Back to Journals » International Medical Case Reports Journal » Volume 13
Pancreatic Tumors Complicating Pregnancy: A Concern for Fetomaternal Well Being
Authors Joshi U , Upadhaya SR , Agrawal V , Rana A
Received 19 May 2020
Accepted for publication 20 June 2020
Published 8 July 2020 Volume 2020:13 Pages 255—259
DOI https://doi.org/10.2147/IMCRJ.S263298
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Ronald Prineas
Utsav Joshi, Sandesh Raj Upadhaya, Vishakha Agrawal, Ashma Rana
Department of Obstetrics and Gynecology, Institute of Medicine, Tribhuvan University, Kathmandu, Nepal
Correspondence: Utsav Joshi
Maharajgunj Medical Campus, Institute of Medicine, Tribhuvan University, PO Box 1524, Kathmandu, Nepal
Tel +977 98 4972 6254
Fax +977 1 4423771
Email [email protected]
Introduction: Hemoperitoneum resulting from the rupture of pancreatic tumors is a rare condition, especially during pregnancy.
Case Presentation: We report a case of a 21-year-old gravida 2, para 1, at 25+5 weeks of gestation, who presented to the hospital with severe epigastric pain and decreased fetal movement. Ultrasonography showed intrauterine fetal death, a retroperitoneal mass in the epigastric region, and hemoperitoneum. Computed tomography scan revealed a heterogeneously enhancing pancreatic mass suggestive of pancreatic neoplasm. However, the late diagnosis and the delay in treatment resulted in a deterioration of maternal status with eventual mortality.
Conclusion: Diagnostic difficulties occur because of the rarity of the condition and vague clinical presentations. In case of a pregnancy complicated by hemoperitoneum, prompt effort to stop the intraperitoneal bleeding is imperative.
Keywords: hemoperitoneum, pancreas, neoplasm, pregnancy, mortality
Introduction
Neoplasms are uncommon during pregnancy, leaving no exception to pancreatic tumors, which affect nearly one in 1000 live births.1 The symptoms of pancreatic neoplasms may simulate symptoms of normal pregnancy, resulting in a delay in diagnosis. Rupture of pancreatic neoplasms with subsequent hemoperitoneum is challenging, even more so during pregnancy as fetal maturation and maternal disease state have to be considered while planning intervention. Here, we discuss a case of suspected pancreatic neoplasm in a 21-year-old female, at 25+5 weeks of gestation, who presented with an acute abdomen and unstable vital signs.
Case Presentation
A 21-year-old gravida 2, para 1, at 25+5 weeks of gestation was referred to the emergency department of a university hospital with complaints of severe epigastric pain, vomiting, and decreased fetal movement for four days. She denied any history of trauma. Regarding menstrual history, she attained her menarche at 12 years, and she had a regular 30-day cycle with bleeding lasting for 3–4 days. Her past medical history was unremarkable. On examination, she was pale, severely dehydrated, tachycardiac (pulse – 140/min), tachypneic (respiratory rate – 35/min), hypotensive (blood pressure – 100/60 mm of Hg), and febrile (temperature – 101°F). Her Glasgow Coma Scale (GCS) score was 15/15. Abdominal examination revealed a palpable epigastric mass that was firm, tender, and measured roughly 10 cm x 8 cm. The uterus was approximately 22 weeks in size. Two contractions, each lasting fifteen seconds, were noted in ten minutes. On auscultation, fetal heart sounds could not be appreciated. Per-vaginal examination revealed a midline cervix of medium consistency, 1 cm dilation, and 20 to 30% effacement with the presence of “show”.
