Back to Journals » Clinical Ophthalmology » Volume 8
Optic neuropathy associated with the use of over-the-counter sexual enhancement supplements
Authors Karli S, Liao S, Carey A, Lam BL, Wester S
Received 22 August 2014
Accepted for publication 16 September 2014
Published 29 October 2014 Volume 2014:8 Pages 2171—2175
DOI https://doi.org/10.2147/OPTH.S73059
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Scott Fraser
Sapir Z Karli, Sophie D Liao, Andrew R Carey, Byron L Lam, Sara T Wester
Bascom Palmer Eye Institute, University of Miami Miller School of Medicine, Miami, FL, USA
Abstract: This case report details an association of the use of over-the-counter sexual enhancement supplements with atypical optic neuropathy. A 42-year-old man presented with right-sided headache and vision loss of the right eye, which deteriorated to a single quadrant of hand motion over 11 days. Serial orbital magnetic resonance imaging scans demonstrated progressive orbital optic nerve enhancement extending into the optic canal despite high-dose steroid treatment. The patient eventually admitted to using several over-the-counter sexual enhancement supplements prior to the onset of symptoms and throughout the course of his steroid treatment, which he subsequently discontinued. His vision improved to 20/200 with an expanded visual field. Anterior ischemic optic neuropathy has been reported in association with phosphodiesterase (PDE)-5 inhibitor use, but visual loss in association with unregulated sexual enhancement supplements has not been studied. While one case cannot establish association, our case is suggestive of potential dangers of over-the-counter sexual enhancement supplements, which may contain PDE-5 inhibitors, “male hormones,” and “substances that enhance blood production.” The case also underscores the importance of obtaining a careful history of supplements in patients with optic neuropathies.
Keywords: optic neuropathy, PDE-5 inhibitor, sildenafil
Introduction
Phosphodiesterase (PDE)-5 inhibitors are commonly prescribed for erectile dysfunction.1–4 Inhibition of PDE-5 leads to vasodilation, which may affect ophthalmic circulation and reduce optic nerve perfusion.2,4,5 Ocular complications have been reported to include anterior ischemic optic neuropathy (AION), posterior ischemic optic neuropathy, color perception impairment, optic atrophy, angle closure glaucoma, branch retinal artery occlusion, and central serous chorioretinopathy.2,3,5–7
Posterior ischemic optic neuropathy has been reported to have a possible association with PDE inhibitors and Chinese “health products” containing PDE-5 inhibitors. Several reported cases have highlighted the importance of considering this in the differential diagnosis of patients with suspected retrobulbar optic neuritis or a compressive lesion when the clinical course is atypical and refractory to corticosteroid treatment.6,7
Initial reports on the link between PDE-5 inhibitors and AION were inconsistent.1,3–5,8 The strongest and most recent data demonstrate a small but clinically and statistically significant increase in risk of AION from PDE-5 inhibitor exposure.9 Several prospective studies are in progress.10,11 Visual loss in association with the use of unregulated sexual enhancement supplements has not been studied. Herein, we describe a case of atypical optic neuropathy associated with the use of over-the-counter sexual enhancement supplements.
Case report
A 42-year-old healthy man was evaluated initially elsewhere for acute right-sided headache followed by visual loss of the right eye with pain on eye movement. Best-corrected visual acuity was 20/400 in the right eye and 20/20 in the left eye, with a right relative afferent pupillary defect. The right eye had 30% red desaturation and an inferonasal visual field defect. Optic disc edema was not detected, and a brain magnetic resonance imaging (MRI) without contrast was unremarkable. After 5 days of deteriorating vision, the patient was prescribed 40 mg prednisone daily.
