. Ophthalmology. 1996;103(8):1196–1203. doi:10.1016/S0161-6420(96)30522-8
27. Mansour AM, Al Dairy M. Standing phacoemulsification in morbid obesity. J Cataract Refract Surg. 2004;30(11):2265–2268. doi:10.1016/j.jcrs.2004.02.088
28. Kelly SP, Steeples LR, Smith R, Azuara-Blanco A. Surgical checklist for cataract surgery: progress with the initiative by the Royal College of Ophthalmologists to improve patient safety. Eye (London). 2013;27(7):878–882. doi:10.1038/eye.2013.101
29. Weigl M, Antoniadis S, Chiapponi C, Bruns C, Sevdalis N. The impact of intraoperative interruptions on surgeons’ perceived workload: an observational study in elective general and orthopedic surgery. Surg Endosc. 2015;29(1):145–153. doi:10.1007/s00464-014-3668-6
30. El Boghdady M, Ewalds-Kvist BM. The influence of music on the surgical task performance: a systematic review. Int J Surg. 2020;73:101–112. doi:10.1016/j.ijsu.2019.11.012
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.