Laboratory investigations revealed severe anemia (4.4 gm%), thrombocytopenia (33,000/cu. mm), deranged prothrombin time (PT – 60 seconds) and international normalized ratio (INR – 5), and hypokalemia (2.4 mEq/L). Amylase and lipase were within normal limits. The standard values of these lab parameters used in our hospital are presented at the end of the text. Ultrasonography (USG) showed intrauterine fetal death (IUFD) of 26–27 weeks of gestation, a complex mass at the epigastric region (retroperitoneal area), and gross hemoperitoneum. Computed Tomography (CT) scan of the abdomen (Figures 1 and 2) showed a well-defined, round to oval, heterogeneously enhancing mass measuring 18 cm x 12.5 cm, originating from the body of the pancreas, suggestive of pancreatic neoplasm. A significant amount of high-density collection in the peritoneal cavity was suggestive of hemoperitoneum.
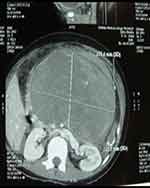 |
Figure 1 CT axial image shows well-defined heterogeneously enhancing mass arising from the body of pancreas. |
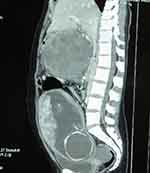 |
Figure 2 CT sagittal image shows heterogeneously enhancing pancreatic mass along with a fetus within the uterus. |
In the emergency room, an intravenous line was opened in both the arms with a wide bore cannula, and the patient was managed with intravenous normal saline, pantoprazole (40 mg), ondansetron (4mg), and antibiotics (ceftriaxone 1 gm and metronidazole 500 mg). A multidisciplinary discussion with the surgical, radiological, and anesthetic team was done. After reviewing the case, the team decided to transfer the patient to radiology a second time to perform a contrast-enhanced computed tomography (CECT) scan, identify the bleeding point, and attempt arterial embolization in the same setting. However, she could not undergo CECT because of her unstable hemodynamic status and was transferred to the surgical intensive care unit (SICU) for further management. On re-evaluation in SICU, she had hematuria, slight vaginal bleeding, bleeding from the nasogastric tube in situ, and her blood pressure was 90/50 mm of Hg. Per-vaginal examination showed progressive dilatation of the cervical os to 4 cm. Repeat evaluation of the coagulation profile demonstrated an INR of 20. Deranged coagulation profile and bleeding from multiple sites raised suspicion of IUFD induced coagulopathy. In the meantime, three units of fresh frozen plasma, three units of whole blood (including one unit of fresh blood), and two units of platelet-rich plasma were transfused over six hours. However, her blood pressure continued to drop, and she was eventually started on noradrenaline support (0.1 micrograms/kg/min). Her oxygen saturation continued to fall and eventually reached 52% in room air. Consequently, she was intubated. Repeat physical examinations were done at regular intervals to evaluate the clinical status of the patient and the progression of labor.
Twenty-one hours after admission to the hospital, there was a spontaneous expulsion of the dead fetus weighing around 900 grams, with no gross anomalies, and a complete and normal-looking placenta. Ten units of oxytocin were administered intravenously following delivery. There was no evidence of active vaginal bleeding postpartum. Yet, her hemoglobin (6.4 gm%) and platelet count (28,000/cu. mm) were low. The surgical team still planned to acquire sufficient information via CECT on the exact source of bleeding before shifting the patient to the operating room for laparotomy. However, multiple attempts to shift the patient to radiology were unsuccessful. Thirty hours after hospitalization, her blood pressure plummeted, and she went into asystole. She was managed with ten cycles of cardiopulmonary resuscitation (CPR), following which her blood pressure reverted to 84/46 mm Hg. Simultaneous inotrope and vasopressor support were barely helpful, as severe bradycardia ensued, followed by asystole. Two hours later, she was pronounced dead.
The family members of the deceased did not provide consent for the autopsy. Thus, postmortem histopathological evaluation of the tumor could not be done. In the end, we were unable to get more details of the tumor or the bleeding site.