The patient was then referred to our clinic for progressive visual loss and persistent pain with eye movement in the right eye. At initial presentation, 7 days after onset of symptoms, vision in the right eye was hand motion temporally with a relative afferent pupillary defect. The optic nerve had mild edema with blurring of the superior margin. Orbit MRI with contrast revealed enhancement of the right optic nerve and sheath (Figure 1A and B), as well as nonspecific periventricular, deep, and subcortical white matter lesions. The patient was started on intravenous methylprednisolone 1 g daily, with a plan to assess clinical response frequently, as the MRI optic nerve and sheath enhancement suggested an inflammatory component.12
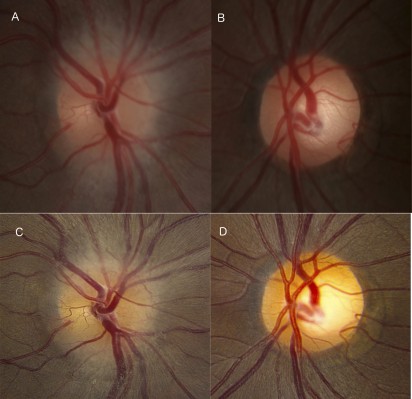
Repeat examination at 11 days after the onset of symptoms demonstrated narrowing of the visual field of the right eye to hand motion in a small temporal island and persistent right afferent pupillary defect. The right optic nerve head was diffusely edematous without hemorrhage (Figure 2A), and the left optic nerve head was normal with a 0.5 cup-to-disc ratio (Figure 2B). A systemic workup for inflammatory etiologies was unremarkable, including anti-aquaporin-4 antibody, anti-neutrophil cytoplasmic antibodies, angiotensin-converting enzyme, rapid plasma reagin, and fluorescent treponemal antibody, as well as Leber hereditary optic neuropathy genotyping, chest X-ray, and cerebral spinal fluid analysis. Due to lack of improvement in vision, intravenous methylprednisolone was discontinued after 8 days and the patient was started on oral prednisone 60 mg daily for 1 week, with a taper of 40 mg daily for 1 week, followed by 20 mg daily for 1 week.
Three weeks after the onset of symptoms, the right eye vision was still hand motion only in the superotemporal quadrant. The right optic nerve head showed persistent edema and the left optic nerve remained normal (Figure 2C and D). Goldmann visual field (GVF) showed severe constriction of the visual field, with a small superotemporal and superonasal island of vision remaining (Figure 3A). Repeat MRI showed enhancement of the right optic nerve extending to the optic canal (Figure 1C and D). Given the progressive clinical course and the lack of significant improvement with intravenous corticosteroids, atypical optic neuritis with perineuritis, ischemic optic neuropathy, and malignant optic glioma were considered. After discussing the case in a multidisciplinary meeting, the decision was made to observe closely and continue prednisone with the aforementioned taper, with the option of an optic nerve or sheath biopsy should the patient’s condition worsen further.
Seven weeks after the onset of symptoms, the vision in the right eye had improved to 20/400; repeat GVF showed expansion of the superior field (Figure 3B), eye pain resolved, and a repeat MRI found decreased right optic nerve enhancement (Figure 1E and F). The patient eventually admitted to intermittent use of several over-the-counter sexual enhancement supplements throughout the course of his steroid treatment without disclosing the exact amount or frequency, which he had discontinued prior to his clinical improvement. Four months after onset of visual loss, his headache and pain with eye movement had resolved. GVF showed improved sensitivity in the superior field and a persistent inferior defect in the right eye (Figure 3C). Visual acuity of the right eye was 20/200, at which point it remained stable.
Discussion
The association between PDE-5 inhibitors prescribed for erectile dysfunction and optic neuropathy is still under investigation.10,11 Recent data demonstrate a small but statistically significant increase in the risk of AION from PDE-5 inhibitor exposure.9 While the role of PDE-5 inhibitors in causing ischemic injury to the optic nerve is unknown, there may be an association between AION and PDE-5 inhibitors due to their vasomotor effects on the nitric oxide/cyclic guanosine monophosphate (NO/cGMP) pathway.4 The NO/cGMP pathway is important for both regulation of normal ocular blood flow and photoreceptor transduction.3 PDE-5 breaks down cGMP and therefore reduces NO-induced vasodilation.3 PDE-5 inhibitors increase cGMP concentration and subsequently may lead to vasodilation and reduction in optic nerve perfusion. Optic nerve hypoperfusion may also occur because PDE-5 is found in smooth muscle cells of the systemic arteries and veins throughout the body, and thus PDE-5 inhibitors may induce systemic hypotension because of the mild systemic vasodilator effects.13
Our patient developed atypical optic neuropathy and eventually admitted to using several over-the-counter “herbal” sexual enhancement supplements prior to his visual symptoms and throughout his steroid treatment, which had a Chinese label claiming it may contain PDE-5 inhibitors, “male hormones,” and “substances that enhance blood production.” He did not disclose the amount or frequency of supplement use. The patient’s progressive clinical course was not typical of optic neuritis or AION. The patient’s age, pain with ocular motility, optic nerve head edema, and optic nerve enhancement on MRI were more consistent with optic neuritis and perineuritis. However, the lack of improvement and progression of the optic nerve enhancement on MRI despite high-dose corticosteroids is not typical for optic neuritis. The MRI findings are also not consistent with AION wherein optic nerve enhancement is not usually present.14–16
We hypothesize that the continued use of over-the-counter sexual enhancement supplements may have led to a reduction of optic nerve perfusion, leading to serial ischemic insults. While one case cannot establish association, our case may suggest the potential dangers of unregulated over-the-counter sexual enhancement supplements, and patients using these supplements should be educated about the potential risks. In the event of optic neuropathy, these medications should likely be discontinued to reduce the possible risk of further visual loss to both the affected and the unaffected eyes. Our case also underscores the importance of obtaining a careful history of supplements in patients with optic neuropathies.