Discussion
In a review of over 4.8 million deliveries in California for nine years, Smith and colleagues (2003) noted the incidence of malignant neoplasms during pregnancy or in the subsequent 12 months to be only about 0.94/1000 live births.1 Pancreatic neoplasms, though rare, can complicate pregnancy as evidenced by a few reported cases in the literature [pancreatic adenocarcinoma (8), cystic pancreatic neoplasms (13), and pancreatic neuroendocrine tumors (3)].2–5 Mucinous cystic neoplasms and adenocarcinoma are the most common forms of pancreatic malignancies, with pancreatic neuroendocrine tumors accounting for less than 5% of all pancreatic tumors.5,6
Patients with pancreatic neoplasms present with epigastric pain, postprandial fullness, palpable abdominal mass, nausea, vomiting, diarrhea, steatorrhea, and/or weight loss.7 These tumors may contain estrogen receptors and be sensitive to estrogen manipulation. This may cause the tumors to be more aggressive during pregnancy, with a higher chance of rupture and intraperitoneal bleed.3,4 This may explain the tumor bleeding in this case. Besides, symptoms like abdominal pain, nausea, vomiting, and backache may be construed to pregnancy and lead to a delay in diagnosis and eventual disease progression.
Several other tumors in pregnancy can also present with symptoms that result in a misdiagnosis. Pheochromocytoma or paraganglioma, which occur in 0.007% of all pregnancies, can present with new-onset hypertension, palpitations, diaphoresis, and headache, all of which may result in a misdiagnosis of hypertensive disorders in pregnancy.8 Renal cell carcinoma often presents with vague and mild abdominal discomfort, which can be falsely labeled as pregnancy-related.9 The pregnancy-related changes in the breast often overshadow the diagnosis of breast cancer and allow these cancers to remain unnoticed until the first postpartum year.10 Cushing’s syndrome due to adrenal adenoma in pregnancy shares many clinical signs and symptoms with a normal pregnant woman like weight gain, hypertension, hyperglycemia, and fatigue.11
Imaging with USG, CT scan, and magnetic resonance imaging (MRI); tumor markers; and biopsy of the mass can help establish a diagnosis of pancreatic neoplasm. The lower risk of fetal radiation exposure compared to a CT scan, and improved quality of imaging compared to a transabdominal USG, makes MRI the preferred imaging modality to decide resectability in pregnant patients.5 Though biopsy is an important tumor diagnostic tool, some authors do not recommend it during pregnancy because of the potential risks of biopsy related bleeding, rupture of the lesion, or peritoneal seeding of the biopsy material.12
Acute hemoperitoneum is a rare obstetrical emergency, and its presence in combination with an epigastric mass is rarer. The differentials are few. Pregnancy is associated with an increased risk of rupture of some liver lesions, hepatic adenoma being the most common benign mass. Pregnancy complicated by hepatic hemangioma, hemolysis, elevated liver enzymes, and low platelet count (HELLP) syndrome, and hepatocellular carcinoma may also result in spontaneous hepatic rupture and subsequent hemoperitoneum.13 Another infrequent differential is an ectopic hepatic pregnancy, with a projected incidence of 1 in 10,000 to 25,000 live births, and it typically presents with acute symptoms like abdominal pain and bleeding. Hemoperitoneum secondary to such a pregnancy may either be a prolonged and gradual drip or a massive hemorrhage resulting in hypovolemic shock. Abdominal USG can identify the hepatic location of the pathology, and laparoscopic surgery can be an amenable option in the first trimester.14
For any surgical emergency in pregnancy, the benefit of delaying surgery for fetal maturation must be balanced with the risk of maternal disease progression. Surgery in the first trimester may cause spontaneous abortion or poor fetal outcomes, including congenital anomalies.5,15 Fetal organogenesis is completed once the pregnancy enters the second trimester and the smaller size of the fetus allows for an easier surgical procedure as compared to the third trimester.5,16,17 With malignancy that is highly aggressive, we should discuss the possibility of termination of pregnancy with the patient in the first trimester itself to pursue further management without any delay.2 Chemoradiation therapy has been proposed as another modality of management for resectable pancreatic cancer, both in adjuvant and neoadjuvant forms.15