Acknowledgments
This work was supported by the National Institutes of Health Center Core (grant P30EY014801), and a Research to Prevent Blindness Unrestricted Grant.
Disclosure
The authors report no conflicts of interest in this work.
References
Gorkin L, Hvidsten K, Sobel RE, Siegel R. Sildenafil citrate use and the incidence of nonarteritic anterior ischemic optic neuropathy. Int J Clin Pract. 2006;60:500–503. | ||
Hayreh SS. Non-arteritic anterior ischaemic optic neuropathy and phosphodiesterase-5 inhibitors. Br J Ophthalmol. 2008;92(12):1577–1580. | ||
Laties AM. Vision disorders and phosphodiesterase type 5 inhibitors: a review of the evidence to date. Drug Safety. 2009;32(1):1–18. | ||
Pomeranz HD, Smith KH, Hart WM, Egan RA. Sildenafil-associated nonarteritic anterior ischemic optic neuropathy. Ophthalmology. 2002;109: 584–587. | ||
Azzouni F, Abu Samra K. Are phosphodiesterase type 5 inhibitors associated with vision-threatening adverse events? A critical analysis and review of the literature. J Sex Med. 2011;8:2894–2903. | ||
Cullen JF, Chung HW. Mistaken diagnosis of optic neuritis and the possible role of phosphodiesterase-5 inhibitors (sildenafil/Viagra). Med J Malaysia. 2010;65(4):317–318. | ||
Su DHW, Ang PS, Tow SLC. Bilateral posterior ischemic optic neuropathy associated with use of sildenafil. J Neuroophthalmol. 2008; 28(1):75. | ||
McCulley TJ, Lam BL, Marmor MF, Hoffman KB, Luu JK, Feuer WJ. Acute effects of sildenafil (Viagra) on blue-on-yellow and white-on-white Humphrey perimetry. J Neuroophthalmol. 2000;20(4):227–228. | ||
Pfizer. A study to assess whether PDE5 inhibitors increase the chance of triggering the onset of acute NAION. In: ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US); 2000 [cited May 7, 2014]. Available from: http://clinicaltrials.gov/show/NCT00759174. NLM Identifier: NCT00759174. | ||
Bayer. PDE5 inhibitor use and non-arteritic anterior ischemic optic neuropathy (NAION). In: ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US); 2000 [cited May 7, 2014]. Available from: http://clinicaltrials.gov/show/NCT00867815. NLM Identifier NCT00867815. | ||
Eli Lilly and Company. A study to determine if there is a possible association between nonarteritic anterior ischemic optic neuropathy (NAION) and phosphodiesterase type 5 (PDE5) inhibitors. In: ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US); 2000 [cited May 7, 2014]. Available from: http://clinicaltrials.gov/show/NCT01131104. NLM Identifier: NCT01131104. | ||
Hoitsma E, Faber CG, Drent M, Sharma OP. Neurosarcoidosis: a clinical dilemma. Lancet Neurol. 2004;3(7):397–407. | ||
Kloner RA. Cardiovascular effects of the 3 phosphodiesterase-5 inhibitors approved for the treatment of erectile dysfunction. Circulation. 2004;110(19):3149–3155. | ||
Rizzo JF 3rd, Andreoli CM, Rabinov JD. Use of magnetic resonance imaging to differentiate optic neuritis and nonarteritic anterior ischemic optic neuropathy. Ophthalmology. 2002;109:1679–1684. | ||
Arnold AC. Ischemic optic neuropathy. In: Miller NR, Newman NJ, Biousse V, Kerrison JB, editors. Walsh & Hoyt’s Clinical Neuro-Ophthalmology. Vol. I. 6th ed. Philadelphia: Lippincott Williams & Wilkins; 2005:349–384. | ||
Ischemic Optic Neuropathy Decompression Trial Research Group. Characteristics of patients with nonarteritic anterior ischemic optic neuropathy eligible for the Ischemic Optic Neuropathy Decompression Trial. Arch Ophthalmol. 1996;114:1366–1374. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.