Non-obstetric surgeries during pregnancy require a multidisciplinary approach involving the obstetrician, general surgeon, anesthesiologist, and neonatologist for the best management. There are several indications for non-obstetric surgeries during pregnancy, with appendicitis, cholecystitis, bowel obstruction, adnexal torsion, and trauma being the most common ones.18 As in the presented case, tumors complicated by hemoperitoneum may also be a rare indication of surgery during pregnancy. The data concerning the effect of these surgeries on the developing fetus and the pregnancy has been conflicting. As mentioned previously, there is a potential increase in the risk of spontaneous abortion among patients undergoing general anesthesia and surgery.5,19 Surgery can also increase the risk of complications like fetal hypoxia, infection, preterm labor, and rate of cesarean delivery.19,20
In our case, the patient initially presented with equivocal symptoms, which could have been a part of her pregnancy. However, the patient had unstable vital signs, which called for urgent diagnosis. The results from the imaging studies alerted us to the presence of a pancreatic mass that probably ruptured, resulting in hemoperitoneum and landed the patient in shock. In a situation like this, a high degree of suspicion is required to make a diagnosis early in the course of the disease. Even in the presence of gross hemoperitoneum, we deferred an exploratory laparotomy with a view of doing so after the expulsion of the dead fetus for the fear of IUFD induced coagulopathy. In retrospect, to avoid maternal morbidity and mortality, the best approach would have been to perform emergency laparotomy to evacuate the blood and control the bleeding site in an early stage of presentation, rather than waiting for the delivery of the fetus.
Conclusion
This case report highlights the fact that pancreatic neoplasms, being uncommon during pregnancy, may pose a diagnostic difficulty. Obstetricians should always keep the possibility of ruptured pancreatic neoplasm in mind when encountered with an acute abdomen in pregnancy. In pancreatic tumors complicating pregnancy, prompt effort to stop the intraperitoneal bleeding must be strongly considered first along with the use of critical resuscitative measures such as vasopressor and inotrope support.
Standard Laboratory Values
Hemoglobin= 12-18 gm%
Platelets= 150,000-400,000/cu mm
Prothrombin time= 11-16 seconds
International normalized ratio= 1-1.2
Potassium= 3.5-5.2 mEq/L
Amylase= <100 U/L
Lipase = < 60 U/L
Abbreviations
GCS, Glasgow Coma Scale; PT, prothrombin time; INR, international normalized ratio; USG, ultrasonography; IUFD, intrauterine fetal death; CT, computed tomography; CECT, contrast enhanced computed tomography; SICU, surgical intensive care unit; CPR, cardiopulmonary resuscitation; MRI, magnetic resonance imaging; HELLP, hemolysis, elevated liver enzymes, and low platelet count.
Data Sharing Statement
Not applicable.
Ethics Approval
Not required.
Consent for Participation and Publication
Written informed consent was obtained from the patient’s parents for the publication of this case report and any accompanying images. A copy can be made available for review upon request.
Author Contributions
UJ and SRU compiled the case and drafted the initial manuscript. VA did the initial literature review. AR did the clinical diagnosis, investigations, and treatment of the case. All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; gave final approval of the version to be published; and agree to be accountable for all aspects of the work.
Funding
There is no funding to report.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Cunningham F, Leveno K, Bloom S, Hauth J, Rouse D, Spong C. Neoplastic disease. In: Williams Obstetrics.
2. Blackbourne LH, Jones RS, Catalano CJ, Iezzoni JC, Bourgeois FJ. Pancreatic adenocarcinoma in the pregnant patient: case report and review of the literature. Cancer. 1997;79(9):1776–1779. doi:10.1002/(SICI)1097-0142(19970501)79:9<1776::AID-CNCR20>3.0.CO;2-U
3. Kosumi K, Takamori H, Hashimoto D, et al. Mucinous cystic neoplasm of the pancreas activated during pregnancy. Surgical Case Rep. 2015;1(1):1–5. doi:10.1186/s40792-014-0012-2
4. Sciscione AC, Villeneuve JB, Pitt HA, Johnson TR. Surgery for pancreatic tumors during pregnancy: a case report and review of the literature. Am J Perinatol. 1996;13(01):21–25. doi:10.1055/s-2007-994197
5. Boyd CA, Benarroch-Gampel J, Kilic G, Kruse EJ, Weber SM, Riall TS. Pancreatic neoplasms in pregnancy: diagnosis, complications, and management. J Gastrointestinal Surgery. 2012;16(5):1064–1071. doi:10.1007/s11605-011-1797-y
6. Zaborowski A, Walsh SM, Ravi N, Reynolds JV. Pancreatic aetiology for massive upper gastrointestinal haemorrhage in pregnancy. Case Rep Surg. 2016;2016.
7. de Jong K, Bruno MJ, Fockens P. Epidemiology, diagnosis, and management of cystic lesions of the pancreas. Gastroenterol Res Pract. 2012;2012.
8. Bapayeva G, Terzic M, Togyzbayeva K, et al. Late diagnosis of pheochromocytoma in pregnancy with poor fetal outcome. Archive Oncol. 2020;2. doi:10.2298/AOO191012002B
9. Tiang KW, Ng KL, Vega-Vega A, Wood S. Rapidly enlarging renal tumor during pregnancy: diagnostic and management dilemma. J Kidney Cancer VHL. 2014;1(1):12. doi:10.15586/jkcvhl.2014.6
10. Botha MH, Rajaram S, Karunaratne K. Cancer in pregnancy. Int J Gynecol Obstet. 2018;143:137–142. doi:10.1002/ijgo.12621
11. LK N. Cushing’s syndrome in pregnancy; 2019. Available from: http://www.uptodate.com/contents/cushings-syndrome-inpregnancy.
12. Søreide JA, Greve OJ, Gudlaugsson E. Adult pancreatic hemangioma in pregnancy–concerns, and considerations of a rare case. BMC Surg. 2015;15(1):119. doi:10.1186/s12893-015-0106-1
13. Lucey BC, Varghese JC, Soto JA. Spontaneous hemoperitoneum: causes and significance. Curr Probl Diagn Radiol. 2005;34(5):182–195. doi:10.1067/j.cpradiol.2005.06.001
14. Garzon S, Raffaelli R, Montin U, Ghezzi F. Primary hepatic pregnancy: report of a case treated with laparoscopic approach and review of the literature. Fertil Steril. 2018;110(5):925–931. doi:10.1016/j.fertnstert.2018.05.020
15. Kakoza RM, Jr CM V, Stuart KE, Takoudes T, Hanto DW. Pancreatic adenocarcinoma in the pregnant patient: a case report and literature review. J Gastrointestinal Surgery. 2009;13(3):535–541. doi:10.1007/s11605-008-0697-2
16. Herring AA, Graubard MB, Gan SI, Schwaitzberg SD. Mucinous cystadenocarcinoma of the pancreas during pregnancy. Pancreas. 2007;34(4):470–473. doi:10.1097/mpa.0b013e31803799d8
17. Ikuta SI, Aihara T, Yasui C, et al. Large mucinous cystic neoplasm of the pancreas associated with pregnancy. World J Gastroenterol. 2008;14(47):7252. doi:10.3748/wjg.14.7252
18. Simonetto C, Garzon S, Laganà AS, et al. Non-obstetric surgery during pregnancy: current perspectives and future directions. Italian J Gynaecol Obstet. 2019;31. 10.14660/2385-0868-125.
19. Yu CH, Weng SF, Ho CH, et al. Pregnancy outcomes following nonobstetric surgery during gestation: a nationwide population-based case-control study in Taiwan. BMC Pregnancy Childbirth. 2018;18(1):460. doi:10.1186/s12884-018-2079-4
20. Newton E. Surgical problems in pregnancy. Glob Libr Women’s Med. 2008. doi:10.3843/GLOWM.10191
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
